Introduction
Colorectal cancer (CRC) is one of the major causes
of cancer-related death. In order to improve the prognosis of CRC
patients, the role of chemotherapy has been considerably expanded
over the past decades. The administration of molecular targeted
therapy, including bevacizumab and anti-epithelial growth factor
receptor (EGFR) antibodies, has further prolonged the survival
times of CRC patients (1–3). The KRAS mutation in CRC is now
a well-established useful marker for predicting tumor
responsiveness, especially involving the use of the anti-EGFR
antibodies cetuximab and panitumumab. Many studies have reported
that 30–40% of CRC patients have KRAS mutations and that
they failed to respond to anti-EGFR antibody therapy (3–10).
Most of the mutations are found in codons 12 and 13. However,
recent reports have suggested that CRC tumors with the codon 13
mutation might show a different response to treatment than tumors
with other KRAS mutations (11,12).
In fact, some reports have suggested that cetuximab but not
panitumumab has shown therapeutic efficacy regarding CRC with codon
13 (11–13).
The KRAS mutations are monoallelic and appear
during carcinogenesis, mostly between the stages of early and
intermediate adenoma, maintaining a constant incidence in late
adenomas and carcinomas. Therefore, many studies have been
conducted to assess the prognostic significance of KRAS
mutations in CRC (14–25). Since the 1990's, it has been
suggested that alteration of codons 12 or 13, both of which are
important for protein functionality, might have a stronger
prognostic value (26–31). However, the prognostic value of
KRAS mutations in CRC remains controversial, especially in
the case of codons 12 or 13, although KRAS mutations have
been associated with a poor response to modern anti-EGFR antibody
therapy in relation to metastatic CRC. In addition, recent
attention has been drawn to the fact that KRAS mutations
could be a useful marker for predicting tumor responsiveness rather
than as a prognostic factor.
More recently, the Japan Study Group of KRAS
Mutation in CRC has reported on the clinicopathological features of
KRAS mutations in a large-scale Japanese study population
(32). They clarified the
clinicopathological features of KRAS mutations, rather than
their prognostic significance, in Japanese patients with CRC. Only
one study has reported the prognostic value of KRAS and
BRAF, which is a downstream KRAS molecule, in
Japanese patients with advanced and recurrent CRC (33). The study concluded that the
KRAS codon 13 mutation showed a trend towards poor patient
survival, and that BRAF mutations and not KRAS
mutations are a powerful prognostic factor in advanced and
recurrent CRC (33). Despite these
studies, the prognostic impact of KRAS mutations in Japanese
patients remains unknown in those patients who had recurrence or
curative surgery for advanced CRC. The aim of the current study was
to evaluate the KRAS genotypes of Japanese patients with
Union for International Cancer Control (UICC) stage I–IV CRC who
did not have anti-EGFR antibody therapy, and to assess the
relationship of these genotypes with clinical outcome.
Patients and methods
Patients and sample collection
A total of 99 patients (56 males and 43 females;
mean age, 66.9 years; age range, 42–91 years) who underwent CRC
resection at the Mie University Hospital between 2002 and 2004, and
from whom resected cancer tissue could be collected with informed
consent for use as samples, were enrolled in this study. All tumors
were histologically confirmed to be adenocarcinoma of the colon and
rectum. Exclusion criteria included a history of previous neoplasm,
patients from families with familial adenomatous polyposis or
hereditary non-polyposis CRC, and patients who had undergone
chemotherapy or radiotherapy prior to surgery. In this study, there
were no patients who received anti-EGFR antibody therapy, because
it had not been approved in Japan. To assess the precise prognostic
value of KRAS status, we also excluded patients with stage
IV disease who had received curative resection for synchronous
metastasis.
All patients were classified according to the UICC
stage classifications using resected specimens. There were 20
patients with stage I disease, 30 patients with stage II disease
and 25 patients with stage III disease. Twenty-four patients with
distant metastases were classified as having stage IV disease. Drug
approval in Japan takes considerably more time than in the Western
world. Molecular targeted agents including bevacizumab, cetuximab
and panitumumab were approved for use in 2007, 2008 and 2010,
respectively. Then, all stage IV patients underwent 5-FU with
irinotecan or oxaliplatin based chemotherapy. None of the patients
received molecular targeting agents including anti-EGFR antibodies
and bevacizumab. Adjuvant chemotherapy using 5-FU based
chemotherapy was introduced for all stage III and some of the stage
II patients. Clinicopathological data, such as age, gender, tumor
size and location were retrieved from patient records. The
specimens used for KRAS genotyping were paraffin-embedded
tissues. At the time of analysis, the median follow-up period for
all patients was 34 months. For the KRAS genotyping,
appropriate approvals were obtained from the institutional review
board.
