Introduction
Cervical carcinoma is the second most common type of
cancer among women worldwide and results in approximately 300,000
deaths annually (1,2). This type of cancer is much more
prevalent in developing countries, reflecting the success of
screening programs on the incidence of cervical cancer in developed
nations. Approximately 85% of pathological type cervical cancer
cases are squamous cell carcinoma (SCC) (3). Although the International Federation
of Gynecology and Obstetrics (FIGO) staging system for cervical
carcinoma does not take into account the pelvic lymphadenopathy
(LN) status, it is the most significant prognostic factor for
patients with stages IB or IIA of the disease; the 5-year survival
rate for patients declines dramatically from approximately 80–95%
in patients without lymph node metastasis (LNM) to approximately
50–65% in patients with positive lymph nodes (3–5).
Currently, there are no effective serum markers with the ability to
predict LNM in cervical SCC patients. The FIGO staging system fails
to detect LNM in approximately 15–20% of patients with early-stage
cervical carcinoma (4). SCC
antigen (SCC-Ag) is currently the most widely used SCC marker; it
is a subfraction of TA-4 and a tumor-associated antigen belonging
to the family of serine protease inhibitors. Serum levels of SCC-Ag
have been found to correlate with tumor stage, tumor size, the
amount of residual tumor remaining after treatment, recurrent or
progressive disease, as well as survival in patients with SCC
(6–10). However, SCC is not organ-specific
(for cervix) or malignancy-specific. In apparently healthy women,
the 99th percentile of circulating SCC-Ag is found at a level of
1.9 μg/l. Elevated levels have been found in patients with SCC of
the vulva, vagina, head and neck, esophagus and lungs, as well as
in patients with benign diseases of the skin (e.g., psoriasis,
eczema), lungs (e.g., sarcoidosis), liver and kidneys (3,11,12). A normal initial SCC-Ag level
cannot exclude the presence of LNM and extra-cervical spread, and
hence is of limited use in treatment planning (13). These factors interfere with the
specificity of serum SCC-Ag levels, particularly at relatively low
threshold levels. According to our previous meta-analysis, the LNM
predictive value of serum SCC-Ag levels in patients with cervical
carcinoma was unable to provide satisfactory reference information
for clinicians (sensitivity, 0.60; specificity, 0.76) (14).
MicroRNAs (also known as miRs) are small non-coding
RNAs that are approximately 22 nt in size. They can modulate
differentiation, growth, apoptosis and the proliferation of cells
by regulating gene expression at the post-transcriptional level.
MicroRNAs have crucial functions in human diseases, including
cancer (15,16). A single microRNA can regulate
hundreds of downstream genes, thus having a broad effect. As a
result, information from microRNA profiling may provide better
classification of cancer stages than the profiles of protein-coding
genes. Due to their stability and presence in almost all body
fluids, microRNAs constitute a novel class of non-invasive
biomarkers (17–21). To date, however, to our knowledge,
there are no reports describing the use of serum microRNA
expression levels in patients with cervical SCC.
We hypothesized that some microRNAs found in tissue
may be used as fingerprints to predict LNM in patients with
early-stage cervical SCC (IB1-IIA1).
Furthermore, their expression levels in serum may serve as
non-invasive biomarkers. To address this hypothesis, we screened
microRNAs in SCC tissue using hybridization arrays. We also
performed quantitative RT-PCR (qRT-PCR) in a cohort of 100 paired
samples (cervical tissue and paired serum from the same woman) to
validate the predictive value of these microRNAs and their ability
to serve as biomarkers.
Materials and methods
Research subjects and clinical
samples
Between March 2010 and May 2012, we collected 100
cervical tissue samples and matched serum samples. In total, 80
patients had cervical SCC and 20 were healthy women. All patients
were classified as FIGO stages IB1-IIA1 and
had received radical hysterectomy and bilateral lymphadenectomy at
the Department of Gynecological Oncology, Affiliated Cancer
Hospital of Guangxi Medical University, Nanning, China. The healthy
controls were women who had received regular health checks and did
not possess any gynecological disease. Sample collections were
performed prior to primary treatment. The study was approved by the
Ethics Committee of Guangxi Cancer Prevention and Control
Committee, and written informed consent was obtained from all
participants. All tissue samples were immediately stored at −80°C
after collection. Blood samples were obtained by venous puncture,
clotted and centrifuged at room temperature. They were then also
stored at −80°C. The personnel responsible for collecting the
samples did not participate in patient grouping or experimental
data analysis.
Each cervical tissue sample (approximately 300 mg)
used for microRNA hybridization array analysis was a mixture from 3
different individuals (100 mg each). In total, 3 groups were
analyzed: cervical SCC tissue from patients with LNM, cervical SCC
tissue from patients without LNM and normal controls.
qRT-PCR was performed on 100 subjects, including 40
cervical SCC patients with LNM, 40 cervical SCC patients without
LNM and 20 healthy women. There was no statistically significant
difference in age among the patients in all 3 groups.
MicroRNA profiling using hybridization
arrays
The test operator and data analyst were blinded to
the original source of samples and patient information. RNA
isolation was completed using TRIzol reagent (Invitrogen/Life
Technologies, Carlsbad, CA, USA). RNA quality and quantity were
evaluated using the 260/280 ratio with a NanoDrop ND-1000
spectrophotometer (NanoDrop Technologies, Inc., Rockland, DE, USA)
and an Agilent 2100 Bioanalyzer (Agilent Technologies, Palo Alto,
CA, USA) (22). The microRNA
expression profiles of the cervical SCC tissues were run on Human
microRNA OneArray® (Phalanx Biotech Group, Inc.,
Hsinchu, Taiwan). Probe design was based on the Sanger miRBase
database, according to the characteristics of each sequence,
corresponding to 1,450 mature microRNAs, and based on strict Tm
values of the control and adjustment. For each chip, each microRNA
sequence has 3 probes (23).
Candidate microRNA confirmation and
quantification
For each patient, we extracted small RNAs (<200
nt) from 50 mg cervical cancer tissue and 500 μl serum. Synthetic
miR-238 from C. elegans was spiked into the serum samples
prior to RNA extraction as an internal control as previously
described (24). Small RNA was
isolated using the miRcute microRNA isolation kit (DP501; Tiangen
Biotech, Beijing, China). cDNA was obtained using the MiraMas™ kit
(no. 5208; Bioo Scientific, Austin, TX, USA), using RNA Ligase to
add the same adapter sequence to the 3′ ends of the small RNAs,
followed by reverse transcription with M-MLV Reverse Transcriptase
(Invitrogen/Life Technologies) to convert the small RNAs to cDNA
templates for qPCR. The qPCR reaction was performed with diluted
cDNA, (For + Rev) primer mixture, and SYBR Premix Ex Taq™ II
(DRR081A; Takara Biotechnology Co., Dalian, China). The reverse
primer was universal (5′-GTCCTTGGTGCCCGAGTG-3′).
We surveyed the literature and selected U6 as the
endogenous control in cervical tissue, C. elegans miR-238
was used as an endogenous control for serum as previously described
(24). The control sample and the
test sample were placed in parallel into each 96-well plate. The
ratios of the microRNAs were calculated using the equation
2−ΔΔCT, in which ΔΔCT = cycle threshold
(CTpatient − CTendogenous control −
ΔCTcontrol sample), ΔCTcontrol sample is the
difference between the CTcontrol sample in one reaction
and the mean of CTcontrol sample in all reactions
(25). The coefficient of
variation (CV) of CT values for the control sample between
different plates for different patients was small and comparable
(CV, 0.046 for miR-16; CV, 0.031 for miR-122). We selected the
minimum microRNA expression sample and set the expression quantity
of this sample as 1. The expression quantities of all the other
samples were equal to the relative ratio.
Statistical analysis
Statistical analysis was performed using Cluster and
TreeView software (http://rana.lbl.gov/EisenSoftware.htm), SPSS software
version 16.0 (SPSS Inc., Chicago, USA) and Rockit 1.1B2 software
(University of Chicago, Chicago, IL, USA). All statistical tests
were two-sided. P-values <0.05 were considered to indicate
statistically significant differences. Due to the magnitude and
range of the observed data, the results were log-transformed for
analysis. For each microRNA, a receiver operating characteristic
(ROC) curve was generated. Spearman correlation analysis was used
to test the consistency of the tissue and serum expression levels
in the same individual. Non-parametric tests were used to analyze
the expression levels in the different groups. To increase the
predictive accuracy of the serum microRNAs, multiple logistic
regression analyses were performed to generate a comprehensive set
of marker microRNAs. The fitted binormal ROC curve is displayed as
the true positive rate (TPR) vs. the false positive rate (FPR).
