Introduction
More than 300 cases of small cell esophageal
carcinoma have been reported since the initial case report by
McKeown in 1952 (1). Small cell
neuroendocrine carcinoma (SCNEC) is a rare disease with aggressive
and malignant biological behavior and a poor prognosis. SCNEC
accounts for 1.0 to 2.8% of all esophageal cancers (2,3). A new
classification of neuroendocrine tumors that included SCNEC was
introduced by the World Health Organization (WHO) in 2010 (4). This new classification distinguishes
between well- and poorly differentiated neuroendocrine tumors in a
different manner than the 1963 classification by Sandler and
Williams, and the 2000 classification by WHO (5–7). Small
and large cell esophageal carcinomas are classified as
neuroendocrine carcinomas (NECs) of proliferative activity grade 3
(Ki-67 >20%). Although SCNEC is a highly proliferative
carcinoma, a standard therapeutic strategy has not yet been
developed. A multidisciplinary treatment approach consisting of
resection, chemotherapy and radiation therapy is recommended for
the treatment of extrapulmonary small cell carcinoma according to
the National Comprehensive Cancer Network (NCCN) guidelines for
small cell lung cancer. In the present study, we review previous
studies and clinical trials of treatments for NECs resistant to
chemotherapy.
Case report
Written informed consent was obtained from the