DNA extraction from formalin-fixed,
paraffin-embedded (FFPE) tumor tissue sections
Tumor cell-rich area in a hematoxylin and
eosin-stained section was marked under a microscope, and tissue was
scratched from the area of another deparaffinized unstained
section. DNA for pieces of the scratched tissue sample was isolated
using the QIAamp FFPE Tissue kit (Qiagen K.K., Tokyo, Japan)
according to the manufacturer's protocol.
The concentration and purity of the extracted DNA
was determined by spectrophotometry (NanoDrop® ND-1000).
The extracted DNA was stocked at −20°C until use.
PCR amplification and direct
sequencing
To detect point mutation at KRAS codons 12
and 13, we used direct sequencing as previously described by
Karapetis et al (4). Exons 2
of the KRAS gene was amplified by PCR. The PCR products were
visualized using agarose gel electrophoresis with ethidium bromide
staining. The PCR DNA fragment was extracted from agarose gel, and
directly sequenced using an ABI 3130×l Genetic Analyzer (Applied
Biosystems, Tokyo, Japan) according to the manufacturer's
instructions.
Statistical analysis
We used JMP version 7 (SAS Institute Inc., Cary, NC,
USA) to perform statistical analyses. Contingency tables were
analyzed using Fisher's exact test or the χ2 test with
Yate's correction. Associations between continuous variables and
categorical variables were evaluated using the Mann-Whitney U test.
Survival curves were constructed according to the Kaplan-Meier
method, and differences were analyzed using the log-rank test. A
P-value of <0.05 was considered as significant. Variables found
to be significant at this level were considered eligible for Cox
proportional hazard regression.
Results
Frequency of KRAS gene mutation in
Japanese CRC patients
KRAS mutations were found in 37 (37.4%) of
