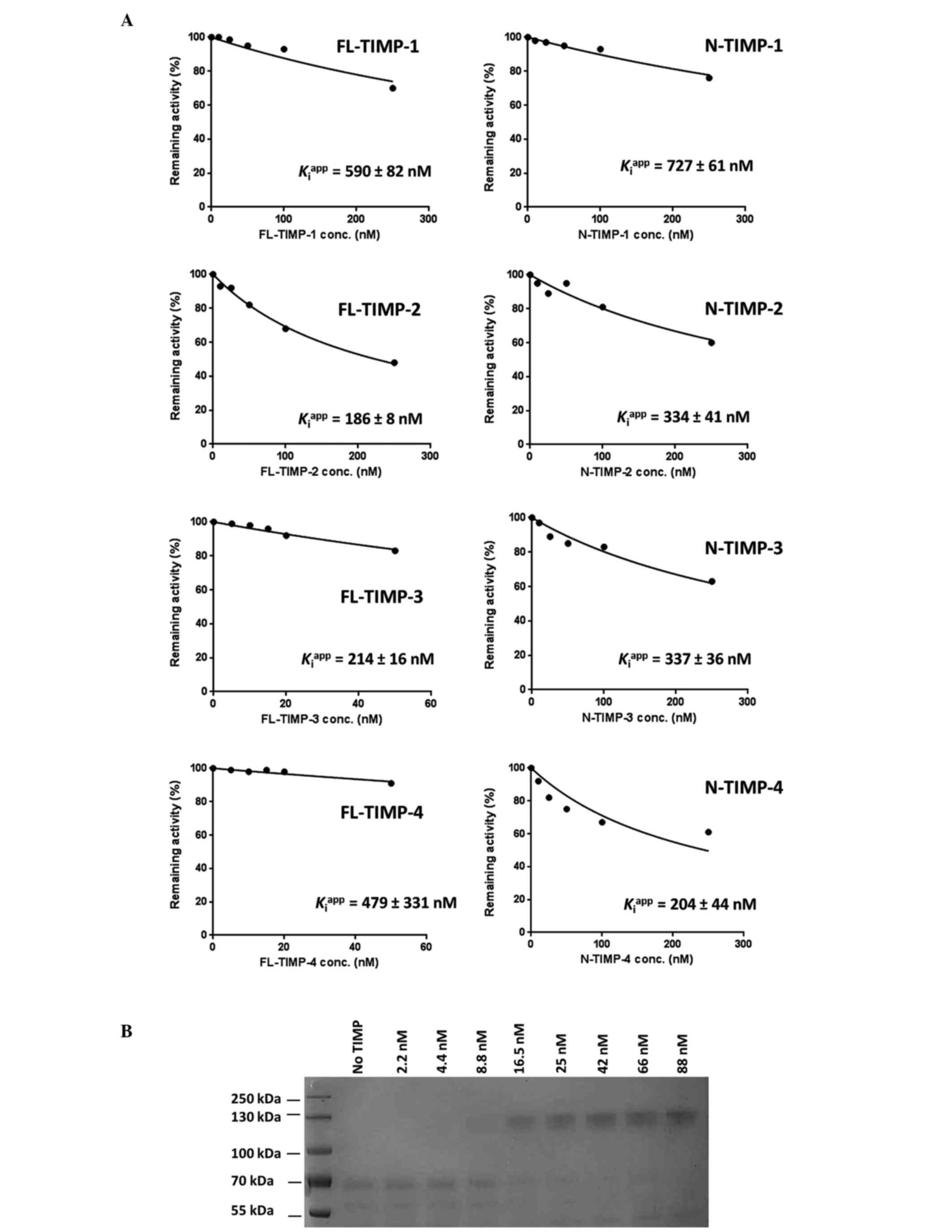
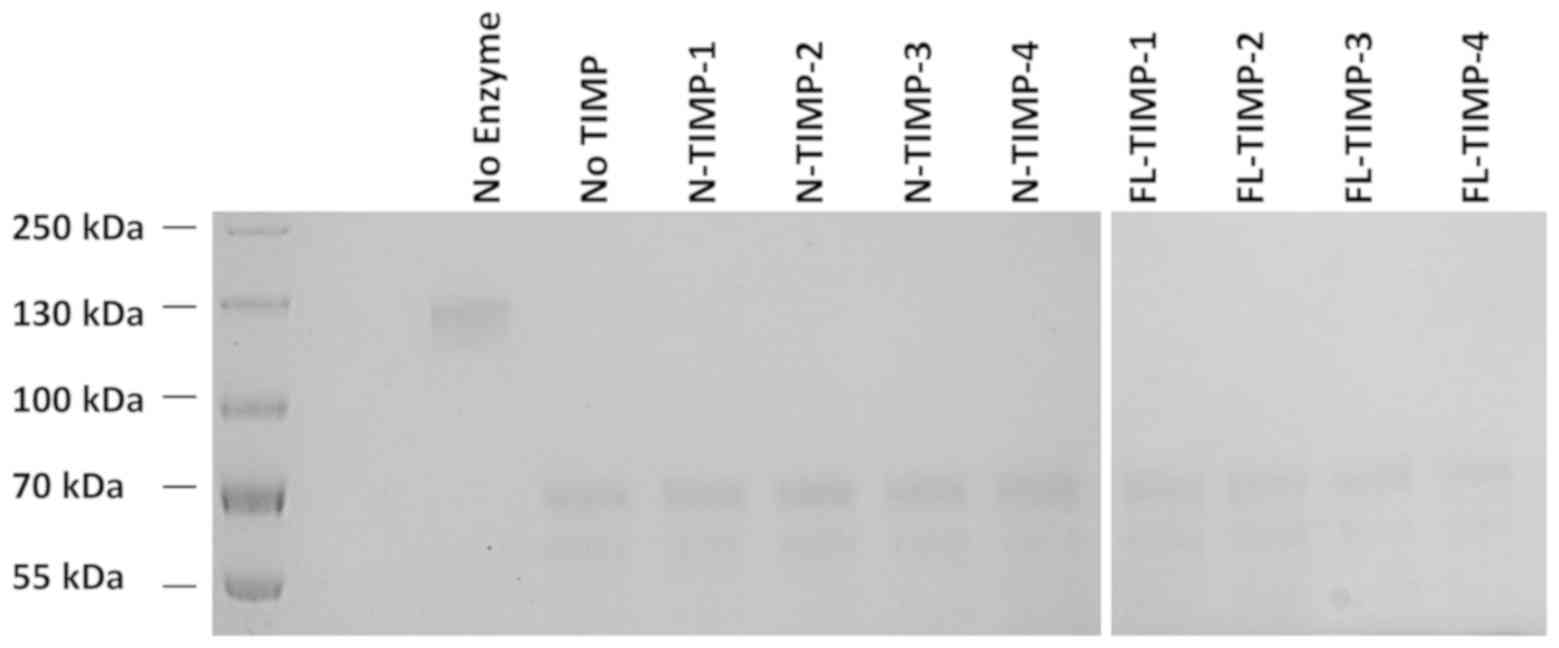
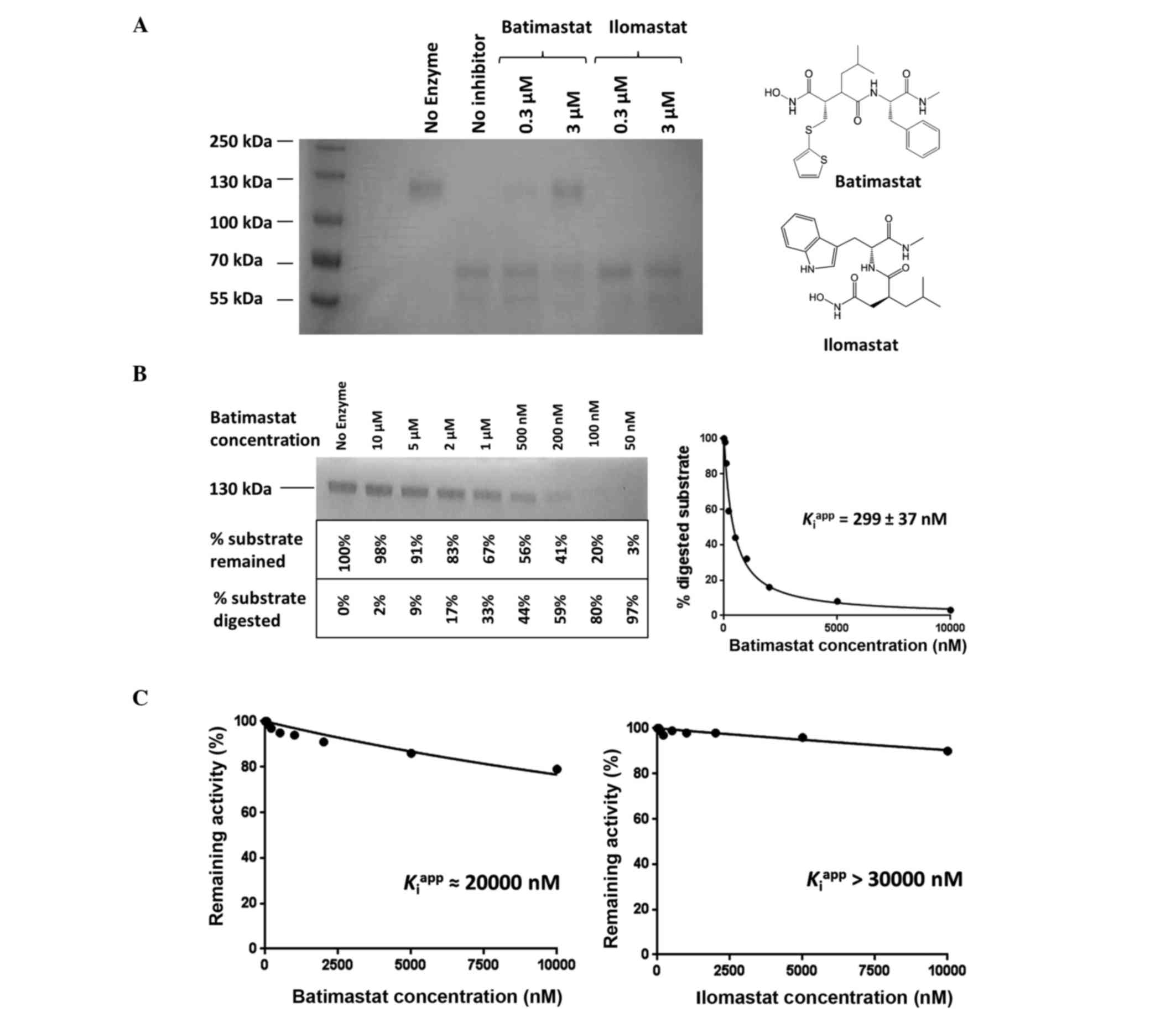
|
1
|
Dubail J and Apte SS: Insights on ADAMTS
proteases and ADAMTS-like proteins from mammalian genetics. Matrix
Biol. 44(46C): 24–37. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cal S, Obaya AJ, Llamazares M, Garabaya C,
Quesada V and López-Otín C: Cloning, expression analysis, and
structural characterization of seven novel human ADAMTSs, a family
of metalloproteinases with disintegrin and thrombospondin-1
domains. Gene. 283:49–62. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lancellotti S and De Cristofaro R:
Structure and proteolytic properties of ADAMTS13, a metalloprotease
involved in the pathogenesis of thrombotic microangiopathies. Prog
Mol Biol Transl Sci. 99:105–144. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Viloria CG, Obaya AJ, Moncada-Pazos A,
Llamazares M, Astudillo A, Capellá G, Cal S and López-Otín C:
Genetic inactivation of ADAMTS15 metalloprotease in human
colorectal cancer. Cancer Res. 69:4926–4934. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Porter S, Span PN, Sweep FC, Tjan-Heijnen
VC, Pennington CJ, Pedersen TX, Johnsen M, Lund LR, Rømer J and
Edwards DR: ADAMTS8 and ADAMTS15 expression predicts survival in
human breast carcinoma. Int J Cancer. 118:1241–1247. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zheng XL: Structure-function and
regulation of ADAMTS-13 protease. J Thromb Haemost. 11(Suppl 1):
11–23. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Dancevic CM, Fraser FW, Smith AD, Stupka
N, Ward AC and McCulloch DR: Biosynthesis and expression of a
disintegrin-like and metalloproteinase domain with thrombospondin-1
repeats-15: A novel versican-cleaving proteoglycanase. J Biol Chem.