Results
Phase I - Marker microRNA
identification
The identification of marker microRNAs was carried
out as previously described (26). First, we determined the expression
levels of 1,450 microRNAs in cervical SCC and normal cervical
tissue (total n=9) using hybridization arrays. MicroRNA expression
was normalized to small nuclear RNA (RNU6B, RNU44 and RNU48), and
the mean expression level was calculated. A total of 89 microRNAs
were differentially expressed (P<0.05) at relatively high levels
(intensity >1,000). Among these, 62 microRNAs were upregulated
and 27 were downregulated. The microRNAs with high levels of
expression in tissue were more easily detectable in serum. As a
result, we selected these 89 microRNAs as our potential candidate
markers for further research (Table
I).
 | Table IRelative expression of the 89
microRNAs up- or downregulated in cervical sample A (cases with
LNM). |
Table I
Relative expression of the 89
microRNAs up- or downregulated in cervical sample A (cases with
LNM).
| Expression | MicroRNAa | Total |
|---|
| Upregulated | miR-1246, miR-1248,
miR-1273f, miR-1275, miR-1290, miR-1321, miR-17, miR-1973,
miR-1976, miR-20a, miR-21, miR-2392, miR-3131, miR-3147, miR-3149,
miR-3154, miR-3162-5p, miR-3175, miR-3176, miR-3202, miR-3679-5p,
miR-3682-3p, miR-3934, miR-3945, miR-4281, miR-4294, miR-4298,
miR-4299, miR-4314, miR-4419b, miR-4433, miR-4455, miR-4484,
miR-4485, miR-4492, miR-4497, miR-4505, miR-4507, miR-4508,
miR-4530, miR-4534, miR-4644, miR-4646-5p, miR-4653-3p,
miR-4667-5p, miR-4669, miR-4689, miR-4695-5p, miR-4706,
miR-4725-3p, miR-4739, miR-4749-5p, miR-4763-5p, miR-4769-5p,
miR-4778-5p, miR-4792, miR-4800-5p, miR-483-5p, miR-513a-5p,
miR-574-5p, miR-765 | 62 |
| Downregulated | Let-7a, Let-7b,
Let-7c, miR-100, miR-103b, miR-125a-5p, miR-125b, miR-126, miR-141,
miR-143, miR-145, miR-200b, miR-200c, miR-203, miR-26a, miR-27b,
miR-30d, miR-3185, miR-3196, miR-34a, miR-375, miR-3960, miR-4324,
miR-4467, miR-4488, miR-451, miR-4525 | 27 |
Subsequently, using qPCR, we measured the expression
of these 89 microRNAs in both tissue and serum from 80 cervical SCC
patients and 20 healthy women. Upon comparing patients with LNM
(n=40) with those without LNM (n=40) and the healthy controls, we
needed to control the isolation efficiency. Thus, tissue microRNA
expression was normalized to U6, and serum microRNA was normalized
to C. elegans miR-238, which was spiked into the serum prior
to RNA isolation. We compared the microRNA expression levels in the
different groups of patients and used the area under the ROC curve
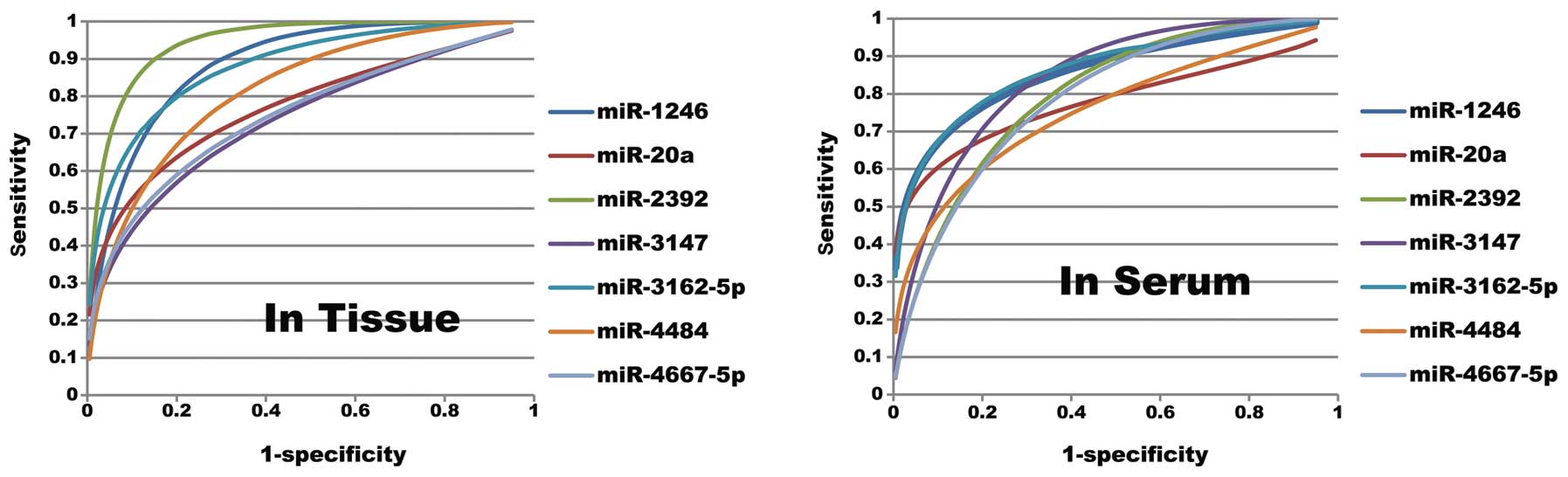
(AUC) >0.70 as our inclusion criteria. In SCC tissue, 17
microRNAs were upregulated and 7 were downregulated. In the serum
from patients, 22 microRNAs were upregulated and all other
microRNAs were expressed at similar levels. We followed-up on 7
candidate microRNAs for further investigation. These included
miR-1246, miR-20a, miR-2392, miR-3147, miR-3162-5p, miR-4484 and
miR-4667-5p. The criteria for further investigation of the 7 most
promising candidates were: i) a high microRNA expression level in
the cancer tissue of patients with LNM, ii) a high microRNA
expression level in the serum of patients with LNM, and iii)
quantification cycle values <35 to enable reliable detection
(Figs. 1 and 2, and Table II).
