Introduction
Enterococcus faecalis
(E. faecalis), a generally commensal organism, has
emerged as the major pathogen for persistent periapical
periodontitis (1). Nonpathogenic
(or commensal) bacterial pathogens can switch from the commensal
stage to a pathogenic state (2).
The virulence of E. faecalis is derived from its ability to
sense and adapt to varying environmental stresses during
colonization and in the host (3).
During long-term interaction with the host, the two-component
signal transduction system (TCS) of E. faecalis regulates
the expression of virulence genes in response to microenvironmental
conditions, including the host environment, and binds to the
regulatory regions of target genes accordingly (4). These two-component signal transduction
systems are involved in various cellular processes, including cell
viability, virulence, biofilm formation, quorum sensing and
antibiotic resistance (5). A
typical TCS consists of a histidine sensory kinase membrane
receptor and its cognate response regulator (6). The histidine kinase is activated by
recognition of environmental stimuli, including pH, oxidative
stress, antibiotic pressure and nutrient starvation, and relays the
activated phosphoryl group to the response regulator for gene
expression modulation by binding at the regulatory regions of
target genes (4).
The WalRK (also known as VicRK or YycFG) TCS
originally from Bacillus subtilis is highly conserved in
Gram-positive bacteria and is annotated as VicRK or YycFG in some
genera, such as Staphylococcus aureus, Enterococcus
faecalis, and Streptococcus mutans (S. mutans)
(6). The system WalRK has been
proposed because of its major roles in the regulation of genes
associated with cell wall synthesis and biofilm formation (7). The essential gene walR, is
closely associated with bacterial growth, virulence and biofilm
extracellular polysaccharide synthesis and aggregation (1). Bacterial growth and biofilm
aggregation are strongly associated with the process of human
infections (4). Consistently, the
ability of E. faecalis to form the three-dimensional biofilm
scaffold contributes to the failure of persistent infected root
canal treatments (8). In E.
faecalis, it was reported that the inability to construct
walR deletion mutants indicated that this regulatory gene
was essential for bacterial viability (4).
A noncoding antisense RNA (AS RNA) can bind to the
target messenger RNA (mRNA) by base-pairing, and their interaction
forms an RNA duplex structure (9).
This AS RNA-induced duplex complex generally inhibits mRNA
transcription or translation and performs regulatory functions
(10). Using high-throughput
transcriptomics analyses, AS RNA regulators can be identified in
bacteria (11). An endogenous
vicR antisense RNA transcript has been identified in S.
mutans (12). In addition, it
was found that the production of VicR protein associated inversely
with different levels of vicR antisense RNA and that the
biofilm biomass decreased in the vicR antisense
overexpression (12). Considering
the close proximity of E. faecalis and S. mutants in
related Gram-positive cocci, it was suggested that there were
similar structural and functional relationships between vicR
in S. mutans and walR in E. faecalis.
Additionally, by mapping the E. faecalis walR gene with
BLAST searches, a high similarity (the DNA sequences are 70%
identical) with the homologous vicR gene in S. mutans
was demonstrated. In the present study, a potential ASwalR
was hypothesized, and whether the potential ASwalR was
specifically associated with regulation of WalR function was
investigated in E. faecalis.
Materials and methods
Bacterial strains and growth
conditions
The bacterial strains and plasmids used in the
present study are listed in Table
SI. E. faecalis strains purchased from Guangdong Huankai
Microbial Science & Technology Co., Ltd. were grown in brain
heart infusion (BHI) broth (BD Biosciences) at 37˚C in a 5%
CO2 atmosphere with 500 µg/ml spectinomycin when
ASwalR overexpression strains were incubated at 37˚C
overnight. E. faecalis strains were cultured to
mid-exponential phase (optical density at a wavelength of 600 nm of
OD value=0.5). For antisense overexpression strain construction,
the shuttle vector pDL278 (Novagen) was used to overexpress
ASwalR or walR sequence under the control of the
walR gene promoter region. First, the antisense walR
or walR sequences were obtained by oligonucleotide synthesis
(Sangon Biotech Co., Ltd.). It was reported that E. faecalis
V583 carrying the empty vector pDL278 has no effects of the vector
itself on the phenotypes examined (13). Next, antisense ASwalR or
walR sequences were cloned into the pDL278 plasmid at
BamHI and EcoRI restriction sites. All recombinant
ASwalR RNA or walR gene overexpression plasmids were
verified by PCR and DNA sequencing analyses. The recombinant
plasmid pDL278ASwalR or pDL278walR was transformed
into E. faecalis V583 strains, as previously described
(1,12).
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNAs were purified from E. faecalis strains
using a MasterPure RNA purification kit (Epicentre; Illumina Inc.).
Residual genomic DNA was removed using Turbo RNase-free DNase I
(Ambion; Thermo Fisher Scientific, Inc.). The quality and integrity
of RNAs were assessed by 1% agarose gel electrophoresis. The
concentration and purity of RNAs were determined using a NanoDrop
8000 spectrophotometer (Thermo Fisher Scientific, Inc.). Total RNAs
were extracted from E. faecalis V583, ASwalR and walR
overexpression strains as aforementioned. Subsequently, purified
RNA was reverse transcribed to cDNA using a PrimeScript RT Reagent
kit (Takara Bio, Inc). RT-PCR was conducted for identification and
detection of antisense RNA. For ASwalR detection, a gene-specific
primer (PCR1) was used for the first strand cDNA synthesis, and
another gene-specific primer (AS2) was used for RT-qPCR analysis
(Supplementary Table II). The
expression of walR and walK and virulent factor genes including ace
(adhesin of collagen), esp (protein surface), epal, epaA
(enterococcal polysaccharide antigen), and gel (gelatinase) of all
E. faecalis strains were also examined. RT-qPCR was performed using
primers listed in Supplementary Table
II on a LightCycler 480 (Roche Diagnostics), following the
instructions of the PrimeScript RT Reagent kit (Takara Bio, Inc.).
RT-qPCR conditions were: 95°C for 30s, followed by 40 cycles at
95°C for 5s; 60°C for 30s, and then dissociation. The threshold
cycle values (CT) were quantified and transcription levels of each
gene were compared to the expression of the 16sR gene, which was
used as a reference gene (1).
Reverse transcription-PCR (RT-PCR)
assays for walR co-transcribed operator and antisense RNA
detection
Conditions for PCR reactions were: 94˚C, 3 min
(initial denaturation); 32 cycles at 94˚C, 30 sec (denaturation);
55˚C, 40 sec (primer annealing); and 72˚C, 1 min (primer
extension). For potential antisense walR RNA detection,
total RNAs were purified from E. faecalis strains after
planktonic growth using a MasterPure RNA purification kit
(Epicentre; Illumina Inc.). Residual genomic DNA was removed using
Turbo RNase-free DNase I (Ambion; Thermo Fisher Scientific, Inc.).