288:37267–37276. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Rodríguez-Manzaneque JC, Westling J, Thai
SN, Luque A, Knauper V, Murphy G, Sandy JD and Iruela-Arispe ML:
ADAMTS1 cleaves aggrecan at multiple sites and is differentially
inhibited by metalloproteinase inhibitors. Biochem Biophys Res
Commun. 293:501–508. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kashiwagi M, Tortorella M, Nagase H and
Brew K: TIMP-3 is a potent inhibitor of aggrecanase 1 (ADAM-TS4)
and aggrecanase 2 (ADAM-TS5). J Biol Chem. 276:12501–12504. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Amour A, Slocombe PM, Webster A, Butler M,
Knight CG, Smith BJ, Stephens PE, Shelley C, Hutton M, Knäuper V,
et al: TNF-alpha converting enzyme (TACE) is inhibited by TIMP-3.
FEBS Lett. 435:39–44. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Amour A, Knight CG, Webster A, Slocombe
PM, Stephens PE, Knäuper V, Docherty AJ and Murphy G: The in vitro
activity of ADAM-10 is inhibited by TIMP-1 and TIMP-3. FEBS Lett.
473:275–279. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lee MH, Verma V, Maskos K, Nath D, Knäuper
V, Dodds P, Amour A and Murphy G: Engineering N-terminal domain of
tissue inhibitor of metalloproteinase (TIMP)-3 to be a better
inhibitor against tumour necrosis factor-alpha-converting enzyme.
Biochem J. 364:227–234. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lee MH, Verma V, Maskos K, Becherer JD,
Knäuper V, Dodds P, Amour A and Murphy G: The C-terminal domains of
TACE weaken the inhibitory action of N-TIMP-3. FEBS Lett.
520:102–106. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lee MH, Rapti M and Murphy G: Delineating
the molecular basis of the inactivity of tissue inhibitor of
metalloproteinase-2 against tumor necrosis factor-alpha-converting
enzyme. J Biol Chem. 279:45121–45129. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lee MH, Rapti M and Murphy G: Total
conversion of tissue inhibitor of metalloproteinase (TIMP) for
specific metalloproteinase targeting: Fine-tuning TIMP-4 for
optimal inhibition of tumor necrosis factor-α-converting enzyme. J
Biol Chem. 280:15967–15975. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hashimoto G, Aoki T, Nakamura H, Tanzawa K
and Okada Y: Inhibition of ADAMTS4 (aggrecanase-1) by tissue
inhibitors of metalloproteinases (TIMP-1, 2, 3 and 4). FEBS Lett.
494:192–195. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lee MH, Rapti M and Murphy G: Unveiling
the surface epitopes that render tissue inhibitor of
metalloproteinase-1 inactive against membrane type 1-matrix
metalloproteinase. J Biol Chem. 278:40224–40230. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Gerhardt S, Hassall G, Hawtin P, et al:
Crystal structures of human ADAMTS-1 reveal a conserved catalytic
domain and a disintegrin-like domain with a fold homologous to
cysteine-rich domains. J Mol Biol. 373:891–902. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mosyak L, Georgiadis K, Shane T, Svenson
K, Hebert T, McDonagh T, Mackie S, Olland S, Lin L, Zhong X, et al:
Crystal structures of the two major aggrecan degrading enzymes,
ADAMTS4 and ADAMTS5. Protein Sci. 17:16–21. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Grillberger R, Gruber B, Skalicky S,
Schrenk G, Knöbl P, Plaimauer B, Turecek PL, Scheiflinger F and
Rottensteiner H: A novel flow-based assay reveals discrepancies in
ADAMTS-13 inhibitor assessment as compared with a conventional
clinical static assay. J Thromb Haemost. 12:1523–1532. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Feys HB, Vandeputte N, Palla R, Peyvandi
F, Peerlinck K, Deckmyn H, Lijnen HR and Vanhoorelbeke K:
Inactivation of ADAMTS13 by plasmin as a potential cause of
thrombotic thrombocytopenic purpura. J Thromb Haemost. 8:2053–2062.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Doldan-Silvero A, Acevedo-Gadea C, Habib
C, Freeman J and Johari V: ADAMTS13 activity and inhibitor. Am J
Hematol. 83:811–814. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lam JK, Chion CK, Zanardelli S, Lane DA
and Crawley JT: Further characterization of ADAMTS-13 inactivation
by thrombin. J Thromb Haemost. 5:1010–1018. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Crawley JT, Lam JK, Rance JB, Mollica LR,
O'Donnell JS and Lane DA: Proteolytic inactivation of ADAMTS13 by
thrombin and plasmin. Blood. 105:1085–1093. 2005. View Article : Google Scholar : PubMed/NCBI
|