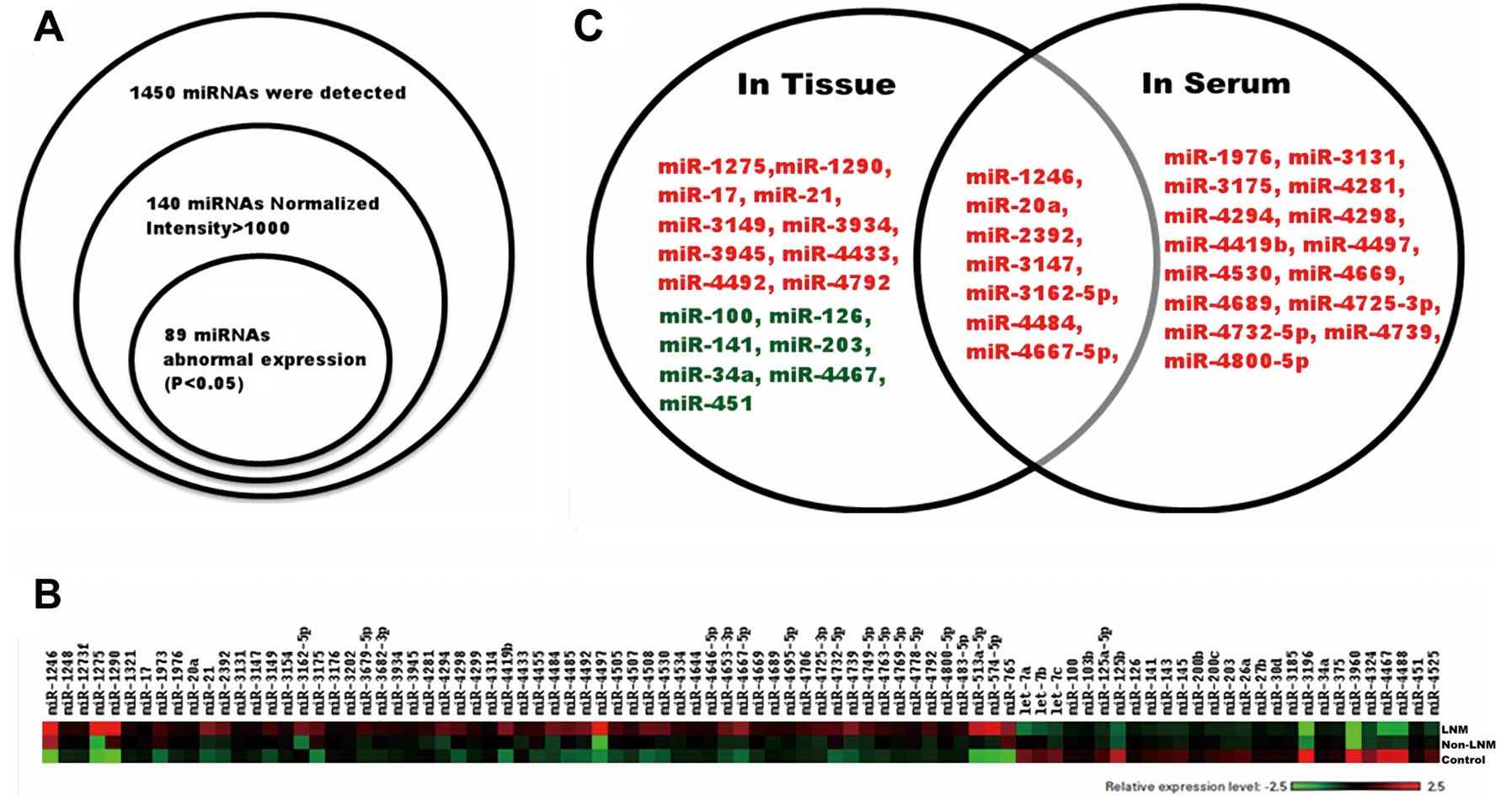 | Figure 1The screening process of the 7
candidate microRNAs. (A) According to the results of hybridization
array (9 cases cervical tissue), we selected 89 microRNAs for
further investigation. (B) Color-gram of the microRNA expression
levels of the 89 microRNAs in the hybridization array samples. The
3 cervical samples from top to bottom were from patients with lymph
node metastasis (LNM), patients without LNM and normal controls in
order. (C) Based on the qPCR results of the 89 microRNAs (100 cases
of tissue samples and 100 paired serum samples), we drew receiver
operating characteristic (ROC) curves for each microRNA and
selected the microRNA whose area under the ROC curve (AUC) was
>70%. Seven microRNAs (miR-1246, -20a, -2392, -3147, -3162-5p,
-4484 and -4667-5p) were selected for additional study and were
found to have diagnostic value both in tissue and serum. Red,
upregulated; green, downregulated. |
 | Table IIDiagnostic value of microRNAs for LNM
in patients with cervical SCC in cancer tissue and serum. |
Table II
Diagnostic value of microRNAs for LNM
in patients with cervical SCC in cancer tissue and serum.
| In cancer
tissue | In serum |
|---|
|
|
|
|---|
| MicroRNA | AUC | 95% CI | SE | Youden | Cut-off | Sen | Spe | AUC | 95% CI | SE | Youden | Cut-off | Sen | Spe |
|---|
| miR-1246 | 0.881a | 0.818–0.944 | 0.032 | 0.611 | 5.055 | 0.861 | 0.750 | 0.847a | 0.767–0.927 | 0.041 | 0.575 | 6.010 | 0.715 | 0.860 |
| miR-20a | 0.761a | 0.657–0.864 | 0.053 | 0.438 | 6.525 | 0.588 | 0.850 | 0.771a | 0.664–0.878 | 0.055 | 0.504 | 5.050 | 0.604 | 0.900 |
| miR-2392 | 0.945a | 0.904–0.985 | 0.021 | 0.748 | 4.055 | 0.898 | 0.850 | 0.784a | 0.695–0.872 | 0.045 | 0.444 | 5.000 | 0.744 | 0.700 |
| miR-3147 | 0.722a | 0.617–0.826 | 0.053 | 0.369 | 5.985 | 0.569 | 0.800 | 0.825a | 0.748–0.903 | 0.040 | 0.521 | 3.975 | 0.821 | 0.700 |
| miR-3162–5p | 0.872a | 0.805–0.940 | 0.035 | 0.597 | 5.775 | 0.797 | 0.800 | 0.869a | 0.796–0.941 | 0.037 | 0.584 | 10.480 | 0.734 | 0.850 |
| miR-4484 | 0.813a | 0.727–0.899 | 0.044 | 0.477 | 9.390 | 0.727 | 0.750 | 0.73a | 0.628–0.832 | 0.052 | 0.400 | 10.490 | 0.600 | 0.800 |
| miR-4667–5p | 0.737a | 0.635–0.839 | 0.052 | 0.389 | 7.000 | 0.589 | 0.800 | 0.78a | 0.689–0.872 | 0.047 | 0.427 | 6.020 | 0.727 | 0.700 |
Phase II - Marker microRNA
elimination
After identifying the candidate microRNAs in step
one, we then eliminated the microRNAs which were susceptible to
fluctuation in serum. As a relatively stable marker, serum
expression should be consistent with the expression in cancer
tissue, and minimal interference from other organs would be ideal.
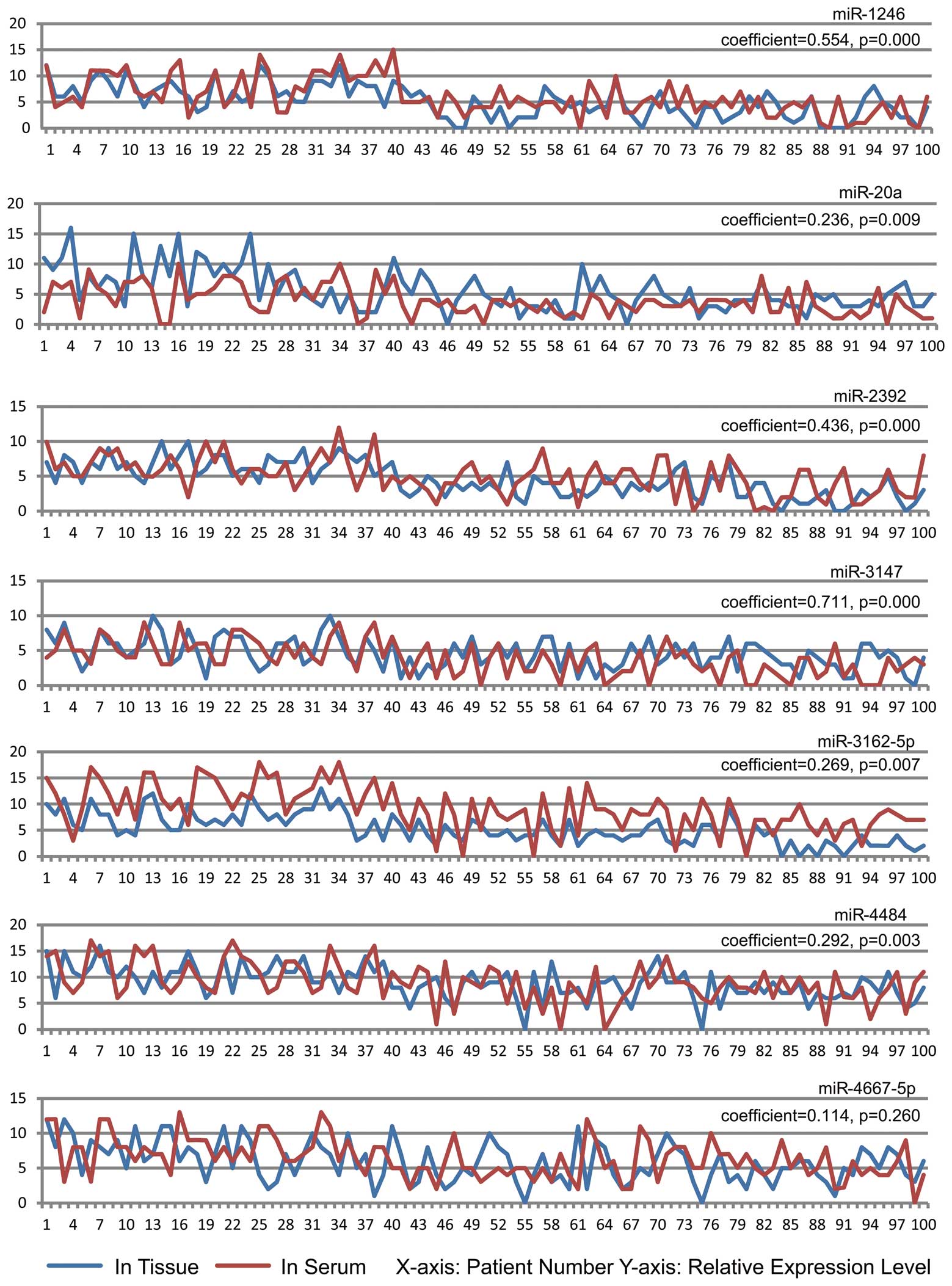
We performed spearman correlation analysis of the microRNA
expression levels in tissue and paired serum samples (total n=100)
in order to examine these 7 microRNAs as serum markers. We wanted
to ensure that serum expression correlated with the expression in
the cervical tissue. The results revealed that, apart from
miR4667-5p, the other 6 microRNAs found in tissue and the paired
serum sampels were positively correlated. Thus, the serum
expression of these 6 microRNAs (miR-1246, miR-20a, miR-2392,
miR-3147, miR-3162-5p and miR-4484) reflects the microRNA
expression in the cervical tissue of the same subject (P<0.05).
