Introduction
Extracellular vesicles (EVs) are a heterogeneous
population of membrane vesicles released by almost all types of
cells. Increased secretion of EVs is observed in cases of
intensively proliferating tumor cells. EVs carry biologically
important components including proteins, nucleic acids, lipids and
glycans (1) located within EVs or
as corona biomolecules surrounding EVs (2). Colon cancer-derived EVs express
parental cancer-associated markers such as epithelial cell adhesion
molecule, glypican 1 and mucin 13 (3–5). After
secretion from cancer cells, tumor-derived EVs (TEVs) can be taken
up by neighboring cells or circulate freely within body fluids
(6). Interest in TEVs has developed
due to their impact on phenotype and function of acceptor/target
cells.
Effective transfer of TEVs to monocytes has been
described (7–9), however, the knowledge of the early
stages of these interactions is limited. In general, membrane
fusion and different types of endocytosis, which may occur
simultaneously but with different effectiveness, are essential for
uptake of TEVs (10). Monocytes and
macrophages preferentially use phagocytosis, a type of endocytosis
dedicated to the internalization of particles in a
receptor-dependent manner (11).
The major phagocytic receptors are Fc receptors (CD64, CD32 and
CD16), complement and scavenger receptors widely distributed on
immune cells (12). In 2006, Vachon
et al (13) showed that CD44
is a competent receptor that efficiently mediates internalization
of hyaluronan (HA)-coated beads. CD44 is the major receptor for HA,
a negatively-charged polysaccharide composed of glucuronic acid and
N-acetylglucosamine units. It is hypothesized that the CD44
molecule may be involved in the engulfment of small-size TEVs
(<200 nm) and that HA carried by TEVs may facilitate TEVs
interactions with CD44-expressing cells. All monocytes express CD44
molecules, with the predominance of short, standard form CD44s
encoded by exons 1–5 and 16–20 (14). Monocytes are a heterogeneous
population of cells differing in phenotype and functions (15,16).
In 2010, Ziegler-Heitbrock et al (15) proposed a nomenclature for monocyte
subpopulations based on expression of markers CD14 and CD16. The
major monocyte subset characterized by high expression of CD14
marker is classical monocytes. The minor monocyte subset with low
CD14 and high CD16 is non-classical monocytes. Cells in between
these two subpopulations are intermediate monocytes (15). Different subsets of monocytes serve
different roles during tumor progression; for example, following
migration from the bloodstream, classical monocytes mainly
differentiate into pro-tumoral tumor-associated macrophages (TAMs),
non-classical monocytes prevent metastasis formation (17) and the intermediate subset supports
angiogenesis (18). The knowledge
concerning CD44 expression in the aforementioned monocyte subsets
(19) is limited, however, it may
be important when considering their interactions with tumor cells
and TEVs.
It was previously shown that blocking monocytic CD44
molecules with anti-CD44 antibodies decreases the engulfment of
large-size (>200 nm) TEVs of pancreatic carcinoma origin
(20). The present study aimed to
investigate the role of CD44 in the internalization of small TEVs
of colon cancer origin. It is hypothesized that CD44 plays a role
in TEVs endocytosis by monocytes, varies between subsets of
monocytes and that the composition of TEVs impacts the rate of this
process.
Material and methods
Cell culture and isolation of
TEVs
TEVs were obtained from supernatant derived from the
culture of the colon cancer cell lines SW1116 and HCT116 (American
Type Culture Collection). Briefly, cells were cultured at 37°C,
without CO2 (SW1116) or with 5% CO2 (HCT116)
in L15 or McCoy's medium (cat. nos. 11415064 and 16600082; Gibco;
Thermo Fisher Scientific, Inc.), respectively, with 10% fetal
bovine serum (FBS; cat. no. S1860; Ultra-low Endotoxin; Biowest
USA). Cells were split twice per week. Bovine-derived EVs were
depleted from FBS by centrifugation at 100,000 × g for 4 h at 4°C
(Sorvall™ WX+ Ultracentrifuge with T-1270 rotor; Thermo
Fisher Scientific, Inc.). PBS used in TEVs isolation was filtered
(0.22 µm; Merck KGaA) and its purity was tested by nanoparticle
tracking analysis (NTA; NanoSight LM10HS equipped with the LM14 488
nm laser module; Malvern Instruments, Ltd.). Cell lines were tested
every month for Mycoplasma sp. contamination using a Mycoplasma PCR
Detection kit according to manufacturer's instructions (cat. no.
G238; Applied Biological Materials, Inc.). Supernatants from
well-grown (confluency >90%) cell cultures were collected and
spun at 500 × g for 5 min at room temperature (RT), and then at
3,200 × g for 12 min at 4°C to remove cell debris. The supernatants
were again centrifuged at 100,000 × g for 2 h at 4°C. Pellets were
washed in PBS to remove FBS and resuspended in filtered PBS.
Quantification of TEVs was performed by NTA and protein measurement
by the Bradford method (cat. no. 5000201; Bio-Rad Laboratories,
Inc.). TEVs were tested for endotoxin contamination by Limulus test
according to the manufacturer's instructions (cat. no. A39553;
Pierce™ Chromogenic Endotoxin Quant kit; Thermo Fisher
Scientific, Inc.), and stored at −80°C.
Isolation of monocytes
Anticoagulated citrate dextrose A-treated blood from
healthy donors was purchased from the Regional Center of Blood
Donation and Blood Therapy (Krakow, Poland; agreement no.
DZM/SAN/CM/U-678/2015; Bioethical Committee of the Jagiellonian
University, Kraków, Poland; approval no. 1072.6120.1.2020). Human
peripheral blood mononuclear cells were isolated from blood by
standard Ficoll/Isopaque (cat. no. 17-1440-03; GE Healthcare)
density gradient centrifugation (30 min, 800 × g, RT). Monocytes
were separated from mononuclear cells by counterflow centrifugal
elutriation with a JE-5.0 elutriation system equipped with a 5-ml
Sanderson separation chamber (Beckman Coulter, Inc.), as previously
described (21). Monocytes were
suspended in RPMI-1640 culture medium supplemented with L-glutamine
(cat. no. 11875093; Gibco; Thermo Fisher Scientific, Inc.) and
gentamicin (cat. no. P06-13021; 50 µl/ml; PAN-Biotech GmbH). The
purity of isolation was ≥95%, determined by staining with an
anti-CD14 monoclonal antibody (cat. no. 555399; mAb; clone no.
M5E2; BD Pharmingen; BD Biosciences). Monocytes were incubated with
anti-CD14 for 30 min at 4°C in the dark (antibody concentration
according to the manufacturer's protocol) and then washed with PBS,
collected by flow cytometry (BD FACSCanto™; BD
Biosciences) and analyzed by FACSDiva Software (version 8.0.1; BD
Biosciences).
Expression of CD44 on monocytes and
subpopulations
To determine the expression of surface markers on
monocytes by flow cytometry, the following mAbs were used: FITC
anti-human CD44s (cat. no. 347943; clone no. G44-26), APC
anti-human CD14 (cat. no. 555399; clone no. M5E2) and PE anti-human
CD16 (cat. no. 555407; clone no. 3G8) (BD Pharmingen; BD
Biosciences). Monocytes were incubated with anti-CD44, -CD14 and
-CD16 for 30 min at 4°C in the dark and then washed with PBS as
aforementioned. A total of 10,000 cells per run were analyzed on a
BD FACSCanto™ Flow Cytometer (BD Biosciences) using Diva
Software (version 8.0.1; BD Biosciences). The monocyte subsets
gating strategy is presented in Fig.
S1. CD44 is used throughout the manuscript in the sense of
CD44s, unless otherwise noted. CD44 expression was analyzed as mean
fluorescence intensity (MFI) because the percentage of cells that
exhibited fluorescence in every subpopulation was ~100%. CD44
expression on monocytes was evaluated after 2 and 18 h of culture
(control), as well as after incubation with TEVs (TEV:monocyte,
5,000:1) after the same times, at 37°C.
TEVs characterization
The size and concentration of TEVs were defined by
NTA. A suspension of 1,000 times diluted TEVs was loaded into the
measuring chamber. The movement of particles was recorded in
triplicates of 1-min videos (Fig.
S2), after which the concentration, average size and mode
values of TEVs were calculated. TEVs membrane structure was
confirmed by MEMGlow staining, as previously described (22). MEMGlow solution (cat. no. MG01-02;
20 µM; Cytoskeleton, Inc.) was diluted in filtered PBS to 0.1 µM
concentration, added to 40X diluted TEVs suspension and incubated
for 30 min at RT in the dark. It was later analyzed by flow
cytometry without washing.
EV markers were detected by western blotting (WB).
Briefly, protein concentration in EV samples was determined by the
Bradford method. Next, 20 µg/lane EV proteins extracted with
Mammalian Protein Extraction Reagent (cat. no. 78501; Thermo Fisher
Scientific, Inc.) were heated with loading buffer at 75°C for 10
min (cat. no. NP0007; 4X sample buffer; and cat. no. NP0004; 10X
sample reducing agent; Invitrogen, Thermo Fisher Scientific, Inc.).
Electrophoresis was performed at 180 V for 45 min on 14%
polyacrylamide gel. Proteins were transferred onto a polyvinylidene
fluoride membrane with semi-dry transfer at 25 V for 1 h, blocked
at RT for 1 h with 1% bovine serum albumin in Tris-buffered saline
with 0.1% Tween and incubated overnight at 4°C with rabbit anti-CD9
(cat. no. 13174S; clone no. D801A), anti-β-actin (cat. no. 8457S;
clone no. D6A8) and mouse anti-ALIX mAb (cat. no. 2171S; clone no.