The quality and integrity of RNAs were assessed by 1% agarose gel
electrophoresis. The concentration and purity of RNAs were
determined using a NanoDrop 8000 spectrophotometer (Thermo Fisher
Scientific, Inc.).
Total RNA was prepared from strain E.
faecalis V583 grown as planktonic culture in BHI and used as
templates to validate walR and adjacent downstream genes,
walK, EF1195, EF1196, and EF1197 were co-transcribed
(12). The primers for PCR
amplification were presented in Table
SIII. For first-strand DNA synthesis, walR
antisense-(PCREf) and sense-specific primers (ASEf) were used. The
ASwalR RT-PCR product was purified with QIAquick PCR
Purification Kit according to the manufacturer's instructions and
subsequently sequenced. Detection of antisense RNA was performed by
1% agarose gel electrophoresis and RT-PCR assays were performed
using a first strand-specific primer for synthesis of cDNA
transcripts using the First Strand cDNA synthesis kit (Thermo
Fisher Scientific, Inc.) (14). The
primers (PCREf) used for first strand synthesis PCR are listed in
Table SII. Following the reverse
transcription reaction, the first-stand cDNA synthesis products
were used as templates. The sense strand primer (ASEf) for PCR
amplification is shown in Table
SIV.
Northern blotting
Northern blotting assays were performed as
previously described with minor modifications (12,15).
In brief, 10 µg of total RNA was purified from E. faecalis
planktonic cultures. The RNA samples were detected by
electrophoresis in 1% formaldehyde-agarose gels and transferred to
nylon membranes (Amersham; Cytiva). The probe was made specifically
(listed in Table SIV) and labeled
using the DIG High Prime DNA Labeling and Detection Starter Kit II
(Roche Diagnostics). Blots were incubated 16 h with 20 ng/ml probes
at 50˚C, and signals were detected using the CSPD Star substrate
(Roche Diagnostics).
5'-rapid amplification of cDNA ends
(5'-RACE) assay
To find the transcription initiation site and
probable termination site of ASwalR, total RNA (20 µg) from
E. faecalis strains was ligated to the 5'-RACE outer adapter
from the FirstChoice RLM-RACE Kit (Ambion; Thermo Fisher
Scientific, Inc.) according to the manufacturer's instructions
(12). The primers used for PCR
assays for 5'-RLM-RACE included 5'-RACE gene-specific outer primer
and 5'-RACE gene-specific inner primer as listed in Table SIV. The nested PCR thermocycling
conditions were as follows: Initial activation of 95˚C for 3 min;
followed by 35 cycles of denaturation at 94˚C for 30 sec, annealing
at 56˚C for 30 sec and extension at 72˚C for 1 min; and a final
extension at 72˚C for 7 min was performed thereafter. An aliquot (6
µl) of the PCR mixture was assessed by 1% agarose gel
electrophoresis using reactants without a cDNA template as the
control and sequencing by Sangon Biotech Co., Ltd..
Western blotting
Protein extraction and western blotting were
proceeded as previously described (12,13).
E. faecalis cells grown as planktonic cultures of
mid-exponential phase were washed and resuspended in 10 mM Tris-HCl
buffer (pH 8.0). Cells were mechanically disrupted by
ultrasonication with glass beads (diameter, 0.1 mm) for three
cycles of 15 sec with 1 min rest on ice. Clear supernatants were
collected by centrifugation (12,000 x g, 2 min, 4˚C) and protein
concentrations were determined using a Bradford assay (BioRad
Laboratories, Inc.). For western blotting, equal amounts of protein
(30 µg) were mixed with Laemmli sample buffer (BioRad Laboratories,
Inc.) in boiling water for 10 min. The protein samples were loaded
on precast 4-20%, gradient gels (Beijing Solarbio Science &
Technology Co., Ltd.) and electrotransferred to PVDF membranes
(Thermo Fisher Scientific, Inc.). Polyclonal antibodies against
r-WalR were produced using the standard 70 days Rabbit Protocol
(AbMax Biotechnology Co., Ltd.). The membranes were blocked, then
probed with purified WalR-specific rabbit antibody (AbMax
Biotechnology Co., Ltd.; http://www.antibodychina.com/, cat. no. scu001;
1:1,000) for 2 h at room temperature and incubated with horseradish
peroxidase-conjugated goat anti-rabbit secondary antibody (HRP
conjugated, cat. no. SSA004; 1:10,000) for 2 h at room temperature.
A BioRad GS-700 Imaging Densitometer (BioRad Laboratories, Inc.)
was used to determine the signal density of protein signals.
Biofilm assessment
E. faecalis V583, ASwalR and walR+
strains were cultured in BHI for 24 h at 37˚C as previously
described (1,12,13).
Crystal violet assay was performed to measure the biomass of each
E. faecalis biofilm. E. faecalis V583 biofilms were
set as controls. Briefly, the biofilms were washed three times with
PBS. Subsequently, the biofilms were stained with 0.1% (w/v)
crystal violet for 15 min at 37˚C and destained with 1 ml
ethanol/acetone (at a ratio of 8:2) solution. The collected
solution was removed to a new plate and measured with a microplate
reader (ELX800; Gene Company Ltd.) at a wavelength of 600 nm. For
confocal laser scanning microscopy (CLSM), the E. faecalis
stains in biofilm were labeled with SYTO9 (Invitrogen; Thermo
Fisher Scientific, Inc.) and the extracellular polymeric substance
(EPS) matrix was stained with an Alexa Fluor 647-labeled dextran
conjugate (Invitrogen; Thermo Fisher Scientific, Inc.) (1). Three-dimensional reconstruction of the
biofilms and the EPS/bacterial ratio were analyzed using Imaris 7.0
software (Bitplane; Oxford Instruments).
Periapical periodontitis lesions in
animal experiments
Animal experiments were approved by the Biomedical
Research Ethics Committee of West China Hospital (approval no.
2019128A) and conducted according to the guidelines of
Institutional Animal Care and Use Committee (IACUC) for animal care
and use of laboratory animals (16). A total of 20 6-week-old female
Sprague-Dawley rats (260-280 g) were included in the study and
anesthetized with ketamine/xylazine (90 and 10 mg/kg, respectively)
by intraperitoneal injection. On the occlusal surface of the left
mandible first molars, an access opening was made using a
high-speed round bur (Aseptico Inc.). The right first mandible
molar was used as a blank control. Next, the root canal preparation
was performed with sterile K-files (0.02 taper, sizes #15-#35).
