Introduction
Liver cancer is one of the most frequent lethal
malignancies worldwide. In 2012, ~782,500 people were diagnosed
with liver cancer and 745,500 mortalities occurred worldwide, with
China alone accounting for ~50% of all cases and mortalities
(1). Its incidence is also expected
to increase in the future (1).
Previous studies have advanced the prevention and treatment of
liver cancer (2). However, excluding
chemotherapy as a conventional option for patients following
surgical treatment, other additional interventions, including
targeted therapy remain limited (3).
It is therefore important to determine the molecular mechanisms
underlying the development and progression of liver cancer for the
development of potential therapeutic targets.
Long non-coding RNAs (lncRNAs), which are defined as
a class of RNA with a length of >200 nucleotides that do not
encode proteins (4), have been
demonstrated to participate in various cellular and molecular
processes, including genomic imprinting, chromatin modification,
transcriptional control and post-transcriptional control (5). Given that lncRNAs exert notable
functions in various biological processes, the abnormal expression
of lncRNAs may therefore be associated with various diseases,
including human cancer (6,7). Furthermore, emerging data have revealed
that the transcriptome of hepatocarcinoma tissues exhibits a
distinct alteration of lncRNAs compared with normal tissues
(8–10). However, the majority of these
abnormally expressed lncRNAs are poorly investigated and their
roles in the development of liver cancer are largely elusive.
Currently, the biological function of microRNAs
(miRNAs or miRs) is widely recognized. Mature miRNAs specifically
bind to the 3′untranslated region (UTR) of target mRNAs for
cleavage or translational inhibition (11). Several lncRNAs have been revealed to
function as competing endogenous RNAs (ceRNAs) that sequester
miRNAs via base-pair targeting, which leads to a loss of miRNA
function along with the upregulated expression of endogenous target
genes (12–14). LncRNAs exhibit a high capacity to
modulate the expression of specific targets that are closely
associated with tumor cell proliferation, survival, apoptosis,
invasion and migration via ceRNA regulation, thus serving crucial
roles in cancer initiation and progression (15).
The current study revealed that a particular lncRNA,
zinc-finger E-box binding homeobox 1 antisense 1 (ZEB1-AS1), was
highly expressed in liver cancer, which may be used to predict a
poor prognosis for patients with liver cancer. Furthermore, the
results demonstrated that ZEB1-AS1 competitively binds to
miR-365a-3p and abolishes the repression on E2F transcription
factor 2 (E2F2) caused by miR-365a-3p, resulting in increased E2F2
expression and enhanced liver cancer cell proliferation. The
current study enhances the understanding of the mechanism that
underlies ZEB1-AS1 in liver cancer progression. Therefore, ZEB1-AS1
maybe a novel target for the therapeutic treatment of liver
cancer.
Materials and methods
Clinical samples
The present study was approved by the Ethics
Committee of Sichuan Provincial People's Hospital (Chengdu, China)
and all patients provided written informed consent. A total of 32
patients (male, n=18 and female, n=14; age range, 49–75 years)
diagnosed with hepatocarcinoma who underwent surgery at the
Department of Hepatobiliary Surgery, Sichuan Provincial People's
Hospital (Chengdu, China) were randomly enrolled between March 2013
and January 2015. Exclusion criteria included radiotherapy or
chemotherapy prior to surgical treatment, a prior history of
cancer, and a lack of the written informed consent. Hepatocarcinoma
tissue and matched adjacent noncancerous tissue samples, 2 cm away
from the edge of the carcinoma lesion, were obtained from patients
during surgery and experienced pathologists confirmed diagnosis.
All procedures were performed in accordance with the Helsinki
Declaration.
Cell culture and transfection
HepG2, 293T and HCCLM6 cell lines were purchased
from the American Type Culture Collection (Manassas, VA, USA).
Cells were cultured in Dulbecco's Modified Eagle's medium (Gibco;
Thermo Fisher Scientific, Inc., Waltham, MA, USA) supplemented with
10% fetal bovine serum (HyClone; GE Healthcare, Chicago, IL, USA)
and maintained at 37°C in a 5% CO2-humidified incubator.
miR-365a-3p mimic (5′-UAAUGCCCCUAAAAAUCCUUAU-3′), mimic negative
control (NC; miR-NC; 5′-AUGUACCUAACUAAUCUACACU-3′), miR-365a-3p
inhibitor (5′-AUUACGGGGAUUUUUAGGAAUA-3′) and an inhibitor negative
control (5′-ACUAUUGGAGAUUAGGUAUGUA-3) were purchased from Shanghai
GenePharma Co., Ltd. (Shanghai, China). The ZEB1-AS1 short hairpin
RNA (shRNA) (5′-GACAGATGTGATCTCTGAACCTGAT-3′), synthesized by
Shanghai GenePharma Co., Ltd. (Shanghai, China), was cloned into
the pGHP1/Neo vector (Shanghai GenePharma Co., Ltd.) to generate
sh-ZEB1-AS1, as previously described (16). A scrambled sh-NC (Shanghai GenePharma
Co., Ltd.) was used as the negative control. Cells were transfected
with 50 nM miRNA mimic, 50 nM miRNA inhibitor or 4.0 µg
corresponding plasmids (sh-NC or sh-ZEB1-AS1) using
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocol.
Following 48-h transfection, cells were harvested for subsequent
experimentation.
To establish stable sh-ZEB1-AS1 cell lines, HepG2
and HCCLM6 cell lines were transfected with sh-ZEB1-AS1 or sh-NC
plasmids, respectively, using Lipofectamine® 2000 and
stable cell lines were selected for using 800 µg/ml neomycin at
37°C for ~3 weeks.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was extracted from cells or tissue samples
using TRIzol® reagent (Invitrogen; Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocol. Total
RNA was reverse transcribed into cDNA using the M-MLV Reverse
Transcriptase kit (Invitrogen; Thermo Fisher Scientific, Inc.),
according to the manufacturer's protocol. qPCR was subsequently
performed using a SYBR Green Master mix (Takara Biotechnology Co.,
Ltd., Dalian, China) and the Biosystems Real Time PCR System
(Thermo Fisher Scientific, Inc.). The following primer pairs were
used for the qPCR: miR-365a-3p forward,
5′-TAATGCCCCTAAAAATCCTTAT-3′ and reverse, miScript universal primer
(Shanghai GenePharma Co., Ltd.); ZEB1-AS1 forward,
5′-CCGTGGGCACTGCTGAAT-3′ and reverse, 5′-CTGCTGGCAAGCGGAACT-3′;
E2F2 forward, 5′-GAGCTCACTCAGACCCCAAG-3′ and reverse,
5′-AACAGGCTGAAGCCAAAAGA-3′; GAPDH forward,
5′-CTGGGCTACACTGAGCACCAG-3′ and reverse, 5′-CCAGCGTCAAAGGTGGAG-3′;
and U6 forward, 5′-CTCGCTTCGGCAGCACA-3′ and reverse,
5′-AACGCTTCACGAATTTGCGT-3′. The following thermocycling conditions
were used for the qPCR: Initial denaturation at 95°C for 3 min; 40
cycles of 95°C for 5 sec and 60°C for 30 sec. A melt curve step
from 65–95°C was performed in increments of 0.5°C per 5 sec.