Although our results indicated that miR-4667-5p in the serum of
cervical SCC patients may predict LNM, the serum expression was not
consistent with the tissue expression (coefficient, 0.114;
P=0.260). Thus, serum miR-4667-5p expression levels may be
influenced by factors other than cervical cancer tissue. As a
result, we ruled out this microRNA as a serum LNM marker in
cervical SCC patients (Fig.
3).
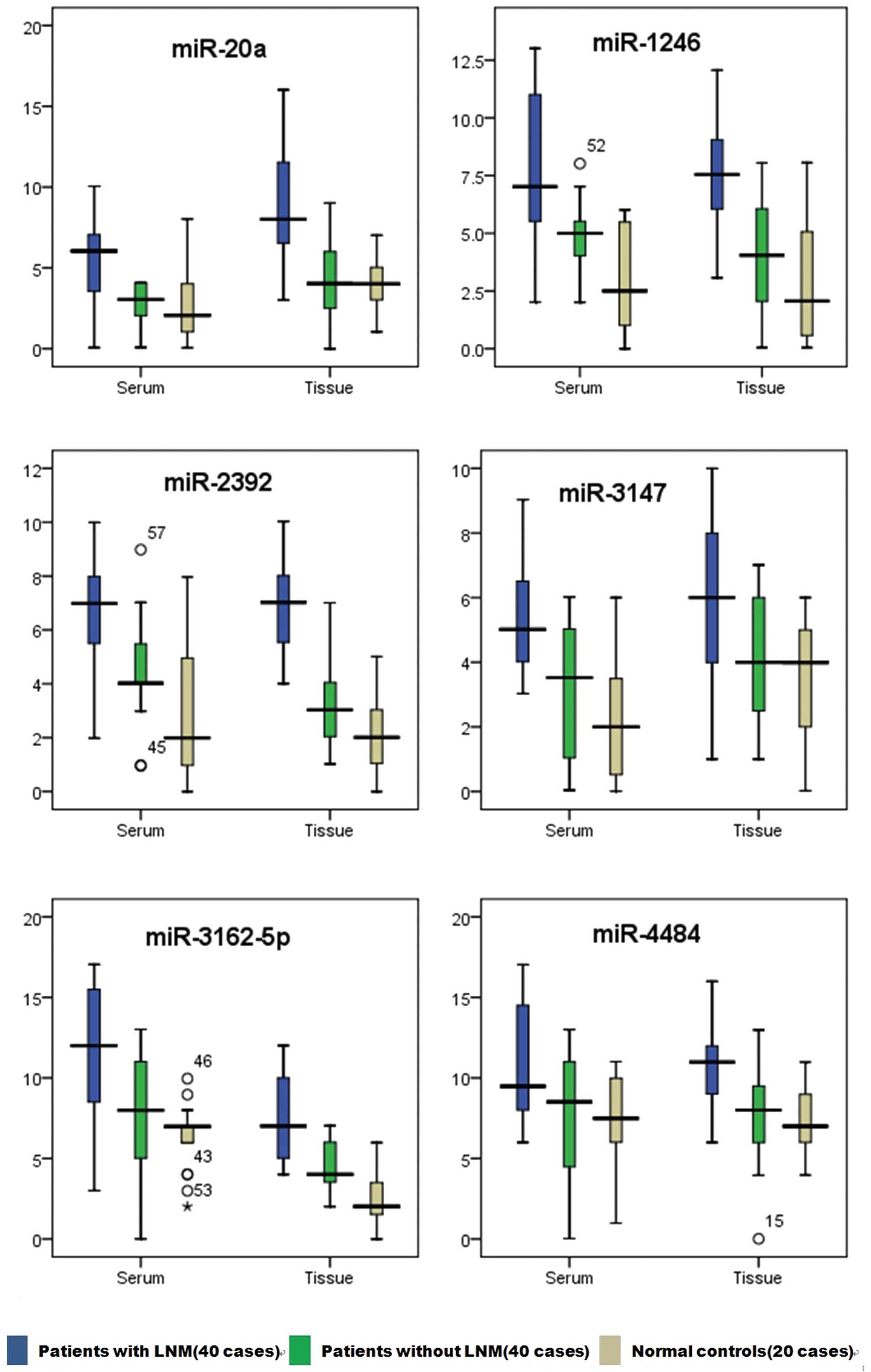
To further refine our set of potential microRNA
biomarkers, we ensured that the difference between patients with
LNM and those without LNM should not be smaller than the difference
between patients with LNM and the normal controls. In this manner,
if the microRNA is a qualified LNM marker in cervical SCC patients,
the expression in patients with LNM should be higher than the
expression in patients without LNM and the expression in normal
controls. Additionally, the expression in patients without LNM
should not be lower than that in normal controls. According to this
principle, using a non-parametric test, we compared the differences
in microRNA expression levels between patients with LNM, those
without LNM and the normal controls. The results revealed that 6
candidate microRNAs conformed to the inclusion criteria, and no
microRNAs were eliminated (Fig. 4
and Table III).
 | Table IIIThe expression level of each
candidate microRNA in the different groups of tissue (or serum)
samples. |
Table III
The expression level of each
candidate microRNA in the different groups of tissue (or serum)
samples.
| MicroRNA | In tissue or
serum | Patients with
LNM | Patients without
LNM | Control | P-value |
|---|
|
|
|
|
|---|
| Median | Interquartile
range | Median | Interquartile
range | Median | Interquartile
range | A/B | A/C | B/C |
|---|
| miR-1246 | Tissue | 7.555 | 6.055–9.060 | 4.040 | 2.060–5.060 | 2.070 | 0.575–5.065 | 0.000 | 0.000 | 0.396 |
| Serum | 10.000 | 6.000–11.000 | 5.000 | 3.990–6.000 | 2.505 | 1.010–5.485 | 0.000 | 0.000 | 0.005 |
| miR-20a | Tissue | 8.010 | 4.525–10.515 | 4.025 | 3.010–5.995 | 4.015 | 3.025–5.030 | 0.000 | 0.001 | 0.747 |
| Serum | 6.060 | 3.565–7.075 | 3.070 | 2.070–4.060 | 2.065 | 1.050–4.020 | 0.000 | 0.016 | 0.001 |
| miR-2392 | Tissue | 7.025 | 6.015–8.015 | 3.040 | 2.040–4.050 | 2.010 | 1.040–3.030 | 0.000 | 0.000 | 0.001 |
| Serum | 6.010 | 5.005–7.500 | 4.020 | 3.475–5.990 | 1.985 | 0.970–4.955 | 0.000 | 0.000 | 0.005 |
| miR-3147 | Tissue | 6.010 | 3.990–7.010 | 3.990 | 2.500–5.985 | 3.990 | 2.005–5.985 | 0.002 | 0.001 | 0.460 |
| Serum | 5.025 | 3.990–7.025 | 3.030 | 1.040–5.025 | 2.005 | 0.520–3.495 | 0.000 | 0.000 | 0.038 |
| miR-3162–5p | Tissue | 8.010 | 6.000–9.005 | 4.020 | 3.010–6.015 | 2.025 | 1.520–3.505 | 0.000 | 0.000 | 0.000 |
| Serum | 12.015 | 9.01–15.010 | 7.995 | 5.020–10.015 | 6.990 | 5.985–7.005 | 0.000 | 0.000 | 0.005 |
| miR-4484 | Tissue | 10.990 | 9.500–12.500 | 8.490 | 6.000–9.500 | 5.995 | 7.000–8.980 | 0.000 | 0.000 | 0.268 |
| Serum | 10.980 | 7.990–13.990 | 8.020 | 5.505–10.010 | 7.490 | 6.010–9.985 | 0.001 | 0.000 | 0.423 |
Phase III - Comprehensive predictive
value of biomarker microRNAs
After confirmation and in order to access the
comprehensive predictive value of biomarker microRNAs, we
integrated these microRNAs (miR-1246, miR-20a, miR-2392, miR-3147,
miR-3162-5p and miR-4484) into a comprehensive factor by regression
analysis, and fitted binormal ROC curves by Rockit software. The
results revealed that the comprehensive set of serum microRNA
markers can predict LNM in cervical SCC patients with a sensitivity
of 0.856, a specificity of 0.850 and at a threshold of 0.551 [AUC
0.932; 95% confidence interval (CI) 0.884–0.980]. However, this
predictive value was inferior to that of the comprehensive set of
tissue microRNA markers (sensitivity, 0.967; specificity, 0.950; at
a threshold of 0.538; AUC 0.992; 95% CI 0.980–1.004). Overall,
serum microRNAs are still reliable markers of LNM (Fig. 5).