Subsequently, 50 µl log-phased E. faecalis V583 bacterial
suspensions were inoculated into the pulp chamber and sealed with
light-cured flowable resin (3M Filtek™ Z350XT; 3M) in the V583
group (n=10). For the ASwalR group (n=10), 50 µl log-phased
ASwalR E. faecalis bacterial suspensions were applied for
establishment of an infection model. After 4 weeks of operation,
the rats were scarified by euthanasia under deep anesthesia using
ketamine/xylazine by cervical dislocation, and the rats were imaged
using the Quantum GX Micro-CT System (PerkinElmer, Inc.). The
scanning conditions were as follows: kV=90; CT µA=72; 360˚ scan
time=8 sec. The reconstructed images were analyzed with Analyze
12.0 (PerkinElmer, Inc.).
HE and Gram staining and PNA-FISH
For histological evaluation and peptide nucleic acid
fluorescence in situ hybridization (PNA-FISH), the
mandibular bone was longitudinally split into two parts. The
specimens were prepared for histological evaluation. The mandibular
bone was fixed in 10% neutral buffered formalin for 72 h at room
temperature, decalcified in 10% EDTA and embedded in paraffin. The
5-µm slices were prepared for hematoxylin and eosin (HE) and Gram
staining for tissue assessment with a light microscope at x40
magnification. For PNA-FISH, bone specimens were also fixed for 72
h in 10% neutral buffered formalin and decalcified in 10% EDTA
decalcified for 3 weeks before being embedded in paraffin blocks
and sectioned to 3 µm thickness. Smears were deparaffinized in
xylene (2x5 mins) and rehydrated by a graded ethanol series (100,
95, 80, 70 and 50%) for 5 min each time and permeated with
proteinase K (Wuhan ServiceBio Technology Co., Ltd.) at 37˚C for 25
min. Finally, all smears were washed up with distilled water for 10
min and allowed to air dry.
The above slides were fixed in 100% methanol for 10
min at 37˚C, followed by immersion in 80% ethanol for 10 min. For
pre-hybridization, one drop of hybridization solution without probe
containing 10% (w/v) dextran sulfate (Thermo Fisher Scientific,
Inc.), 10 mM NaCl (Panreac), 0.2% (w/v) polyvinylpyrrolidone
(Sigma-Adrich; Merck KGaA), 0.2% (w/v) Ficoll (Thermo Fisher
Scientific, Inc), 5 mM disodium EDTA (PanReac AppliChem), 0.1%
(v/v) Triton X-100 (PanReac AppliChem), 50 mM Tris-HCl (pH 7.5;
Thermo Fisher Scientific, Inc.) was applied for 1 h at 37˚C. From
the template of genomic DNA a fluorescein amidite-labeled PNA probe
(5'-GGTGTTGTTAGCATTTCG-3') targeting E. faecalis 16S rRNA
(Wuhan Servicebio Technology Co., Ltd.) was applied. The slides
were then incubated with hybridization solution containing 200 nM
PNA probe overnight at 37˚C. Subsequently, the slides were
co-cultured in saline sodium citrate washing buffer (Wuhan
ServiceBio Technology Co., Ltd.) at 37˚C for 30 min and dried in
air. Bone smears were then stained with 100 µl DAPI (Thermo Fisher
Scientific, Inc.) in the dark for 30 min at room temperature. The
samples were washed with PBS buffer twice and dried in air. The
slides were mounted with Gold Antifade reagent (Thermo Fisher
Scientific, Inc.) and stored at 4˚C for further investigation with
fluorescent microscope at 40 magnification.
Statistical analysis
Bartlett's test was conducted to analyze the
homogeneity of data variances and Shapiro-Wilk test was applied to
determine the normal distribution of data. For parametric testing,
one-way ANOVA was performed followed by pairwise multiple
comparisons of Tukey's test using SPSS software 18.0 (SPSS, Inc.).
Data are presented as the mean ± SD. P<0.05 was considered to
indicate a statistically significant difference.
Results
WalR gene operon contains an antisense
walR RNA
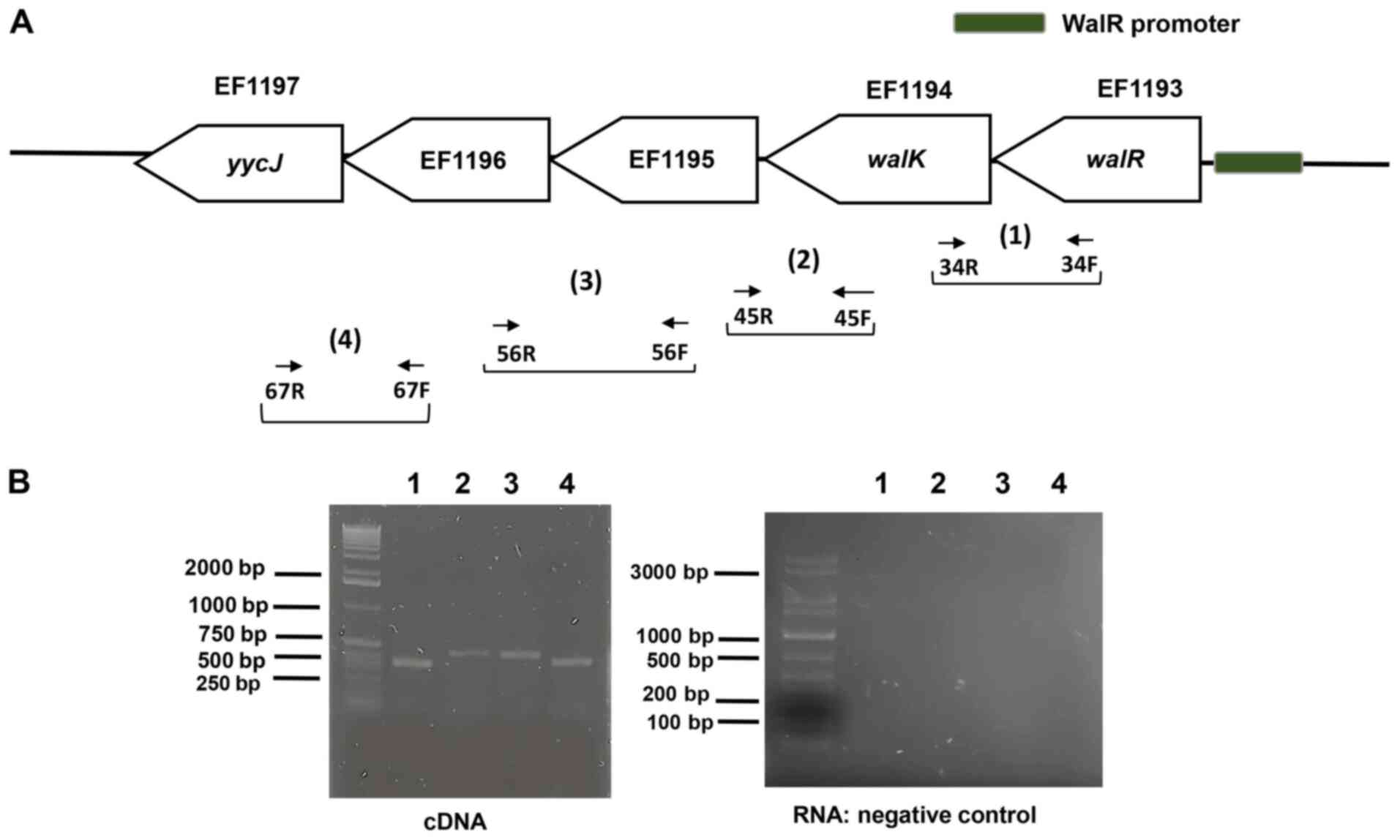
Downstream of the walR (EF1193) in E.
