Introduction
Lange initially described obliterative bronchiolitis
in 1901 (1) and the unique clinical
and histological characteristics of this pulmonary disease are
well-known. Epler et al coined the term ‘bronchiolitis
obliterans organizing pneumonia’ (BOOP) to describe this condition.
In 1985, the authors described 50 patients with BOOP and showed
that it was a clinicopathological entity that was distinct from
irreversible pulmonary fibrosis (2). Histologically, BOOP is characterized
by granulation tissue and polypoid masses in the lumens of the
small airways, alveolar ducts and certain alveoli. Radiographs show
an unusual pattern of patchy densities with a ‘ground glass’
appearance. In 2001, the American Thoracic Society and the European
Respiratory Society standardized the classification of idiopathic
interstitial pneumonia with the presence of BOOP as cryptogenic
organizing pneumonia (COP) (3).
Idiopathic interstitial pneumonia may be associated
with post-respiratory infection, drug addiction, connective tissue
disease, organ transplantation, inhalation of fumes and
radiotherapy (4,5). Crestani et al defined COP
associated with radiotherapy for breast cancer as the presence of
the following conditions: i) radiation therapy to the breast within
12 months of symptom onset; ii) general and/or respiratory symptoms
lasting for ≥2 weeks; iii) infiltrations outside the irradiated
volume; and iv) no other specific cause of the symptoms (6). Although there are a number of previous
studies that have reported COP following radiotherapy for breast
cancer, reports of COP following radiotherapy for non-small cell
lung cancer (NSCLC) are rare. COP associated with radiotherapy is
occasionally diagnosed as radiation pneumonitis or bacterial
pneumonia. The current study presents two cases of COP following
radiotherapy, one breast cancer patient and the other a patient
with NSCLC. Written informed consent was obtained from the
patients.
Case reports
Breast cancer case
A 48-year-old premenopausal female with cancer in
the left breast underwent breast conservative surgery and sentinel
lymph node biopsy. The pathological examination led to a diagnosis
of stage I (pT1cN0M0) invasive ductal carcinoma that was estrogen
receptor-positive and Her2 receptor-positive. The patient underwent
a second surgery following observations of a positive surgical
margin, and, three months following the initial surgery, underwent
radiotherapy of 50 Gy in 25 fractions to the left whole breast
using a 4-MV X-ray and a tangential field technique. An additional
dose of 10 Gy in five fractions to the tumor bed was used as a
boost therapy with 9 MeV electrons (Fig. 1A). The patient was treated with
anti-estrogen therapy consisting of oral tamoxifen plus
subcutaneous luteinizing hormone-releasing hormone agonist.
The patient experienced a fever (38.8°C) and
developed a productive cough four months following the completion
of radiotherapy. The patient’s laboratory results are shown in
Table I. A chest X-ray showed an
infiltrating shadow in the upper lobe of the left lung. Computed
tomography (CT) showed consolidation with air bronchogram in the
left upper lobe and ground glass densities in the left lower and
right upper lobes (Fig. 2A and 2B).
As these shadows appeared outside of the irradiated volume, the
patient was diagnosed with bacterial pneumonia, despite no
pathological confirmation. The patient was treated with oral
levofloxacin (500 mg for 14 days). Four weeks following the
completion of levofloxacin therapy, a CT scan showed that the
original consolidation and ground glass densities had resolved, but
new ground glass densities had appeared in the right upper lobe.
Transbronchial lung biopsy revealed alveolar septal and focal
intra-alveolar fibrosis; these observations were consistent with an
observation of organizing bronchopneumonia. Examination of
bronchoalveolar lavage (BAL) fluid showed the following: 34%
lymphocytes, 5% neutrophils, 19% eosinophils and 42% macrophages.
The CD4/CD8 ratio was 0.78. Based on these results, the patient was
diagnosed with COP. After 8 months, all symptoms were spontaneously
resolved (Fig. 2C and 2D). A CT
showed only slight fibrosis in the lung that was consistent with
the irradiated volume. The patient began taking herbal medicine
containing Scutellaria root to treat menopause symptoms.