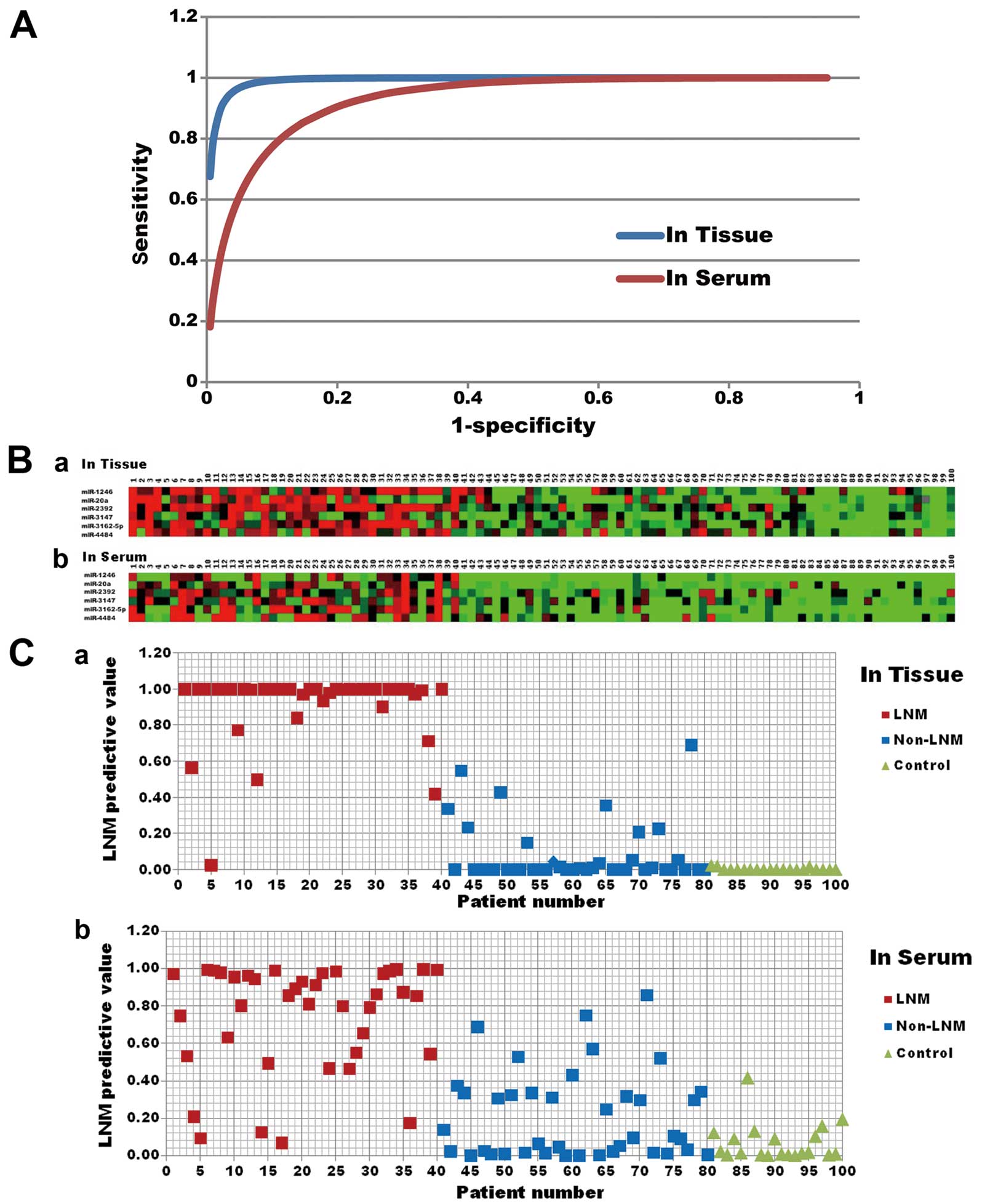 | Figure 5The lymph node metastasis (LNM)
predictive ability of the comprehensive set of microRNAs. (A) The
fitted binormal receiver operating characteristic (ROC) curves of
the comprehensive set of microRNAs. The comprehensive set was made
up of miR-1246, miR-20a, miR-2392, miR-3147, miR-3162-5p and
miR-4484 and was generated through multiple regression analysis and
integration. The ROC curves were drawn by Rockit software. In
cancer tissue, the area under the ROC curve (AUC) was 0.992, and
the maximum Youden index of this comprehensive set was 0.917
(sensitivity, 0.967; specificity, 0.950). In serum, the AUC was
0.932, and the maximum Youden index was 0.706 (sensitivity, 0.856;
specificity, 0.850). (B) Color-gram of 6 candidate microRNAs in
tissue and serum. (a) Marker microRNA expression levels in tissue.
(b) Marker microRNA expression levels in serum. Numbers 1–40 were
patients with LNM, numbers 41–80 were patients without LNM and
numbers 81–100 were the normal controls. (C) Scatter diagram of the
LNM predictive value of the comprehensive set of microRNAs. (a) LNM
predictive value of the comprehensive set in tissue. (b) LNM
predictive value of the comprehensive set in serum. Shown is the
predictive value of the comprehensive set for each patient. The
patients with higher values are those whose pelvic lymph nodes are
more likely to be positive. Numbers 1–40 were patients with LNM,
numbers 41–80 were patients without LNM and numbers 81–100 the were
normal controls. |
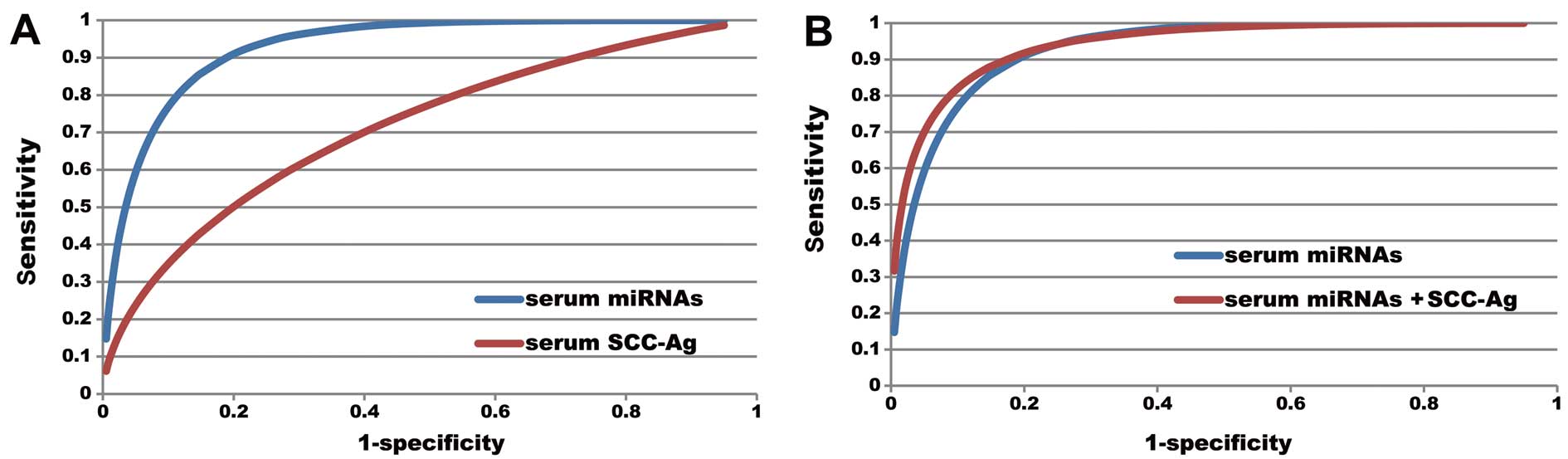
We compared the LNM predictive value of serum
microRNAs and SCC-Ag by drawing fitted binormal ROC curves. The AUC
of serum SCC-Ag was 0.713 and the maximum Youden index was 0.312
(sensitivity, 0.612; specificity, 0.700). Comparing the AUC of the
serum SCC-Ag and serum microRNA, the predictive accuracy of serum
microRNA was by far superior to that of SCC-Ag (P<0.0001, Rockit
software). In order to further increase the serum LNM predictive
value, we integrated the serum SCC-Ag into the comprehensive set of
serum microRNAs by regression analysis, and the revealed that
although the AUC increased from 0.932 to 0.940, there was no
statistically significant difference (P=0.321). Thus, the
combination of serum SCC-Ag with serum microRNA levels does not
increase the LNM predictive value in cervical SCC patients
(Fig. 6).