faecalis V583, a cluster of four genes (EF1194 to EF1197) is
co-transcribed (Fig. 1A). Using a
series of overlapping primers, RT-PCR indicated co-transcription
comprising walR and the four downstream genes (Fig. 1B), indicating that walR,
walK, EF1195, EF1196, and EF1197 are part of the WalR
regulon. RNA agarose gel electrophoresis for northern blotting was
included in Fig. S1.
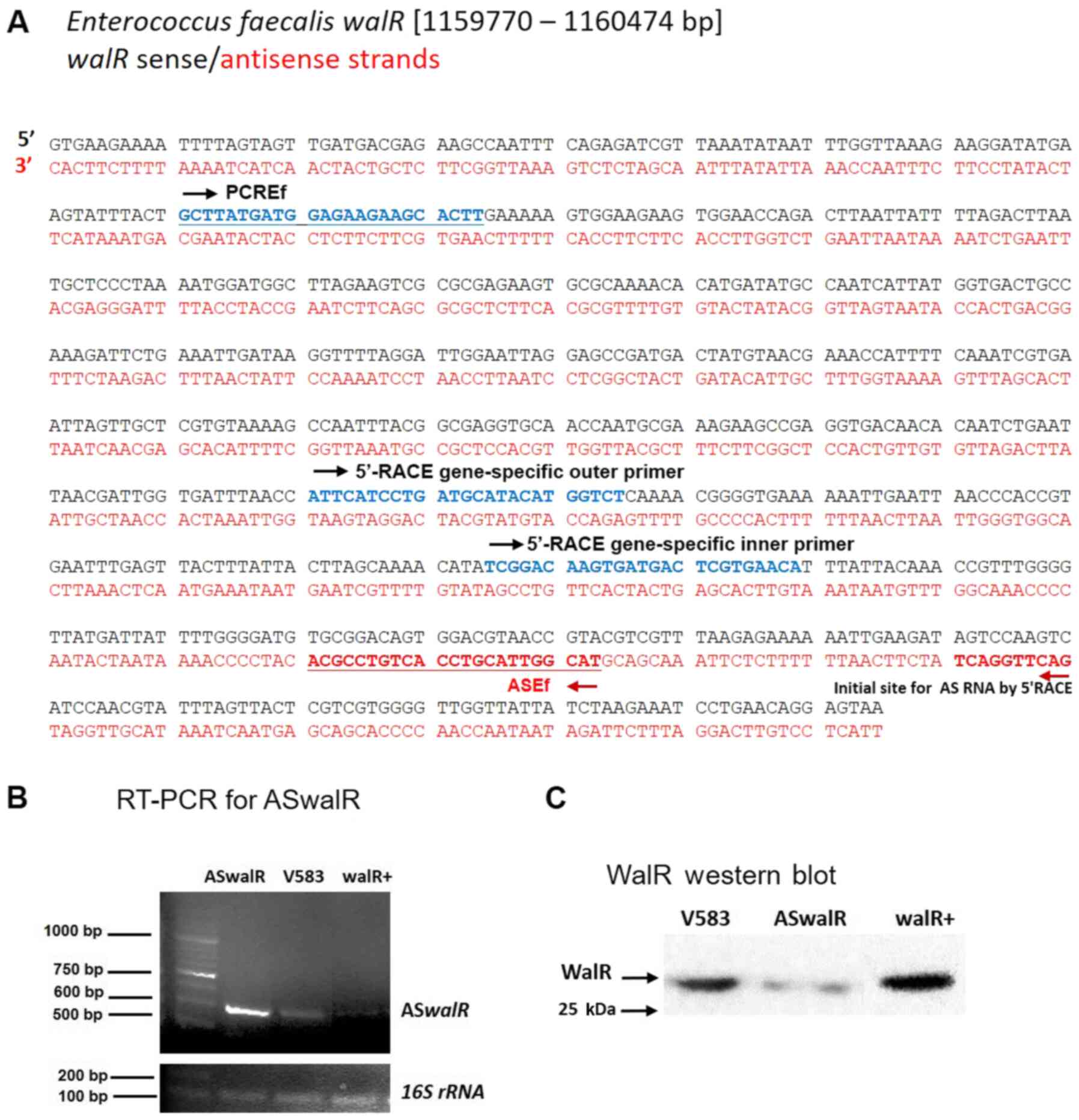
To investigate whether a potential antisense RNA was
specifically associated with walR, first strand cDNA
synthesis was performed to detect the transcript in E.
faecalis V583. The sense and antisense sequences of the
walR gene (EF1193) are shown in Fig. 2A. To determine the 5'-terminus of
ASwalR RNA, 5'-RACE assays of ASwalR were conducted.
The primers used and the design and position of primers for 5'-RACE
is indicated in Fig. 2A. The
sequencing analyses indicated that the potential ASwalR
contained 550 bp of antisense sequence in the E. faecalis
V583 genome by RT-PCR assays (Fig.
2B). Western blotting showed that the expression of WalR
protein was inversely associated with different levels of
ASwalR transcripts (Fig.
