Introduction
Infection with the hepatitis C virus (HCV) affects
approximately 200 million people worldwide. It is a leading cause
of chronic hepatitis, cirrhosis and hepatocellular carcinoma (HCC).
It is estimated that 25% of HCC worldwide is related to HCV. HCV
may be implicated in the development of HCC in an indirect way
through the induction of chronic inflammation, or directly by means
of viral proteins activating several signaling pathways. For
patients with chronic hepatitis C, there is a widespread agreement
that antiviral therapy is mandatory in order to eliminate the
virus. The current standard of care is a combination of pegylated
interferon (IFN) and ribavirin for 24–48 weeks, depending on the
genotype. Previous studies have demonstrated that a high HCV load
prior to treatment and infection with the HCV genotype 1b (HCV-1b)
are independent predictive factors for poor response to IFN
treatment (1,2). The majority of Chinese patients are
infected with HCV-1b (3). In
addition, the response to IFN treatment may vary in patients
infected with the same genotype and with similar viral loads,
indicating that other factors are involved.
A strong characteristic of HCV infection is its
significant genetic diversity, the consequence of the absence of
proofreading activity in RNA-dependent RNA polymerase (4), and the high level of viral
replication during its life cycle (5). As a result, the infecting HCV clones
in each patient invariably demonstrate population diversity with a
high degree of genetic heterogeneity. The collection of viruses in
a population of closely related but non-identical genomes is
referred to as a quasispecies (6,7),
and the dominant viral population may be evolve as a result of its
viral replicative fitness and concurrent immune selection pressures
that drive clonal selection.
It is reasonable to assume that the viral
pathogenesis and sensitivity to treatment are affected by the
generation of escape mutants through immune evasion and the
modification of virulence characteristics by anti-viral treatment
(8). Thus, certain viral
mutations have important implications for the pathogenesis of viral
disease and the sensitivity to antiviral therapy.
Previous reports have suggested that the degree of
quasi-species heterogeneity of the hypervariable region (HVR)1 and
NS5A regions is predictive of the effectiveness of IFN therapy
(9,10); however, but there have been
conflicting opinions (11,12).
In this study, mutations in different regions of the HCV genome,
including HVR1, HVR2 and NS5A, were identified in 14 Chinese
patients with chronic HCV-1b infection, with the objective of
assessing the correlation between mutations and the therapeutic
efficacy of IFN.
Patients and methods
Patients
Fourteen patients infected with HCV-1b were enrolled
in the study. Patients included were 8 men and 6 women, with ages
ranging from 23 to 45 years. This study was conducted in accordance
with the Declaration of Helsinki and approval from the Ethics
Committee of Sheng Jing Hospital of China Medical University.
Written informed consent was obtained from all participants. The
patients received pegylated IFN-α-2a (180 μg/week) and
ribavirin (1,000 mg/day) for 24 weeks. This was the first treatment
for each patient. After the completion of treatment, patients with
normal levels of serum alanine aminotransferase (ALT) and who were
negative for HCV RNA for longer than 6 months were classified as
sustained virological responders, and the others were classified as
non-responders. Prior to treatment, sera were obtained from the
patients and stored at −70°C until analysis.