However, two weeks following this, the patient experienced symptoms
similar to those experienced with COP. The CT scan showed new
ground glass densities in the right upper lobe and the patient was
suspected to have a recurrence of COP. The patient stopped taking
the herbal medicine, and symptoms and shadows on the CT
resolved.
 | Table IPatient laboratory results. |
Table I
Patient laboratory results.
| Parameters | Breast cancer
patient | Lung cancer
patient |
|---|
| Hematology |
| WBC count,
cells/μl |
9.9×103 |
6.2×103 |
| Neutrophils, % | 86.7 | 74.4 |
| Lymphocytes, % | 6.8 | 15.2 |
| Eosinophils, % | 2.4 | 2.4 |
| RBC, cells/μl |
4.55×106 |
4.24×106 |
| Hb, g/dl | 12.8 | 13.7 |
| Ht, % | 38.5 | 41.1 |
| Plt, cells/μl |
3.79×105 |
2.11×105 |
| Serology |
| CRP, mg/dl | 9.5 | 11.8 |
| KL-6, U/ml | 310 | 833 |
| Biochemistry |
| TP, g/dl | 7.3 | 7.0 |
| T-Bil, mg/dl | 0.35 | 0.97 |
| GOT, IU/l | 30 | 20 |
| GPT, IU/l | 39 | 24 |
| LDH, IU/l | 215 | 168 |
| ALP, IU/l | 750 | 274 |
| γ-GTP, IU/l | 177 | 54 |
| BUN, mg/dl | 8.5 | 19.6 |
| Cr, mg/dl | 0.54 | 0.66 |
| Na, mEq/l | 143 | 141 |
| K, mEq/l | 4.0 | 4.3 |
| Cl, mEq/l | 106 | 104 |
| BAL fluid |
| Total cell count,
cells/ml |
2.5×107 | - |
| Macrophages, % | 42.4 | - |
| Lymphocytes, % | 34.0 | - |
| Neutrophils, % | 4.8 | - |
| Eosinophils, % | 18.8 | - |
| CD4/CD8 | 0.78 | - |
NSCLC case
An 84-year-old male exhibited clinical stage IIA
(cT1bN1M0) squamous cell lung cancer in the right upper lobe and
was considered unsuitable for surgery due to poor pulmonary
function. The patient underwent radiotherapy to the primary tumor
and metastatic lymph node, consisting of 60 Gy in 30 fractions
using 10-MV X-rays (Fig. 1B). The