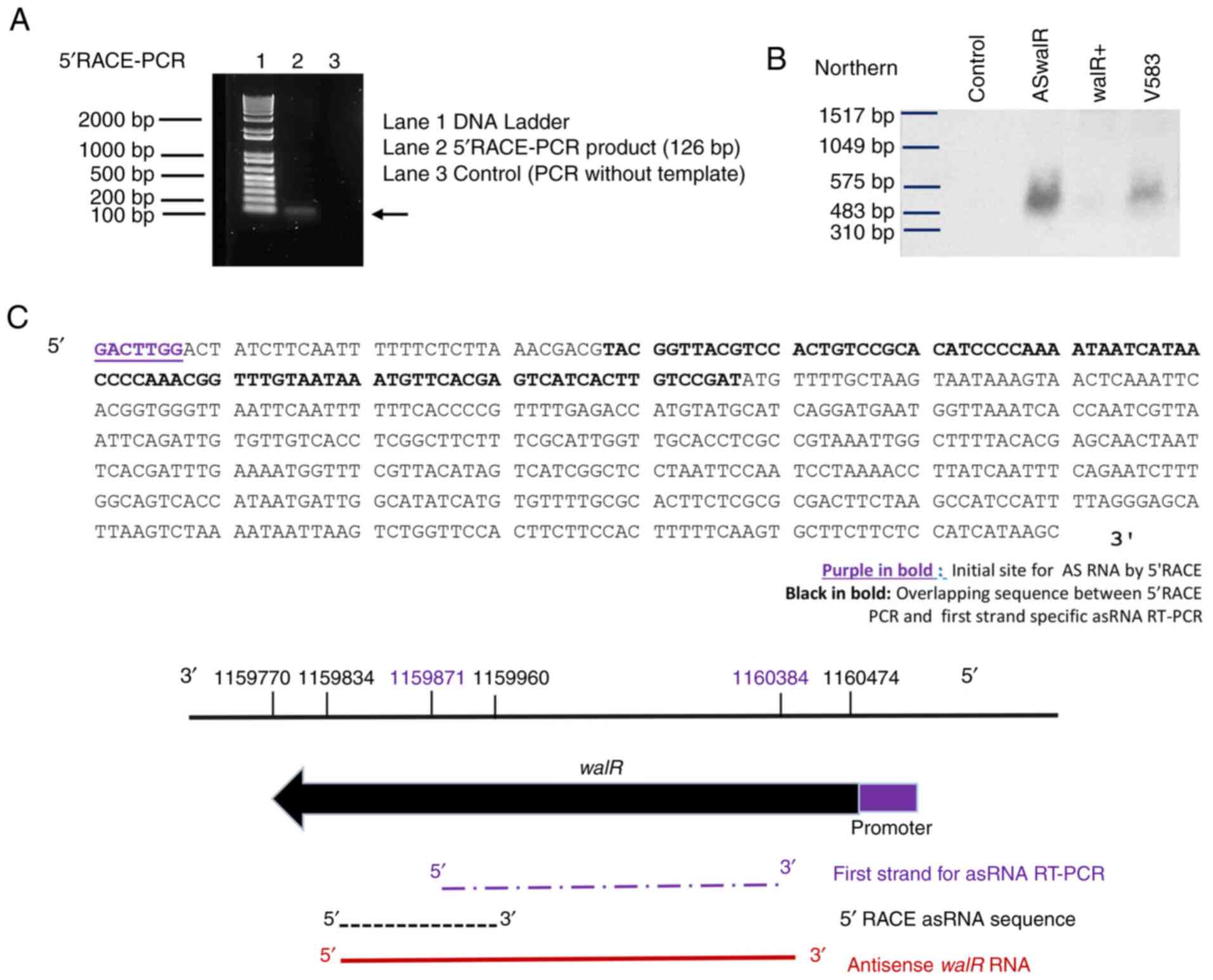
2C). The size of the 5'-RACE PCR product was ~120 bp (Fig. 3A). Additionally, the differences in
ASwalR abundance in E. faecalis planktonic growth
were confirmed by northern blotting (Fig. 3B). The sequence predicted that the
5'-terminus of ASwalR begins at and covers most of the
walR coding sequence (Fig.
3C).
Antisense walR RNA negatively affects
the production of WalR and suppresses biofilm formation
Using CLSM visualization of double-stained biofilms,
it was found that overproducing ASwalR markedly decreased
EPS production compared with E. faecalis V583 strain, while
biofilm cells were packed within the enriched EPS matrix, which was
stained red in the walR+ strains, similar to the results obtained
with the V583 parent strain (Fig.
4A). These findings were further confirmed by quantitation of
data revealing that ASwalR exhibited the lowest
EPS/bacterial biomass volume ratio, indicating a role for the
walR gene in EPS architecture development (Fig. 4B). Next, the effects of
ASwalR overexpression on the ability to form biofilms were
evaluated. E. faecalis V583, ASwalR and walR+ strains
were allowed to form biofilms in BHI for 24 h. The biomass was
quantified by a crystal violet microtiter assay, and the results
indicated that overexpression of ASwalR resulted in a 45%
decrease in biofilm growth compared with the V583 parent strain
(Fig. 4C). However, the biomass of
the walR+ strain was slightly but non-significantly increased
compared with the E. faecalis V583 biofilm (Fig. 4C). The expression levels of
ASwalR and walR were quantified, which indicated that
the transcripts of ASwalR in the ASwalR strain were
2.7-fold higher compared with V583 parent cells, while the
transformation of a pDL278 empty vector did not affect the levels
of walR mRNA (1). The results
showed that the levels of transcripts of two glycosyltransferase
genes, epal and epaA, were significantly decreased in
the ASwalR strain when compared with E. faecalis V583
strain (Fig. 4D).