Discussion
To our knowledge, this is the first study of a
comprehensive interrogation of circulating microRNA expression
levels in cervical SCC patients with LNM. MicroRNAs are an abundant
class of small, non protein-coding RNAs that function as negative
regulators of gene expression. Recent research shows that certain
microRNAs are associated with cancer and may correlate with tumor
stage (27,28). Endogenous circulating microRNAs
are stable, well protected from RNases, and remain stable even
after being subjected to harsh conditions. Due to their stability
and presence in almost all body fluids, microRNAs constitute a
novel class of non-invasive biomarkers. A number of studies have
described the usefulness of microRNAs as markers in a variety of
diseases. For example, serum miR-21 levels have been described as a
marker for necroinflammation in hepatitis C patients and miR-214
levels in the blood have been linked to breast cancer (29,30). Moreover, circulating microRNA
levels have been specifically linked to LNM. For example, high
serum miR-10b levels have been associated with LNM in lung cancer
(31); miR-199a-3p levels in
plasma have been associated with gastric cancer invasion and LNM
stage (32); and serum microRNA
levels have been shown to correlate with the tumor-node-metastasis
stage in papillary thyroid carcinoma (33). However, to our knowledge, there
are no studies concerning circulating microRNA expression levels
and LNM in cervical SCC patients.
Currently, LNM in cervical SCC patients is
identified by surgery, pathological analysis and imaging (34). However, these techniques have
their limitations. SCC-Ag is the only marker which the National
Academy of Clinical Biochemistry (NACB) recommends for cervical SCC
patients. High SCC-Ag levels prior to treatment may indicate the
presence of LNM or extra-cervical spread. Importantly, normal
SCC-Ag levels do not exclude the presence of LNM (3,8–10,14). Other cervical cancer markers, such
as carcinoembryonic antigen (CEA) and CA125, are primarily
associated with cervical adenocarcinoma. Tissue polypeptide antigen
(TPA) and tissue polypeptide-specific antigen (TPS) may have
predictive value in cervical cancer; however, data concerning these
2 antigens are still conflicting and thus further investigation is
required (3,35,36).
Pioneering studies on cervical carcinoma microRNAs
have demonstrated that tissue microRNAs are potential tumor
markers. miR-372 has been found to be downregulated in cervical
carcinoma tissues compared with adjacent normal cervical tissues
(37). miR-23b/uPA is involved in
HPV-16 E6-associated cervical carcinoma development (38). The expression of miR-21 has been
shown to increase with worsening clinical diagnosis (39). miR-34a suppresses cervical
carcinoma invasion through the downregulation of Notch1 and Jagged1
(40). miR-20a promotes migration
and invasion by regulating tankyrase, TRF1-interacting
ankyrin-related ADP-ribose polymerase 2 (TNKS2) expression in human
cervical carcinoma cells (41).
The concordance of the microRNAs (miR-21, miR-20a and miR-34a) in
our study with those from previous studies supports the validity of
our findings in Phase I. Some tissue microRNAs, including miR-21
and miR-34a, were not good serum markers. This is possibly due to
the fact that the cervix is not a vital metabolic or endocrine
organ, and finding cervical lesions in serum is extremely
difficult. There may also be other reasons for this: i)
experimental errors from tissue RNA isolation, such as sample size
errors or RNA degradation; ii) different normalization strategies
in serum (C. elegans miR-238) and tissue (U6) may also
contribute to the different numbers of unregulated microRNAs in
tissue and serum.
The tumor specificity of a single microRNA may be
limited by biological characteristics. However, microRNAs can
regulate hundreds of downstream genes. Thus, the information
obtained from microRNA profiling may provide a more accurate
classification of cancer stages than the profiles obtained from
protein-coding genes. In order to evaluate the predictive value of
our comprehensive set of microRNAs, we integrated these marker
microRNAs (miR-1246, miR-20a, miR-2392, miR-3147, miR-3162-5p and
miR-4484) into a comprehensive set by regression analysis. The
fitted binormal ROC curves showed that the AUC of the serum
microRNAs was 0.932 (sensitivity, 0.856; specificity, 0.850).
Although this predictive value was inferior to the value of these
microRNAs in tissue (AUC 0.917; sensitivity, 0.967; specificity,
0.950), it was still superior to the predictive value of serum
SCC-Ag (AUC 0.713; sensitivity, 0.612; specificity, 0.700) and is a
good predictive marker of LNM in cervical SCC patients.
Before beginning the experiments, the study design
was strictly reviewed. First, we screened the microRNAs in cervical
tissue but not in serum in order to reduce the number of
potentially interfering microRNAs from other organs. Second, each
study participant had a clearly defined responsibility, including
specimen collection and data analysis. The experimental operator
did not participate in any of the experiments; thus, this is a
double-blinded study. Third, in order to avoid repeated freezing
and thawing of the RNA samples, we added the same adapter sequence
to the 3′ ends of the small RNAs via reverse transcription, and all
89 microRNAs were subject to PCR in a single 96-well plate.
Finally, we only selected patients with stage
IB1-IIA1 SCC for our study in order to
guarantee experimental accuracy. Lesions with a stage lower than
stage IB1 are constantly mixed with surrounding tissue
during specimen collection. Tumors with a stage higher than
IIA1 usually receive neoadjuvant chemotherapy prior to
radical surgery, and patients with these tumors were not included
in this study.
Our results revealed that all microRNAs
downregulated in cancer tissue were unable to predict LNM in the
serum samples. This may be due to the fact that serum microRNAs
originating from the cervix account for only a small portion of
total serum microRNAs. We measured the microRNA expression levels
in both tissue and serum. The results demonstrated that miR-1246,
miR-20a, miR-2392, miR-3147, miR-3162-5p, miR-4484 and miR-4667-5p
were all expressed in the serum, and that their expression levels
in serum were consistent with their expression levels in cervical
tissue. Unlike the others, the expression of miR-4667-5p in serum
was not consistent with its expression in cervical tissue. As a
result, we eliminated this microRNA from further analysis.
To our knowledge, our study is the first to describe
the correlation between miR-1246 and cervical carcinoma. Pioneering
studies suggest that miR-1246 is a novel target of p53, and that
dual specificity tyrosine-phosphorylation-regulated kinase 1A
(DYRK1A), a down syndrome-associated protein kinase, is a target of
miR-1246. Thus, there may be a p53-miR-1246-DYRK1A-nuclear factor
of activated T-cells (NFAT) pathway in cancer (42,43). Baek et al(44) reported that DYRK1A along with down
syndrome candidate region 1 (DSCR1), which encodes a protein that
suppresses vascular endothelial growth factor (VEGF)-mediated
angiogenic signaling by the calcineurin pathway, may be sufficient
to markedly diminish angiogenesis. On the other hand, Pigati et
al(45) reported that the
extracellular and cellular microRNA profiles in cancer tissue are
different. For example, the bulk of miR-1246 produced by malignant
mammary epithelial cells was released. However, the majority of
these microRNAs produced by non-malignant mammary epithelial cells
were retained. Therefore, microRNAs released into the serum do not
necessarily reflect the abundance of microRNAs in the cell of
origin. Recently, Baraniskin et al(46) reported that serum hsa-miR-1246 is
likely a pseudo microRNA derived from U2 small nuclear RNA (snRNA)
fragments (RNU2-1f). It is highly stable in serum and plasma and
may serve as a novel diagnostic biomarker for pancreatic ductal
adenocarcinoma and colorectal carcinoma patients for future
prospective screening studies.
miR-20a, another marker microRNA identified in our
study, has been more extensively investigated in terms of tumor
biology. It is an important member of the miR-17-92 cluster, which
is amplified and overexpressed in lung cancer, ovarian cancer and
osteosarcoma (47-49). Several targets of miR-20 have been
identified and include signal transducer and activator of
transcription 3 (STAT3), BCL2/adenovirus E1B 19 kDa interacting
protein 2 (BNIP2), connexin 43 (CX43), ephrin (EPH)B2, EPHB4, LRF
and Unc-51-like kinase 1 (C. elegans) (ULK1) (50–55). Recently, Elkayam et
al(56) reported that the
structure of human argonaute 2 (Ago2) bound to miR-20a, at 2.2 Å
resolution, is anchored at both ends by the Mid and PAZ domains and
makes several kinks and turns along the binding groove. Thus,
miR-20a binding confers remarkable stability on hAgo2, locking this
otherwise flexible enzyme into a stable conformation. In cervical
carcinoma, Kang et al(41)
reported that miR-20a is overexpressed in cervical
carcinoma-derived cell lines (HeLa and C-33A), where it enhances
long-term cellular proliferation, migration and invasion.