patient for publication of this case report and any accompanying
images. A 66-year-old male was referred to our hospital in October
2009 with an initial diagnosis of esophageal cancer based on the
results of gastrointestinal endoscopy. The patient had not
experienced dysphagia, weight loss or retrosternal/epigastric pain.
The patient had a history of tobacco and alcohol use. There was no
personal or family history of malignancy. A physical examination
revealed no abnormalities. A routine complete blood count revealed
normochromic anemia with a hemoglobin level of 10.2 g/dl. Blood
chemistry findings revealed hypoalbuminemia (albumin, 3.2 g/dl).
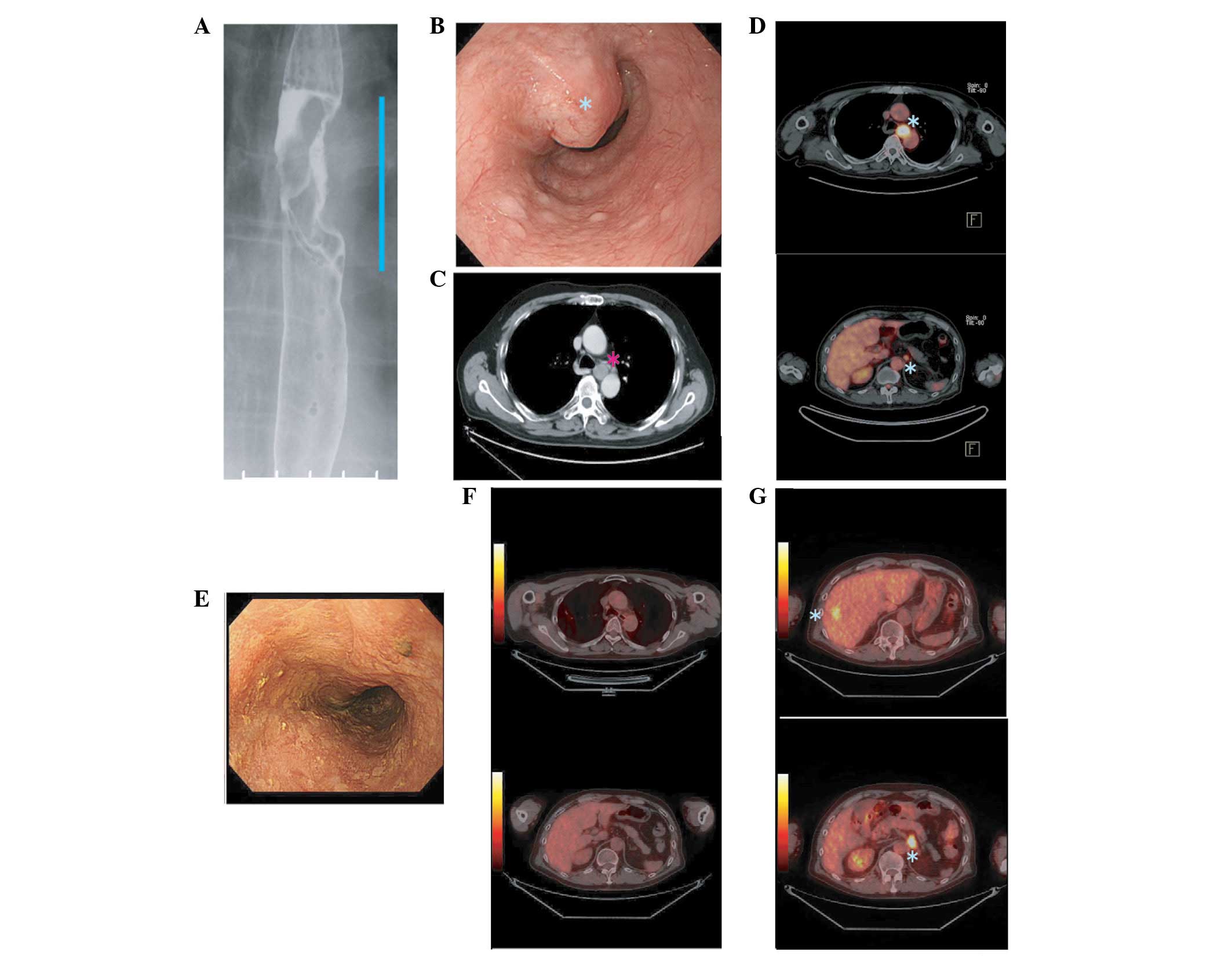
Barium studies and endoscopic examination of the upper
gastrointestinal tract revealed a type 2 tumor in the middle of the
esophagus (Fig. 1A and B). The
tumor contained a deep central ulceration 4.5 cm in length.
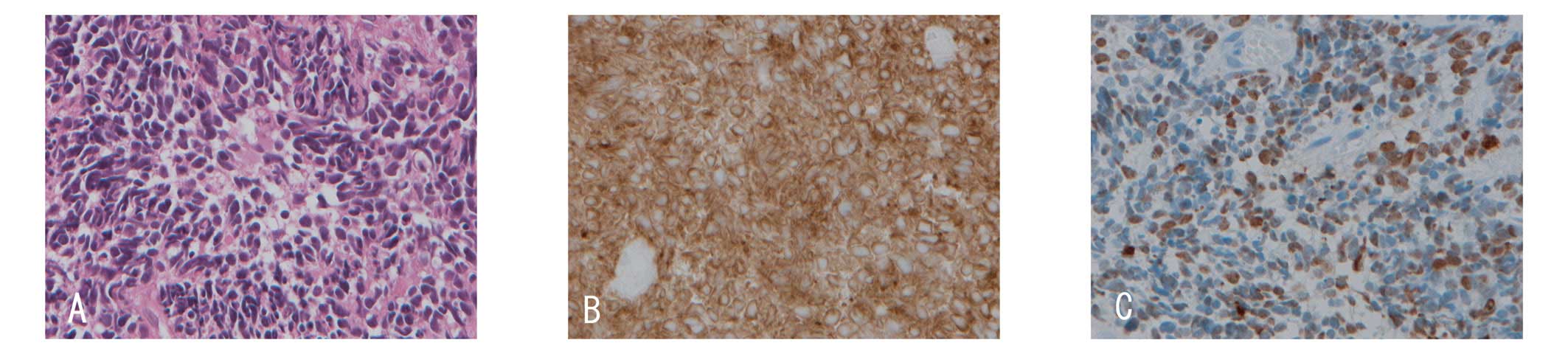
Hematoxylin-eosin (HE) and immunohistochemical staining were
performed on biopsy samples. Microscopic examination of endoscopic
biopsy specimens revealed a small cell carcinoma that had invaded
into the submucosal region. The tumor cells showed a high
nuclear-cytoplasmic ratio, hyperchromic nuclei and absent or
inconspicuous nucleoli, which are typical features of small cell
lung carcinoma (SCLC; Fig. 2A).