Amplification of E2/NS1 and NS5A gene
segments
HCV RNA was extracted from serum samples, and the
E2/NS1 and NS5A gene segments were amplified using
reverse-transcription polymerase chain reaction (RT-PCR), following
previously published methods (13). The primer sequences are listed in
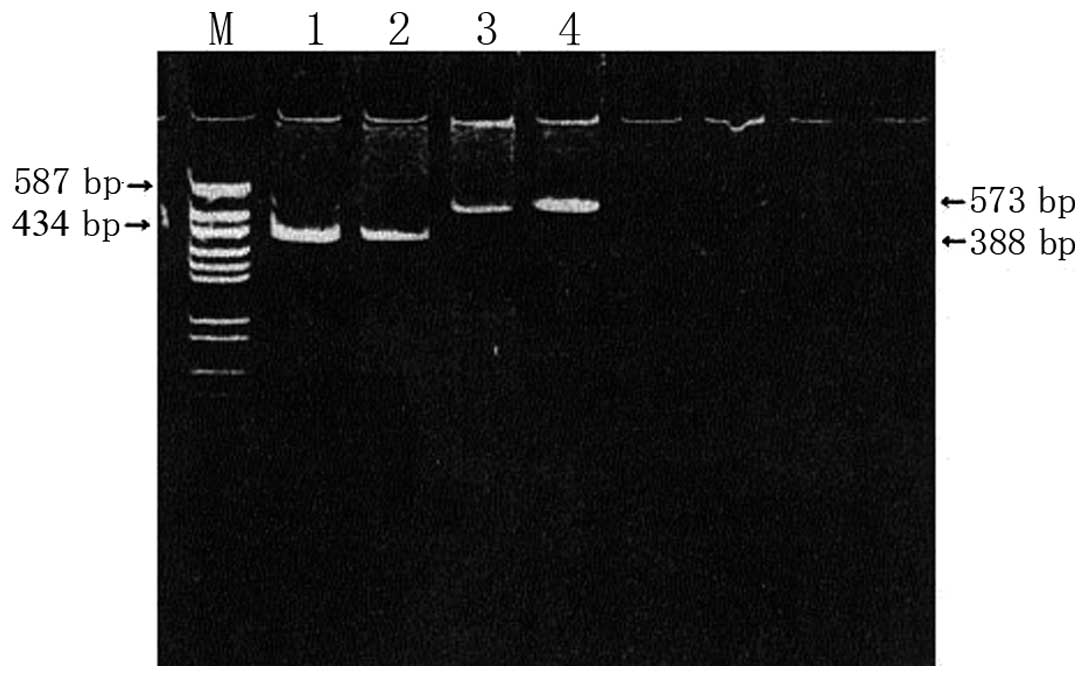
Table I; the sizes of the E2/NS1
and NS5A products were 388 and 573 bp, respectively (Fig. 1).
 | Table IPrimer sequences for E2/NS1 and
NS5A. |
Table I
Primer sequences for E2/NS1 and
NS5A.
| Region | Primer
sequences | Nucleotide
position |
|---|
| E2/NS1 | Outer primer 1 |
5′-CTACTCCGGATCCCACAAGC-3′ | 1009–1028 |
| Outer primer 2 |
5′-GGGCTCGGAGTGAAGCAATA-3′ | 1519–1538 |
| Inner primer 1 |
5′-GGATTCATTCCTTGGTGGGGAACTGG-3′ | 1085–1104 |
| Inner primer 2 |
5′-GAATCCGCAATACACTGGACCACACA-3′ | 1504–1523 |
| NS5A | Outer primer 1 |
5′-TTGGCCAGCTCTTCAGCTAGC-3′ | 6921–6936 |
| Outer primer 2 |
5′-CCAGGACTCTAGCAGTGGAC-3′ | 7211–7232 |
| Inner primer 1 |
5′-TCAGCTAGCCAATTGTCTGC-3′ | 6933–6952 |
| Inner primer 2 |
5′-CAGTGGAGGGTTGTAGTCTG-3′ | 7200–72200 |
Quasispecies determination for HCV E2/NS1
regions
We used primer 5.0 software to design the primers
for E2/NS1 PCR. The forward primer contained a BamHI (Takara
D1010A) digestion site, and the reverse primer contained an
EcoRI (Takara D1040A) digestion site. The sequences of both
primers were the following for RT-PCR (Takara DR027A): P1, 5′-GGA
TTCATTCCTTGGTGGGGAACTGG-3′; and P2, 5′-GAAT
CCGCAATACACTGGACCACACA-3′. The PCR product was identified by 1.5%
agarose gel electrophoresis, and a corresponding DNA band of
interest was purified using the DNA gel purification kit (Takara
DV805A). The concentration of purified DNA was measured.
The purified DNA of interest (0.3 pmol) was ligated
with the pMD18-T simple vector (Takara D103A) at 16°C overnight.
The ligated product was transformed into E. coli EZ
competent cells. Single positive clone colonies were selected to
extract the plasmids of interest using the plasmid miniprep kit
(Takara DV801A). BamHI and EcoRI were used for double
digestion. Agarose gel electrophoresis was used for identification
before and after digestion.