the 99 patients. The most prevalent mutations were 10 (27.0%)
patients with GGT→GAT (G12D) and eight (21.6%) with GGT→GTT (G12V)
within codon 12, and 11 (29.7%) patients with GGC→GAC (G13D) within
codon 13. In addition, there were three (8.1%) patients with
GGT→TGT (G12c), three (8.1%) with GGT→AGT (G12S) and two (5.4%)
with GGT→GAT (G12D) mutations. Of these, 11% were the KRAS
p.G13D mutation and the remaining 26.2% were other KRAS
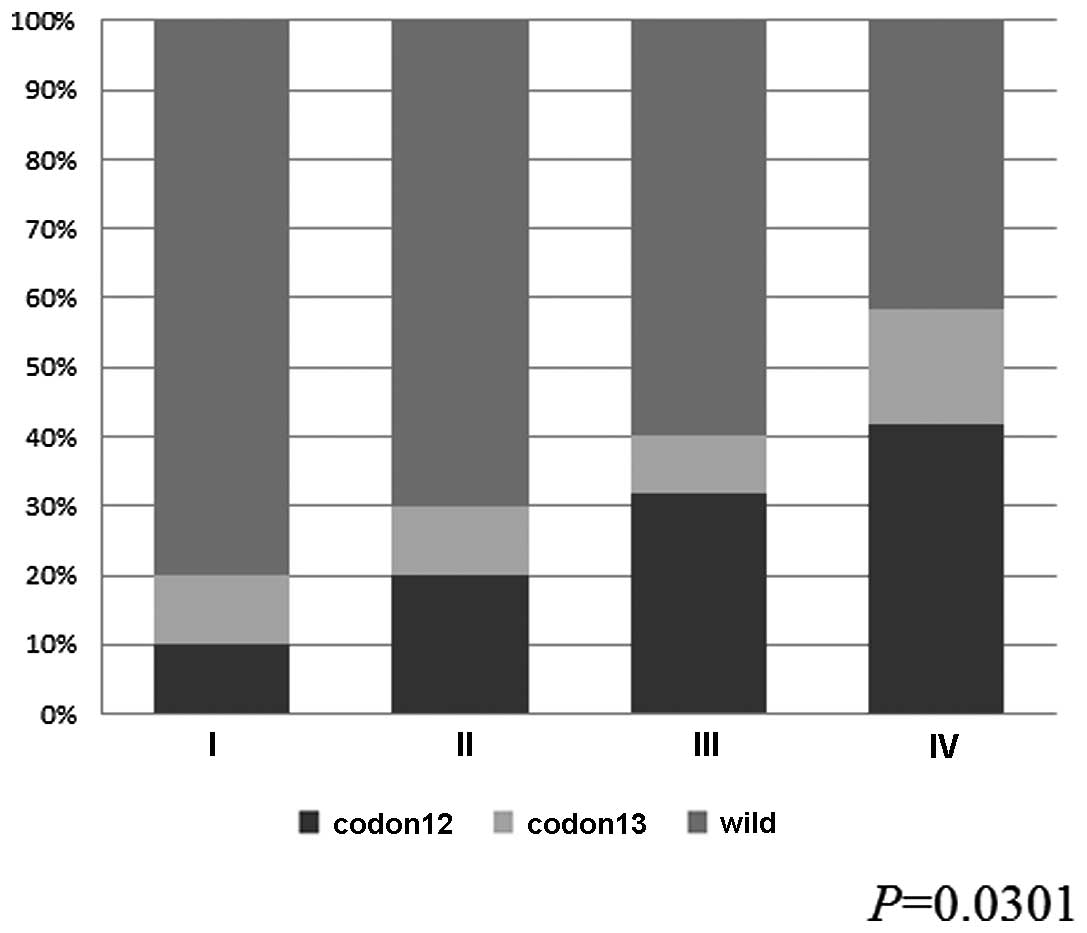
mutations. The frequency of KRAS mutation according to UICC
stage was 20% in stage I, 30% in stage II, 40% in stage III and 58%
in stage IV. The frequency of KRAS mutations was positively
correlated with stage classification mutations (P=0.0301), which
resulted in a trend towards a high frequency of KRAS 12
mutations in advanced stage disease (Fig. 1); while the KRAS p.G13D
mutation occurred at a constant rate in all disease stages.
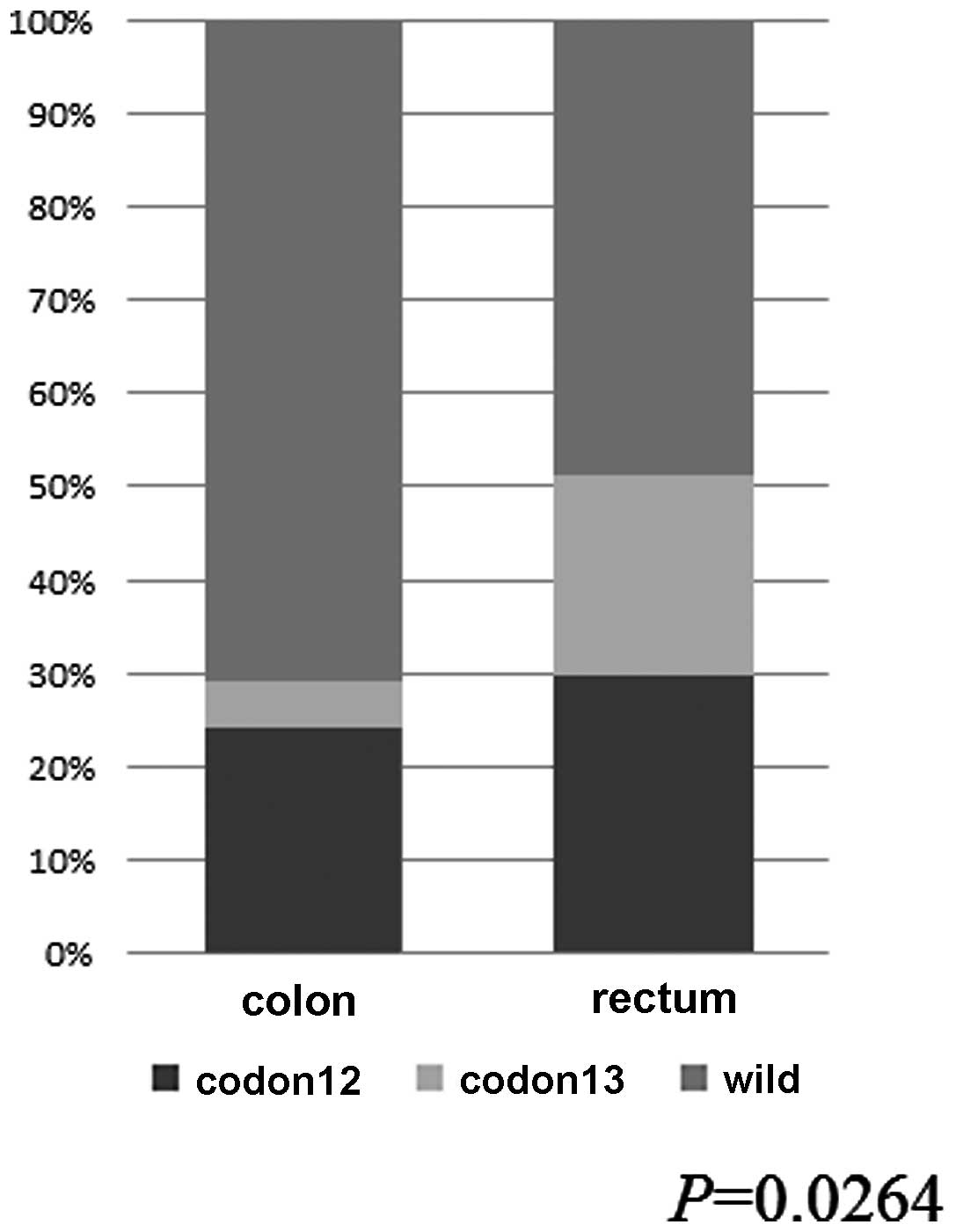
Furthermore, the KRAS p.G13D mutation was found more
frequently in the rectum than in the colon (21.6 vs. 4.8%;
P=0.0102), whereas KRAS 12 mutations were found at
comparable levels in the rectum and colon (29.7 vs. 24.2%;
P=0.5448) (Fig. 2).
Association of KRAS mutations with
clinicopathological features
When all patients were divided into three groups
according to their KRAS status (wild-type vs. KRAS 12
mutations vs. KRAS p.G13D), a significant association
emerged between KRAS mutations and lymph node metastases
(P=0.0076), and distant metastases (P=0.0289) (Table I). There were no significant
differences in age, gender, primary site, tumor size, pathological
type, tumor invasion, vessel and lymphatic invasion.
 | Table IRelationship of KRAS status
with clinicopathological features. |
Table I