Tumor cells were positive for AE1/AE3, synaptophysin (Fig. 2B) and CD56. Chromogranin-positive
cells were scattered. More than 40% of tumor cells were positive
for Ki-67 (Fig. 2C). The
pathological diagnosis of the tumor was NEC, G3, small cell type.
The patient was finally diagnosed with SCNEC of the esophagus. A
contrast-enhanced computed tomography (CT) scan of the chest and
abdomen revealed an area of poorly enhancing esophageal wall
thickening, which was 4.5 cm long, concomitant with enlarged lymph
nodes (Fig. 1C). Fluorodeoxyglucose
positron emission tomography/CT (FDG PET-CT) revealed an
18F-FDG-accumulated primary lesion and multiple lymph
nodes around the mediastinal locoregional lesion and the celiac
artery with maximal standardized uptake value (SUVmax)
of 3.0–9.0. The clinical stage was classified as cT3N2M0, stage
IIIB [2010 Seventh Edition of the American Joint Committee on
Cancer (AJCC)/International Union Against Cancer (UICC)] (8,9), and
it was considered to be extensive disease according to the
Veterans’ Administration Lung Study Group (VALSG) Staging System
used for small cell lung cancer (10). Laboratory tests revealed an elevated
level of serum progastrin-releasing peptide (ProGRP, 105 pg/ml;
normal range, <81.0 pg/ml) and nonspecific enolase (NSE; 21.1
ng/ml; normal range, <16.3 ng/ml). The serum carcinoembryonic
antigen (CEA) and squamous cell carcinoma antigen (SCC) levels were
within normal limits. The patient was in good condition with an
Eastern Cooperative Oncology Group performance status of 0.
Combination chemotherapy consisting of 60
mg/m2 cisplatin on day 1 plus 60 mg/m2
irinotecan administered intravenously on days 1, 8 and 15 was
administered every 4 weeks (IP) from Dec 2010 until Aug 2011.
During the first chemotherapy course, the patient developed
bacterial pneumonia with neutropenia. Therefore, the total dose of
cisplatin was reduced by 20% for the remaining 5 courses. After the
third course of IP, the primary lesion disappeared and multiple
lymph nodes that had been previously enlarged were reduced to
normal size, as indicated by GI endoscopy and PET-CT. No cancer
cells were observed in an endoscopic biopsy specimen, indicating a
positive therapeutic response and complete remission of the tumor.
However, lymph nodes around the celiac artery increased in size
again in October 2011 immediately following cessation of the sixth
course of IP. These enlarged lymph nodes were considered to
indicate refractory relapse cancer. The ProGRP and NSE levels
increased further. The axial PET-CT image revealed intense FDG
avidity in lymph nodes around the celiac artery with an
SUVmax of 8.3. The patient was treated with amrubicin
(AMR, 35 mg/m2) administered intravenously for 3
consecutive days every 3 weeks. In the first round of AMR therapy,
the patient experienced febrile neutropenia that was treated with
ciprofloxacin. Hence, the total dose of AMR was reduced by 20% for
the remaining 2 courses. Although the ProGRP and NSE levels
returned to normal 1 month after the initiation of AMR
chemotherapy, they were elevated after the third course of AMR, and
PET-CT revealed progressive disease with liver metastasis and
expanded lymph node metastasis. As the patient remained
asymptomatic, paclitaxel (100 mg/m2) was started as
third-line chemotherapy.
Extrapulmonary SCNECs are relatively rare tumors
that occur in almost every organ, including the cervix, esophagus,
pharynx, larynx, lymph node, pancreas, colon and rectum. According
to the epidemiological data, the prevalence and incidence of
gastroentero-pancreatic neuroendocrine tumors have been increasing,
which is likely due to improved detection methods (11). Although certain extrapulmonary
SCNECs have slow progressive behavior, most are aggressive and have
a poor prognosis despite various multidisciplinary treatments. In
particular, gastrointestinal SCNECs, including esophageal SCNECs,
have a worse prognosis compared with tumors in other sites
(12).
