Introduction
Lung cancer is the leading cause of cancer-related
mortality in the world. Non-small cell lung cancer (NSCLC) accounts
for approximately 85% of all lung cancer cases (1). The oral small molecule epidermal
growth factor receptor (EGFR) tyrosine kinase inhibitors (TKIs),
such as gefitinib and erlotinib, promote responses in 10–18% of
patients who had a failed response to prior chemotherapy. Erlotinib
was found to have a 2-month median survival advantage over a
placebo (2), and gefitinib did not
exhibit an inferior efficacy when compared with docetaxel (3).
Treatment with an EGFR TKI is effective in women,
Asians, non-smokers, and patients with adenocarcinoma. An EGFR
mutation was found to be the most important predictive factor for
patient response to an EGFR TKI (4). However, acquiring adequate tissue for
an EGFR mutational analysis is often not feasible, particularly in
patients with advanced disease (2–4).
Therefore, the identification of clinical parameters that can serve
as surrogates markers for an EGFR mutation may prove useful when
mutational analysis is not feasible. A recent study reported that
the molecular analysis of circulating tumor cells from the
peripheral blood of patients with lung cancer was useful in
monitoring changes in epithelial tumor genotypes during the course
of treatment (5). However, this
molecular analysis may prove to be difficult as a specific
microfluidic-based device, the CTC chip, is required.
Therefore, a marker that is easily analyzed and
predicts the responses to EGFR TKI treatment is needed. Several
serum markers have been considered potentially prognostic and
predictive in NSCLC. Among these NSCLC markers, carcinoembryonic
antigen (CEA) and cytokeratin-19 fragments (CYFRA 21-1) have been
considered sensitive and valuable tumor markers for diagnosis,
prognosis, and the monitoring of therapy (6–10).
According to recent reports, CEA and CYFRA 21-1 were significant
predictors of sensitivity and survival in patients treated with
gefitinib (11–13). Therefore, we investigated the
clinical significance of the pre-treatment serum levels of CEA and
CYFRA 21-1 in advanced NSCLC patients who were treated with
gefitinib or erlotinib.
Materials and methods
We retrospectively collected clinical data on 123
NSCLC patients whose pre-treatment levels of CEA and CYFRA 21-1 had
been measured and who received gefitinib or erlotinib treatment at
Severance Hospital, Yonsei University Health System, Seoul, Korea,
from January 2006 to December 2008. Variables used in the
pre-treatment analysis were age, gender, clinical stage, Eastern
Cooperative Oncology Group (ECOG) performance status (PS),
histological type, smoking history, number of prior chemotherapy
regimens, and EGFR mutation if possible. Serum CEA (normal range,
0–5 ng/ ml) and CYFRA 21-1 (normal range, 0–3.3 ng/ml) were
measured using a chemiluminescence enzyme immunoassay kit (Beckman
Coulter, MN, USA) and an electrochemiluminescence immunoassay on an
automatic analyzer (Elecsys 200; Roche Diagnostics Mannheim, Basel,
Switzerland), respectively, before TKI treatment. Histological
analysis of the tumors was based on the WHO classification of cell
types (14). The clinical response
to the drug was defined according to the response evaluation
criteria of RECIST 1.0 for patients with measurable disease
(15). Nucleotide sequencing of
the kinase domain of EGFR (exons 18 to 21) was performed using
nested polymerase chain reaction (PCR) amplification of individual
exons. The details of sequencing have been described previously
(16). This study was approved by
the Institutional Review Board of the Yonsei University Health
System (approval no. 4-2009-0700).
Statistical methods
The association between pre-treatment levels of CEA
and CYFRA 21-1 and other categorical clinical variables were
compared using the Pearson's Chi-square test. Progression-free
survival (PFS) was defined as the time from the start day of TKI
treatment until the date of tumor progression or death. Overall
survival (OS) was measured from the date of diagnosis to the date
of death or final follow-up. The survival data were estimated using
a Kaplan-Meier curve and compared using the log-rank test.
Multivariate analyses were performed to find prognostic markers
using Cox's proportional hazards model. A P-value <0.05 was
considered statistically significant.
Results
Patient characteristics
The clinicopathological characteristics of the 123
patients are summarized in Table
I. Notably, a a high serum CEA (h-CEA) level (≥5 ng/ml) was
observed in 70 (56.9%) patients, and was significantly more
frequent in females, patients with adenocarcinoma and patients
without a history of smoking. On the other hand, 64 (52%) patients
had an elevated serum CYFRA 21-1 (h-CYFRA 21-1) level (≥3.3 ng/ml),
which was significantly more frequent in patients with a poor ECOG
PS (P=0.017) and in those with a history of smoking (P=0.072).
There was no difference in either CEA or CYFRA 21-1 levels in terms
of EGFR mutation status.
 | Table I.Comparison of pre-treatment
clinicopathological characteristics according to CEA and CYFRA 21-1
levels. |
Table I.