Relationship of KRAS status
with clinicopathological features.
| Wild-type (n=62)
(%) | KRAS 12
(n=26) (%) | KRAS p.G13D
(n=11) (%) | P-value |
|---|
| Age (years) | 67.9 | 65 | 65.9 | ns |
| Gender
(male/female) | 40/22 | 9/16 | 6/5 | ns |
| Colon/rectum | 44/18 | 15/11 | 3/8 | ns |
| Tumor size
(mm) | 40.4 | 50.2 | 41.4 | ns |
| Pathology |
| Well | 15 | 6 | 2 | |
| Moderately | 41 | 17 | 8 | ns |
| Poorly | 3 | 1 | 0 | |
| Mucinous | 3 | 2 | 1 | |
| pT |
| T1 | 11 | 1 | 0 | |
| T2 | 10 | 5 | 2 | ns |
| T3 | 29 | 13 | 5 | |
| T4 | 12 | 7 | 4 | |
| pN |
| N0 | 41 (66) | 10 (38) | 5 (46) | |
| N1 | 12 (19) | 11 (42) | 3 (27) | 0.0076 |
| N2 | 8 (13) | 1 (5) | 3 (27) | |
| N3 | 1 (2) | 4 (15) | 0 | |
| Lymphatic
invasion |
| 0–1/2–3 | 24/38 | 8/18 | 3/8 | ns |
| Venous
invasion |
| 0–1/2–3 | 50/12 | 19/7 | 9/2 | ns |
| Distant
metastasis |
| Yes/no | 9/62 (15) | 10/26 (38) | 4/11 (36) | 0.0289 |
Prognostic analyses
Fig. 3 shows the
cumulative overall survival (OS) curves for all patients subdivided
according to KRAS status. The cumulative 5-year survival
rate of KRAS wild-type and KRAS 12 mutations, and the
KRAS p.G13D mutation was 81.4, 61.4 and 42.0%, respectively
(P=0.0397). On the basis of Cox univariate proportional hazards
analysis, tumor size (>5 vs. <5 cm) [hazard ratio (HR),
2.674; 95% confidence interval (CI), 1.155–6.188; and P=0.0216],
pathological T stage (T4 vs. others) (HR, 15.761; 95% CI,
6.122–40.577; and P<0.0001), lymphatic vessel involvement (HR,
5.600; 95% CI, 1.312–23.895; and P=0.020), vessel involvement (HR,
2.848; 95% CI, 1.232–6.582; and P=0.0144), distant metastasis (HR,
14.910; 95% CI, 5.802–38.314; and P<0.0001), KRAS
mutation (mutation vs. wild-type) (HR, 2.884; 95% CI, 1.247–6.668;
and P=0.0133) and KRAS p.G13D mutation (p.G13D vs.