In the present study, we reviewed the literature
regarding characteristics of SCNEC. We searched PubMed and
Ichushi-Web with a combination of three terms, ‘small cell
carcinoma’, ‘neuroendocrine carcinoma’ and ‘esophagus’, for studies
published in or after 2000. Our search yielded 229 reports of
SCNEC, from which we extracted the data on diagnosis, age, gender,
disease stage, treatment and survival time. The data (including the
present case) are summarized in Table
I. The median age was 64 years (interquartile range, 23–90
years) and the majority (73%) of the patients were male.
 | Table IPatient characteristics and treatment
course for the 9 cases of small cell carcinoma of the esophagus
that relapsed after complete remission with chemotherapy or
resection by surgery in the literature between 2000 and 2011. |
Table I
Patient characteristics and treatment
course for the 9 cases of small cell carcinoma of the esophagus
that relapsed after complete remission with chemotherapy or
resection by surgery in the literature between 2000 and 2011.
| Case/(Ref.) | Gender | Age (years) | Type | Location | Tumor size (mm) | VALSG stage | Sensitive
relapse | Refractory
relapse | CT regimen | Radiotherapy
(Gy) | Treatment course | Survival time after
recurrence (months) | Survival time
(months) | Outcome |
|---|
| 1a | Male | 66 | 2 | Mt | 30 | ED | | 1 | IPx6, AMRx3,
TXLx1 | - | CT, CT, CT | >6 | 17 | Alive |
| 2 (16) | Male | 53 | NA | MtLt | NA | ED | 1 | | EPx4, sur, RT, EPx7,
topotecan/paclitaxelx1, CPT-11/paclitaxelx1 | 40 | CT, sur, RT, CT, CT,
CT | NA | 44 | Deceased |
| 3 (17) | Male | 66 | 3 | MtLt | NA | ED | | 1 | IPx4, RT/TXL,
CBDCA/VP16 | 60 | CT, CRT, CT | >9 | 16 | Alive |
| 4 (18) | Male | 59 | 2 | UtMt | 60 | ED | | 1 | IPx6, CBDCA,
VP16x3 | - | CT-CT | 6 | 18 | Deceased |
| 5 (19) | Male | 53 | 1 | MtLt | 100 | ED | 1 | | CDDP, etoposide,
FPx2, DOC, IP | 45 | CCRT, sur, CT,
hepatectomy | >48 | 56 | Alive |
| 6 (20) | Male | 65 | 2 | Mt | NA | ED | | 1 | CDDP/5FU, CPT-11,
GEM, TXL | 60 | CCRT, CT, CT | >10 | 18 | Alive |
| 7 (21) | Male | 59 | 3 | Lt | 90 | ED | | 1 | FPx10, ETPx6,
ETP/CPA/EPIx3, FPx4/RT | - | CT, CT, CT,
CCRT | 12 | 24 | Deceased |
| 8 (22) | Male | 65 | NA | Lt | 95 | ED | | 1 | PVP/CAV | 60 | CRT, sur, CT | NA | 17 | Deceased |
| 9 (22) | Female | 61 | 2 | Mt | 58 | LD | 1 | | PVP/CEV, 5FU,
CBDCA | - | CT, sur, CT | 12 | 21 | Deceased |
Macroscopic tumor characteristics and
clinical stage
Most esophageal SCNECs occur in the middle (50%) or
lower (42%) sites of the esophagus, as shown in Table I. The average tumor length was 6.5
cm (±2.3 cm) and 66% (88/133) of the tumors were longer than 5 cm.
The most frequent gross appearance of SCNEC was type 2 localized
ulcerated type, which accounted for 57% (55/96) of cases. According
to the VALSG criteria, 53% (121 of 229) of all cases had limited
disease and 47% (108 of 229) had extensive disease (ED).
Histology and
immunohistochemistry
In the 21st century, the WHO classification for
neuroendocrine tumors (NETs) has been revised twice. The WHO 2000
classification focused on tumor stage, including lymph node or
distant metastasis, while the WHO 2010 classification is more
practical and reflects clinical prognosis. SCNEC is to be included
in NEC for high grade (G3) moderately to poorly differentiated
neuroendocrine neoplasms. In addition, extrapulmonary SCNECs,
including SCNECs of the esophagus, are indistinguishable from SCLC
in histological and immunohistochemical features (13,14).