3A9) (Cell Signaling Technology, Inc.) diluted 1,000 times. Next,
the membrane was incubated with secondary anti-rabbit (cat. no.
sc-2357; Santa Cruz Biotechnology, Inc.) or anti-mouse (cat. no.
31430; Invitrogen; Thermo Fisher Scientific Inc.) antibodies
conjugated with horseradish peroxidase and diluted 2,000 times for
1 h at RT. The protein bands were visualized with
SuperSignal™ West Pico PLUS Chemiluminescent Substrate
(cat. no. 34578; Thermo Fisher Scientific, Inc.) by ChemiDoc
Imaging System (Bio-Rad Laboratories, Inc.).
The expression of CD44 and CD44v6 on TEVs was
determined by flow cytometry with mAbs: FITC anti-human CD44 and PE
anti-human CD44v6 (cat. no. 566803; clone no. 2F10). TEVs were
incubated with CD44 and CD44v6 for 30 min at 4°C in the dark and
analyzed on a BD FACSCanto™ Flow Cytometer (BD
Biosciences).
TEVs staining
TEVs suspension was incubated with SYTO RNASelect
(cat. no. S32703; Thermo Fisher Scientific, Inc.) at 37°C for 20
min in the dark. Excess dye was removed with Exosome Spin Columns
(cat. no. 4484449; Invitrogen, Thermo Fisher Scientific, Inc.)
according to the manufacturer's protocol. The effectiveness of
labeling was determined by flow cytometry (FACS Canto II; BD
Biosciences) in the FL1 channel (Fig.
S3). SYTO RNASelect-labeled TEVs were termed SYTO RNA-labeled
TEVs.
HA content measurement
HA concentration in cell culture supernatants, TEVs
and culture supernatants after TEVs isolation was measured by
Quantikine ELISA Hyaluronan kit (cat. no. DHYAL0; R&D Systems,
Inc.) according to the manufacturer's protocol. Briefly, samples
were diluted 20–50-fold, depending on the HA content. The optical
density was determined with ELX800NB, Universal Microplate Reader
(450 nm; BioTek Instruments, Inc.). The concentration of HA was
calculated by linear standard curve. The minimum detectable dose of
HA was 0.068 ng/ml.
Transfer of TEVs to monocytes and
monocyte subsets
SYTO RNA-labeled TEVs were incubated with monocytes
at a ratio of 1,000:1; 5,000:1 and 10,000:1 for 30 min, 2 h and
overnight at 37°C. Following incubation, cells were washed and
analyzed by flow cytometry. The binding of SYTO RNA-labeled TEVs to
monocytes was determined by analysis of green fluorescence
intensity and percentage of positive cells. Vital dye trypan blue
was used for quenching extracellular fluorescence, as previously
described (23). Briefly, 100 µl
cell suspension was mixed with trypan blue solution (ratio, 1:1;
final concentration 0.25 mg/ml), and reanalyzed by flow cytometry
after 5 min. The fluorescence of monocytes incubated with SYTO
RNA-labeled TEVs was compared with control monocytes that were not
incubated with TEVs. The autofluorescence of TEVs was
negligible.
Flow cytometry analysis of TEVs
transfer to monocyte subsets
Monocytes were stained with anti-CD14 and anti-CD16
antibodies, washed and incubated with SYTO RNA-labeled TEVs
(TEV:monocyte, 5,000:1) for 15 min, 30 min or 2 h at 37°C. Excess
TEVs were washed and cells were assessed by flow cytometry by
analyzing the shift in green fluorescence intensity. Due to
antibody labeling, the extracellular fluorescence quenching with
trypan blue was not performed. MFI and the percentage of positive
cells in the FL1 channel were determined for each gated
subpopulation. To determine engulfment of TEVs, monocytes stained
with mAbs were separated using FACS Aria II (BD Biosciences) into
three populations: i) classical
(CD14++/CD16−); ii) non-classical
(CD14+/CD16++) and iii) intermediate
(CD14++/CD16+). Sorted cells were collected
into polystyrene tubes and incubated with SYTO RNA-labeled TEVs
(TEV:monocyte, 5,000:1). The experiments were performed only with
HA-rich TEVsHCT116 and limited to the shorter contact
time of either 15 min or 1 h due to the small number of sorted
non-classical and intermediate monocytes. The engulfment of TEVs
was analyzed by quenching extracellular fluorescence with trypan
blue; data are presented as a percentage of fluorescence-positive
cells containing TEVs.
Blocking of CD44
The role of CD44 in TEVs endocytosis was
investigated by blocking this receptor on monocytes. A total of
2×106/ml monocytes were incubated with anti-CD44 mAb
(cat. no. BMS113; clone no. SFF-2; 10 µg/ml; Invitrogen; Thermo
Fisher Scientific, Inc) or appropriate IgG1 isotype control (cat.
no. 14-4714-82; 10 µg/ml; Invitrogen; Thermo Fisher Scientific,
Inc.) for 30 min at 4°C. Monocytes were washed with RPMI, and
incubated with SYTO RNA-labeled TEVs (TEV:monocyte, 5,000:1) for 30
min to 18 h at 37°C. The binding of SYTO RNA-labeled TEVs was
determined by flow cytometry. To distinguish surface-bound and
internalized TEVs, the quenching of extracellular fluorescence
signals by trypan blue was used. MFI value of control monocytes
(without TEVs) was subtracted from the MFI of monocytes incubated
with SYTO RNA-labeled TEVs (elimination of autofluorescence
effect), then the MFI of monocytes incubated with SYTO RNA-labeled
TEVs without blocking CD44 was established as 100% and relative to
this value, the percentage change of MFI (%MFI) for monocytes with
blocked CD44 or with isotype control was calculated.
In the case of monocyte subsets, binding of TEVs to
monocytes for 15 min or 1 h was determined by the percentage of
fluorescence-positive cells (FL1 channel) for each gated
subpopulation. The engulfment of TEVs was analyzed in sorted
subsets after trypan blue treatment and presented as the percentage
of fluorescence-positive cells.
Statistical analysis
Statistical analysis was performed by Statistica v.
13.3 (TIBCO Software Inc.). Mann-Whitney U, Wilcoxon signed rank,
Student's t-test or Welch's test were used. For multiple group
comparisons, one-way ANOVA followed by Tukey's post hoc test was
used. Graphs were constructed in GraphPad Prism (version 8.0.1;
Dotmatics). Detailed information about the statistical tests and
the number of experiments is included in the respective figure
legends. P<0.05 was considered to indicate a statistically
significant difference.
Results
TEVs derived from colon cancer cell
lines carry HA
TEVsHCT116 and TEVsSW1116 were
isolated from cell culture supernatants by ultracentrifugation. The
size distribution of TEVs was similar between TEVsHCT116
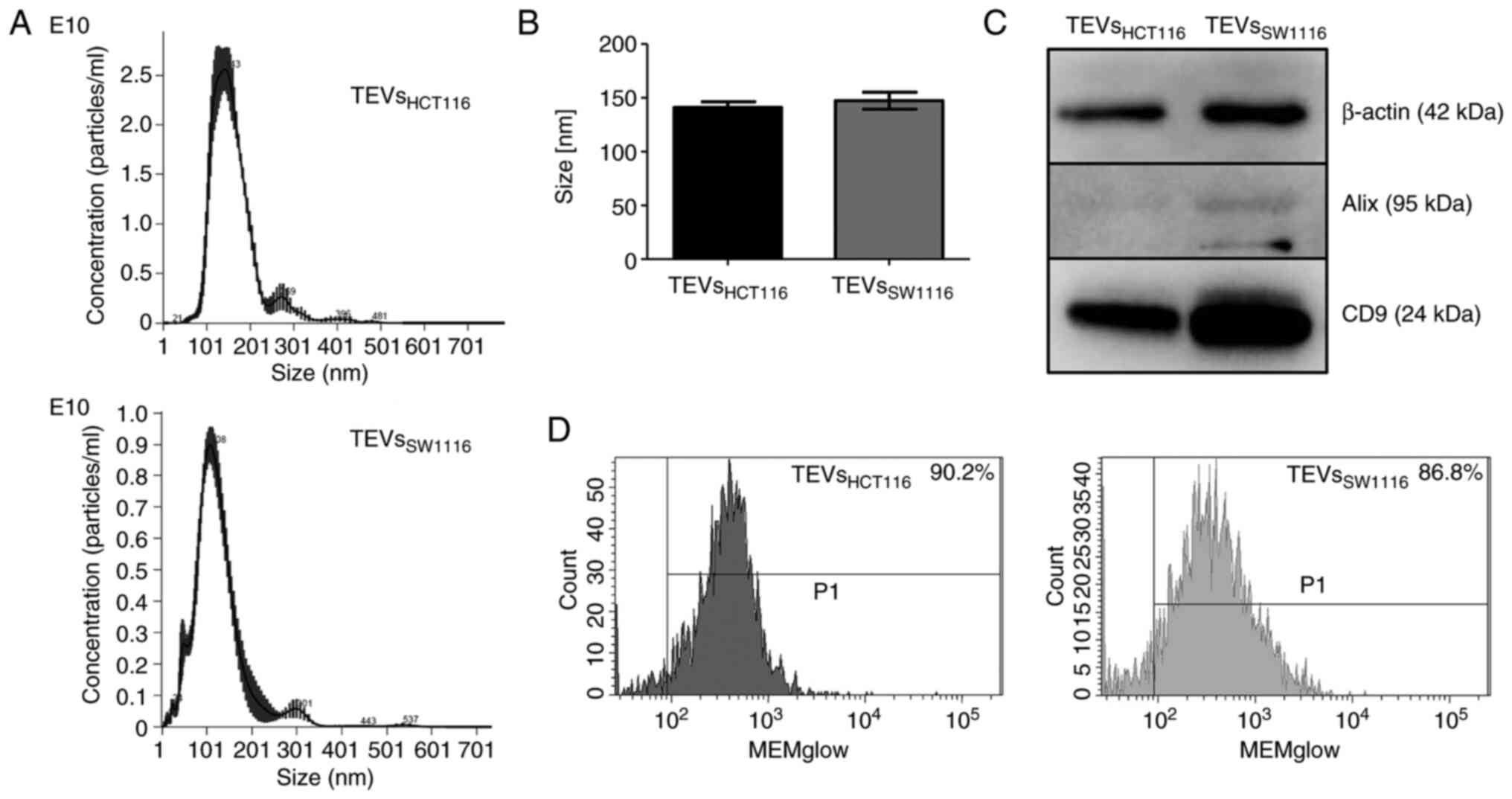
and TEVsSW1116 (Fig. 1A and
B). Both tested TEVs expressed CD9 and β-actin as shown by WB.