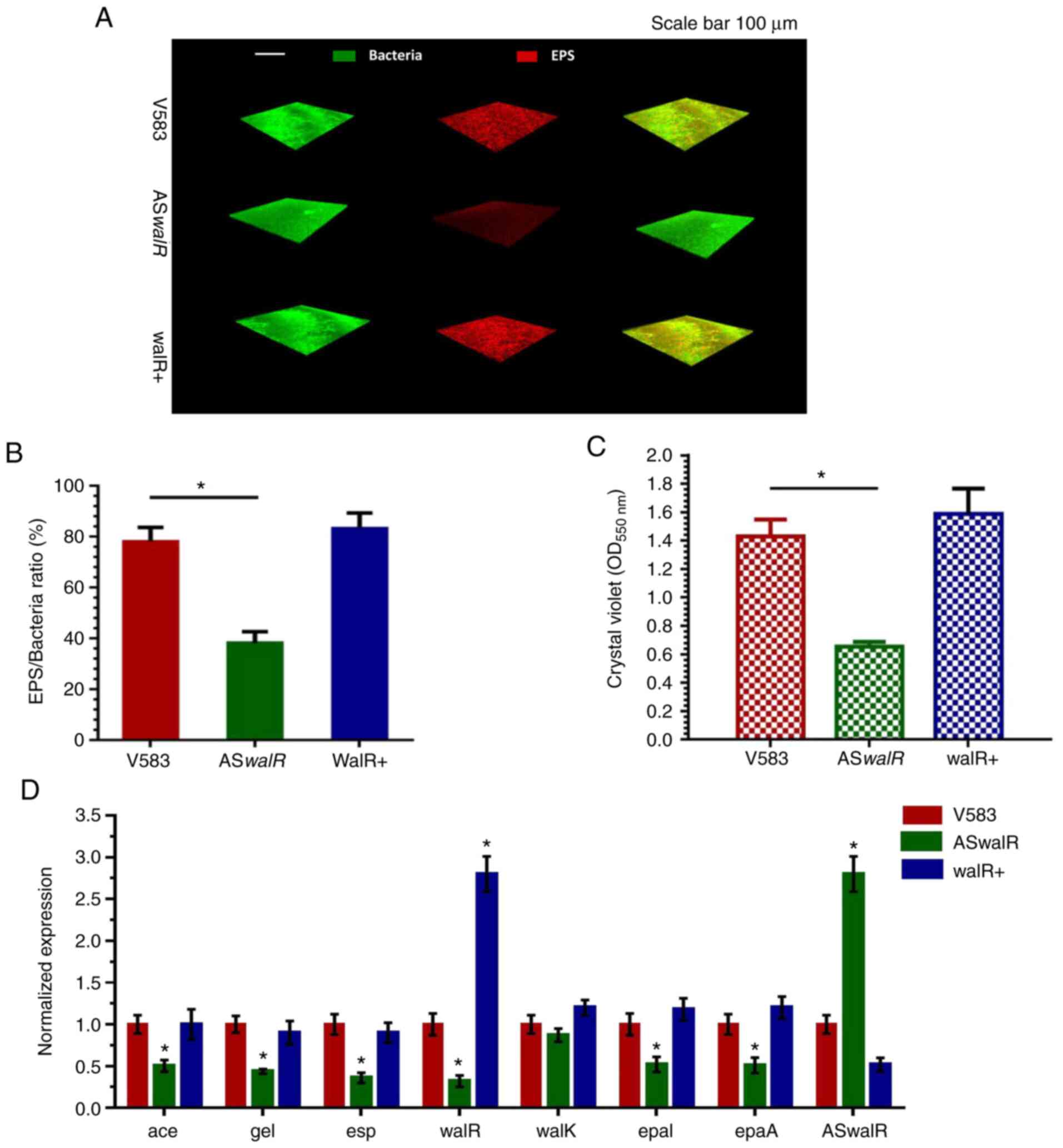 | Figure 4ASwalR overexpression
suppresses E. faecalis biofilm aggregation and EPS
production. (A) Double labeling of the biofilms in the E.
faecalis V583, ASwalR and walR+ strains. Green,
bacterial cells (SYTO9); red, EPS matrix (propidium iodide, PI).
Scale bar, 100 µm. (B) Percentage of EPS matrix/bacteria ratio in
biofilm growth. n=10. *P<0.05. (C) Biomass was
quantified by crystal violet staining. n=10. *P<0.05.
(D) Reverse transcription-PCR analysis showed the gene transcripts
in E. faecalis V583, ASwalR, and walR+ strains. E.
faecalis gene expression was quantified using 16sR as an
internal control and calculated based on the E. faecalis
V583 expression, which was set as 1.0. n=10. *P<0.05
when compared to the E. faecalis V583. ASwalR,
walR antisense RNA; EPS, exopolysaccharide; E.
faecalis, Enterococcus faecalis; OD, optical density;
Ace, adhesin of collagen; esp, protein surface; epal,
epaA, enterococcal polysaccharide antigen; gel,
gelatinase. |
WalR antisense inhibited the
pathogenicity and reduced Periapical Lesion Size
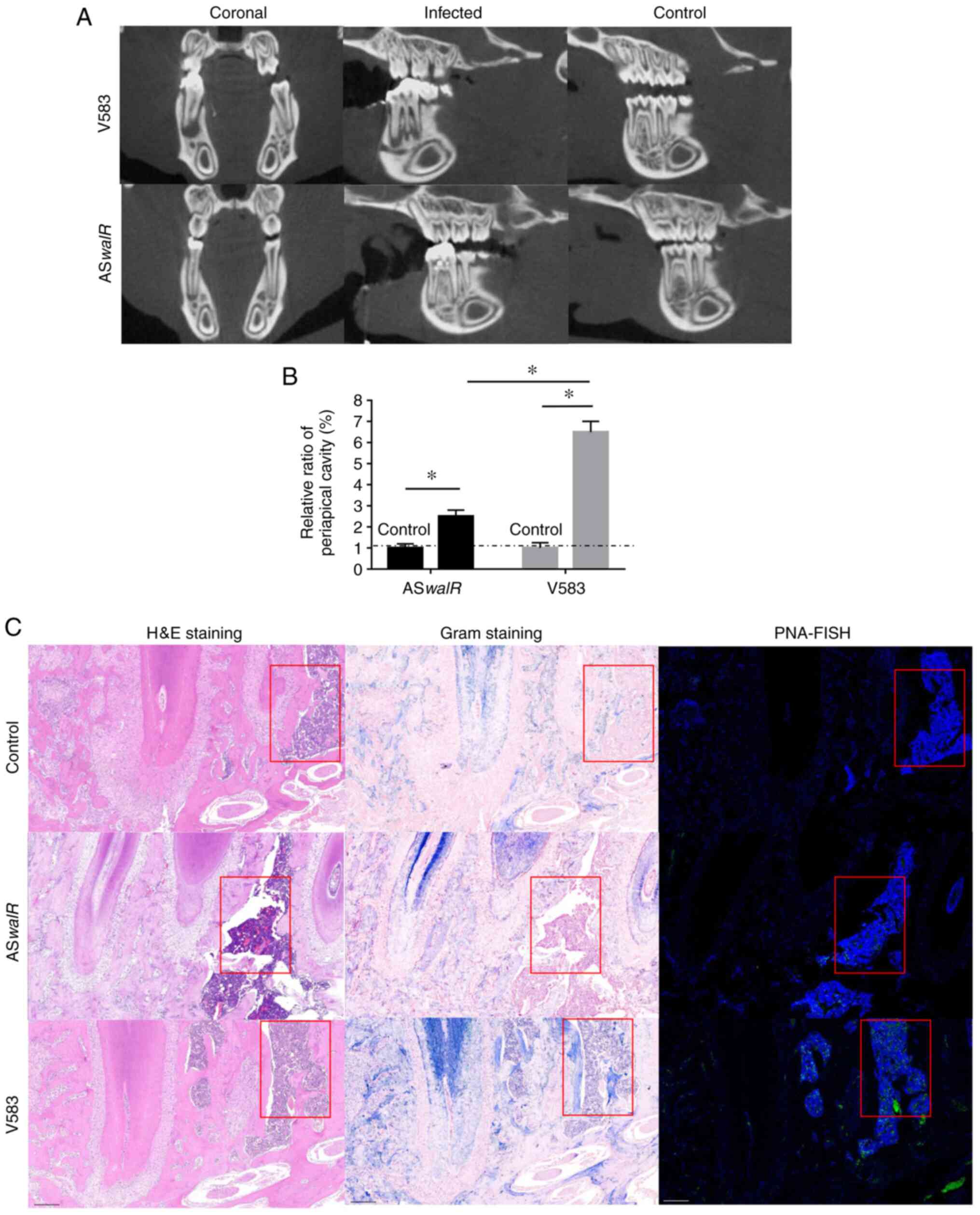
The micro-CT results showed that the severity of
bone destruction was markedly higher in the E. faecalis
V583-infected group compared with the ASwalR-treated group
(Fig. 5A). Quantitatively, the
average relative ratio of the periapical cavity was ~2.5% in the
ASwalR-treated group compared with the right first mandible
molar as a blank control, which was significantly lower than that
of E. faecalis V583-treated group (Fig. 5B). This trend indicated that
ASwalR-transformed stains presented a limited capability to
infarct infected bone tissues. Histological assessments in
HE-stained samples showed bone tissue absorption and inflammatory
infiltration in the E-faecalis V583-colonized groups, which
were more severe compared with control and ASwalR groups (Fig. 5C; left column). Gram staining
results showed that more Gram-positive cells were identified within
the bone of the E-faecalis V583 group compared with the
ASwalR group (Fig. 5C;
middle column). After labeling with PNA fluorescent probes,
bacterial cells were specifically detected by green fluorescence.