Due to their histological similarity, protocols for SCLC have been
recommended for the diagnosis of extrapulmonary SCNEC (15). Accurate histological diagnosis is
critical as systemic chemotherapy recommended for SCNEC differs
from therapies for squamous cell carcinoma or adenocarcinoma of the
esophagus. To diagnose the tumor as SCNEC, it is essential to
identify typical small cell carcinoma histology and
immunohistochemical evidence of epithelial differentiation.
Although positive staining for neuroendocrine markers is not
necessary, it makes a supplementary contribution to the diagnosis.
Li et al reported that the incidence for positive
immunohistochemical reactivity for CK8, synaptophysin, NSE and CD56
in gastrointestinal SCNEC was >90% and that these markers were
useful in diagnosis (14,23), whereas Yun et al reported
that the percentages of SCNEC samples with positive
immunoreactivity were: Syn, 95.2%; CD56, 76.2%; TTF-1, 71.4%; NSE,
61.9%; CgA, 61.9%; CK, 57.1%; EMA, 61.9%; and S100, 19.0% (14). A multivariate analysis by Shia et
al identified the following three factors as having an adverse
impact on 2-year disease-specific survival: the absence of an
associated adenocarcinoma component (P=0.04), the presence of
synaptophysin staining (P=0.05) and high disease stage
(P<0.0001) (24). The present
case has all three of these factors; thus, a poor prognosis may be
predicted.
Imaging for diagnosis and the
evaluation of the response to treatment
As reported by Howard et al(12), little information has been reported
regarding the CT findings of extrapulmonary SCNEC. The use of
high-resolution CT is the current standard approach to assess tumor
spreading. However, PET-CT may be useful for both staging and
restaging by detecting new lesions as SCNEC is typically
18F-FDG avid (12,25).
In the present case, the results of PET-CT influenced the decision
to initiate second-line chemotherapy after disease recurrence was
detected, concomitant with the elevation of NSE and ProGRP levels.
Careful evaluation is necessary as SCNEC tends to recur repeatedly
at distant sites, including the brain. It remains to be determined
if PET-CT will lead to improved patient outcome in restaging after
treatment. Further investigations are needed to confirm the benefit
and cost effectiveness of PET-CT compared with conventional imaging
techniques such as contrast-enhanced CT.
Treatments
Surgery
Surgery is one of the mainstay treatments for
neuroendocrine tumors (NETs) (G1, G2), with the exception of cases
with distant metastasis, whereas there is no definitive evidence
that surgery is optimal for NEC (G3), including esophageal disease
(11). However, certain studies
advocate surgery as a treatment for SCNEC cases with limited
disease, but not with ED, due to the possibility of a benefit to
long-term survival (2). The outcome
for SCNEC treated with surgery alone is extremely poor due to a
high likelihood of disease recurrence, even following complete
resection; therefore, chemotherapy and chemoradiation before or
after surgery may be critical for improved survival.
Radiotherapy
Radiotherapy alone is insufficient to improve the
survival of SCNEC patients. Rather, radiotherapy has been
successfully used to treat bone and brain metastases.
Radioembolization using Yttrium-90 microspheres has been revealed
as an optional treatment for tumor control of NET with liver
metastasis (25).
Chemotherapy
The therapeutic strategies for slowly progressive
NET (G1, G2) and rapidly progressive NEC (G3) are fundamentally
different. NCCN guidelines recommend: i) a watch-and-wait approach
every 3 to 6 months until the disease progresses; ii) enrollment in
a clinical trial; or iii) the administration of octreotide, which
is a somatostatin analog and a biotherapeutic agent (26). In February 2011, the New England
Journal of Medicine published reports that everolimus and sunitinib
demonstrated significant clinical benefit in a randomized phase 3
trial for the treatment of advanced pancreatic neuroendocrine
tumors (26,27). However, according to SCLC
guidelines, systemic chemotherapy is recommended for the treatment
of NEC. A regimen of etoposide and cisplatin (EP) is most
frequently used in patients with SCLC. Furthermore, EP plus
concurrent thoracic radiotherapy are recommended for SCLC with
limited disease (15). A randomized
phase 3 trial for SCLC with extensive disease in Japan demonstrated
a significant difference in survival between patients treated with
IP and EP (28). Unfortunately, two
subsequent phase 3 trials performed in the USA found no benefit in
the use of IP therapy instead of EP therapy (29,30).