The expression of Alix was limited to TEVsSW1116
(Fig. 1C). The MEMglow-positive
fraction represented ~90% of both tested TEVs (Fig. 1D). The TEVs derived from HCT116
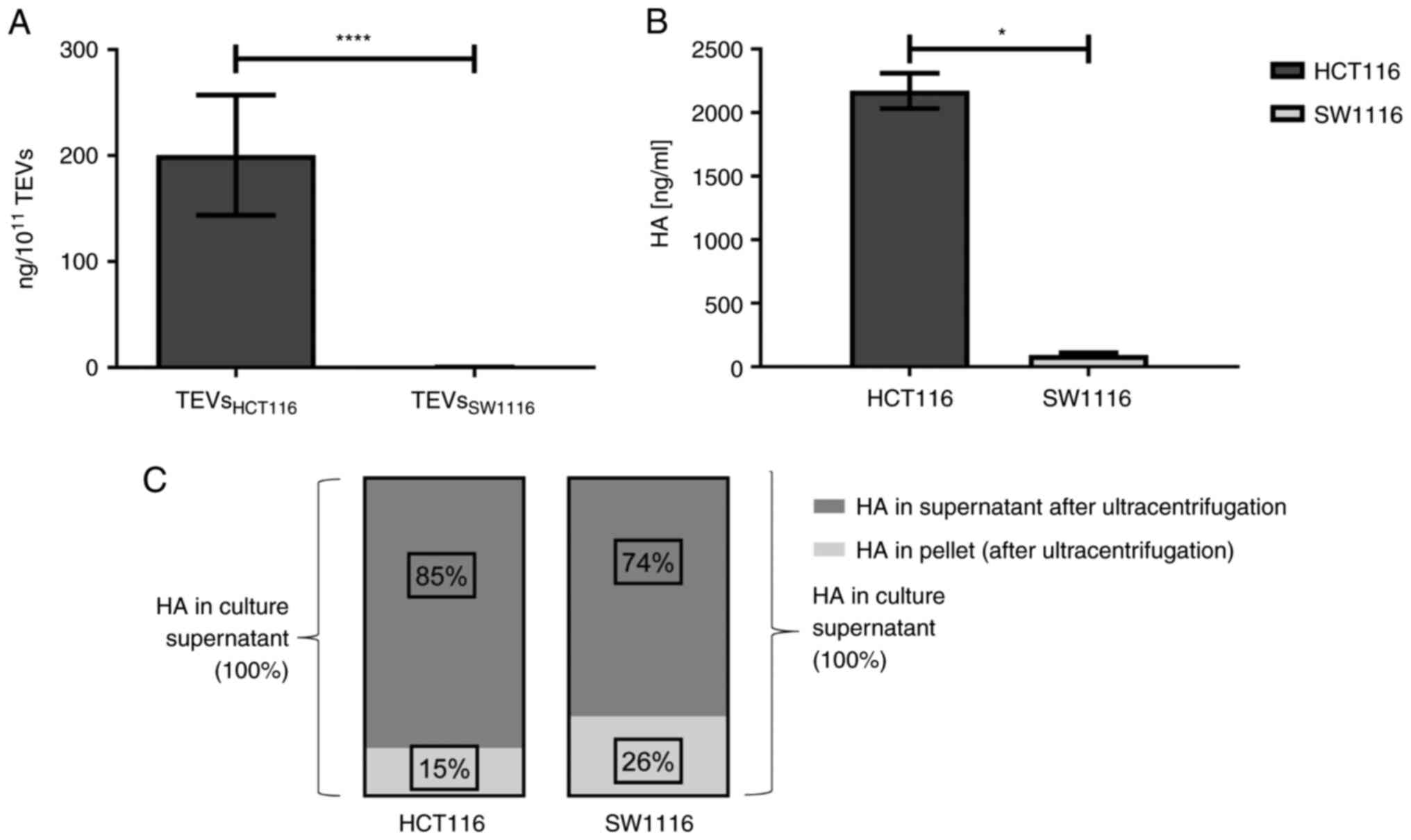
cells were significantly enriched in HA compared with those derived
from SW1116 cells (200.43 vs. 0.26 ng/1×1011 TEVs; n=13;
Fig. 2A) which was associated with
HA content in cell culture supernatants (Fig. 2B; n=4). The comparison of HA levels
in cultured supernatants before and after TEVs isolation indicated
that HA was primarily present in soluble form. HA detected in TEVs
represented only 15 and 26% of the total HA in the culture
supernatants of HCT116 and SW1116 cells, respectively (Fig. 2C; n=2). There were at least two
forms of CD44 (CD44s and CD44v6) present on TEVsHCT116
(~42 and ~58%, respectively) and TEVsSW1116 (~32 and
~44%, respectively) (data not shown).
Monocyte subsets differ in CD44
expression
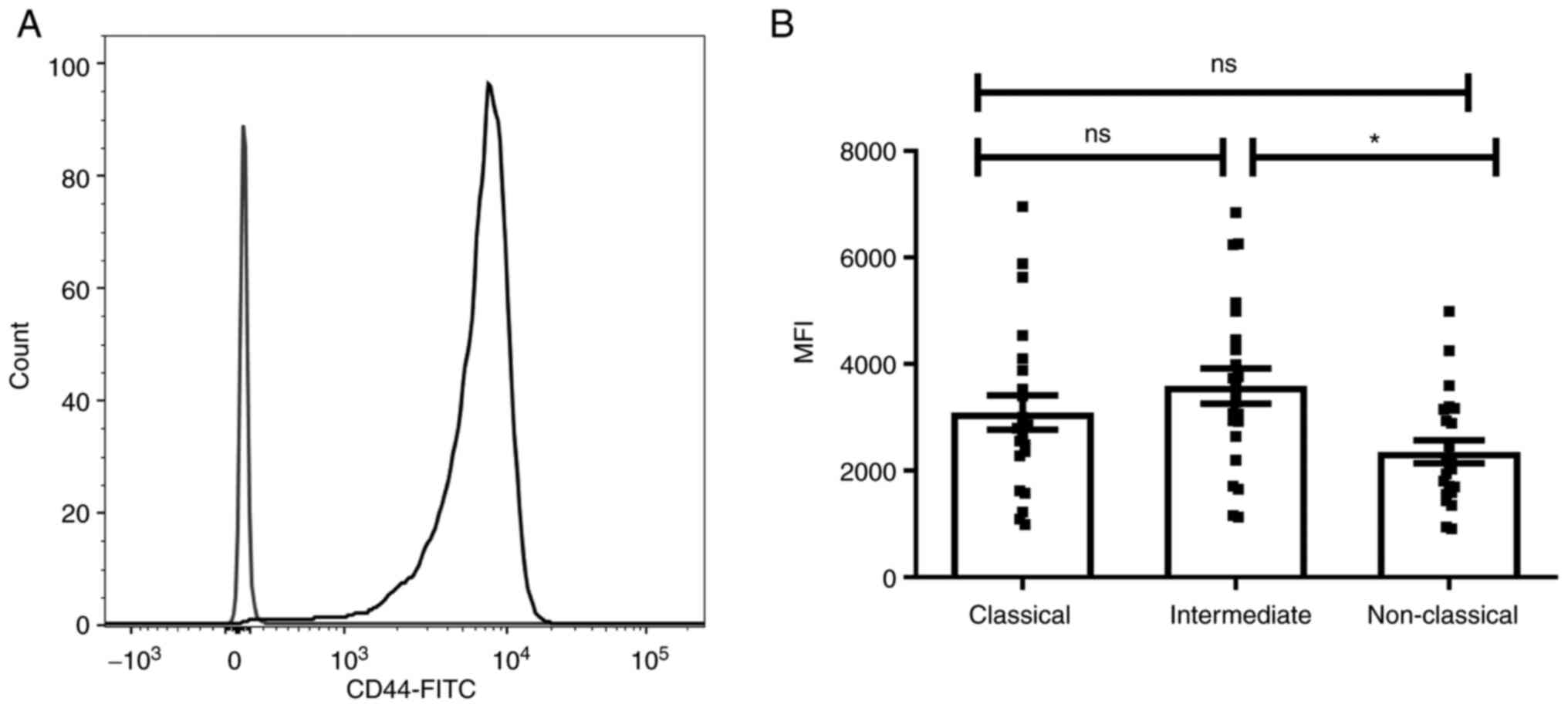
To investigate the role of CD44 in interaction of
monocytes with TEVs, expression of CD44 on monocytes and their
subsets was determined by flow cytometry (Fig. 3A). The three monocyte subsets,
classical CD14++/CD16−, intermediate
CD14++/CD16+ and non-classical
CD14+/CD16++, were determined based on
expression of the CD14 and CD16 markers, as described by
Ziegler-Heitbrock (16). Since all
monocytes are CD44+, to assess differences in CD44
expression between subsets, MFI was evaluated in comparison to the
appropriate isotype control (Fig.