Relative mRNA and miRNA levels were quantified using the
2−ΔΔCq method and normalized to GAPDH and U6,
respectively (17).
Cell proliferation and cell cycle
analysis
Cell proliferation was analyzed using the Cell
Counting Kit-8 (CCK-8; Dojindo Molecular Technologies, Inc.,
Kumamoto, Japan), according to the manufacturer's protocol.
Transfected cells were seeded into 96-well plates at a density of
3.0×103 cells/well and cultured for 24, 48, 72 and 96 h,
after which 10 µl CCK-8 reagent was added to each well. Cells were
incubation for a further 2 h at 37°C. The optical density of each
well was measured at a wavelength of 450 nm using a microplate
reader (Model 550; Bio-Rad Laboratories, Inc., Hercules, CA, USA).
Each experiment was performed in triplicate.
Proliferating cells were analyzed using the
Cell-Light EdU DNA Cell Proliferation kit (Guangzhou Ribobio Co.,
Ltd., Guangzhou, China), according to the manufacturer's protocol.
Proliferating cells were stained with fluorescent-labeled EdU for 2
h at room temperature. For the cell cycle assay,
~1.0×106 stained cells were fixed using 70% ethanol
overnight at 4°C. Subsequently, cells were stained with propidium
iodide (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) at 37°C for
1 h. Cells were analyzed using a flow cytometer (BD Biosciences,
Franklin Lakes, NJ, USA) and FlowJo software (version 7.6.1; Tree
Star, Inc., Ashland, OR, USA). Each experiment was performed in
triplicate (data not shown).
RNA pull-down assay
Biotinylated miR-365a-3p-wild-type (WT)
(5′-UAAUGCCCCUAAAAAUCCUUAU-3′), miR-365a-3p-mutant (Mut)
(5′-UUUACGGGCUAAAAAUCCUUAU-3′) and biotinylated mimic-NC
(5′-AUGUACCUAACUAAUCUACACU-3′) were synthesized by Shanghai
GenePharma Co., Ltd. (Shanghai, China). As previously described
(18), HepG2 cells were transfected
with the aforementioned biotinylated miRNA mimics (50 nM) using
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocol.
Following 48-h transfection, cells were harvested and lysed on ice
with a buffer containing 200 mM NaCl, 2.5 mM MgCl2, 20
mM Tris, pH 7.5, 0.05% Igepal, 60 U/ml Superase-In (Ambion; Thermo
Fisher Scientific, Inc.), 1 mM DTT and protease inhibitors (Roche
Applied Science, Rotkreuz, Switzerland) for 10 min. Subsequently,
cell lysates were incubated with M-280 streptavidin magnetic beads
(Sigma-Aldrich; Merck KGaA) for 3 h at 4°C. To avoid non-specific
binding of protein complexes and RNA, the beads were coated with
yeast tRNA and RNase-free bovine serum albumin (Sigma-Aldrich;
Merck KGaA). The beads were washed three times with low salt buffer
(0.1% SDS, 1% Trition X-100, 2 mM EDTA, 20 mM TrisHCl pH 8.0 and
150 mM NaCl) and once with high salt buffer (0.1% SDS, 1% Trition
X-100, 2 mM EDTA, 20 mM Tris-HCl pH 8.0 and 500 mM NaCl), Following
washing steps, bound RNA was extracted using TRIzol®
reagent, prior to RT-qPCR analysis, as previously mentioned.
Dual-luciferase reporter assay
Bioinformatic analysis was used to predict target
genes of miR-365a-3p using the miRDB database (http://mirdb.org). The WT ZEB1-AS1 cDNA fragment or WT
E2F2 3′UTR cDNA containing potential binding sites for miR-365a-3p
were amplified by PCR using the PfuUltra II Fusion HS DNA
Polymerase (Stratagene; Agilent Technologies, Inc., Santa Clara,
CA, USA). These fragments were subsequently cloned into the pmirGLO
vector (Promega Corporation, Madison, WI, USA) and designated
pmirGLO-ZEB1-AS1 WT and pmirGLO-E2F2 3′UTR WT, respectively. In
addition, the ZEB1-AS1 cDNA or E2F2 3′UTR cDNA fragments with point
mutations in the response element of the miR-365a-3p seed sequence
region were synthesized by Shanghai GenePharma Co., Ltd. (Shanghai,
China) and cloned into the pmirGLO vector and designated
pmirGLO-ZEB1-AS1 Mut and pmirGLO-E2F2 3′UTR Mut, respectively.
Cells were seeded into 96-well plates at a density of
1.5×104 293T cells/well and luciferase reporter vectors
were co-transfected with miR-365a-3p mimic or miR-NC using
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocol.
Following 48-h incubation at 37°C, cells were collected and the
relative firefly and Renilla luciferase activities were
detected using a Dual-Luciferase Reporter assay system (Promega
Corporation), according to the manufacturer's protocol. Firefly
luciferase activity was normalized to Renilla luciferase
activity.
Western blotting
Western blotting was performed as previously
described (19). Total cellular
protein was extracted using radioimmunoprecipitation assay buffer
(Beyotime Institute of Biotechnology, Haimen, China) containing a
proteinase inhibitor (Sigma-Aldrich; Merck KGaA). Total protein was
quantified using a bicinchoninic acid assay (Beyotime Institute of
Biotechnology), following the manufacturer's protocol using a
microplate reader (Model 550; Bio-Rad Laboratories, Inc.) In total
10 µg protein/lane was separated via SDS-PAGE on a 10% gel. The
separated proteins were transferred onto polyvinylidene difluoride
membranes and blocked for 1 h at 25°C with 5% non-fat milk. The
membranes were incubated with primary antibodies against E2F2
(1:500; cat. no. SAB4500684; Sigma-Aldrich; Merck KGaA) and GAPDH
(1:1,000; cat. no. 2118; Cell Signaling Technology, Inc., Danvers,
MA, USA) overnight at 4°C. Following primary incubation, membranes
were incubated with horseradish peroxidase-conjugated secondary
antibodies (1:1,000; cat. no. A0216; Beyotime Institute of
Biotechnology) for 2 h at room temperature. Protein bands were
visualized using the Pierce ECL Western Blotting kit (Pierce;
Thermo Fisher Scientific, Inc.). Protein expression was quantified
using Image-Pro® Plus software (version 6.0; Media
Cybernetics, Inc., Rockville, MD, USA).
Statistical analysis
All data are presented as the mean ± standard
deviation from three independent experiments. The overall survival
was analyzed using the Kaplan-Meier method, and patients were
classified as higher or lower than the mean value of ZEB1-AS1
expression. In addition, a Log-rank test was used to analyzed the
overall survival data. Pearson's Coefficient was applied to
evaluate the correlation between ZEB1-AS1 levels and E2F2 levels. A
two-tailed Student's t-test was performed to determine differences
between two groups, and paired Student's t-tests were used to
determine differences between hepatocarcinoma tissues and matched
adjacent noncancerous tissues. P<0.05 was considered to indicate
a statistically significant difference.