The control group indicated no Gram-positive cells were inoculated
(Fig. 5C; right column). The
fluorescence intensity of the E. faecalis group was higher
compared with ASwalR strain-infected samples. The control
group indicated no fluorescence intensity of the E. faecalis
were observed.
Discussion
The WalRK (also known as VicRK or YycFG)
two-componen t signal transduction system is highly conserved in
Gram-positive bacteria, which originated from Bacillus
subtilis (6). In E.
faecalis, the downregulation of walR led to a reduction
in the dextran-dependent aggregation in the biofilm and inhibited
the transcription of virulence genes (1). Several genes in the region of the
walR locus were assigned to the WalR operon as RT-PCR
results indicated co-transcription comprising walR and the
four downstream genes, including walK, EF1195, EF1196, and
EF1197. The proximity of walK to walR suggested a
close interaction and role in the regulation of WalRK TCS (17). The function annotated for the EF1195
gene, a hypothetical protein belonging to the conserved protein
domain family YycH, is associated with the regulatory protein YycH
in the two-component signal transduction system YycFG (18). The most distal genes, EF1196 and
EF1197, in the extended WalR regulon encoded a putative Yycl
protein and a metallo-beta-lactamase, YycJ, which probably plays a
role in regulating protein function by lactamization (19). Although the information of these
genes regulated by WalR remains limited, the close proximity of the
other genes in the extended WalR regulon suggested a functional
relationship, including cell wall biogenesis and biofilm formation,
which also needs further investigation.
The evidence has revealed that substantial antisense
transcripts, including intergenic and intragenic transcription,
exist in the bacteria (11). A cis
antisense noncoding RNA to the walR gene (ASwalR) was
identified and confirmed by northern blotting and 5'-RACE. The
effect of ASwalR on decreasing and inhibiting walR
gene expression and associated production of WalR was further
detected using following RT-PCR and western blotting. Similarly, it
was shown that antisense RNA could combine with its reverse
complementary sequences and restricted the expression of targeted
mRNA (12). Based on this, the
present study hypothesized an inverse association between
ASwalR levels and walR transcripts and WalR
production. The inverse association observed is consistent with the
ASwalR-walR mRNA complex blocking its transcription
and translation (20).
To determine whether ASwalR RNA activity
played a role in biofilm formation, the present study constructed
ASwalR and walR overexpression mutant strains to
inhibit ASwalR levels. Our previous results suggested that
the interference of the walR gene suppressed the expression
of virulent genes associated with cellular adhesion and biofilm
growth (1). In E. faecalis,
glycosyltransferase is a type of enzyme involved in the synthesis
of polysaccharides by the transfer of a sugar precursor onto sugar
residues (21). Biofilm is one of
major factors for infection development and processes (1,13). By
overexpression of ASwalR, the levels of two
glycosyltransferase genes, epal and epaA were
significantly reduced, which contributed to impaired EPS production
in the ASwalR biofilm and decreased biomass of ASwalR
overproducing strains. In addition, the transcripts of virulence
genes were lower in all ASwalR-treated groups. Therefore,
the ability of ASwalR strains to induce periapical
infections was reduced compared to the parent V583 strains,
demonstrated by histological evidence from HE and Gram staining
(1). The effects of walR
overexpression on both bacterial growth and EPS production were
similar to the V583 parental strain levels, justifying additional
studies.
These analyses of biofilm formation again confirm
that the ASwalR RNA functions in biofilm formation by E.
faecalis. Next, the role of ASwalR RNA in the
pathogenicity of E. faecalis was validated using a
Sprague-Dawley rat model. The results indicated that ASwalR
overexpression markedly reduced the periapical lesion size at 4
weeks compared to the E. faecalis V583 group, which
presented large periapical lesions. In the present study,
restricting the periapical lesions in ASwalR-treated group
was considered an important step for stimulating the periapical
repair process (22). Additionally,
histological evidence from HE and Gram staining demonstrated that
the ability of ASwalR strains to cause periapical infections
was reduced compared with the parent V583 strains. Furthermore, the
presence of E. faecalis bacterial cells involved in
periapical tissues was identified by PNA-FISH, which showed higher
fluorescence intensity in the parent V583 strains. It was
hypothesized that the pathogenicity of E. faecalis was
markedly decreased by ASwalR overexpression in periapical
periodontitis. However, one limitation of the present study is the
lack of direct evidence showing whether ASwalR-mRNA duplexes
can be disrupted by additional RNase activities. Bioinformatic
studies for the ASwalR transcript started in the opposite
direction to walR, which was reverse complementary to the
walR transcript (BPROM at http//:Linux1.softberry.com/berry.phtml). A
putative-35 box, TTCACA, and a-10 box, TTCTAATCT. Future research
is needed to construct a vector which contains the entire
ASwalR gene and the promoter of the ASwalR to
identify the transcriptional initial site of ASwalR.
In summary, the downstream genes walK,
EF1195, EF1196, and EF1197 together with the walR gene are
co-transcribed in E. faecalis V583. The present study
detected and confirmed a 550-bp noncoding antisense RNA with the
potential to attenuate the activities of the essential response
regulator WalR. The levels of antisense walR RNA transcripts
were inversely associated with the production of WalR protein. In
addition, the present study showed that overexpression of
ASwalR led to a reduction in biofilm formation and EPS
synthesis. Furthermore, the results of animal experiments revealed
that the pathogenicity of E. faecalis was markedly decreased
by ASwalR overexpression in periapical periodontitis. Collectively,
the data suggested a role for ASwalR as a
post-transcriptional modulator of the WalR regulator in E.
faecalis and revealed that preserving the novel antisense
walR RNA will be a potential substitutive therapy for E.
faecalis infections, especially in the treatment of periapical
periodontitis.