3B). The highest MFI of CD44 was observed in the intermediate
subpopulation and only the difference between intermediate and
non-classical monocytes was statistically significant (n=23). The
expression of CD44 on monocytes was not affected by incubation with
TEVs (data not shown) for 2 and 18 h, however, an increase in CD44
MFI was observed after 2 and 18 h of culture (Fig. S4).
Attachment and engulfment of TEVs
depend on their availability, time of exposition and TEVs
origin
TEVs were labeled with green fluorescent SYTO RNA
Select dye. The labeling did not affect the size distribution and
concentration of TEVs (Fig. S2).
The efficiency of TEVs labeling was high and controlled by flow
cytometry (Fig. S2). TEVs were
incubated with monocytes for 30 min and 2 and 18 h, and the
attachment of TEVs to monocytes was analyzed by flow cytometry.
More than 90% of monocytes was fluorescence-positive after 30 min
incubation with TEVs from both cell lines, thus analysis of MFI
shift was considered more informative than percentage of positive
cells. The increase in monocyte MFI was associated with the number
of TEVs/monocyte and exposition time (Fig. S5; n=3). A TEV:monocyte ratio of
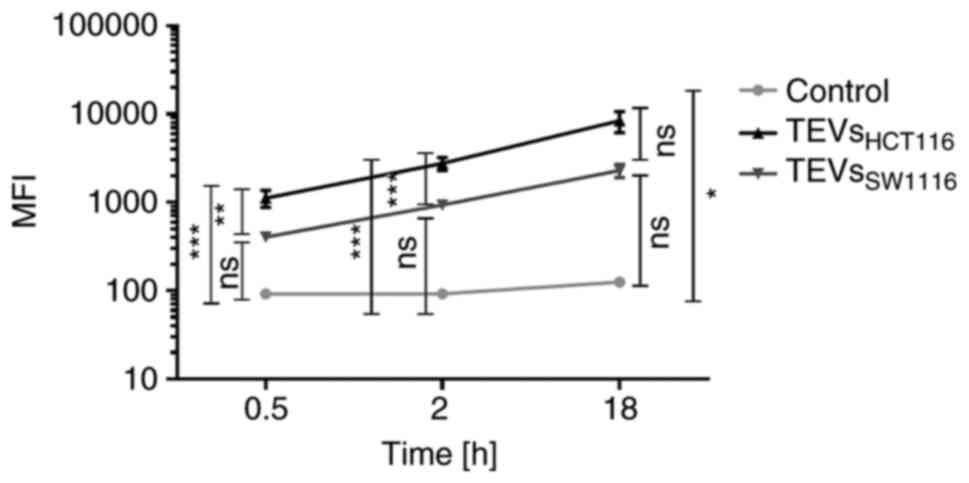
5,000:1 was chosen for further experiments. The kinetics of TEVs
attachment to monocytes are shown in Fig. 4. MFI of monocytes interacting with
TEVsHCT116 increased in a shorter time compared with
TEVsSW1116. This observation may indicate higher,
statistically significant efficiency of attachment of TEVs derived
from HCT116 cells compared with that of SW1116 cells (n=10;
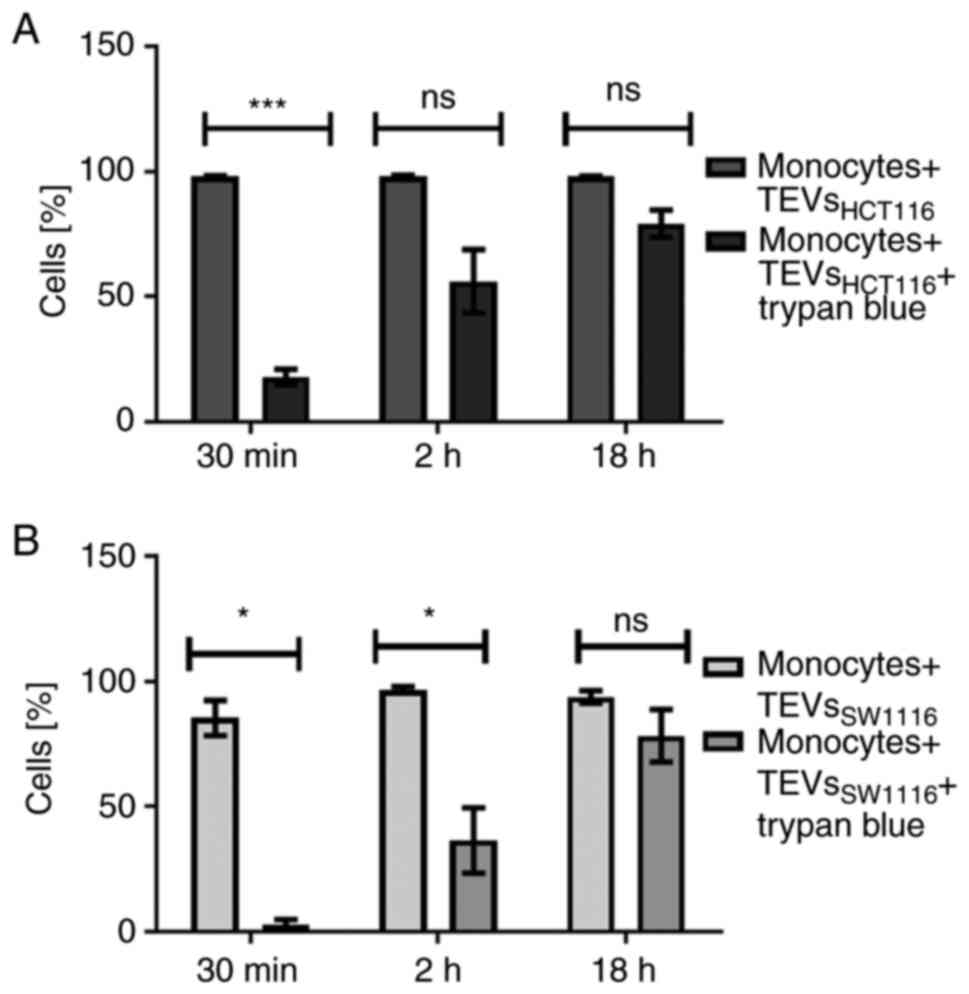
P<0.001). The percentage of monocytes showing fluorescence after
30 min incubation with TEVsHCT116 was 5 times reduced
after quenching with trypan blue (Fig.
5A). This suggested that approximately 20% of monocytes
exhibited TEVs in the cytoplasmic area after that time. After 2 h
incubation, ≥50% of monocytes engulfed TEVs. The internalization of
TEVsSW116 was not effective, as the uptake of TEVs after
30 min was ~3%. After 2 h, ~37% of monocytes contained
TEVsSW1116 (Fig. 5B).
The internalization outcome/level of TEVsHCT116 and
TEVsSW1116 after overnight incubation was comparable; in
~80% of monocytes, TEVs were located inside (Fig. 5; n=6).
Blocking of CD44 partially inhibits
HA-rich TEVs engulfment by monocytes
TEVs carry HA and are effectively engulfed by
monocytes, which is associated with high expression of major HA
receptor CD44. To evaluate the role of CD44 in engulfment, control
monocytes and monocytes with blocked CD44 molecules were incubated
with SYTO RNA-labeled TEVs. MFI of green fluorescence channel was
used for quantification of TEVs attachment and engulfment following
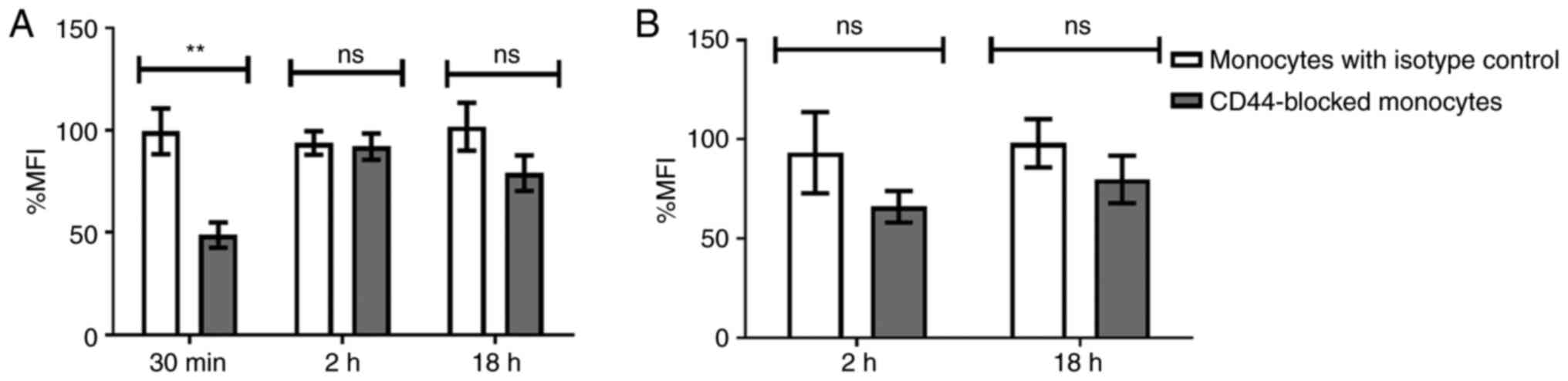
trypan blue treatment. The attachment of TEVs decreased following
CD44 blocking, however this was not significant (not shown). In
turn, blocking of CD44 molecules on monocytes reduced
TEVsHCT116 engulfment by 50.75% compared with that of
the isotype control (P=0.00739) after 30 min incubation with TEVs;
after 2 and 18 h, the reduction was 1.8 and 22.75%, respectively,
but this was not significant (Fig.