wild-type) (HR, 3.462; 95% CI, 1.158–10.351; and P=0.0262) were
significant prognostic factors for OS. However, no KRAS
mutations were identified as independent risk factors for
predicting poor prognosis using multivariate analysis.
Next, we evaluated the prognostic value of
KRAS mutations according to CRC stage classification. In
this study, all stage IV patients underwent therapeutic
chemotherapy using irinotecan (CPT-11) and/or oxaliplatin (L-OHP)
based chemotherapy without any molecular agents including cetuximab
or panitumumab. There were no differences in the median survival
times (MST) of patients between KRAS and wild-type mutations
(16.2 vs. 17.8 months). Regardless of KRAS mutant status,
KRAS genotype had no effect upon the OS of stage IV patients
without anti-EGFR antibody therapy (KRAS p.G13D, MST 15.7
months; KRAS 12 mutations, MST 17.8 months; wild-type, MST
16.2 months).
To further investigate the prognostic value of
KRAS mutation, we evaluated the correlation between
KRAS mutations and DFS in patients with stage I–III disease.
Significant prognostic factors or a trend in prognostic factors for
DFS were pathological T stage (T4 vs. others; P=0.0015), lymphatic
vessel involvement (P=0.0645) and KRAS p.G13D (p.G13D vs.
wild-type; P=0.0895). Furthermore, multivariate analysis identified
pathological T4 stage (HR, 6.993; 95% CI, 1.678–29.145; and
P=0.0076) and the KRAS p.G13D mutation (HR, 5.934; 95% CI,
1.001–35.174; and P=0.0499) as the most significant independent
prognostic factors associated with DFS (Table II).
 | Table IIMultivariate analysis of factors
associated with disease-free survival. |
Table II
Multivariate analysis of factors
associated with disease-free survival.
| Variables | HR | 95% CI | P-value |
|---|
| Pathological T (T4
vs. others) | 6.993 | 1.678–29.145 | 0.0076 |
| Lymphatic vessel
involvement (ly 2–3 vs.ly 0–1) | 4.807 | 0.583–36.639 | 0.1447 |
| KRAS
mutations |
| KRAS 12
mutations vs. wild-type | 3.288 | 0.691–15.640 | 0.1346 |
| KRAS p.G13D
vs. wild-type | 5.934 | 1.001–35.174 | 0.0499 |
Discussion
KRAS is a proto-oncogene encoding a small 21
kD guanosine triphosphate (GTP)/guanosine diphosphate (GDP) binding
protein, and is responsible for the regulation of cellular response
to many extracellular stimuli (34). The major signal transduction
pathways, including the RAS-RAF mitogen activated protein kinase
(MAPK), lead to the expression of proteins involved in cell
proliferation, differentiation and survival (35,36).