Results
High ZEB1-AS1 expression contributes
to liver cancer cell proliferation
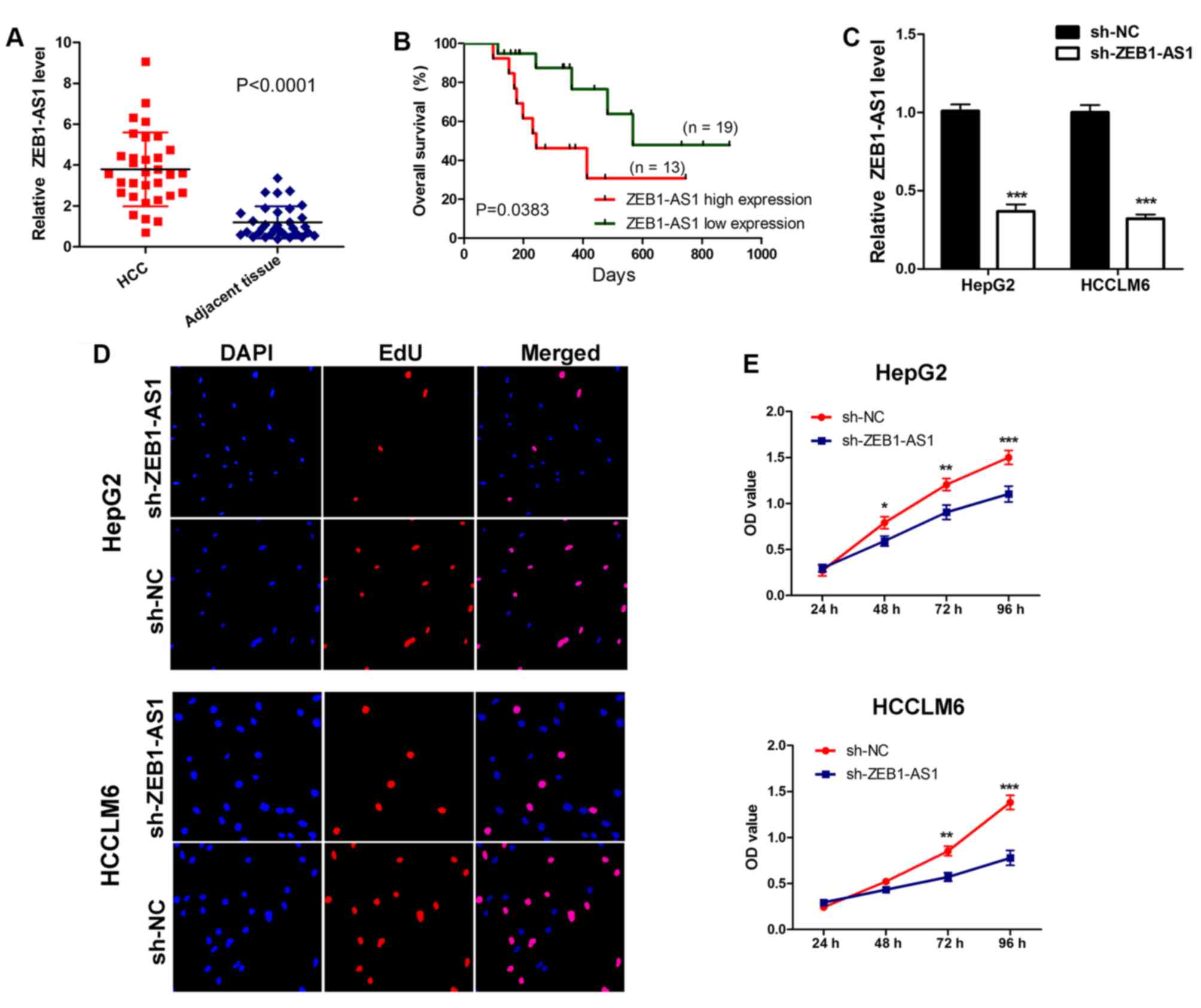
Initially, the expression of ZEB1-AS1 was assessed
in 32 pairs of hepatocarcinoma tissues and adjacent non-cancerous
tissues collected from patients. The results demonstrated that
ZEB1-AS1 levels were significantly upregulated in hepatocarcinoma
tissues compared with normal adjacent tissues (Fig. 1A). Subsequently, survival analysis
was performed on patients. It was revealed that patients with a
higher ZEB1-AS1 expression exhibited a significantly poorer overall
survival than those with a lower expression (Fig. 1B). These results indicate that
ZEB1-AS1 is highly expressed in hepatocarcinoma and may exhibit
oncogenic potency. Therefore, the current study aimed to determine
the role of ZEB1-AS1 in liver cancer. The expression of ZEB1-AS1 in
HepG2 and HCCLM6 cells was knocked down by stably transfecting
sh-RNA plasmids (Fig. 1C). Using a
CCK-8 assay and EdU staining, it was determined that the depletion
of ZEB1-AS1 suppressed cell proliferation and decreased EdU
incorporation, indicating that the downregulation of ZEB1-AS1 may
inhibit liver cancer cell proliferation (Fig. 1D and E). These results imply that
ZEB1-AS1 may be required for liver cancer cell proliferation.
ZEB1-AS1 is associated with
miR-365a-3p
miRDB target prediction revealed that the sequence
of ZEB1-AS1 possessed a response element to the seed sequence of
miR-365a-3p, indicating that a direct interaction between ZEB1-AS1
and miR-365a-3p may exist (Fig. 2A).
Subsequently, to determine whether miR-365a-3p interacts with
ZEB1-AS1 via the seed sequence region, a luciferase reporter assay
was performed using reporter vectors containing WT ZEB1-AS1 or Mut
ZEB1-AS1 (Fig. 2B). The results
demonstrated that the overexpression of miR-365a-3p decreased the
luciferase activity of WT reporters but not of empty or Mut
reporters (Fig. 2C). To assess
whether ZEB1-AS1 directly binds to miR-365a-3p, a biotin-avidin
pull-down assay was performed. Biotinylated miR-365a-3p mimics were
transfected into HepG2 cells to pull-down associated ZEB1-AS1
(Fig. 2D) and RT-qPCR was
subsequently performed to assess ZEB1-AS1 expression. The results
demonstrated that ZEB1-AS1 was enriched in the group transfected
with WT miR-365a-3p (Bio-miR-365a-3p-wt) but not in the group
transfected with Mut miR-365a-3p (Bio-miR-365a-3p-mut; Fig. 2E). These data indicated that
miR-365a-3p is able to bind to ZEB1-AS1. The effect of ZEB1-AS1 on
the expression of miR-365a-3p was assessed. The results
demonstrated that there was no marked difference in the expression
of miR-365a-3p in the ZEB1-AS1knockdown and negative control groups
(Fig. 2F). Thus, these findings
demonstrate that ZEB1-AS1 binds to miR-365a-3p but does not affect
the expression of miR-365a-3p. Therefore, ZEB1-AS1 might function
as a ceRNA for miR-365a-3p.