6A). For TEVsSW1116, the CD44 blocking with the
short time of incubation (30 min) was skipped due to the lack of
engulfment; almost all fluorescence was derived from outside of
monocytes (Fig. 5B). Blocking of
CD44 molecules, followed by 2 or 18 h incubation with TEVs resulted
in ~27 and ~6% reduction in the % MFI, respectively, compared with
the isotype control. However, the differences were not
statistically significant (Fig.
6B). Isotype control did not significantly diminish TEVs
engulfment compared with control monocytes (n=4).
Differences in TEVs
attachment/engulfment by monocyte subsets: The role of CD44
To determine whether differences in CD44 expression
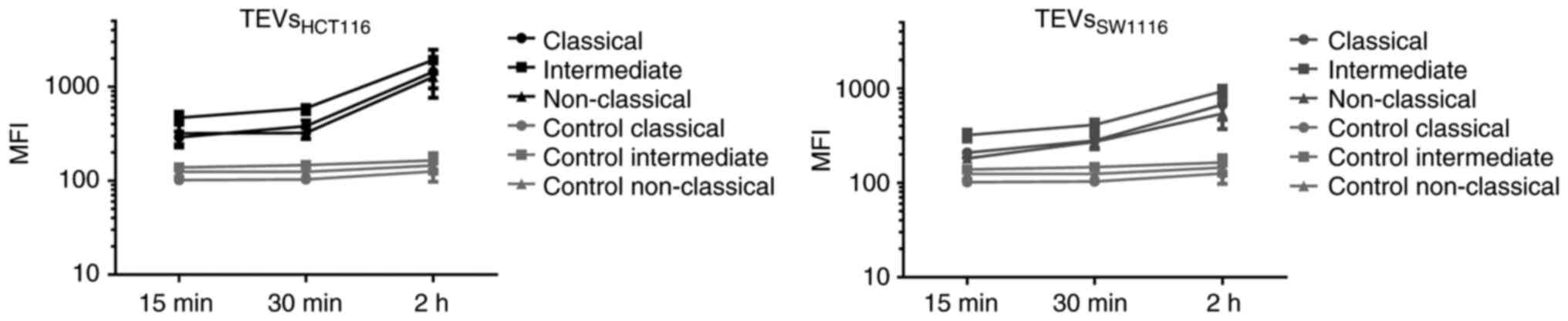
on monocyte subsets affect their interactions with TEVs, MFI value
of subsets was analyzed after incubation with SYTO RNA-labeled
TEVs. The highest increase in MFI was observed in the intermediate
subset of monocytes, which may suggest that TEVs preferentially
adhere to them (Fig. 7; n=5). This
observation was similar for both types of TEVs; however, MFI after
incubation with TEVsSW1116 was lower (Fig. 7), which in turn, corroborates
results obtained in previous experiments (Figs. 4 and S5). Next, the percentage of monocytes in
subsets that showed green fluorescence after incubation with SYTO
RNA-labeled TEVs was analyzed. Following 15 min incubation with
TEVsHCT116, ~46% of classical, ~29% of intermediate and
~20% of non-classical monocytes showed green fluorescence (Fig. 8A). In the case of
TEVsSW1116, the percentages of monocytes showing green
fluorescence were 7.7, 12.5 and 7.5% for classical, intermediate
and non-classical monocytes, respectively (Fig. 8B). The percentages of monocytes
showing green fluorescence increased over time (n=3). Blocking of
CD44 on monocytes resulted in a decrease in TEVsHCT116,
but not TEVsSW1116 binding in all three subsets by 10.1,
9.94 and 8.63% for classical, intermediate and non-classical
monocytes, respectively, however, the differences were not
significant and observed only after 15 min of exposure to TEVs
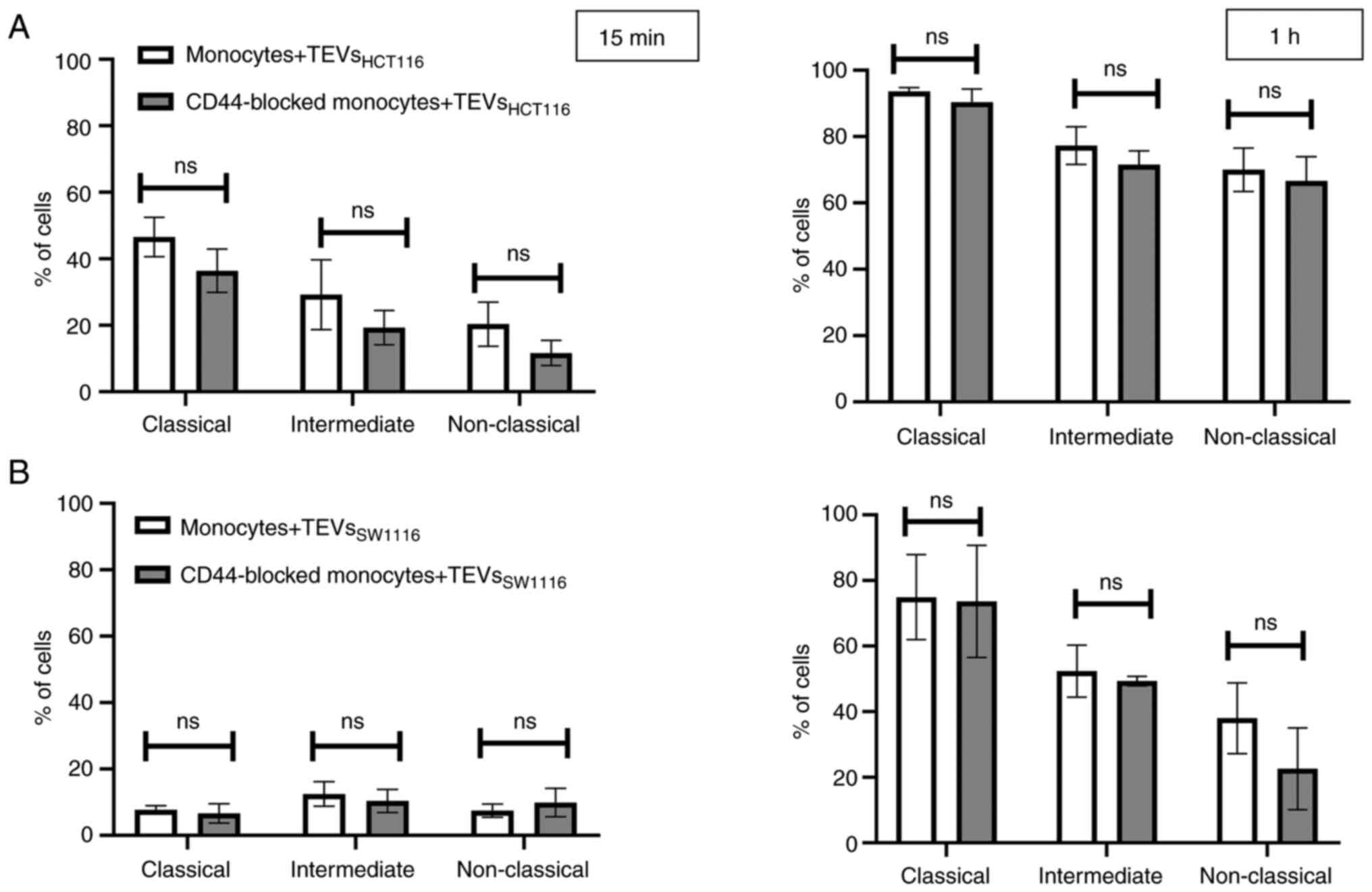
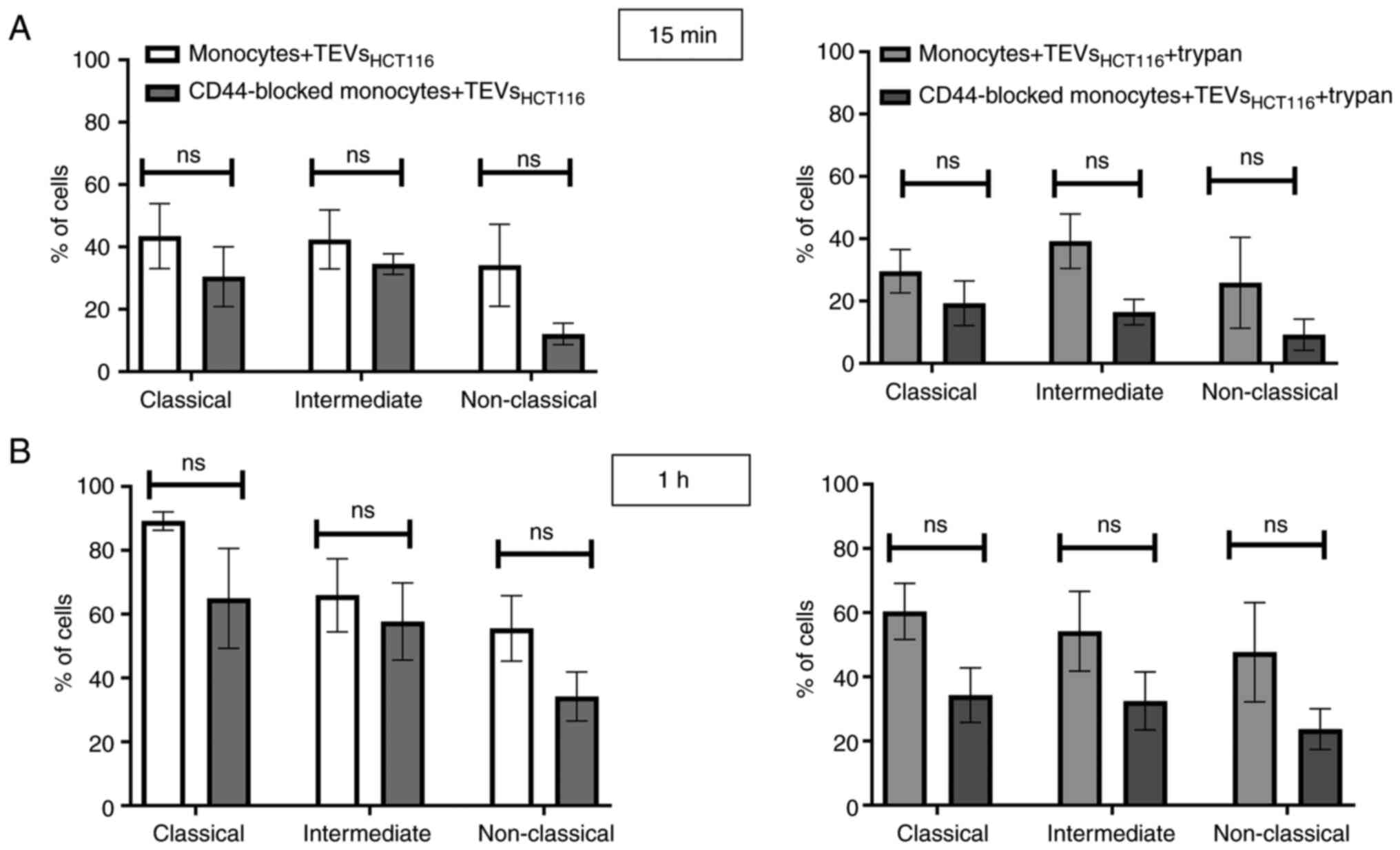
(Fig. 8). To exclude competition
between subsets of monocytes in TEVs attachment/engulfment,
experiments were repeated on sorted cells. TEVs were attached and
internalized by all three subsets of monocytes. After 15 min
contact, the differences in TEVs attachment were slight (Fig. 9A), with a non-significant
predominance of intermediate monocytes. Over time, higher binding
of TEVs was observed in classical monocytes compared with that in
other subsets (91 vs. 60% for classical vs. intermediate monocytes
and 91 vs. 54% for classical vs. non-classical monocytes (Fig. 9B). The highest engulfment of TEVs
was observed in intermediate monocytes after 15 min of incubation
(Fig. 9A); the percentage of
positive intermediate cells was similar before and after trypan
treatment which may suggest that TEVs were effectively internalized
(47 vs. 43%, respectively). Blocking of CD44 resulted in a slight
reduction in binding of TEVs to classical and intermediate
monocytes and stronger to non-classical monocytes (decreased by 8,
13 and 27%, respectively). The internalization of TEVs was
decreased following CD44 blocking; the effect was observed for all
three subsets after 15 min and 1 h of contact, however this was not
significant. The percentage of fluorescence-positive classical and
intermediate monocytes with blocked CD44 receptors was reduced by
~42% after quenching of extracellular fluorescence. In
non-classical monocytes, the decrease in the percentage of
fluorescence-positive cells was ~13%.
Discussion
The present study was designed to evaluate the role
of CD44 in the interactions of monocytes with colon cancer-derived
EVs. TEVs released by the two colon cancer cell lines HCT116 and
SW1116 were characterized. No differences in size, mode and range
between TEVsHCT116 and TEVsSW1116 were
observed; these corresponded to small EVs (24). The selective expression of EV
markers suggests their mixed origin from outer and inner cell
membranes (25–27). In the present study, the
investigated cell lines differed in HA secretion: HCT116 cells
produced more HA than SW1116 cells. Colorectal cancer cells release
HA primarily in a soluble form and also in a form bound with TEVs.
A major part of the sensitive cargo of TEVs is located inside and
covered by membrane sheaths; however, some of the cargo may be
attached to the surface. Recently, Buzas (2) and Tóth et al (28) described proteins and proteoglycans
that ‘decorate’ the surface of EVs and named them ‘corona’. The
present study extends this observation on HA, a component of the
extracellular matrix, that structurally belongs to polysaccharides.
HA content in TEVs was demonstrated using ELISA and it was
consistent with previous results obtained by using atomic force
microscopy combined with spectroscopy (29). Paul et al (29) showed that TEVsHCT116
exhibits notably increased HA surface density compared with the EVs
derived from normal colon epithelium.
Elevated levels of low molecular weight HA are
detected in the serum of patients with cancer, including colon
cancer (30–32). According to the presented data, at
least part of HA is linked with TEVs and may impact target cells
via interaction with CD44. CD44 is expressed at high levels in
cancer, as well as immune cells. HA supports tumor progression by
promoting tumor proliferation or influencing anti-apoptotic
activity (33), which is also
attributed to TEVs (34). Moreover,