The DNA of interest was sent to be sequenced by the
Shanghai United Gene Technology Group Company, and the sequencing
results were analyzed using computer software.
Evaluation of NS5A genetic variation
PCR products of NS5A were purified, and the
nucleotide sequences were determined by direct sequencing. The
evaluation of NS5A genetic variation was finished by the Shanghai
United Gene Technology Group Company.
Statistical analysis
Statistical analysis was performed with the aid of
SPSS 15.0 software. Differences between 2 groups were performed
using the Chi-square test and t-test. P-values <0.05 were
considered to indicate a statistically significant difference.
Results
E2/NS1 quasispecies heterogeneity and IFN
plus ribavilin therapy
PCR amplification of the E2/NS1 region was positive
in 10 of 14 patients. Out of the 10 patients, 4 were responders and
6 were non-responders. A total of 93 sequences were obtained among
100 clones. The amino acid sequences of HVR1 and HVR2 are shown in
Figs. 2 and 3. The degree of complexity (number of
quasispecies) and nucleotide diversity (mean nucleotide variation
rate/site) of HVR1 quasispecies was greater in the non-responder
group than in the responder group. There were no significant
differences observed for HVR2 quasispecies between the responders
and non-responders (Tables II and
III).
 | Table IIHVR1 and HVR2 quasispecies in HCV 1b
patients undergoing IFN therapy. |
Table II
HVR1 and HVR2 quasispecies in HCV 1b
patients undergoing IFN therapy.
| Group | No. of
patients | Clone no. | HVR1 quasispecies
(n) | HVR2 quasispecies
(n) |
|---|
| Responder | 4 | 38 | 7 | 6 |
| Non-responder | 6 | 55 | 32a | 11b |
 | Table IIIGenomic diversity of responders and
non-responders. |
Table III
Genomic diversity of responders and
non-responders.
| Group | HVR1
| HVR2
|
|---|
| Mean nucleotide
variation rate/site | Mean amino acid
variation rate/site | Mean nucleotide
variation rate/site | Mean amino acid
variation rate/site |
|---|
| Responder |
1.146×10−2 |
4.156×10−2 |
0.89×10−2 |
2.55×10−2 |
| Non-responder |
3.563×10−2a |
7.625×10−2a |
1.19×10−2 |
3.26×10−2 |
NS5A gene variation and IFN therapy
Out of the 14 patients, 11 were positive for NS5A
RT-PCR amplification. The number of mutant nucleotides before IFN
treatment is shown in Table IV.
Patients 3 and 4 were responders to the treatment and the other
patients were non-responders. There was no significant difference
in the number of nucleotide mutations between the responder and
non-responder groups (t=0.86, P>0.05). Compared with the HCV-J
strain, the number of nucleotide mutations was not >10%, and
most were synonymous mutations. These results indicate that the
NS5A region is highly conserved. There was no association between
mutations leading to amino acid changes in NS5A2209–2248
and response to IFN therapy (Fig.
4).
 | Table IVNucleotide sequences of NS5A of
patients compared with HCV-J. |
Table IV
Nucleotide sequences of NS5A of
patients compared with HCV-J.
| No. of
patients | No. of nucleotide
mutations | Nucleotide mutation
rate (%) |
|---|
| 1 | 9 | 5.2 |
| 2 | 10 | 5.8 |
| 3 | 10 | 5.8 |
| 4 | 9 | 5.2 |
| 5 | 13 | 7.6 |
| 6 | 8 | 4.7 |
| 7 | 8 | 4.7 |
| 8 | 15 | 8.8 |
| 9 | 11 | 6.4 |
| 10 | 9 | 5.2 |
| 11 | 9 | 5.2 |
Discussion
To date, there is no effective vaccine for the
prevention of HCV infection. IFN plus ribavirin remains the primary
treatment. The rates of sustained virological response to IFN
therapy is approximately 70%. Certain refractory patients do not
respond to the treatment or relapse after treatment. To improve the
response rate, the prediction of treatment efficacy to obtain
satisfactory treatment options is critical. Among the HCV
genotypes, HCV-1b, which is the predominant cause of infection in
China, has a lower response rate to IFN treatment compared to other
genotypes. Therefore, predicting the response to IFN treatment in
patients with HCV-1b infection is clinically important.