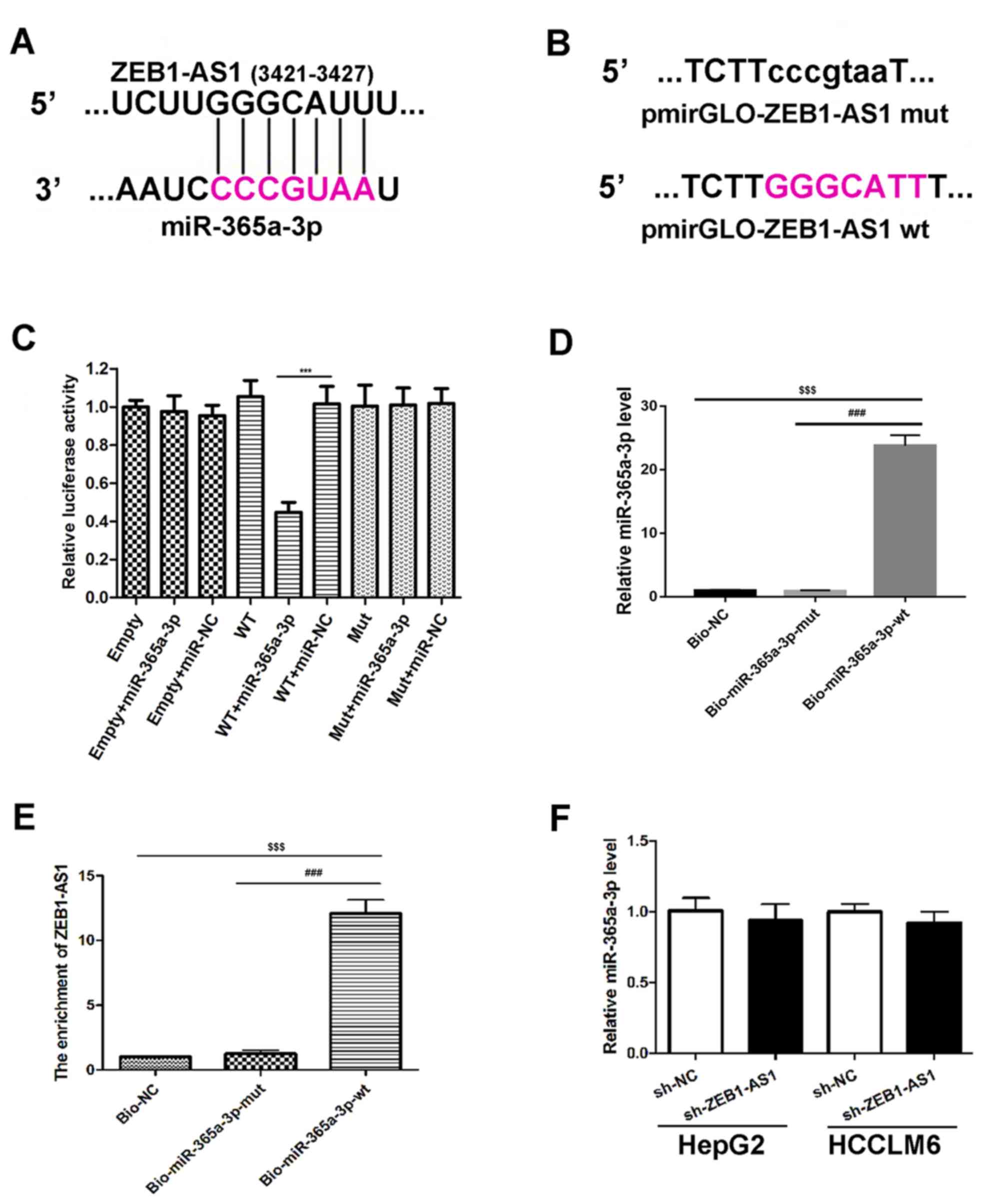 | Figure 2.Association between ZEB1-AS1 and
miR-365a-3p. (A) Schematic diagram presenting the putative
miR-365a-3p recognition site in the ZEB1-AS1 sequence. The
miR-365a-3p seed sequence region is marked in pink. (B) Schematic
diagram of indicated luciferase reporter vector constructs. The
un-mutated bases are highlighted in pink. (C) Luciferase activity
of 293T cells co-transfected with miRNA mimics, empty luciferase
pmirGLO reporters and luciferase pmirGLO reporters containing
ZEB1-AS1 wtor ZEB1-AS1 mut. ***P<0.001 vs. WT+miR-NC. (D)
Biotin-based miRNA mimics were transfected into HepG2 cells,
followed by RT-qPCR to assess miR-365a-3p expression. (E) RT-qPCR
and a biotin-based pull-down assay were utilized to assess ZEB1-AS1
expression in Bio-365a-3p-wt or Bio-365a-3p-mut transfected HepG2
cells. Bio-NC served as the negative control and the relative
enrichment of ZEB1-AS1 is presented. $$$P<0.001 vs.
Bio-NC and ###P<0.001 vs. Bio-miR-365a-3p-mut. (F)
RT-qPCR analysis of miR-365a-3p levels in HepG2 and HCCLM6 cells.
Cells transfected with sh-NC served as the control. Data are
presented as the mean ± standard deviation (n=3). ***P<0.001 vs.