patient was referred to Tokyo Medical University (Tokyo, Japan) due
to a non-productive cough four months following the completion of
radiotherapy. The patient was afebrile, but blood CRP levels were
elevated (11.8 mg/dl). The patient’s laboratory results are shown
in Table I. The chest radiograph
showed a patchy shadow in the right middle lobe. CT disclosed
consolidation in the right lower lobe that was consistent with the
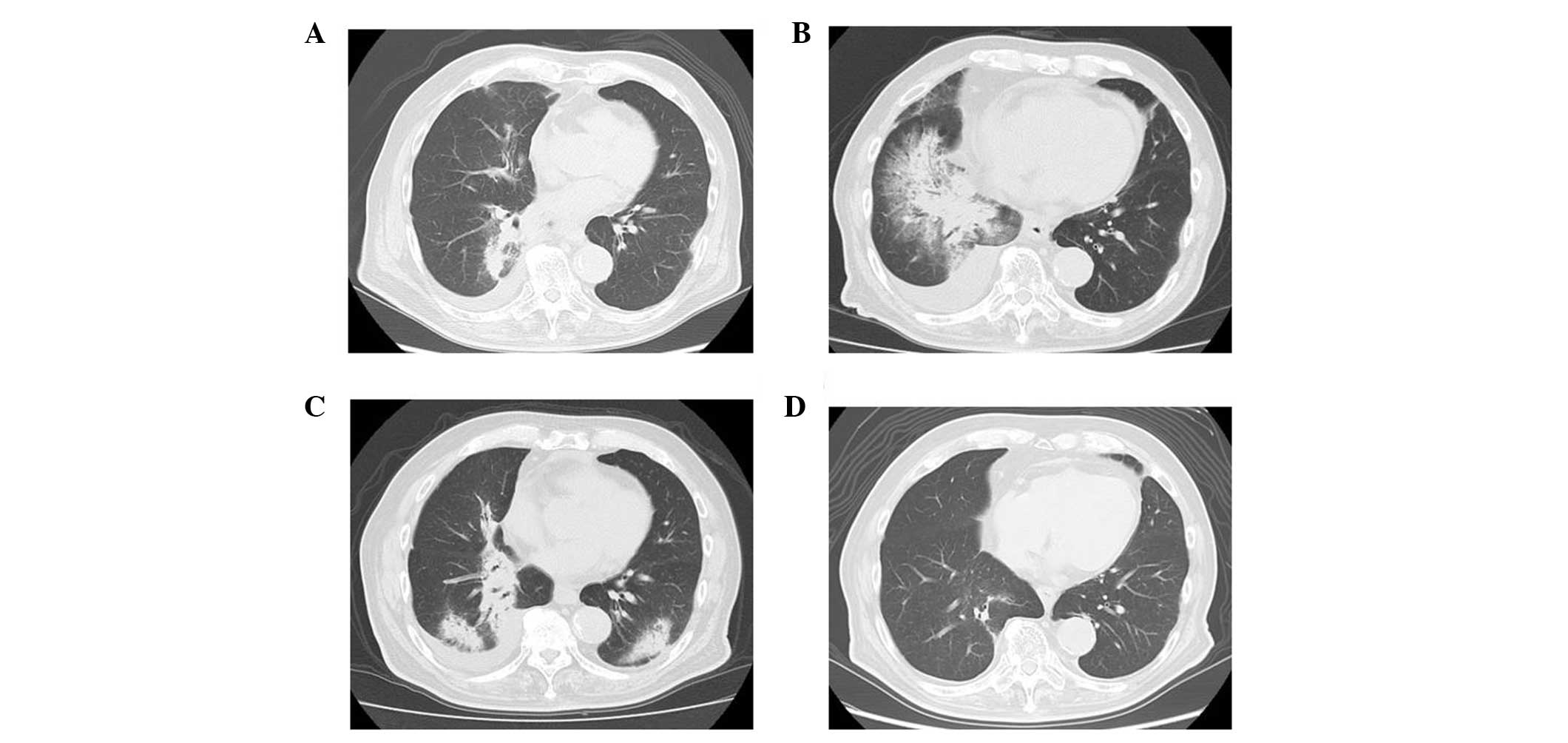
irradiation volume (Fig. 3A). The
patient was diagnosed with radiation pneumonitis and prescribed
oral prednisone at a dose of 30 mg per day. After two weeks, the
patient’s symptoms and laboratory results improved, and the patchy
shadow on the chest radiograph disappeared. The prednisone dose was
tapered and discontinued after two months. The patient again
developed a non-productive cough without fever three months
following the first presentation with a cough. CT showed an
extended consolidated shadow outside the irradiated volume
(Fig. 3B) that was resolved
following the administration of oral prednisone. The prednisone was
discontinued and 11 months following the completion of
radiotherapy, a follow-up CT showed an infiltrated shadow in the
lower lobes (Fig. 3C). The white
blood cell count was 4.8×103 cells/μl and the C-reactive
protein levels were 1.1 mg/dl. The patient was diagnosed with COP
due to their responsiveness to prednisone, migration of the
consolidation on CT and resolution of fibrosis following shadow
consolidation. The patient again received daily oral prednisone (20
mg/day) for four months, which was tapered over seven months. The
infiltrated shadows disappeared following 18 months of radiotherapy
(Fig. 3D).