Consistent with these results, the inhibition of miR-20a suppressed
these functions. Oncogenic TNKS2 is directly regulated by miR-20a,
and the suppression of TNKS2 inhibits the colony formation,
migration and invasion of cervical carcinoma cells. Taken together,
these data demonstrate that miR-20a is a SCC-associated
oncomir.
In addition to miR-1246 and miR-20a, this is the
first clinical report describing 4 biomarker microRNAs (miR-2392,
miR-3147, miR-3162-5p and miR-4484). Each of these 4 microRNAs was
identified in 2010. While their exact biological function is
unknown, deep sequencing of tumor tissue has revealed that they are
expressed in cancer: miR-3162-5p and miR-3147 are found in melanoma
(57,58), and miR-2392 and miR-4484 are
expressed in malignant human B cells (59). In this study, we confirmed that
they are associated with cervical carcinoma, which indicates that
they should be further investigated as tumor-associated
factors.
This had some limitations. While the presence of LNM
is the most important prognostic factor associated with recurrent
disease and poor survival, serum microRNA biomarkers of LNM cannot
directly serve as prognostic factors of cervical SCC. Long-term
follow-up studies are still required to confirm the correlation
between serum microRNA levels and patient outcome. While our data
is applicable to early-stage cervical SCC patients, it does not
represent the condition of advanced cervical SCC patients.
Additionally, all patients and healthy controls were from southern
China. Thus, the results of this study may be influenced by local
characteristics and may not represent the characteristics of
patients of other races or who live in other areas.
Based on the data presented in this, as well as
other studies, unique patterns of serum microRNAs may serve as
non-invasive biomarkers for cancer development and prognosis.
Therefore, using qRT-PCR to determine microRNA signatures in
patients is a clinically applicable procedure. Due to the
simplicity and reproducibility of obtaining a blood sample,
biomarkers found in blood serum can be easily examined and may have
great potential for cancer diagnosis. These serum markers, if
validated in other populations and in prospective studies, may
provide the opportunity for a randomized clinical trial to evaluate
cervical SCC patients in which pelvic lymph nodes have been
affected and who may have poor prognosis. It is important to expand
the research scope, increase the number of subjects, and adjust the
inclusion criteria appropriately in order to identify additional
marker microRNAs and further increase the LNM prediction accuracy.
In addition, due to non-invasive characteristics, serum microRNA
signatures may be used to investigate the effects of surgery and
chemo-radiotherapy by observing their dynamic expression levels and
profiles.
In conclusion, the results from our study suggest
that serum microRNAs may have great potential to serve as novel,
non-invasive biomarkers for LNM in early-stage cervical SCC.
miR-1246, miR-20a, miR-2392, miR-3147, miR-3162-5p and miR-4484 are
potential biomarkers. Additional large-scale studies are required
to fully explore the role of serum microRNAs in cervical SCC
patients with LNM.
Acknowledgements
The present study was supported by grants from the
Natural Science Foundation of Guangxi Zhuang Autonomous Region,
China (2011GXNSFA018184, 2013GXNSFBA019130 and 2013GXNSFBA019132.
www.gxsti.net), and the Public Health self-financing
project research project of Guangxi Zhuang Autonomous Region
(Z2012071).
References
|
1
|
Hu X, Schwarz JK, Lewis JS Jr, et al: A
microRNA expression signature for cervical cancer prognosis. Cancer
Res. 70:1441–1448. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar
|
|
3
|
Sturgeon CM, Duffy MJ, Hofmann BR, et al:
National Academy of Clinical Biochemistry Laboratory Medicine
Practice Guidelines for use of tumor markers in liver, bladder,
cervical, and gastric cancers. Clin Chem. 56:e1–e48. 2010.
View Article : Google Scholar
|
|
4
|
Hacker NF: Cervical cancer. Practical
Gynecologic Oncology. Berek JS and Hacker NF: 3rd edition.
Lippincott Williams and Wilkins; Philadelphia, PA: pp. 345–405.
2000
|
|
5
|
Neoadjuvant chemotherapy for locally
advanced cervical cancer: a systematic review and meta-analysis of
individual patient data from 21 randomised trials. Eur J Cancer.
39:2470–2486. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lee YY, Choi CH, Sung CO, et al:
Prognostic value of pre-treatment circulating monocyte count in
patients with cervical cancer: comparison with SCC-Ag level.
Gynecol Oncol. 124:92–97. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Huang EY, Wang CJ, Chen HC, et al:
Multivariate analysis of para-aortic lymph node recurrence after
definitive radiotherapy for stage IB-IVA squamous cell carcinoma of
uterine cervix. Int J Radiat Oncol Biol Phys. 72:834–842. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ogino I, Nakayama H, Okamoto N, Kitamura T
and Inoue T: The role of pretreatment squamous cell carcinoma
antigen level in locally advanced squamous cell carcinoma of the
uterine cervix treated by radiotherapy. Int J Gynecol Cancer.
16:1094–1100. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nakamura K, Okumura Y, Kodama J, Hongo A,
Kanazawa S and Hiramatsu Y: The predictive value of measurement of
SUVmax and SCC-antigen in patients with pretreatment of primary
squamous cell carcinoma of cervix. Gynecol Oncol. 119:81–86. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
van de Lande J, Davelaar EM, von
Mensdorff-Pouilly S, et al: SCC-Ag, lymph node metastases and
sentinel node procedure in early stage squamous cell cervical
cancer. Gynecol Oncol. 112:119–125. 2009.PubMed/NCBI
|
|
11
|
Matsuda H, Mori M, Tsujitani S, Ohno S,
Kuwano H and Sugimachi K: Immunohistochemical evaluation of
squamous cell carcinoma antigen and S-100 protein-positive cells in
human malignant esophageal tissues. Cancer. 65:2261–2265. 1990.
View Article : Google Scholar
|
|
12
|
Krzystek-Korpacka M, Matusiewicz M,
Diakowska D, Grabowski K, Blachut K and Banas T: Upregulation of
VEGF-C secreted by cancer cells and not VEGF-A correlates with
clinical evaluation of lymph node metastasis in esophageal squamous
cell carcinoma (ESCC). Cancer Lett. 249:171–177. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gaarenstroom KN, Bonfrer JM, Korse CM,
Kenter GG and Kenemans P: Value of Cyfra 21-1, TPA, and SCC-Ag in
predicting extracervical disease and prognosis in cervical cancer.
Anticancer Res. 17:2955–2958. 1997.PubMed/NCBI
|
|
14
|
Chen J, Yao D and Wu Z: Diagnostic value
of serum squamous cell carcinoma antigen on lymphatic metastasis in
early-stage cervical cancer: a meta-analysis. Cancer Res Prev
Treat. 39:811–817. 2012.(In Chinese).
|
|
15
|
Enfield KS, Stewart GL, Pikor LA, et al:
MicroRNA gene dosage alterations and drug response in lung cancer.
J Biomed Biotechnol. 2011:4746322011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Medina PP, Nolde M and Slack FJ: OncomiR
addiction in an in vivo model of microRNA-21-induced pre-B-cell
lymphoma. Nature. 467:86–90. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang XW, Heegaard NH and Orum H: MicroRNAs
in liver disease. Gastroenterology. 142:1431–1443. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liu R, Chen X, Du Y, et al: Serum microRNA
expression profile as a biomarker in the diagnosis and prognosis of
pancreatic cancer. Clin Chem. 58:610–618. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Jurmeister S, Baumann M, Balwierz A, et
al: MicroRNA-200c represses migration and invasion of breast cancer
cells by targeting actin-regulatory proteins FHOD1 and PPM1F. Mol
Cell Biol. 32:633–651. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hennessey PT, Sanford T, Choudhary A, et
al: Serum microRNA biomarkers for detection of non-small cell lung
cancer. PLoS One. 7:e323072012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wu Q, Lu Z, Li H, Lu J, Guo L and Ge Q:
Next-generation sequencing of microRNAs for breast cancer
detection. J Biomed Biotechnol. 2011:5971452011.PubMed/NCBI
|
|
22
|
Santos CF, Kurhanewicz J, Tabatabai ZL, et
al: Metabolic, pathologic, and genetic analysis of prostate
tissues: quantitative evaluation of histopathologic and mRNA
integrity after HR-MAS spectroscopy. NMR Biomed. 23:391–398.