WT+miR-NC; $$$P<0.001 vs. Bio-NC and
###P<0.001 vs. Bio-miR-365a-3p-mut. ZEB1-AS1,
zinc-finger E-box binding homeobox 1 antisense 1; miR, microRNA; WT
or wt, wild type; Mut or mut, mutant; Bio, biotinylated; RT-qPCR,
reverse transcription quantitative polymerase chain reaction; NC,
negative control; sh, short hairpin. |
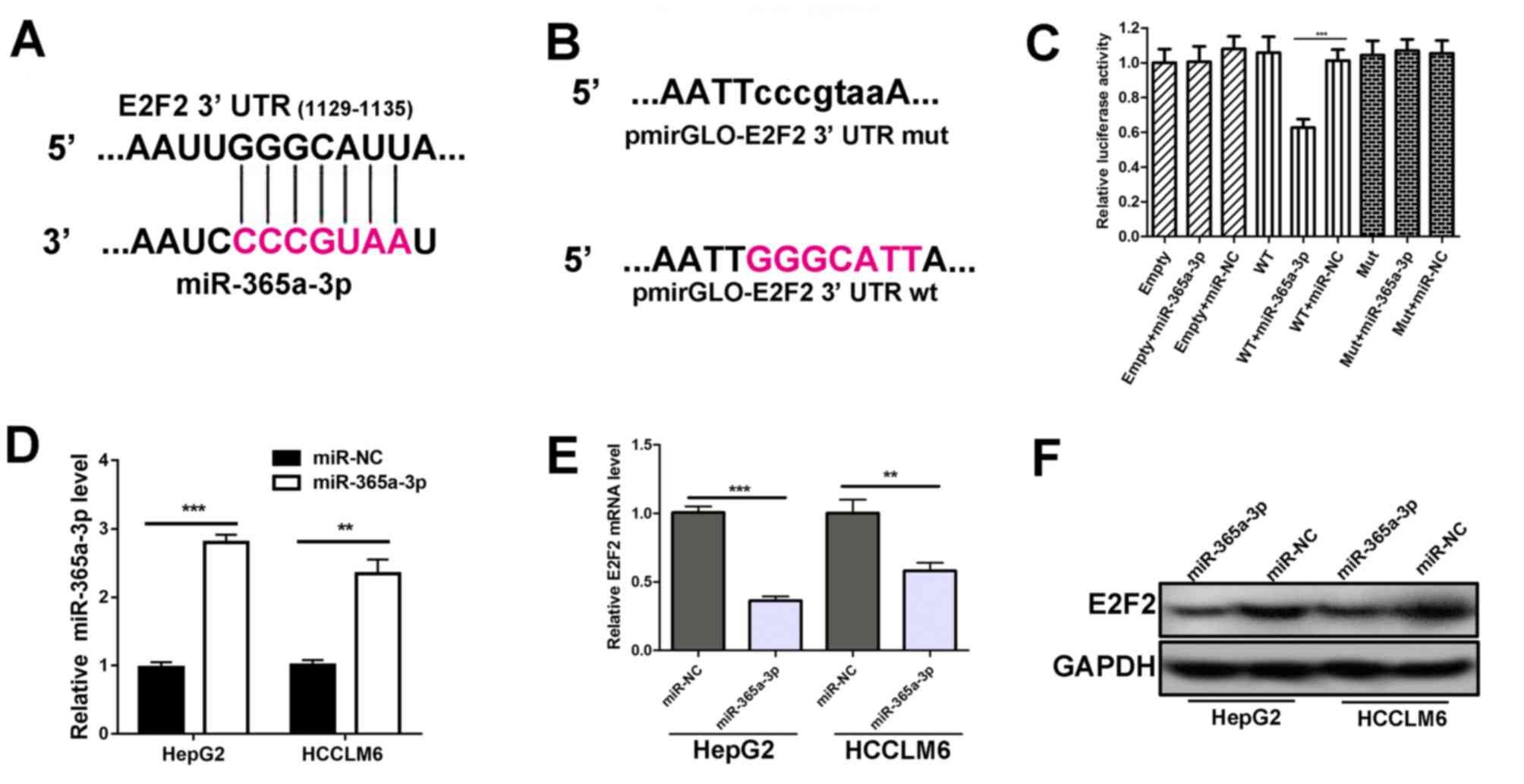
miR-365a-3p targets the 3′UTR of E2F2
and negatively regulates the expression of E2F2
Through miRDB database prediction, it was revealed
that the E2F2 3′UTR possessed a putative recognition site for
miR-365a-3p (Fig. 3A). Therefore,
luciferase reporter vectors were generated containing either WT
E2F2 3′UTR or Mut E2F2 3′UTR which lacked the response element to
the seed sequence region of miR-365a-3p (Fig. 3B). Subsequently, luciferase plasmids
were co-transfected with miR-365a-3p mimics or miR-NC into 293T
cells. The results demonstrated that the luciferase activity of
co-transfectedmiR-365a-3p mimics and WT E2F2 3′UTR was decreased
compared with the controls, indicating that miR-365a-3p targets the
E2F2 3′UTR at the seed sequence region (Fig. 3C). Furthermore, RT-qPCR demonstrated
that transfecting miR-365a-3p mimics without miR-NC led to reduced
E2F2 mRNA expression in HepG2 and HCCLM6 cells (Fig. 3D and E). In addition, western
blotting results were consistent with RT-qPCR data, indicating that
the overexpression of miR-365a-3p resulted in the downregulation of
E2F2 protein (Fig. 3F). These
results indicate that miR-365a-3p targets E2F2 3′UTR and inhibits
E2F2 expression.
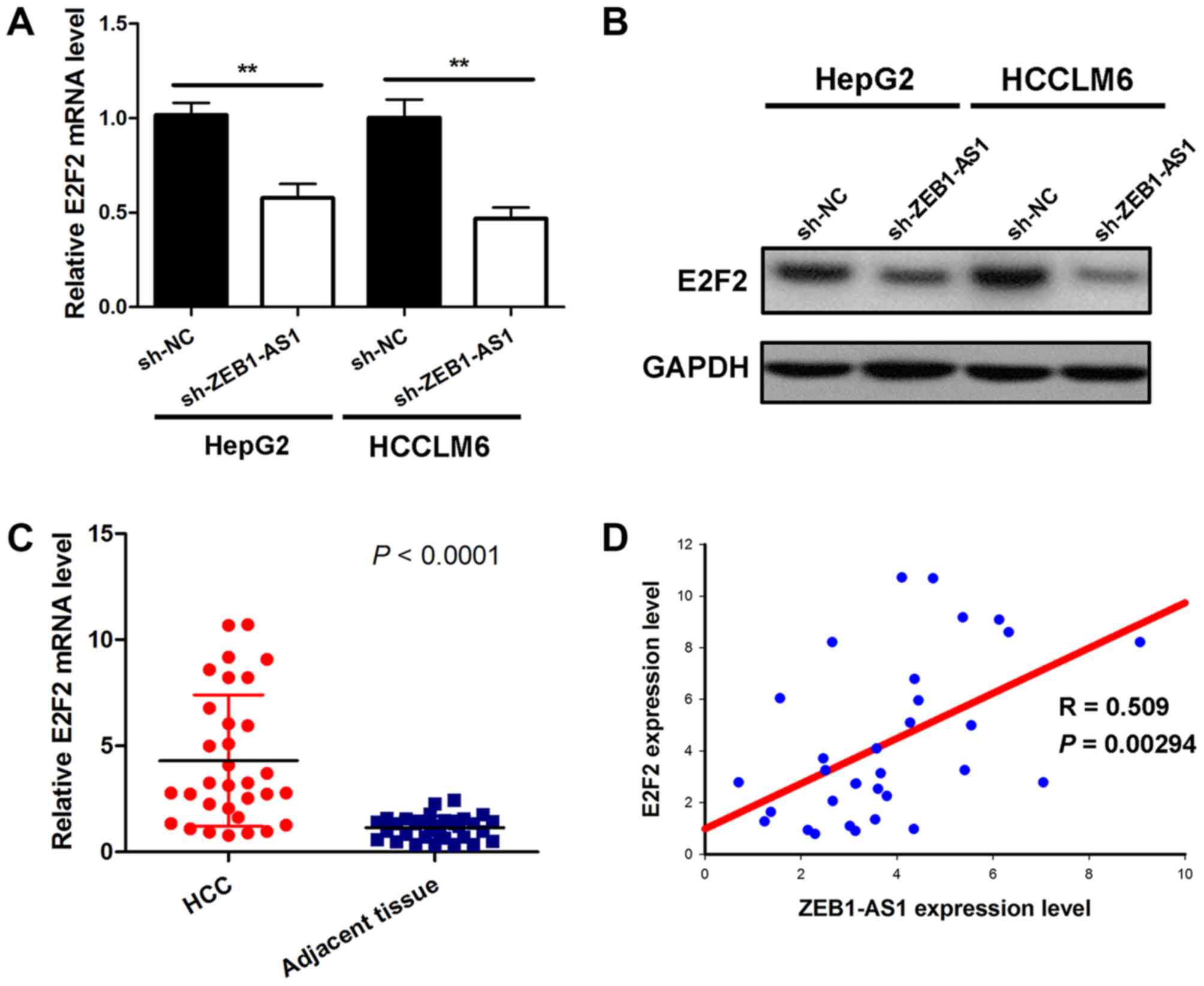
ZEB1-AS1 positively modulates E2F2
expression
Given that ZEB1-AS1 was demonstrated to bind to
miR-365a-3p without affecting the expression of miR-365a-3p, the
current study subsequently assessed whether ZEB1-AS1 indirectly
affected the target gene of miR-365a-3p by serving as a ceRNA. The
results demonstrated that ZEB1-AS1knockdown induced a significant
decrease of E2F2 mRNA in HepG2 and HCCLM6 cells (Fig. 4A). Additionally, depleting ZEB1-AS1
also reduced E2F2 protein expression in liver cancer cells
(Fig. 4B). Subsequently, the
expression of E2F2 mRNA in hepatocarcinoma and matched
para-carcinoma tissues from 32 patients was assessed. The results
demonstrated that E2F2 levels in tumor tissues were significantly
higher than those in adjacent normal tissues (Fig. 4C). A correlation analysis was then
performed on ZEB1-AS1 and E2F2 levels. The results demonstrated
that ZEB1-AS1 levels were positively correlated with E2F2 levels in
hepatocarcinoma tissues (Fig. 4D).