HA promotes monocyte recruitment and redifferentiation from M1 into
M2 macrophages (35), which is
consistent with M2 polarization of macrophages observed after
contact with TEVs (36). HA carried
by TEVs may trigger changes in cell activity. Therefore, HA-rich
nanoparticles may be used in targeted antitumor therapy (37), which may also impact the functions
of other CD44-positive cells such as monocytes/macrophages. It was
previously shown that HA-decorated liposomes improve cellular
uptake and markedly inhibit proliferation of pancreatic cancer stem
cells. Also, encapsulated microRNA-125b in HA-poly
(ethylenimine)-based nanoparticles repolarizes TAMs from M2 to M1
in lung cancer (38).
In colon adenocarcinoma cells, HA is associated with
cell surface receptors, primarily CD44 and CD44v (39). Expression of CD44 and CD44v6 on
TEVsHCT116 and TEVsSW1116 was similar to that
of EVs of other origins (data not shown) (37). TEVs from both cell lines were able
to adhere to monocytes and be engulfed in a time- and
dose-dependent manner. The number of TEVs/monocytes used was
determined based on preliminary experiments. It was below the
saturation point, however, it fit the reported average number of
EVs and TEVs in plasma, the average number of monocytes in the
blood and the limit of detection of the flow cytometer (40,41).
Both processes of attachment and engulfment have
proceeded faster and at an efficient level, as indicated by MFI
value for HA-rich TEVsHCT116, compared with HA-poor
TEVsSW1116. This observation was consistent with
previous reports that the uptake of TEVs depends on their cargo.
For example, expression of mucin-1 facilitates binding to dendritic
cell-specific intercellular adhesion molecule-3-grabbing
non-integrin (CD209) receptors on monocyte-derived dendritic cells
(42) and fibronectin carried by
EVs enables interaction with target cells via heparan sulfate
chains (43). Previously, the
spontaneous rate of EV internalization was estimated as 1% at 1 h
in HeLa cells (44). The more
intensive uptake of TEVs by monocytes results from high phagocytic
potential and clearance capacity of pathogens and debris of
cellular or other origins. TEVs are cellular derivatives, however,
the mechanism of their uptake is still unclear. After 30 min
incubation, ~20% of monocytes exhibited TEVsHCT116 but
not TEVsSW1116 in the cytoplasmic area. The fast
interaction corroborates the half-time of exosomes circulating in
the blood (≤30 min) (45). The
preferential engulfment of HA-rich TEVs directed our attention to
CD44 the major HA receptor able to bind HA on immune cells
(46–49). More than 90% of monocytes express
the standard CD44 isoform (50).
Because of the prevalence of CD44 and the absence of CD44 variant
isoforms on non-activated cells, the present study was limited to
this isoform. CD44 expression on monocytes was not affected by
contact with TEVs (data not shown) for 2 or 18 h but increased over
the culture time. Blocking of CD44 on monocytes diminished the
attachment of TEVs to the cell surface and significantly decreased
TEVsHCT116 endocytosis after a short time of incubation.
Previous studies suggested that HA-CD44 interaction promotes
endocytosis of HA in various cell lines such as chondrocytes,
keratinocytes and cancer cells (51). However, in macrophages derived from
THP-1 cells, Rios de la Rosa et al (52) showed that the binding but not the
engulfment of HA or HA-linked nanoparticles was positively
associated with the expression of CD44. The differences may be due
to different types of HA carriers (artificial nanoparticles vs.
TEVs), the specification of cells (primary vs. differentiated from
THP-1) or different dynamics of HA-CD44 complex internalization. In
the present study, blocking CD44 decreased internalization of
HA-rich TEVs but only in the first 30 min after contact. The lack
of effect after longer contact may be due to experimental
conditions such as constant concentration of blocking anti-CD44
antibody or the aforementioned increase in CD44 expression on
monocytes during culture, such as caused via enhanced expression or
turnover (53,54). The role of other types of
endocytosis including phagocytosis, dynamin-dependent endocytosis
and pinocytosis, which replace the CD44-dependent mechanism,
increases over time (55–57). In TEVsSW1116, blocking of
CD44 resulted in reduced engulfment after 2 h; however, this was
not statistically significant. The kinetics of the
monocyte/TEVsSW1116 interaction is similar to previously
presented interactions with TEVsHPC derived from the
pancreatic carcinoma cell line HPC-4 (20). In both TEVsSW1116 and
TEVsHPC, the concentration of HA is notably lower
compared with that in TEVsHCT116, which results in
slower internalization by monocytes (58). HA concentration as well as size of
TEVs, cellular origin of HA (pancreatic vs. colon), affinity to
CD44 and availability of HA (thickness of corona, presence of HA in
soluble form) may impact the interactions between TEVs and target
cells. The observed interaction may be important clinically as the
increasing concentration of HA in the blood is considered a marker
of different types of tumors, including colon cancer (30). HA-rich TEVs may be internalized in
preponderance and affect polarization/functional activity of
monocytes and macrophages. The present data are limited to TEVs in
an in vitro model, whereas, in the blood, monocytes are
exposed also to soluble HA, which is internalized faster than
HA-coated nanoparticles (52) and
may inhibit endocytosis of TEVs (59).