Supplementary Material
RNA agarose gel electrophoresis for
northern blotting. Lane M, DL2000 Plus DNA marker; lane 1, total
RNA of walR antisense RNA; lane 2, total RNA of walR+; lane
3, total RNA of V583.
Bacterial strains and plasmids used in
the present study.
Oligonucleotide primers used for
RT-qPCR.
Oligonucleotide primers used for
reverse transcription-PCR of the WalR regulon.
Oligonucleotide primers used for
5’-RACE
Acknowledgements
Not applicable.
Funding
This study was supported by the Natural Science
Foundation of China (grant no. 81800964), the Sichuan Provincial
Natural Science Foundation of China (grant nos. 2018SZ0125 and
2019YFS0270).
Availability of data and materials
The datasets used and/or analyzed during the
current study are available from the corresponding author on
reasonable request.
Authors' contributions
SW contributed to conception, design and
interpretation of data, drafted and critically revised the
manuscript. YL contributed to design, and data interpretation,
drafting manuscript and critical revision the manuscript. LL
contributed to conception, design, and interpretation of data,
drafted and critically revised the manuscript. HZ contributed to
conception, design, and interpretation of data, drafted and
critically revised the manuscript. All authors gave their final
approval and agree to be accountable for all aspects of the work.
All authors read and approved the final manuscript.
Ethics approval and consent to
participate
The West China Hospital of Sichuan University
Biomedical Research Ethics Committee approved the animal
experiments for this investigation (approval no. 2019128A).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Wu S, Liu Y, Zhang H and Lei L: The
susceptibility to calcium hydroxide modulated by the essential
walR gene reveals the role for Enterococcus faecalis
biofilm aggregation. J Endod. 45:295–301.e2. 2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Dewhirst FE, Chen T, Izard J, Paster BJ,
Tanner AC, Yu WH, Lakshmanan A and Wade WG: The human oral
microbiome. J Bacteriol. 192:5002–5017. 2010.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Colomer-Winter C, Flores-Mireles AL, Baker
SP, Frank KL, Lynch AJL, Hultgren SJ, Kitten T and Lemos JA:
Manganese acquisition is essential for virulence of Enterococcus
faecalis. PLoS Pathog. 14(e1007102)2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Hancock LE and Perego M: Systematic
inactivation and phenotypic characterization of two-component
signal transduction systems of Enterococcus faecalis V583. J
Bacteriol. 186:7951–7958. 2004.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Gotoh Y, Eguchi Y, Watanabe T, Okamoto S,
Doi A and Utsumi R: Two-component signal transduction as potential
drug targets in pathogenic bacteria. Curr Opin Microbiol.
13:232–239. 2010.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Fukuchi K, Kasahara Y, Asai K, Kobayashi
K, Moriya S and Ogasawara N: The essential two-component regulatory
system encoded by yycF and yycG modulates expression of the ftsAZ
operon in Bacillus subtilis. Microbiology. 146:1573–1583.
2000.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Dubrac S, Bisicchia P, Devine KM and
Msadek T: A matter of life and death: Cell wall homeostasis and the
WalKR (YycGF) essential signal transduction pathway. Mol
Microbiol. 70:1307–1322. 2008.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Fisher K and Phillips C: The ecology,
epidemiology and virulence of Enterococcus. Microbiology.
155:1749–1757. 2009.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Krinke L and Wulff DL: RNase III-dependent
hydrolysis of lambda cII-O gene mRNA mediated by lambda OOP
antisense RNA. Genes Dev. 4:2223–2233. 1990.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Saberi F, Kamali M, Najafi A, Yazdanparast
A and Moghaddam MM: Natural antisense RNAs as mRNA regulatory
elements in bacteria: A review on function and applications. Cell
Mol Biol Lett. 21(6)2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Rasmussen S, Nielsen HB and Jarmer H: The
transcriptionally active regions in the genome of Bacillus
subtilis. Mol Microbiol. 73:1043–1057. 2009.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Lei L, Stipp RN, Chen T, Wu SZ, Hu T and
Duncan MJ: Activity of Streptococcus mutans VicR is
modulated by antisense RNA. J Dent Res. 97:1477–1484.
2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Ho EC, Donaldson ME and Saville BJ:
Detection of antisense RNA transcripts by strand-specific RT-PCR.
Methods Mol Biol. 630:125–138. 2010.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Stipp RN, Boisvert H, Smith DJ, Höfling
JF, Duncan MJ and Mattos-Graner RO: CovR and VicRK regulate cell
surface biogenesis genes required for biofilm formation in
Streptococcus mutans. PLoS One. 8(e58271)2013.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Wu S, Huang F, Zhang H and Lei L:
Staphylococcus aureus biofilm organization modulated by YycFG
two-component regulatory pathway. J Orthop Surg Res.
14(10)2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Couto M and Cates C: Laboratory guidelines
for animal care. Methods Mol Biol. 1920:407–430. 2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Aakra A, Vebø H, Snipen L, Hirt H,
Aastveit A, Kapur V, Dunny G, Murray BE and Nes IF: Transcriptional
response of Enterococcus faecalis V583 to erythromycin.
Antimicrob Agents Chemother. 49:2246–2259. 2005.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Paulsen IT, Banerjei L, Myers GS, Nelson
KE, Seshadri R, Read TD, Fouts DE, Eisen JA, Gill SR, Heidelberg
JF, et al: Role of mobile DNA in the evolution of
vancomycin-resistant Enterococcus faecalis. Science.
299:2071–2074. 2003.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Wecker P, Klockow C, Ellrott A, Quast C,
Langhammer P, Harder J and Glöckner FO: Transcriptional response of
the model planctomycete Rhodopirellula baltica SH1(T) to changing
environmental conditions. BMC Genomics. 10(410)2009.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Krinke L and Wulff DL: OOP RNA, produced
from multicopy plasmids, inhibits lambda cII gene expression
through an RNase III-dependent mechanism. Genes Dev. 1:1005–1013.
1987.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Dale JL, Cagnazzo J, Phan CQ, Barnes AM
and Dunny GM: Multiple roles for Enterococcus faecalis
glycosyltransferases in biofilm-associated antibiotic resistance,
cell envelope integrity, and conjugative transfer. Antimicrob
Agents Chemother. 59:4094–4105. 2015.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Siddiqui YD, Omori K, Ito T, Yamashiro K,
Nakamura S, Okamoto K, Ono M, Yamamoto T, Van Dyke TE and Takashiba
S: Resolvin D2 induces resolution of periapical inflammation and
promotes healing of periapical lesions in rat periapical
periodontitis. Front Immunol. 10(307)2019.PubMed/NCBI View Article : Google Scholar
|