Thus, these results support the hypothesis that ZEB1-AS1 positively
regulates E2F2 expression in liver cancer.
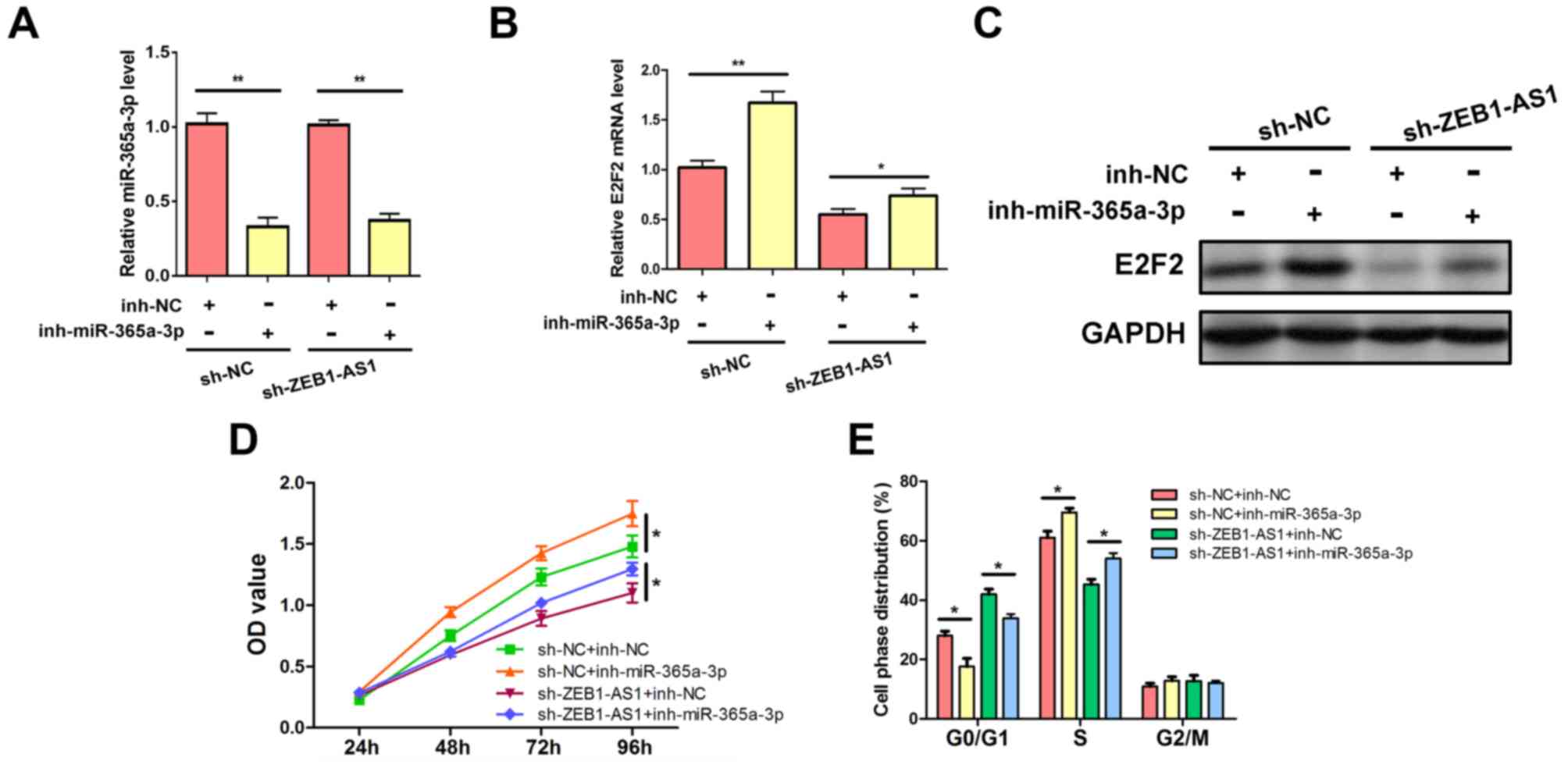
ZEB1-AS1 regulates E2F2 expression and
liver cancer cell proliferation by targeting miR-365a-3p
To ascertain whether ZEB1-AS1 modulates E2F2
expression by affecting miR-365a-3p repression activity, rescue
experiments were performed in which the effects of
ZEB1-AS1silencing were reversed by the inhibition of miR-365a-3p.
The expression of miR-365a-3p was suppressed by the miR-365a-3p
inhibitor (Fig. 5A). The results
revealed that the inhibition of miR-365a-3p abrogated the decreased
expression of E2F2 mRNA and protein, which was caused by
ZEB1-AS1silencing in HepG2 cells (Figs.
5B and C). The associated phenotypes were also examined. The
knockdown of ZEB1-AS1 suppressed HepG2 cell viability; however, the
rescue of miR-365a-3p inhibition abolished this effect and
re-elevated cell proliferation (Fig.