As monocytes are a heterogeneous population of
cells, the adherence of SYTO RNA-labeled TEVs to monocyte subsets
was determined. Within 1 h, almost all classical monocytes showed
green fluorescence, suggesting that both types of TEVs were
attached or internalized by them. Non-classical monocytes, which
are poor phagocytes, interact with TEVs less effectively,
especially in the case of TEVsSW1116. TEVs were attached
by an intermediate monocyte subset, whose endocytic potential was
previously described (60,61). This was verified by sorted monocyte
subsets and highlighted the competition between cells that may take
place in the blood; classical monocytes are the most abundant
subset with great phagocytic potential (60). By using sorted cells, competition
between subsets of monocytes incubated with TEVs was avoided as
subsets were separately incubated with TEVs. The attachment of
HA-rich TEVs in subsets was comparable with a slight predominance
of intermediate cells, confirming the aforementioned MFI shift
after contact with SYTO RNA-labeled TEVs. Also, the highest
efficiency of TEVs engulfment was observed in intermediate cells,
where the smallest difference in the percentage of fluorescent
cells was observed before and after trypan blue treatment after
short contact with HA-rich TEVs.
Blocking of CD44 on monocytes decreased TEVs
attachment in all subsets, however this was not significant. This
trend was observed after a short time of contact with TEVs of 15
min, and faded over 1 h when the total population of monocytes was
studied. The decrease in the attachment of TEVs to non-classical
monocytes after CD44 blocking was observed only on sorted cells and
may have resulted from suboptimal conditions of experiments
(suboptimal concentration of blocking Ab). It is possible the
concentration of anti-CD44 mAbs, which was optimized for the whole
population of monocytes, was too high for non-classical cells (low
CD44 expression) and suboptimal for classical and intermediate
cells (high CD44 expression). Another explanation may be the
different kinetics of CD44 turnover in different cells/monocytes.
The endocytosis of TEVs was decreased after CD44 blocking in all
subsets and associated with CD44 expression (stronger in
intermediate and classical cells). It may indicate that the CD44
mechanism is more important for these subtypes of monocytes at the
early stages of contact with TEVs and less notable over time,
especially in classical monocytes, where it may be rapidly replaced
by other types of endocytosis.
To conclude, TEVs derived from different colon
cancer cell lines vary in amount of carried HA, a new ‘corona’
component. The presence of HA on TEVs and other EVs might
facilitate contact with target cells and trigger intracellular
signaling pathways. The findings of the present study suggested
that CD44 is a surface receptor important for targeting and
capturing HA-rich TEVs.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by the National Science Centre,
Poland (grant no. 2019/33/B/NZ5/00647).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author upon reasonable
request.
Authors' contributions
MBK conceptualized the study. AB and MBK performed
the experiments (acquisition of data), collected and analyzed data
and wrote the manuscript. MS participated in the analysis and
interpretation of data. AGB isolated monocytes (acquisition of
data). KW performed the flow cytometry (acquisition of data). ABM,
MS and KW were involved in drafting the manuscript. MBK supervised
the study. MBK and MS revised the manuscript critically for
important intellectual content. MBK acquired funding. MBK and AB
confirm the authenticity of all the raw data. All authors have read
and approved the final manuscript.
Ethics approval and consent to
participate
Blood from healthy donors was purchased from the
Regional Center of Blood Donation and Blood Therapy (Krakow,
Poland; agreement no. DZM/SAN/CM/U-678/2015; Bioethical Committee
of the Jagiellonian University, Kraków, Poland; approval no.
1072.6120.1.2020).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ping JYX, Neupane YR and Pastorin G:
Extracellular Vesicles and Their Interplay with Biological
Membranes. Manash KP: Extracellular Vesicles Role in Diseases,
Pathogenesis and Therapy. IntechOpen Series Physiology. Volume 13.
IntechOpen; London, UK: pp. 27–59. 2021
|
|
2
|
Buzas EI: Opportunities and challenges in
studying the extracellular vesicle corona. Nat Cell Biol.
24:1322–1325. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen Y, Xie Y, Xu L, Zhan S, Xiao Y, Gao
Y, Wu B and Ge W: Protein content and functional characteristics of
serum-purified exosomes from patients with colorectal cancer
revealed by quantitative proteomics. Int J Cancer. 140:900–913.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Glass SE and Coffey RJ: Recent advances in
the study of extracellular vesicles in colorectal cancer.
Gastroenterology. 163:1188–1197. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Xiao Y, Zhong J, Zhong B, Huang J, Jiang
L, Jiang Y, Yuan J, Sun J, Dai L, Yang C, et al: Exosomes as
potential sources of biomarkers in colorectal cancer. Cancer Lett.
476:13–22. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Russell AE, Sneider A, Witwer KW, Bergese
P, Bhattacharyya SN, Cocks A, Cocucci E, Erdbrügger U, Falcon-Perez
JM, Freeman DW, et al: Biological membranes in EV biogenesis,
stability, uptake, and cargo transfer: An ISEV position paper
arising from the ISEV membranes and EVs workshop. J Extracell
vesicles. 8:16848622019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Czernek L, Chworos A and Duechler M: The
uptake of extracellular vesicles is affected by the differentiation
status of myeloid cells. Scand J Immunol. 82:506–514. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Popēna I, Ābols A, Saulīte L, Pleiko K,
Zandberga E, Jēkabsons K, Endzeliņš E, Llorente A, Linē A and
Riekstiņa U: Effect of colorectal cancer-derived extracellular
vesicles on the immunophenotype and cytokine secretion profile of
monocytes and macrophages. Cell Commun Signal. 16:172018.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Baj-Krzyworzeka M, Szatanek R, Weglarczyk
K, Baran J, Urbanowicz B, Brański P, Ratajczak MZ and Zembala M:
Tumour-derived microvesicles carry several surface determinants and
mRNA of tumour cells and transfer some of these determinants to
monocytes. Cancer Immunol Immunother. 55:808–818. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kwok ZH, Wang C and Jin Y: Extracellular
vesicle transportation and uptake by recipient cells: A critical
process to regulate human diseases. Processes (Basel). 9:2732021.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Dale DC, Boxer L and Conrad Liles W: The
phagocytes: Neutrophils and monocytes. Blood. 112:935–945. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Uribe-Querol E and Rosales C:
Phagocytosis: Our current understanding of a universal biological
process. Front Immunol. 11:10662020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Vachon E, Martin R, Plumb J, Kwok V,
Vandivier RW, Glogauer M, Kapus A, Wang X, Chow CW and Grinstein S:
CD44 is a phagocytic receptor. Blood. 107:4149–4158. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Szatanek R and Baj-Krzyworzeka M: CD44 and
tumor-derived extracellular vesicles (TEVs). Possible gateway to
cancer metastasis. Int J Mol Sci. 22:14632021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ziegler-Heitbrock L, Ancuta P, Crowe S,
Dalod M, Grau V, Hart DN, Leenen PJ, Liu YJ, MacPherson G, Randolph
GJ, et al: Nomenclature of monocytes and dendritic cells in blood.
Blood. 116:e74–e80. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ziegler-Heitbrock L: Blood monocytes and
their subsets: Established features and open questions. Front
Immunol. 6:4232015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Fu X and Yin M: Monocytes in tumor: The
perspectives of single-cell analysis. Tumor Discov. 1:42022.
View Article : Google Scholar
|
|
18
|
Turrini R, Pabois A, Xenarios I, Coukos G,
Delaloye JF and Doucey MA: Tie-2 expressing monocytes in human
cancers. Oncoimmunology. 6:e13035852017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Cormican S and Griffin MD: Human monocyte
subset distinctions and function: Insights from gene expression
analysis. Front Immunol. 11:10702020. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Baj-Krzyworzeka M, Weglarczyk K, Szatanek
R, Mytar B, Baran J and Siedlar M: The role of CD44H molecule in
the interactions between human monocytes and pancreatic
adenocarcinoma-derived microvesicles. Folia Histochem Cytobiol.
57:28–34. 2019.PubMed/NCBI
|
|
21
|
Zembala M, Siedlar M, Ruggiero I,
Wieckiewicz J, Mytar B, Mattei M and Colizzi V: The MHC class-II
and CD44 molecules are involved in the induction of tumour necrosis
factor (TNF) gene expression by human monocytes stimulated with
tumor cells. Int J Cancer. 56:269–274. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hyenne V, Ghoroghi S, Collot M, Bons J,
Follain G, Harlepp S, Mary B, Bauer J, Mercier L, Busnelli I, et
al: Studying the fate of tumor extracellular vesicles at high
spatiotemporal resolution using the zebrafish embryo. Dev Cell.
48:554–572.e7. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chaka W, Scharringa J, Verheul AFM,
Verhoef J, Van Strijp AG and Hoepelman IM: Quantitative analysis of
phagocytosis and killing of Cryptococcus neoformans by human
peripheral blood mononuclear cells by flow cytometry. Clin Diagn
Lab Immunol. 2:753–759. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Théry C, Witwer KW, Aikawa E, Alcaraz MJ,
Anderson JD, Andriantsitohaina R, Antoniou A, Arab T, Archer F,
Atkin-Smith GK, et al: Minimal information for studies of
extracellular vesicles (MISEV2018): A position statement of the
international society for extracellular vesicles and update of the
MISEV2014 guidelines. J Extracell vesicles. 7:15357502018.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kowal J, Arras G, Colombo M, Jouve M,
Morath JP, Primdal-Bengtson B, Dingli F, Loew D, Tkach M and Théry
C: Proteomic comparison defines novel markers to characterize
heterogeneous populations of extracellular vesicle subtypes. Proc
Natl Acad Sci USA. 113:E968–E977. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Khan SA, Tyagi M, Sharma AK, Barreto SG,
Sirohi B, Ramadwar M, Shrikhande SV and Gupta S: Cell-type
specificity of β-actin expression and its clinicopathological
correlation in gastric adenocarcinoma. World J Gastroenterol.