Discussion
The two cases presented in the current study
exhibited CTs that showed consolidations with a ground glass
appearance. In the two cases, the shadows migrated and were finally
resolved without any fibrotic changes in the CT examination. No
observations of bacterial or viral infection were identified. These
observations were compatible with COP according to the criteria of
Crestani et al(6), who
defined COP to be associated with radiotherapy.
Takigawa et al reported that four out of 157
(2.5%) patients who underwent radiotherapy following breast
conservative surgery developed COP (7). The use of tamoxifen, chemotherapy and
breast position were factors that were not associated with
developing COP. However, the eosinophil and neutrophil counts were
increased in the BAL fluid and the CD4/CD8 ratio was >2.0. In
the two present cases, the eosinophil count was elevated but the
CD4/CD8 ratio was <1.0. Toma et al previously reported
that there were increased mast cells in the BAL fluid of five
patients who had developed COP following radiotherapy (8). In addition, Roberts et al
reported that the CD4/CD8 ratio in the BAL fluid was elevated in
one patient with COP following radiotherapy (9), while Nagai et al found that a
decreased CD4/CD8 ratio was consistent with idiopathic COP
(10). Overall, these disparate
observations suggested that the CD4/CD8 ratio in the BAL fluid of
patients with COP associated with radiotherapy is uncertain, as
detailed subset examinations of lymphocytes in BAL fluid have not
been previously conducted.
Previously, Ogo et al conducted a
multi-institutional survey that found that 37 out of 2,056 (1.8%)
patients with breast cancer exhibited COP (11). However, the authors did not identify
any correlation between patient characteristics and the occurrence
of COP. Kubo et al reported that 12 out of 413 (2.9%)
patients exhibited COP (12). The
central lung distance (>1.8 cm) was the only significant risk
factor; notably, nodal status, location, field size, use of
chemotherapy and ipsilateral V20 were not risk factors. Katayama
et al reported that 16 out of 702 (2.3%) patients developed
COP following breast conservative surgery (13). In that study, multivariate and
univariate analyses showed that an age of >50 years and
endocrine therapy were risk factors for COP. The authors noted that
radiation-induced COP occurred >4 months following radiotherapy,
consistent with the cases of the present study. These results
suggested that the COP occurred in 2–3% of patients with tangential
breast irradiation.
Murai et al previously reported that nine out
of 189 patients developed COP following stereotactic radiotherapy
of the lung (14) and calculated
the COP incidence at 1 and 2 years to be 4.0 and 5.2%,
respectively. No association was found between COP and radiation
dose, but prior radiation pneumonitis was a marked risk factor for
COP. The authors suggested that following stereotactic
radiotherapy, patients must be carefully followed for ≥2 years,
particularly those with symptoms consistent with radiation
pneumonitis. To the best of our knowledge, COP following
conventional irradiation of the lung has not been reported
previously. In the current case, a consolidation shadow was
identified in the right lower lobes below the irradiated volume.
Previously, Oie et al observed that COP appeared near the
area affected by radiation pneumonitis (15).
Numerous studies have previoulsy reported that a
marked improvement may be achieved by the administration of
corticosteroids, but relapses often occur when the dose is tapered.
Katayama et al reported that two out of 16 patients required
corticosteroids for >3 years (13), while Narabayashi et al
described radiation-induced COP that was refractory to
corticosteroids (16). Stover et
al suggested that macrolides may be useful in these cases
(17). By contrast, Ogo et
al reported that clinical and radiology observations improved
in 50% of patients without corticosteroid treatment (11), as was demonstrated in the current
breast cancer case.
In conclusion, when an infiltrating shadow is
present outside of the irradiated field, COP must be included in
the differential diagnosis.
References
|
1
|
Lange W: A rare disease of small bronchi
and bronchioles. Deutsche Arch Klin Med. 70:342–364. 1901.
|
|
2
|
Epler GR, Colby TV, McLoud TC, Carrington
CB and Gaensler EA: Bronchiolitis obliterans organizing pneumonia.