We selected the IP regimen for first-line therapy in the present
case (28).
Most patients eventually relapse after initial
chemotherapy and require second-line chemotherapy. As shown in
Table I, only a few studies
published between 2000 and 2011 have reported the treatment course
for patients with SCNEC that relapsed after a complete remission
with chemotherapy or resection by surgery. In general, the
prognosis at relapse is extremely poor, and the response to
second-line chemotherapy tends to be limited. There are occasions
when this rule does not apply. Several phase 2 studies have
demonstrated that single-agent amrubicin (AMR) had promising
effects on patients with refractory SCLC (31,32).
Results from these studies showed overall response rates of 21.3 to
53% and median survival periods of 5.7 to 10.3 months in refractory
SCLC. AMR is a promising agent for second-line therapy in patients
with platinum-refractory SCLC. Furthermore, Asayama et al
have reported the achievement of an objective response in two of
three patients with refractory or recurrent SCNEC treated with AMR
as a second- or third-line therapy (35).
As candidates for single-agent, second-line
chemotherapy in SCLC, etoposide, topotecan, paclitaxel,
gemcitabine, pemetrexed and picoplatin may be considered in
addition to AMR. Although etoposide, pemetrexed and gemcitabine
failed to demonstrate survival benefits, better response rates were
achieved compared with the response rates of 47% for irinotecan and
29% for paclitaxel (36). Moreover,
clinical trials for bortezomib, bendamustine, sunitinib,
rebeccamycin analog BI 2536, Hsp90 inhibitor, STA-9090, BIBF 1120,
ADI-PEG 20, chloroquinoxaline sulfonamide, FR901228 and NK012 are
currently underway throughout the world.
The candidates for combination second-line
chemotherapy in SCLC include cisplatin + etoposide, cisplatin +
irinotecan, carboplatin + irinotecan, cisplatin + etoposide +
irinotecan, carboplatin + paclitaxel, paclitaxel + everolimus,
pasireotideLAR + topotecan, paclitaxel + gemcitabine, topotecan +
bevacizumab and vorinostat + topotecan. The response rates for
these treatments have varied, and further investigations are
necessary to identify their effectiveness for
chemotherapy-resistant SCLC. Molecular-targeted agents, including
imatinib, bevacizumab, cediranib, sorafenib and gefitinib, have not
been promising treatments for SCLC (36). The development of various
chemotherapeutic agents for NEC is expected.
Conclusion
Patients with SCNEC of the esophagus with ED should
be treated with aggressive chemotherapy rather than surgery or
radiation monotherapy. In this case, tumor markers such as ProGRP,
NSE and PET-CT detected disease relapse. Further research may be
required to exploit the promising agents for treatment of
chemotherapy-resistant SCNEC.
Acknowledgements
HT and TH coordinated and assisted in
drafting the manuscript. The authors are grateful to Akinori
Hashiguchi for performing the histological and immunohistochemical
analyses in this case. The authors would also like to thank Kana
Teramoto, Naoki Miyata, Satoshi Kurita, Masayuki Adachi, Yasuo
Hamamoto and Hajime Higuchi for additional assistance. All authors
read and approved the final manuscript.
References
|
1
|
McKeown F: Oat-cell carcinoma of the
oesophagus. J Pathol Bacteriol. 64:889–891. 1952. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Brenner B, Tang LH, Klimstra DS and Kelsen
DP: Small-cell carcinomas of the gastrointestinal tract: a review.