5D). In addition, flow cytometric analysis demonstrated that
the downregulation of ZEB1-AS1 resulted in the decrease of HepG2 S
phase populations and increased G0/G1 period proportions, which
were also abrogated by the inhibition of miR-365a-3p (Fig. 5E). These results demonstrated that
ZEB1-AS1 competitively binds to miR-365a-3p and inhibits the
repressive effect of miR-365a-3p on E2F2, which contributes to
elevated E2F2 expression and enhanced cell proliferation in liver
cancer.
Discussion
The ZEB1-AS1 transcript is a non-coding antisense
RNA that originates from the ZEB1 promoter region, which was
initially reported in hepatocellular cancer (20). Previous studies revealed that
ZEB1-AS1 is located in physical contiguity with ZEB1 and that it
positively modulates ZEB1, induces epithelial to mesenchymal
transition (EMT) and drives cancer metastasis (20,21).
Previous studies have also demonstrated that ZEB1-AS1 functions as
an oncogene and promotes the progression of certain tumors
including bladder cancer, osteosarcoma, glioma and esophageal
squamous cell carcinoma (16,22,23).
However, the exact mechanism underlying the oncogenic role of
ZEB1-AS1 has not yet been fully elucidated. In the present study,
consistent with a previous report, ZEB1-AS1 was highly expressed in
liver cancer tissue samples (21).
In addition, the high level of ZEB1-AS1 may be a predictor of poor
prognosis in patients with liver cancer. The present study also
revealed that ZEB1-AS1 exerted oncogenic effects in vitro by
enhancing liver cancer cell proliferation and promoting cell cycle
progression.
Previous studies have primarily elucidated the
mechanism by which ZEB1-AS1 exhibits tumor-promoting effects by
serving as a crucial regulator of ZEB1 expression (16,21,24).
Currently, three mechanisms by which ZEB1-AS1 controls the
expression of ZEB1 have been proposed. The first suggests that
ZEB1-AS1 may function as an enhancer and thus positively regulates
ZEB1 promoter activity (21). In the
second theory, ZEB1-AS1 directly binds and recruits p300 to the
ZEB1 promoter region, activating ZEB1 transcription (16). Thirdly, ZEB1-AS1 may serve as a
molecular sponge for miR-101 to relieve the inhibition of ZEB1
caused by miR-101 (24). Increased
ZEB1-AS1 expression has been demonstrated to contribute to the
upregulated expression of ZEB1, the progression to EMT and cancer
metastasis (20). Furthermore it is
also significantly associated with tumor progression and patient
survival, in many different types of cancer (25). However, few studies have examined
other mechanisms that involve the oncogenic role of ZEB1-AS1. Given
the complexity of tumor genetics, there may exist other pathways
and mechanisms by which ZEB1-AS1 contributes to tumor
progression.
In the present study, the ceRNA role of ZEB1-AS1 in
liver cancer progression was assessed. The results revealed that
ZEB1-AS1 interacts with miR-365a-3p and inhibits miR-365a-3p
function. Many previous studies have also demonstrated that miR-365
functions as a tumor-suppressor in various types of cancer
(26,27). A previous study also revealed that
the overexpression of miR-365 suppressed hepatocellular carcinoma
growth and induced apoptosis by directly targeting Bcl-2 (19). The results of the current study
demonstrated that ZEB1-AS1 serves as a ceRNA for miR-365a-3p to
positively regulate the expression of E2F2, which is a target of
miR-365a-3p. It was determined that ZEB1-AS1 levels were positively
correlated with E2F2 expression in liver cancer tissues, which
indicated a potential positive regulation between ZEB1-AS1 and E2F2
transcripts. Furthermore, bioinformatics analysis indicated that
ZEB1-AS1 and E2F2 3′UTR shared identical miR-365a-3p recognition
sites. These results indicated that ZEB1-AS1 is physically
associated with miR-365a-3p, while miR-365a-3p targets E2F2 3′UTR
for the degradation of RNA or the inhibition of translation.
Additionally, ZEB1-AS1 silencing led to reduced E2F2 mRNA and
protein levels, which was abolished by the inhibition of
miR-365a-3p. This may be due to ZEB1-AS1 competitively binding to
miR-365a-3p, causing a loss of miR-365a-3p function in the
repression of E2F2. Although depleting ZEB1-AS1 exhibited little
effect on total miR-365a-3p expression, low ZEB1-AS1 might allow
for increased levels of free miR-365a-3p, reducing the level of
E2F2 expression. Thus, miR-365a-3p inhibition may increase the
level of E2F2 expression.
The mammalian E2F family of transcription factors
(E2Fs) regulate a variety of cellular functions associated with
cell proliferation, differentiation, apoptosis and cell cycle
progression (28). Among these
family members, E2F2, as a transcriptional activator of E2F target
genes, exerts critical effects on the regulation of G1/S transition
as well as DNA replication in mammalian cells (29). Increasing evidences have revealed
that E2F2 functions as a tumor activator in gastric, breast and
non-small cell lung cancer (30–32).
Furthermore, it has been revealed that E2F2 is a tumor-promoter in
liver cancer (33). The current
study demonstrated that the ZEB1-AS1/miR-365a-3p axis serves an
important role in the regulation of E2F2 expression as well as in
liver cancer cell proliferation and cell cycle progression,
indicating that targeting this axis maybe a novel approach for the
efficacious intervention of liver cancer.
However, there are limitations in the current study.
The expression of ZEB1-AS1 and E2F2 were analyzed, and survival
analysis was performed using limited sample sizes. In addition,
corresponding knockout experiments were not performed. Thus,
further studies are required to validate the expression of ZEB1-AS1
and E2F2 in a large sample size and to further assess their
correlation and clinical significance. Knockout experiments should
also be considered in future studies to elucidate the role of
ZEB1-AS1 in liver cancer in vivo or in vitro.
Based on the results of the current study, ZEB1-AS1
was determined to positively modulate E2F2 expression and enhance
cell proliferation by competitively binding tomiR-365a-3p. The
current study further elucidated the mechanism underlying ZEB1-AS1
in liver cancer progression. The results demonstrated that ZEB1-AS1
maybe a potential target for liver cancer therapy.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Youth Science
Fund of the Sichuan Provincial People's Hospital (grant no.
30305030611).
Availability of data and materials
All datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
LM and GX designed the study. LM and GH analyzed the
data and prepared the figures. GX and LY prepared the manuscript.
All authors read and approved the final manuscript.
Ethics approval and consent to
participate
This study was approved by the Ethics Committee of
Sichuan Provincial People's Hospital (Chengdu, China).
Patient consent for publication
All the patients provided written informed consent
for the publication of any associated data and accompanying
images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Li L and Wang H: Heterogeneity of liver
cancer and personalized therapy. Cancer Lett. 379:191–197. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Forner A, Llovet JM and Bruix J:
Hepatocellular carcinoma. Lancet. 379:1245–1255. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Batista PJ and Chang HY: Long noncoding
RNAs: Cellular address codes in development and disease. Cell.
152:1298–1307. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Guttman M and Rinn JL: Modular regulatory
principles of large non-coding RNAs. Nature. 482:339–346. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wapinski O and Chang HY: Long noncoding
RNAs and human disease. Trends Cell Biol. 21:354–361. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Geisler S and Coller J: RNA in unexpected
places: Long non-coding RNA functions in diverse cellular contexts.