N Engl J Med. 312:152–158. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
American Thoracic Society and European
Respiratory Society. American Thoracic Society/European Respiratory
Society International Multidisciplinary Consensus Classification of
the Idiopathic Interstitial Pneumonias. In: This joint statement of
the American Thoracic Society (ATS), and the European Respiratory
Society (ERS) was adopted by the ATS board of directors, June 2001
and by the ERS Executive Committee; June 2001; Am J Respir Crit
Care Med. 165. pp. 277–304. 2002, View Article : Google Scholar
|
|
4
|
Epler GR: Bronchiolitis obliterans
organizing pneumonia, 25 years: a variety of causes, but what are
the treatment options? Expert Rev Respir Med. 5:353–361. 2011.
|
|
5
|
Gibson PG, Bryant DH, Morgan GW, et al:
Radiation-induced lung injury: a hypersensitivity pneumonitis? Ann
Intern Med. 109:288–291. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Crestani B, Valeyre D, Roden S, Wallaert
B, Dalphin JC and Cordier JF: Bronchiolitis obliterans organizing
pneumonia syndrome primed by radiation therapy to the breast. The
Groupe d’Etudes et de Recherche sur les Maladies Orphelines
Pulmonaires (GERM’O’P). Am J Respir Crit Care Med. 158:1929–1935.
1998. View Article : Google Scholar
|
|
7
|
Takigawa N, Segawa Y, Saeki T, et al:
Bronchiolitis obliterans organizing pneumonia syndrome in
breast-conserving therapy for early breast cancer:
radiation-induced lung toxicity. Int J Radiat Oncol Biol Phys.
48:751–755. 2000. View Article : Google Scholar
|
|
8
|
Toma CL, Serbescu A, Alexe M, et al: The
bronchoalveolar lavage pattern in radiation pneumonitis secondary
to radiotherapy for breast cancer. Maedica (Buchar). 5:250–257.
2010.
|
|
9
|
Roberts CM, Foulcher E, Zaunders JJ,
Bryant DH, Freund J, Cairns D, et al: Radiation pneumonitis: a
possible lymphocyte-mediated hypersensitivity reaction. Ann Intern
Med. 118:696–700. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Nagai S, Handa T, Ito Y, Takeuchi M and
Izumi T: Bronchoalveolar lavage in idiopathic interstitial lung
diseases. Semin Respir Crit Care Med. 28:496–503. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ogo E, Komaki R, Fujimoto K, et al: A
survey of radiation-induced bronchiolitis obliterans organizing
pneumonia syndrome after breast-conserving therapy in Japan. Int J
Radiat Oncol Biol Phys. 71:123–131. 2008. View Article : Google Scholar
|
|
12
|
Kubo A, Osaki K, Kawanaka T, Furutani S,
Ikushima H and Nishitani H: Risk factors for radiation pneumonitis
caused by whole breast irradiation following breast-conserving
surgery. J Med Invest. 56:99–110. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Katayama N, Sato S, Katsui K, et al:
Analysis of factors associated with radiation-induced bronchiolitis
obliterans organizing pneumonia syndrome after breast-conserving
therapy. Int J Radiat Oncol Biol Phys. 73:1049–1054. 2009.
View Article : Google Scholar
|
|
14
|
Murai T, Shibamoto Y, Nishiyama T, et al:
Organizing pneumonia after stereotactic ablative radiotherapy of
the lung. Radiat Oncol. 7:1232012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Oie Y, Saito Y, Kato M, et al:
Relationship between radiation pneumonitis and organizing pneumonia
after radiotherapy for breast cancer. Radiat Oncol. 8:562013.
View Article : Google Scholar
|
|
16
|
Narabayashi M, Mitsumori M, Araki N, et
al: A case of metachronous bilateral breast cancer with bilateral
radiation pneumonitis after breast-conserving therapy. Breast
Cancer. 13:313–316. 2006. View Article : Google Scholar
|
|
17
|
Stover DE, Milite F and Zakowski M: A
newly recognized syndrome - radiation-related bronchiolitis
obliterans and organizing pneumonia. A case report and literature
review. Respiration. 68:540–544. 2001. View Article : Google Scholar
|