In this study, the association between HCV gene
variation in different regions of the genome and response to IFN
therapy was assessed in 14 patients infected with genotype 1b. We
found that the degree of HVR1 quasispecies heterogeneity in the
non-responders was higher than in the responders, and there was no
significant difference observed for HVR2 quasispecies
heterogeneity. These results indicate that the greater the degree
of HVR1 quasispecies heterogeneity in an infected patient, the more
likely the patient will not respond to IFN treatment. However,
Abbate et al (11)
reported that the complexity of HVR1 quasispecies can only be used
to predict earlier response rates to IFN therapy and is not
associated with sustained virological response rates.
Patients with a higher degree of HVR1 quasispecies
heterogeneity before treatment had lower IFN response rates,
suggesting that there may be strains resistant to IFN in these
infected patients. The development of resistant strains may be
associated with immune selective pressure, and HVR1 is located on
the virus where it may be subjected to immune pressure (14,15). IFN therapy may enhance the
antiviral immune response, but also increases immune selective
pressure against HVR1, leading to the development of greater
variation. When the replication of IFN-sensitive strains is
inhibited, mutant strains that adapt to the new environment become
dominant.
In the E2/NS1 region, the degree of variation in
HVR2 is second to variation in HVR1. HVR2 may be associated with
immune recognition. In this study, the genomic complexity and
diversity of HVR2 quasispecies were less than those observed for
HVR1, and had no association with response to IFN. These findings
should be confirmed by additional studies with larger numbers of
patients.
Previously, Enomoto et al (16) reported that a small region in NS5A
(NS5A2209–2248) of HCV-1b was associated with
sensitivity to IFN. Compared with the HCV-J strain, mutant-type
NS5A2209–2248 regions (number of amino acid mutations
≥3) were sensitive to IFN therapy; intermediate-types (amino acid
mutations =1–3) and wild-types were resistant to IFN. They
designated NS5A2209–2248 the IFN sensitivity-determining
region (ISDR). However, these results have not been confirmed in
European countries (17–19). In our study, we found that
NS5A2209–2248 was highly conserved; among 11 patients
with HCV-1b infection, 10 patients were infected with wild-type HCV
and 1 was infected with the intermediate type. The 2 responders
were in the wild-type group. There were no significant differences
in the neucleotide and amino acid sequences between the responders
and non-responders. Therefore, the amino acid sequence of
NS5A2209–2248 may not be predictive of response to IFN
therapy in Chinese patients. Chayama et al (20) also found that substitutions in
NS5A2209–2248 were rare and that therefore the concept
of ISDR was not applicable.
Since the number of patients in our study was very
small, and no mutant-type infections were observed, whether or not
mutant-type HCV is sensitive to IFN treatment could not be
ascertained. By contrast, the study reported by Enomoto et
al (16) had many patients
infected with mutant-type HCV-1b. These differences may be a result
of the geographical variation in HCV-1b infections. These
differences need to be further investigated.
Acknowledgements
This study was supported by the
National Major Special Infectious Disease Foundation of the
National Eleventh Five-Year Plan Period of China
(2008ZX10002-013).
References
|
1.
|
M MasaroneM PersicoAntiviral therapy: why
does it fail in HCV-related chronic hepatitis?Expert Rev Anti
Infect Ther9535543201110.1586/eri.11.1021609265
|
|
2.
|
M RipoliV PazienzaImpact of HCV genetic
differences on pathobiology of diseaseExpert Rev Anti Infect
Ther9747759201110.1586/eri.11.9421905784
|
|
3.
|
GG XuSM WuXQ ZhouClinical characteristics
of 638 patients infected with hepatitis C virus by post blood
transfusion, in ShanghaiZhonghua Liu Xing Bing Xue Za
Zhi323883912011(In Chinese)
|
|
4.