2010.
|
|
23
|
Hsieh CH, Rau CS, Jeng JC, et al: Whole
blood-derived microRNA signatures in mice exposed to
lipopolysaccharides. J Biomed Sci. 19:692012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Cermelli S, Ruggieri A, Marrero JA,
Ioannou GN and Beretta L: Circulating microRNAs in patients with
chronic hepatitis C and non-alcoholic fatty liver disease. PLoS
One. 6:e239372011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Hu Z, Chen X, Zhao Y, et al: Serum
microRNA signatures identified in a genome-wide serum microRNA
expression profiling predict survival of non-small-cell lung
cancer. J Clin Oncol. 28:1721–1726. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wulfken LM, Moritz R, Ohlmann C, et al:
MicroRNAs in renal cell carcinoma: diagnostic implications of serum
miR-1233 levels. PLoS One. 6:e257872011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Iorio MV, Ferracin M, Liu CG, et al:
MicroRNA gene expression deregulation in human breast cancer.
Cancer Res. 65:7065–7070. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Jamieson NB, Morran DC, Morton JP, et al:
MicroRNA molecular profiles associated with diagnosis,
clinicopathologic criteria, and overall survival in patients with
resectable pancreatic ductal adenocarcinoma. Clin Cancer Res.
18:534–545. 2012. View Article : Google Scholar
|
|
29
|
Bihrer V, Waidmann O, Friedrich-Rust M, et
al: Serum microRNA-21 as marker for necroinflammation in hepatitis
C patients with and without hepatocellular carcinoma. PLoS One.
6:e269712011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Schwarzenbach H, Milde-Langosch K,
Steinbach B, Muller V and Pantel K: Diagnostic potential of
PTEN-targeting miR-214 in the blood of breast cancer patients.
Breast Cancer Res Treat. 134:933–941. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Roth C, Kasimir-Bauer S, Pantel K and
Schwarzenbach H: Screening for circulating nucleic acids and
caspase activity in the peripheral blood as potential diagnostic
tools in lung cancer. Mol Oncol. 5:281–291. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Li C, Li JF, Cai Q, et al: MiRNA-199a-3p
in plasma as a potential diagnostic biomarker for gastric cancer.
Ann Surg Oncol. Sep 7–2012.(Epub ahead of print).
|
|
33
|
Yu S, Liu Y, Wang J, et al: Circulating
microRNA profiles as potential biomarkers for diagnosis of
papillary thyroid carcinoma. J Clin Endocrinol Metab. 97:2084–2092.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sakuragi N: Up-to-date management of lymph
node metastasis and the role of tailored lymphadenectomy in
cervical cancer. Int J Clin Oncol. 12:165–175. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Juang CM, Wang PH, Yen MS, Lai CR, Ng HT
and Yuan CC: Application of tumor markers CEA, TPA, and SCC-Ag in
patients with low-risk FIGO stage IB and IIA squamous cell
carcinoma of the uterine cervix. Gynecol Oncol. 76:103–106. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Gaarenstroom KN, Kenter GG, Bonfrer JM, et
al: Can initial serum cyfra 21-1, SCC antigen, and TPA levels in
squamous cell cervical cancer predict lymph node metastases or
prognosis? Gynecol Oncol. 77:164–170. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Tian RQ, Wang XH, Hou LJ, et al:
MicroRNA-372 is down-regulated and targets cyclin-dependent kinase
2 (CDK2) and cyclin A1 in human cervical cancer, which may
contribute to tumorigenesis. J Biol Chem. 286:25556–25563. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Au Yeung CL, Tsang TY, Yau PL and Kwok TT:
Human papillomavirus type 16 E6 induces cervical cancer cell
migration through the p53/microRNA-23b/urokinase-type plasminogen
activator pathway. Oncogene. 30:2401–2410. 2011.PubMed/NCBI
|
|
39
|
Deftereos G, Corrie SR, Feng Q, et al:
Expression of mir-21 and mir-143 in cervical specimens ranging from
histologically normal through to invasive cervical cancer. PLoS
One. 6:e284232011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Pang RT, Leung CO, Ye TM, et al:
MicroRNA-34a suppresses invasion through downregulation of Notch1
and Jagged1 in cervical carcinoma and choriocarcinoma cells.
Carcinogenesis. 31:1037–1044. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kang HW, Wang F, Wei Q, et al: miR-20a
promotes migration and invasion by regulating TNKS2 in human
cervical cancer cells. FEBS Lett. 586:897–904. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zhang Y, Liao JM, Zeng SX and Lu H: p53
downregulates Down syndrome-associated DYRK1A through miR-1246.
EMBO Rep. 12:811–817. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Liao JM, Zhou X, Zhang Y and Lu H:
MiR-1246: a new link of the p53 family with cancer and Down
syndrome. Cell Cycle. 11:2624–2630. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Baek KH, Zaslavsky A, Lynch RC, et al:
Down’s syndrome suppression of tumour growth and the role of the
calcineurin inhibitor DSCR1. Nature. 459:1126–1130. 2009.
|
|
45
|
Pigati L, Yaddanapudi SC, Iyengar R, et
al: Selective release of microRNA species from normal and malignant
mammary epithelial cells. PLoS One. 5:e135152010. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Baraniskin A, Nopel-Dunnebacke S, Ahrens
M, et al: Circulating U2 small nuclear RNA fragments as a novel
diagnostic biomarker for pancreatic and colorectal adenocarcinoma.
Int J Cancer. 132:E48–E57. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Matsubara H, Takeuchi T, Nishikawa E, et
al: Apoptosis induction by antisense oligonucleotides against
miR-17-5p and miR-20a in lung cancers overexpressing miR-17-92.
Oncogene. 26:6099–6105. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Huang G, Nishimoto K, Zhou Z, Hughes D and
Kleinerman ES: miR-20a encoded by the miR-17-92 cluster increases
the metastatic potential of osteosarcoma cells by regulating Fas
expression. Cancer Res. 72:908–916. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Fan X, Liu Y, Jiang J, et al: miR-20a
promotes proliferation and invasion by targeting APP in human
ovarian cancer cells. Acta Biochim Biophys Sin (Shanghai).
42:318–324. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Chai H, Liu M, Tian R, Li X and Tang H:
miR-20a targets BNIP2 and contributes chemotherapeutic resistance
in colorectal adenocarcinoma SW480 and SW620 cell lines. Acta
Biochim Biophys Sin (Shanghai). 43:217–225. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Zhang M, Liu Q, Mi S, et al: Both
miR-17-5p and miR-20a alleviate suppressive potential of
myeloid-derived suppressor cells by modulating STAT3 expression. J
Immunol. 186:4716–4724. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Li X, Pan JH, Song B, et al: Suppression
of CX43 expression by miR-20a in the progression of human prostate
cancer. Cancer Biol Ther. 13:890–898. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Wang W, Feng L, Zhang H, et al:
Preeclampsia up-regulates angiogenesis-associated microRNA (i.e.,
miR-17, -20a, and -20b) that target ephrin-B2 and EPHB4 in human
placenta. J Clin Endocrinol Metab. 97:E1051–E1059. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Poliseno L, Pitto L, Simili M, et al: The
proto-oncogene LRF is under post-transcriptional control of
MiR-20a: implications for senescence. PLoS One. 3:e25422008.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Wu H, Wang F, Hu S, et al: MiR-20a and
miR-106b negatively regulate autophagy induced by leucine
deprivation via suppression of ULK1 expression in C2C12 myoblasts.
Cell Signal. 24:2179–2186. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Elkayam E, Kuhn CD, Tocilj A, et al: The
structure of human argonaute-2 in complex with miR-20a. Cell.
150:100–110. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Stark MS, Tyagi S, Nancarrow DJ, et al:
Characterization of the melanoma miRNAome by deep sequencing. PLoS
One. 5:e96852010. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Persson H, Kvist A, Rego N, et al:
Identification of new microRNAs in paired normal and tumor breast
tissue suggests a dual role for the ERBB2/Her2 gene. Cancer Res.
71:78–86. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Jima DD, Zhang J, Jacobs C, et al: Deep
sequencing of the small RNA transcriptome of normal and malignant
human B cells identifies hundreds of novel microRNAs. Blood.
116:e118–e127. 2010. View Article : Google Scholar : PubMed/NCBI
|