Nat Rev Mol Cell Biol. 14:699–712. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yang Y, Chen L, Gu J, Zhang H, Yuan J,
Lian Q, Lv G, Wang S, Wu Y, Yang YT, et al: Recurrently deregulated
lncRNAs in hepatocellular carcinoma. Nat Commun. 8:144212017.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Esposti DD, Hernandez-Vargas H, Voegele C,
Fernandez-Jimenez N, Forey N, Bancel B, Le Calvez-Kelm F, McKay J,
Merle P and Herceg Z: Identification of novel long non-coding RNAs
deregulated in hepatocellular carcinoma using RNA-sequencing.
Oncotarget. 7:31862–31877. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cui H, Zhang Y, Zhang Q, Chen W, Zhao H
and Liang J: A comprehensive genome-wide analysis of long noncoding
RNA expression profile in hepatocellular carcinoma. Cancer Med.
6:2932–2941. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Seitz H, Youngson N, Lin S, Dalbert S,
Paulsen M, Bachellerie JP, Ferguson-Smith AC and Cavaille J:
Imprinted microRNA genes transcribed antisense to a reciprocally
imprinted retrotransposon-like gene. Nat Genet. 34:261–262. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tay Y, Rinn J and Pandolfi PP: The
multilayered complexity of ceRNA crosstalk and competition. Nature.
505:344–352. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Salmena L, Poliseno L, Tay Y, Kats L and
Pandolfi PP: A ceRNA hypothesis: The Rosetta stone of a hidden RNA
language? Cell. 146:353–358. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Luo J, Qu J, Wu DK, Lu ZL, Sun YS and Qu
Q: Long non-coding RNAs: A rising biotarget in colorectal cancer.
Oncotarget. 8:22187–22202. 2017.PubMed/NCBI
|
|
15
|
Qi X, Zhang DH, Wu N, Xiao JH, Wang X and
Ma W: ceRNA in cancer: Possible functions and clinical
implications. J Med Genet. 52:710–718. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu C and Lin J: Long noncoding RNA
ZEB1-AS1 acts as an oncogene in osteosarcoma by epigenetically
activating ZEB1. Am J Transl Res. 8:4095–4105. 2016.PubMed/NCBI
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wang K, Liu CY, Zhou LY, Wang JX, Wang M,
Zhao B, Zhao WK, Xu SJ, Fan LH, Zhang XJ, et al: APF lncRNA
regulates autophagy and myocardial infarction by targeting
miR-188-3p. Nat Commun. 6:67792015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li M, Yang Y, Kuang Y, Gan X, Zeng W, Liu
Y and Guan H: miR-365 induces hepatocellular carcinoma cell
apoptosis through targeting Bcl-2. Exp Ther Med. 13:2279–2285.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Li J, Li Z, Leng K, Xu Y, Ji D, Huang L,
Cui Y and Jiang X: ZEB1-AS1: A crucial cancer-related long
non-coding RNA. Cell Prolif. 51:e124232018. View Article : Google Scholar
|
|
21
|
Li T, Xie J, Shen C, Cheng D, Shi Y, Wu Z,
Deng X, Chen H, Shen B, Peng C, et al: Upregulation of long
noncoding RNA ZEB1-AS1 promotes tumor metastasis and predicts poor
prognosis in hepatocellular carcinoma. Oncogene. 35:1575–1584.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lv QL, Hu L, Chen SH, Sun B, Fu ML, Qin
CZ, Qu Q, Wang GH, He CJ and Zhou HH: A long noncoding RNA ZEB1-AS1
promotes tumorigenesis and predicts poor prognosis in glioma. Int J
Mol Sci. 17:E14312016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang YL, Bai Y, Yao WJ, Guo L and Wang ZM:
Expression of long non-coding RNA ZEB1-AS1 in esophageal squamous
cell carcinoma and its correlation with tumor progression and
patient survival. Int J Clin Exp Pathol. 8:11871–11876.
2015.PubMed/NCBI
|
|
24
|
Xiong WC, Han N, Wu N, Zhao KL, Han C,
Wang HX, Ping GF, Zheng PF, Feng H, Qin L and He P: Interplay
between long noncoding RNA ZEB1-AS1 and miR-101/ZEB1 axis regulates
proliferation and migration of colorectal cancer cells. Am J Transl
Res. 10:605–617. 2018.PubMed/NCBI
|
|
25
|
Ni Y, Fang J, Zhu L, Jiang H, Liu Y, Miao
R, Shao C and Shao S: The significant prognostic value of ZEB1-AS1
up-regulation in patients with cancer. J Cancer. 9:2502–2509. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang Y, Xu C, Wang Y and Zhang X:
MicroRNA-365 inhibits ovarian cancer progression by targeting
Wnt5a. Am J Cancer Res. 7:1096–1106. 2017.PubMed/NCBI
|
|
27
|
Nie J, Liu L, Zheng W, Chen L, Wu X, Xu Y,
Du X and Han W: microRNA-365, down-regulated in colon cancer,
inhibits cell cycle progression and promotes apoptosis of colon
cancer cells by probably targeting Cyclin D1 and Bcl-2.
Carcinogenesis. 33:220–225. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chen HZ, Tsai SY and Leone G: Emerging
roles of E2Fs in cancer: An exit from cell cycle control. Nat Rev
Cancer. 9:785–797. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
29
|
Reimer D, Sadr S, Wiedemair A, Goebel G,
Concin N, Hofstetter G, Marth C and Zeimet AG: Expression of the
E2F family of transcription factors and its clinical relevance in
ovarian cancer. Ann N Y Acad Sci. 1091:270–281. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wang H, Zhang X, Liu Y, Ni Z, Lin Y, Duan
Z, Shi Y, Wang G and Li F: Downregulated miR-31 level associates
with poor prognosis of gastric cancer and its restoration
suppresses tumor cell malignant phenotypes by inhibiting E2F2.
Oncotarget. 7:36577–36589. 2016.PubMed/NCBI
|
|
31
|
Nguyen-Vu T, Vedin LL, Liu K, Jonsson P,
Lin JZ, Candelaria NR, Candelaria LP, Addanki S, Williams C,
Gustafsson JA, et al: Liver × receptor ligands disrupt breast
cancer cell proliferation through an E2F-mediated mechanism. Breast
Cancer Res. 15:R512013. View
Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chen L, Yu JH, Lu ZH and Zhang W: E2F2
induction in related to cell proliferation and poor prognosis in
non-small cell lung carcinoma. Int J Clin Exp Pathol.
8:10545–10554. 2015.PubMed/NCBI
|
|
33
|
Zhan L, Huang C, Meng XM, Song Y, Wu XQ,
Miu CG, Zhan XS and Li J: Promising roles of mammalian E2Fs in
hepatocellular carcinoma. Cell Signal. 26:1075–1081. 2014.
View Article : Google Scholar : PubMed/NCBI
|