|
DA SteinhauerE DomingoJJ HollandLack of
evidence for proofreading mechanisms associated with an RNA virus
polymeraseGene122281288199210.1016/0378-1119(92)90216-C1336756
|
|
5.
|
A NeumannN LamH DahariHepatitis C viral
dynamics in vivo and the antiviral efficacy of interferon-alpha
therapyScience282103107199810.1126/science.282.5386.103
|
|
6.
|
M MartellJI EstebanJ QuerHepatitis C virus
(HCV) circulates as a population of different but closely related
genomes: quasispecies nature of HCV genome distributionJ
Virol663225322919921313927
|
|
7.
|
JM PawlotskyHepatitis C virus population
dynamics during infectionCurr Top Microbiol
Immunol299261284200616568902
|
|
8.
|
C ArgentiniD GenoveseS DettoriM
RapicettaHCV genetic variability: from quasispecies evolution to
genotype classificationFuture
Microbiol4359373200910.2217/fmb.09.819327119
|
|
9.
|
MM ElHefnawiS ZadaIA El-AzabPrediction of
prognostic biomarkers for interferon-based therapy to hepatitis C
virus patients: a meta-analysis of the NS5A protein in subtypes 1a,
1b, and 3aVirol J15130201010.1186/1743-422X-7-13020550652
|
|
10.
|
BI YehKH HanHW LeeFactors predictive of
response to interferon-alpha therapy in hepatitis C virus type 1b
infectionJ Med Virol66481487200210.1002/jmv.216911857525
|
|
11.
|
I AbbateO Lo IaconoR Di StefanoHVR-1
quasispecies modifications occur early and are correlated to
initial but not sustained response in HCV-infected patients treated
with pegylated-or standard-interferon and ribavirinJ
Hepatol40831836200410.1016/j.jhep.2004.01.019
|
|
12.
|
G SquadritoG RaffaT RestucciaIs
investigation of hepatitis C virus NS5A gene heterogeneity a tool
for predicting long-lasting response to interferon therapy in
patients with HCV-1b chronic hepatitis?J Viral
Hepat9360369200210.1046/j.1365-2893.2002.00379.x12225331
|
|
13.
|
L ZhangGZ ZhaoY LiDynamic changes of HVR1
quasispecies in chronic hepatitis C after IFN therapyWorld Chin J
Digestol111821842003(In Chinese)
|
|
14.
|
P FaciA ShimodaD WongPrevention of
hepatitis C virus infection in chimpanzees by hyperimmune serum
against the hypervariable region 1 of the envelope 2 proteinProc
Natl Acad Sci
USA931539415399199610.1073/pnas.93.26.153948986822
|
|
15.
|
YK ShimizuH IgarashiT KiyoharaA
hyperimmune serum against a synthetic peptide corresponding to the
hyper-variable region 1 of hepatitis C virus can prevent viral
infection in cell
culturesVirology223409412199610.1006/viro.1996.0497
|
|
16.
|
N EnomotoI SakumaY AsahinaMutations in the
nonstructural protein 5A gene and response to interferon in
patients with chronic hepatitis C 1b infectionN Engl J
Med3347781199610.1056/NEJM1996011133402038531962
|
|
17.
|
M GerottoF Dal PeroP PontissoTwo PKR
inhibitor HCV proteins correlate with early but not sustained
response to
interferonGastroenterology11916491655200010.1053/gast.2000.2023011113086
|
|
18.
|
H KhorsiS CsatelainA WyseurMutations of
hepatitis C virus 1b NS5A 2209–2248 amino acid sequence
do not predict the response to recombinant interferon-alfa therapy
in French patientsJ Hepatol27727719979252076
|
|
19.
|
S ZeuzemJH LeeWK RothMutations in the
nonstructural protein 5A gene of European hepatitis C virus
isolates and response to interferon
alphaHepatology25740744199710.1002/hep.5102503419049228
|
|
20.
|
K ChayamaF SuzukiA TsubotaAssociation of
amino acid sequence in the PKR-eIF2 phosphorylation homology domain
and response to interferon
therapyHepatology3211381144200010.1053/jhep.2000.1936411050067
|