20:12202–12211. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Pols MS and Klumperman J: Trafficking and
function of the tetraspanin CD63. Exp Cell Res. 315:1584–1592.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Tóth EÁ, Turiák L, Visnovitz T, Cserép C,
Mázló A, Sódar BW, Försönits AI, Petővári G, Sebestyén A, Komlósi
Z, et al: Formation of a protein corona on the surface of
extracellular vesicles in blood plasma. J Extracell vesicles.
10:e121402021. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Paul D, Roy A, Nandy A, Datta B, Borar P,
Pal SK, Senapati D and Rakshit T: Identification of biomarker
hyaluronan on colon cancer extracellular vesicles using correlative
AFM and spectroscopy. J Phys Chem Lett. 11:5569–5576. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhang G, Lu R, Wu M, Liu Y, He Y, Xu J,
Yang C, Du Y and Gao F: Colorectal cancer-associated ~ 6 kDa
hyaluronan serves as a novel biomarker for cancer progression and
metastasis. FEBS J. 286:3148–3163. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ropponen K, Tammi M, Parkkinen J,
Eskelinen M, Tammi R, Lipponen P, Agren U, Alhava E and Kosma VM:
Tumor cell-associated hyaluronan as an unfavorable prognostic
factor in colorectal cancer. Cancer Res. 58:342–347.
1998.PubMed/NCBI
|
|
32
|
Josefsson A, Adamo H, Hammarsten P,
Granfors T, Stattin P, Egevad L, Laurent AE, Wikström P and Bergh
A: Prostate cancer increases hyaluronan in surrounding nonmalignant
stroma, and this response is associated with tumor growth and an
unfavorable outcome. Am J Pathol. 179:1961–1968. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Karousou E, Misra S, Ghatak S, Dobra K,
Götte M, Vigetti D, Passi A, Karamanos NK and Skandalis SS: Roles
and targeting of the HAS/hyaluronan/CD44 molecular system in
cancer. Matrix Biol. 59:3–22. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Baj-Krzyworzeka M, Mytar B, Weglarczyk K,
Szatanek R, Kijowski J and Siedlar M: Protumorogenic potential of
pancreatic adenocarcinoma-derived extracellular vesicles. Folia
Biol (Praha). 66:104–110. 2020.PubMed/NCBI
|
|
35
|
Liu M, Tolg C and Turley E: Dissecting the
dual nature of hyaluronan in the tumor microenvironment. Front
Immunol. 10:9472019. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Baj-Krzyworzeka M, Mytar B, Szatanek R,
Surmiak M, Węglarczyk K, Baran J and Siedlar M: Colorectal
cancer-derived microvesicles modulate differentiation of human
monocytes to macrophages. J Transl Med. 14:362016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Rilla K, Siiskonen H, Tammi M and Tammi R:
Hyaluronan-coated extracellular vesicles-a novel link between
hyaluronan and cancer. Adv Cancer Res. 123:121–148. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Luo Z, Dai Y and Gao H: Development and
application of hyaluronic acid in tumor targeting drug delivery.
Acta Pharm Sin B. 9:1099–1112. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Heider KH, Hofmann M, Hors E, van den Berg
F, Ponta H, Herrlich P and Pals ST: A human homologue of the rat
metastasis-associated variant of CD44 is expressed in colorectal
carcinomas and adenomatous polyps. J Cell Biol. 120:227–233. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
He M and Zeng Y: Microfluidic exosome
analysis toward liquid biopsy for cancer. J Lab Autom. 21:599–608.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Johnsen KB, Gudbergsson JM, Andresen TL
and Simonsen JB: What is the blood concentration of extracellular
vesicles? Implications for the use of extracellular vesicles as
blood-borne biomarkers of cancer. Biochim Biophys Acta Rev Cancer.
1871:109–116. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Näslund TI, Paquin-Proulx D, Paredes PT,
Vallhov H, Sandberg JK and Gabrielsson S: Exosomes from breast milk
inhibit HIV-1 infection of dendritic cells and subsequent viral
transfer to CD4+ T cells. AIDS. 28:171–180. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Purushothaman A, Bandari SK, Liu J, Mobley
JA, Brown EE and Sanderson RD: Fibronectin on the surface of
myeloma cell-derived exosomes mediates exosome-cell interactions. J
Biol Chem. 291:1652–1663. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Bonsergent E, Grisard E, Buchrieser J,
Schwartz O, Théry C and Lavieu G: Quantitative characterization of
extracellular vesicle uptake and content delivery within mammalian
cells. Nat Commun. 12:18642021. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Yamashita T, Takahashi Y, Nishikawa M and
Takakura Y: Effect of exosome isolation methods on physicochemical
properties of exosomes and clearance of exosomes from the blood
circulation. Eur J Pharm Biopharm. 98:1–8. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Gouëffic Y, Guilluy C, Guérin P, Patra P,
Pacaud P and Loirand G: Hyaluronan induces vascular smooth muscle
cell migration through RHAMM-mediated PI3K-dependent Rac
activation. Cardiovasc Res. 72:339–348. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Schledzewski K, Falkowski M, Moldenhauer
G, Metharom P, Kzhyshkowska J, Ganss R, Demory A, Falkowska-Hansen
B, Kurzen H, Ugurel S, et al: Lymphatic endothelium-specific
hyaluronan receptor LYVE-1 is expressed by stabilin-1+, F4/80+,
CD11b+ macrophages in malignant tumours and wound healing tissue in
vivo and in bone marrow cultures in vitro: Implications for the
assessment of lymphangiogene. J Pathol. 209:67–77. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Taylor KR, Yamasaki K, Radek KA, Nardo AD,
Goodarzi H, Golenbock D, Beutler B and Gallo RL: Recognition of
hyaluronan released in sterile injury involves a unique receptor
complex dependent on Toll-like receptor 4, CD44, and MD-2. J Biol
Chem. 282:18265–18275. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Lee-Sayer SSM, Dong Y, Arif AA, Olsson M,
Brown KL and Johnson P: The where, when, how, and why of hyaluronan
binding by immune cells. Front Immunol. 6:1502015. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Khaldoyanidi S, Achtnich M, Hehlmann R and
Zöller M: Expression of CD44 variant isoforms in peripheral blood
leukocytes in malignant lymphoma and leukemia: Inverse correlation
between expression and tumor progression. Leuk Res. 20:839–851.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Choi KY, Saravanakumar G, Park JH and Park
K: Hyaluronic acid-based nanocarriers for intracellular targeting:
Interfacial interactions with proteins in cancer. Colloids Surf B
Biointerfaces. 99:82–94. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Rios de la Rosa JM, Tirella A, Gennari A,
Stratford IJ and Tirelli N: The CD44-mediated uptake of hyaluronic
acid-based carriers in macrophages. Adv Healthc Mater.
6:16010122017. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Goebeler M, Kaufmann D, Bröcker EB and
Klein CE: Migration of highly aggressive melanoma cells on
hyaluronic acid is associated with functional changes, increased
turnover and shedding of CD44 receptors. J Cell Sci. 109:1957–1964.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Shi M, Dennis K, Peschon JJ,
Chandrasekaran R and Mikecz K: Antibody-induced shedding of CD44
from adherent cells is linked to the assembly of the cytoskeleton.
J Immunol. 167:123–131. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Parada N, Romero-Trujillo A, Georges N and
Alcayaga-Miranda F: Camouflage strategies for therapeutic exosomes
evasion from phagocytosis. J Adv Res. 31:61–74. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Mulcahy LA, Pink RC and Carter DR: Routes
and mechanisms of extracellular vesicle uptake. J Extracell
vesicles. 3:10.3402/jev.v3.24641. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Bjørnetrø T, Steffensen LA, Vestad B,
Brusletto BS, Olstad OK, Trøseid AM, Aass HCD, Haug KBF, Llorente
A, Bøe SO, et al: Uptake of circulating extracellular vesicles from
rectal cancer patients and differential responses by human monocyte
cultures. FEBS Open Bio. 11:724–740. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Lenart M, Rutkowska-Zapala M,
Baj-Krzyworzeka M, Szatanek R, Węglarczyk K, Smallie T,
Ziegler-Heitbrock L, Zembala M and Siedlar M: Hyaluronan carried by
tumor-derived microvesicles induces IL-10 production in classical
(CD14++CD16-) monocytes via PI3K/Akt/mTOR-dependent signaling
pathway. Immunobiology. 222:1–10. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Jordan AR, Racine RR, Hennig MJ and
Lokeshwar VB: The role of CD44 in disease pathophysiology and
targeted treatment. Front Immunol. 6:1822015. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Kapellos TS, Bonaguro L, Gemünd I, Reusch
N, Saglam A, Hinkley ER and Schultze JL: Human monocyte subsets and
phenotypes in major chronic inflammatory diseases. Front Immunol.
10:20352019. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Kwong C, Gilman-Sachs A and Beaman K: An
independent endocytic pathway stimulates different monocyte subsets
by the a2 N-terminus domain of vacuolar-ATPase. Oncoimmunology.
2:e229782013. View Article : Google Scholar : PubMed/NCBI
|