Despite the many studies that have investigated the
prognostic value of KRAS mutations in CRC treatment
(14–25), uncertainty remains. Large cohort
studies such as the collaborative RASCAL study have indicated that
KRAS mutations increased the risk of recurrence and death
(37). A second RASCAL II study
involving 3,498 CRC patients found that of the 12 possible
KRAS mutations on codons 12 and 13, only the substitution of
glycine to valine on codon 12 had a significant impact on failure
free survival and OS. Although the various KRAS mutations
did not have prognostic value in relation to patients with
metastasis, the mutation had greater impact in Dukes' C than in
Dukes' B cancers (25). These
studies did not identify the KRAS 13 mutation as being
prognostic. However, a recent report by De Roock et al
(11) suggested that tumors with
the KRAS p.G13D mutation may have a worse prognosis, and
that the use of cetuximab was associated with longer OS and
progression-free survival (PFS) among patients with tumors
containing the KRAS p.G13D mutation than with tumors
containing other KRAS mutations.
In order to evaluate the precise prognostic value of
KRAS status for CRC patients, we need to exclude several
biases including the use of anti-EGFR antibody therapy for stage IV
disease and curability of the patients. Therefore, our exclusion
criteria were patients with stage IV disease who had received
curative metastasectomy, and who had undergone chemotherapy with
anti-EGFR antibody therapy. Furthermore, we evaluated the prognosis
of patients with stage I–III and IV disease separately. Our study
found that the DFS of stage I–III patients with KRAS p.G13D
mutated tumors showed a worse trend than was the case for
KRAS wild-type mutated tumors, and that the KRAS
p.G13D mutation was the most significant independent prognostic
factor associated with DFS. However, there were no significant
correlations between KRAS mutations including the
KRAS p.G13D mutation and OS in Japanese CRC patients, with
respect to stage IV patients who underwent therapeutic chemotherapy
without anti-EGFR antibodies. These results suggest that the
KRAS p.13D mutation may be a possible prognostic factor for
cancer recurrence in Japanese CRC patients via enhanced tumor
growth by mechanisms such as cell proliferation. It is still not
clear whether none or only one of the two codons is important in
the prognosis of CRC patients. According to Guerrero et al
(38), in vitro data
suggested that KRAS 13 mutated tumors had a higher level of
apoptosis and were less aggressive as compared with KRAS 12
mutated tumors. This observation contradicts our prognostic
findings that suggested that the KRAS p.G13D mutation was an
independent prognostic factor associated with DFS; although it was
in agreement with our finding of a positive correlation between
KRAS 12 mutations and advanced disease stage
classification.
In 2011, the Japan Study Group of KRAS
Mutation in CRC reported the results from a large observation study
with information on 5,887 CRC patients (32). The frequency of KRAS
mutations and the KRAS p.G13D mutation in this study was
37.6 and 7.3%, respectively, which was similar to our results (37.4
and 11.1%, respectively). The study suggested that the
clinicopathological features of tumors with the KRAS p.G13D
mutation were not similar to those of the KRAS wild-type
mutation and other mutations. Tumors with the KRAS p.G13D
mutation might show a different response to treatment than tumors
with other KRAS mutations. Thus, KRAS p.G13D and
other KRAS mutations may have differing usefulness as
prognostic indicators in Japanese CRC patients because of their
differing biological effects. The major limitations of our study
were the small number of patients enrolled, and the fact that it
was retrospective.
In conclusion, we found that in Japanese CRC
patients the KRAS p.G13D mutation had prognostic impact in
relation to DFS in stage I–III disease, while the prognosis of
stage IV patients without anti-EGFR antibody therapy was unaffected
by KRAS status. Elucidation of the exact effects of
KRAS 12 mutations and the KRAS p.G13D mutation on
survival in Japanese CRC patients will require further study.
References
|
1
|
Hurwitz H, Fehrenbacher L, Novotny W, et
al: Bevacizumab plus irinotecan, fluorouracil, and leucovorin for
metastatic colorectal cancer. N Engl J Med. 350:2335–2342. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Van Cutsem E, Köhne CH, Hitre E, et al:
Cetuximab and chemotherapy as initial treatment for metastatic
colorectal cancer. N Engl J Med. 360:1408–1417. 2009.PubMed/NCBI
|
|
3
|
Fuchs CS, Marshall J, Mitchell E, et al:
Randomized, controlled trial of irinotecan plus infusional, bolus,
or oral fluoropyrimidines in first-line treatment of metastatic
colorectal cancer: results from the BICC-C Study. J Clin Oncol.