J Clin Oncol. 22:2730–2739. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lv J, Liang J, Wang J, et al: Primary
small cell carcinoma of the esophagus. J Thorac Oncol. 3:1460–1465.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Klöppel G, Rindi G, Perren A, Komminoth P
and Klimstra DS: The ENETS and AJCC/UICC TNM classifications of the
neuroendocrine tumors of the gastrointestinal tract and the
pancreas: a statement. Virchows Arch. 456:595–597. 2010.PubMed/NCBI
|
|
5
|
Williams E and Sandler M: The
classification of carcinoid tumours. Lancet. 7275:238–239. 1963.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Solcia E, Klöppel G and Sobin LH:
Histological Typing of Endocrine Tumours. World Health Organization
- International Histological Classification of Tumours. 2nd
edition. Springer; Berlin, Germany: 2000, View Article : Google Scholar
|
|
7
|
Klöppel G: Classification and pathology of
gastroenteropancreatic neuroendocrine neoplasms. Endocr Relat
Cancer. 18(Suppl 1): S1–S16. 2011.
|
|
8
|
Rice TW: Staging of esophageal cancer: TNM
and beyond. Esophagus. 7:189–195. 2010. View Article : Google Scholar
|
|
9
|
Sobin LH, Gospodarowicz MK and Wittekind
C: TNM Classification of Malignant Tumors. 7th edition.
Wiley-Blackwell; Oxford, UK: 2009
|
|
10
|
Stahel RA, Ginsberg R, Havemann K, et al:
Staging and prognostic factors in small cell lung cancer; a
consensus report. Lung Cancer. 5:119–126. 1989. View Article : Google Scholar
|
|
11
|
Rindi G and Wiedenmann B: Neuroendocrine
neoplasms of the gut and pancreas: new insights. Nat Rev
Endocrinol. 8:54–64. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Howard S, O’Regan K, Jagannathan J,
Krajewski K, Giardino A and Ramaiya N: Extrapulmonary small cell
carcinoma: a pictorial review. AJR Am J Roentgenol. 197:W392–W398.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Frazier SR, Kaplan PA and Loy TS: The
pathology of extrapulmonary small cell carcinoma. Semin Oncol.
34:30–38. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Li AF, Li AC, Hsu CY, Li WY, Hsu HS and
Chen JY: Small cell carcinomas in gastrointestinal tract:
immunohistochemical and clinicopathological features. J Clin
Pathol. 63:620–625. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
NCCN Clinical Practice Guidelines in
Oncology (NCCN Guidelines): Small Cell Lung Cancer. 2012.http://www.nccn.org/professionals/physician_gls/pdf/sclc.pdf.
Accessed October 2, 2012.
|
|
16
|
Kanzaki M, Muto Y, Yoshinouchi S, Maeda M,
Miyoshi S and Teshima S: A case of esophageal small cell carcinoma
with syndrome of inappropriate antidiuretic hormone secretion. Gan
To Kagaku Ryoho. 37:1941–1944. 2010.PubMed/NCBI
|
|
17
|
Mizuiri H, Hihara J and Okada M:
CDDP+CPT-11 therapy is useful for stage IVb esophageal small cell
carcinoma. Gan To Kagaku Ryoho. 36:831–834. 2009.
|
|
18
|
Nakajima S, Hidaka H, Umekita Y, Uchiyama
S, Kataoka H and Chijiiwa K: A 4-year survivor of small cell
carcinoma of the esophagus with liver metastases managed by
multidisciplinary treatment. J Jpn Surg Assoc. 64:1605–1610.
2009.