25:4779–4786. 2007. View Article : Google Scholar
|
|
4
|
Karapetis CS, Khambata-Ford S, Jonker DJ,
et al: K-ras mutations and benefit from cetuximab in advanced
colorectal cancer. N Engl J Med. 359:1757–1765. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Amado RG, Wolf M, Peeters M, et al:
Wild-type KRAS is required for panitumumab efficacy in patients
with metastatic colorectal cancer. J Clin Oncol. 26:1626–1634.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cappuzzo F, Finocchiaro G, Rossi E, et al:
EGFR FISH assay predicts for response to cetuximab in chemotherapy
refractory colorectal cancer patients. Ann Oncol. 19:717–723. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lièvre A, Bachet JB, Boige V, et al: KRAS
mutations as an independent prognostic factor in patients with
advanced colorectal cancer treated with cetuximab. J Clin Oncol.
26:374–379. 2008.
|
|
8
|
Di Fiore F, Blanchard F, Charbonnier F, et
al: Clinical relevance of KRAS mutation detection in metastatic
colorectal cancer treated by Cetuximab plus chemotherapy. Br J
Cancer. 96:1166–1169. 2007.PubMed/NCBI
|
|
9
|
Khambata-Ford S, Garrett CR, Meropol NJ,
et al: Expression of epiregulin and amphiregulin and K-ras mutation
status predict disease control in metastatic colorectal cancer
patients treated with cetuximab. J Clin Oncol. 25:3230–3237. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lièvre A, Bachet JB, Le Corre D, et al:
KRAS mutation status is predictive of response to cetuximab therapy
in colorectal cancer. Cancer Res. 66:3992–3995. 2006.PubMed/NCBI
|
|
11
|
De Roock W, Jonker DJ, Di Nicolantonio F,
et al: Association of KRAS p. G13D mutation with outcome in
patients with chemotherapy-refractory metastatic colorectal cancer
treated with cetuximab. JAMA. 304:1812–1820. 2010.PubMed/NCBI
|
|
12
|
Tejpar S, Bokemeyer C, Celik I,
Schlichting M, Sartorius U and Van Cutsem E: Influence of KRAS G13D
mutations on outcome in patients with metastatic colorectal cancer
(mCRC) treated with first-line chemotherapy with or without
cetuximab. J Clin Oncol. 29:35112010.
|
|
13
|
Peeters M, Douillard JY, Cutsem E, et al:
Evaluation of individual codon 12 and 13 mutant (MT) KRAS alleles
as prognostic and predictive biomarkers of response to panitumumab
(pmab) in patients with metastatic colorectal cancer (mCRC). Eur J
Cancer. 47(Suppl 2): 162011. View Article : Google Scholar
|
|
14
|
Dix BR, Robbins P, Soong R, et al: The
common molecular genetic alterations in Dukes' B and C colorectal
carcinomas are not short-term prognostic indicators of survival.
Int J Cancer. 59:747–751. 1994.
|
|
15
|
Iniesta P, Caldés T, Vega FJ, et al:
Prevalence of p53 overexpression or mutations, but not K-ras
mutations, in recurrent patients affected by colorectal carcinoma.
Int J Oncol. 7:319–325. 1995.PubMed/NCBI
|
|
16
|
Andersen SN, Lovig T, Breivik J, et al:
K-ras mutations and prognosis in large-bowel carcinomas. Scand J
Gastroenterol. 32:62–69. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tortola S, Marcuello E, Gonzalez I, et al:
p53 and K-ras gene mutations correlate with tumor aggressiveness
but are not of routine prognostic value in colorectal cancer. J
Clin Oncol. 17:1375–1381. 1999.PubMed/NCBI
|
|
18
|
Hirvikoski P, Auvinen A, Servomaa K, et
al: K-ras and p53 mutations and overexpressions as prognostic
factors in female rectal carcinoma. Anticancer Res. 19:685–691.