|
|
19
|
Matsuoka M, Boku N, Yoshino T, Hironaka S,
Onozawa Y, Fukutomi A, Zenda S, Yamazaki K, Yasui H, Hasuike N,
Inui T, Yamaguchi Y, Ono H and Kamata M: Small cell carcinoma of
the esophagus responding to fourth-line chemotherapy with weekly
paclitaxel. Int J Clin Oncol. 10:429–432. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tsujie M, Shibata N, Nomura T, Tanaka T,
Morimoto T, Fujita S, Kitani K, Nakahira S, Okuda H and Takeda M: A
patient with stage IVb small cell carcinoma of the esophagus who
survived 23 months after systemic cancer chemotherapy. Gan To
Kagaku Ryoho. 30:271–275. 2003.PubMed/NCBI
|
|
21
|
Koide N, Hiraguri M, Kishimoto K, Nakamura
T, Adachi W, Miyabayashi H, Terai N and Amano J: Small cell
carcinoma of the esophagus with reference to alternating multiagent
chemotherapy: report of two cases. Surg Today. 33:294–298. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yun JP, Zhang MF, Hou JH, et al: Primary
small cell carcinoma of the esophagus: clinicopathological and
immunohistochemical features of 21 cases. BMC Cancer. 7:382007.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Shia J, Tang LH, Weiser MR, et al: Is
nonsmall cell type high-grade neuroendocrine carcinoma of the
tubular gastrointestinal tract a distinct disease entity? Am J Surg
Pathol. 32:719–731. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gregory DL, Brennan SM, Stillie A, et al:
Impact of 18F-fluorodeoxyglucose positron emission tomography in
the staging and treatment response assessment of extra-pulmonary
small-cell cancer. J Med Imaging Radiat Oncol. 54:100–107. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Vyleta M and Coldwell D: Radioembolization
in the treatment of neuroendocrine tumor metastases to the liver.
Int J Hepatol. 2011:7853152011.PubMed/NCBI
|
|
26
|
NCCN Clinical Practice Guidelines in
Oncology (NCCN Guidelines): Neuroendocrine Tumors. 2011.http://www.nccn.org/professionals/physician_gls/pdf/neuroendocrine.pdf.
Accessed October 2, 2012.
|
|
27
|
Raymond E, Dahan L, Raoul JL, et al:
Sunitinib malate for the treatment of pancreatic neuroendocrine
tumors. N Eng J Med. 364:501–513. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yao JC, Shah MH, Ito T, et al; RAD001 in
Advanced Neuroendocrine Tumors, Third Trial (RADIANT-3) Study
Group: Everolimus for advanced pancreatic neuroendocrine tumors. N
Eng J Med. 364:514–523. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Noda K, Nishiwaki Y, Kawahara M, et al;
Japan Clinical Oncology Group: Irinotecan plus cisplatin compared
with etoposide plus cisplatin for extensive small-cell lung cancer.
N Eng J Med. 346:85–91. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hanna N, Bunn PA Jr, Langer C, et al:
Randomized phase III trial comparing irinotecan/cisplatin with
etoposide/cisplatin in patients with previously untreated
extensive-stage disease small-cell lung cancer. J Clin Oncol.
24:2038–2043. 2006. View Article : Google Scholar
|
|
31
|
Lara PN Jr, Natale R, Crowley J, et al:
Phase III trial of irinotecan/cisplatin compared with
etoposide/cisplatin in extensive-stage small-cell lung cancer:
clinical and pharmacogenomic results from SWOG S0124. J Clin Oncol.
27:2530–2535. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ettinger DS, Jotte R, Lorigan P, et al:
Phase II study of amrubicin as second-line therapy in patients with
platinum-refractory small-cell lung cancer. J Clin Oncol.
28:2598–2603. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Onoda S, Masuda N, Seto T, et al; Thoracic
Oncology Research Group Study 0301: Phase II trial of amrubicin for
treatment of refractory or relapsed small-cell lung cancer:
Thoracic Oncology Research Group Study 0301. J Clin Oncol.
24:5448–5453. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Asayama M, Fuse N, Yoshino T, et al:
Amrubicin for the treatment of neuroendocrine carcinoma of the
gastrointestinal tract: a retrospective analysis of five cases.
Cancer Chemother Pharmacol. 68:1325–1330. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kim YH and Mishima M: Second-line
chemotherapy for small-cell lung cancer (SCLC). Cancer Treat Rev.
37:143–150. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kuo CH, Hsieh CC, Chan ML, Li AF, Huang
MH, Hsu WH and Hsu HS: Small cell carcinoma of the esophagus: a
report of 16 cases from a single institution and literature review.
Ann Thorac Surg. 91:373–378. 2011. View Article : Google Scholar : PubMed/NCBI
|