1999.PubMed/NCBI
|
|
19
|
Esteller M, Gonzalez S, Risques RA, et al:
K-ras and p16 aberrations confer poor prognosis in human colorectal
cancer. J Clin Oncol. 19:286–288. 2001.PubMed/NCBI
|
|
20
|
Laurent-Puig P, Olschwang S, Delattre O,
et al: Survival and acquired genetic alterations in colorectal
cancer. Gastroenterology. 102:1136–1141. 1992.PubMed/NCBI
|
|
21
|
Bell SM, Scott N, Cross D, et al:
Prognostic value of p53 overexpression and c-Ki-ras gene mutations
in colorectal cancer. Gastroenterology. 104:57–64. 1993.PubMed/NCBI
|
|
22
|
Lee JC, Wang ST, Lai MD, et al: K-ras gene
mutation is a useful predictor of the survival of early stage
colorectal cancers. Anticancer Res. 16:3839–844. 1996.PubMed/NCBI
|
|
23
|
Ahnen DJ, Feigl P, Quan G, et al: Ki-ras
mutation and p53 overexpression predict the clinical behavior of
colorectal cancer: a Southwest Oncology Group study. Cancer Res.
58:1149–1158. 1998.PubMed/NCBI
|
|
24
|
Bouzourene H, Gervaz P, Cerottini JP, et
al: p53 and Ki-ras as prognostic factors for Dukes' stage B
colorectal cancer. Eur J Cancer. 36:1008–1015. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Andreyev HJ, Norman AR, Cunningham D, et
al: Kirsten ras mutations in patients with colorectal cancer: the
‘RASCAL II’ study. Br J Cancer. 85:692–696. 2001.
|
|
26
|
Benhattar J, Losi L, Chaubert P, et al:
Prognostic significance of K-ras mutations in colorectal carcinoma.
Gastroenterology. 104:1044–1048. 1993.PubMed/NCBI
|
|
27
|
Cerottini JP, Caplin S, Saraga E, et al:
The type of K-ras mutation determines prognosis in colorectal
cancer. Am J Surg. 175:198–202. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Span M, Moerkerk PTM, De Goeij AFPM and
Arends JW: A detailed analysis of K-ras point mutations in relation
to tumor progression and survival in colorectal cancer patients.
Int J Cancer. 69:241–245. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Pajkos G, Kiss I, Sandor J, et al: The
prognostic value of the presence of mutations at the codons 12, 13,
61 of K-ras oncogene in colorectal cancer. Anticancer Res.
20:1695–1701. 2000.PubMed/NCBI
|
|
30
|
Samowitz WS, Curtin K, Schaffer D, et al:
Relationship of Ki-ras mutations in colon cancers to tumor
location, stage, and survival: a population-based study. Cancer
Epidemiol Biomarkers Prev. 9:1193–1197. 2000.PubMed/NCBI
|
|
31
|
Al-Mulla F, Going JJ, Sowden ET, et al:
Heterogeneity of mutant versus wild-type in primary and metastatic
colorectal carcinomas, and association of codon 12 valine with
early mortality. J Pathol. 185:130–138. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Uetake H, Watanabe T, Yoshino T, et al:
Clinicopathological features of patients with colorectal cancer
among KRAS wild type p. G13D and other mutations: results from a
multicenter, cross-sectional study by the Japan Study Group of KRAS
Mutation in Colorectal Cancer. J Clin Oncol. 29:36052011.
|
|
33
|
Yokota T, Ura T, Shibata N, et al: BRAF
mutation is a powerful prognostic factor in advanced and recurrent
colorectal cancer. Br J Cancer. 104:856–862. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Schubbert S, Shannon K and Bollag G:
Hyperactive Ras in developmental disorders and cancer. Nat Rev
Cancer. 7:295–308. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
35
|
Harari PM: Epidermal growth factor
receptor inhibition strategies in oncology. Endocr Relat Cancer.
11:689–708. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kiaris H and Spandidos DA: Mutations of
ras genes in human tumours (Review). Int J Oncol. 7:413–421.
1995.
|
|
37
|
Andreyev HJ, Norman AR, Cunningham D,
Oates JR and Clarke PA: Kirsten ras mutations in patients with
colorectal cancer: the multicenter ‘RASCAL’ study. J Natl Cancer
Inst. 90:675–684. 1998.
|
|
38
|
Guerrero S, Casanova I, Farré L, Mazo A,
Capellà G and Mangues R: K-ras codon 12 mutation induces higher
level of resistance to apoptosis and predisposition to
anchorage-independent growth than codon 13 mutation or
proto-oncogene overexpression. Cancer Res. 60:6750–6756. 2000.
|