Comparison of pre-treatment
clinicopathological characteristics according to CEA and CYFRA 21-1
levels.
| | CEA, n (%)
| CYFRA 21-1, n (%)
|
|---|
| Patient
characteristics | n (%) | <5 ng/ml | ≥5 ng/ml | P-value | <3.3 ng/ml | ≥3.3 ng/ml | P-value |
|---|
| Total | 123 (100) | 53 (43.1) | 70 (56.9) | | 59 (48) | 64 (52) | |
| Age (years) | | | | | | | |
| Median (range) | 55 (34–88) | | | 0.265 | | | 0.451 |
| <65 | 81 (65.9) | 32 (39.5) | 49 (60.5) | | 41 (50.6) | 40 (49.4) | |
| ≥65 | 42 (34.1) | 21 (50.0) | 21 (50.0) | | 18 (42.9) | 24 (57.1) | |
| Gender | | | | <0.001 | | | 0.192 |
| Male | 70 (56.9) | 40 (57.2) | 30 (42.9) | | 30 (42.9) | 40 (57.1) | |
| Female | 53 (43.1) | 13 (24.5) | 40 (75.5) | | 29 (54.7) | 24 (45.3) | |
| Histologic
type | | | | 0.043 | | | 0.995 |
|
Adenocarcinoma | 73 (59.3) | 26 (35.6) | 47 (64.4) | | 35 (47.9) | 38 (52.1) | |
|
Non-adenocarcinoma | 50 (40.7) | 27 (54.0) | 23 (46.0) | | 24 (48.0) | 52 (52.0) | |
| Clinical stage | | | | 0.439 | | | 0.628 |
| IIIB | 35 (18.5) | 17 (48.6) | 18 (51.4) | | 18 (51.4) | 17 (48.6) | |
| IV | 88 (71.5) | 36 (40.9) | 52 (59.1) | | 41 (46.6) | 47 (53.4) | |
| Performance
status | | | | 0.100 | | | 0.017 |
| 0–1 | 83 (67.5) | 40 (48.2) | 43 (51.8) | | 46 (55.4) | 37 (44.6) | |
| 2 | 40 (32.5) | 13 (32.5) | 27 (67.5) | | 13 (32.5) | 27 (67.5) | |
| Smoking
history | | | | 0.036 | | | 0.072 |
| None | 59 (47.5) | 19 (32.8) | 39 (67.2) | | 33 (56.9) | 25 (43.1) | |
| Current +
former | 64 (52.5) | 33 (51.6) | 31 (48.2) | | 26 (40.6) | 38 (59.4) | |
| No. of prior
regimens | | | | 0.631 | | | 0.485 |
| ≤1 | 40 (32.5) | 16 (40.0) | 24 (60.0) | | 21 (52.5) | 19 (47.5) | |
| ≥2 | 83 (67.5) | 37 (44.6) | 46 (55.4) | | 38 (45.8) | 45 (54.2) | |
| TKI | | | | 0.669 | | | 0.203 |
| Gefitinib | 72 (58.5) | 29 (40.3) | 43 (61.4) | | 37 (51.4) | 35 (48.6) | |
| Erlotinib | 51 (41.5) | 24 (47.1) | 27 (52.9) | | 22 (43.1) | 29 (56.9) | |
| EGFR mutation
(n=84) | | | | 0.418 | | | 0.789 |
| Negative | 47 (38.2) | 19 (40.4) | 28 (59.6) | | 23 (48.9) | 24 (51.1) | |
| Positive | 37 (30.1) | 14 (37.8) | 23 (62.2) | | 19 (51.4) | 18 (48.6) | |
| Unknown | 39 (31.7) | 20 (51.3) | 19 (48.7) | | 17 (43.6) | 22 (56.4) | |
Association of serum markers with
responses to EGFR TKIs
The median follow-up duration was 9.0 months (range,
0.2–43 months). The median PFS was 5.0 months (95% CI, 3.3–6.7
months), and the median OS was 16.0 months (95% CI, 8.7–23.3
months). Responses were not assessable in 7 patients; 4 patients
died and 3 patients refused treatment before response evaluation.
Thirty-two of the evaluable 116 (27.6%) patients showed partial
responses. The response rate to EGFR TKIs was significantly higher
in patients with adenocarcinoma, an EGFR mutation, and a h-CEA (≥5
ng/ ml) serum level. The disease control rate in the patients with
h-CEA levels was significantly higher than those with low CEA
(l-CEA) levels (75 vs. 51.9%, P=0.034). There were no differences
in the response rates according to gender, smoking history, or the
number of prior chemotherapy regimens. There was a trend towards a
better response rate in patients with low CYFRA 21-1 (l-CYFRA)
levels (P=0.104). To evaluate whether the combination of CEA and
CYFRA 21-1 levels improved the prediction accuracy, patients were
divided into three groups according to their CEA and CYFRA 21-1
levels. Patients with a l-CEA and a h-CYFRA 21-1 level were defined
as group A (CEA <5 ng/ml and CYFRA 21-1 ≥3.3 ng/ ml, n=24),
while those with l-CEA and l-CYFRA 21-1 levels or h-CEA and h-CYFRA
21-1 levels were considered group B (CEA <5 ng/ml and CYFRA 21-1
<3.3 ng/ml, or CEA ≥5 ng/ ml and CYFRA 21-1 ≥3.3 ng/ml, n=66).
Finally, patients with h-CEA and l-CYFRA 21-1 levels were defined
as group C (CEA ≥5 ng/ml and CYFRA 21-1 <3.3 ng/ml, n=26). The
three groups showed significantly different response rates, with
the most favorable responses noted in group C (42.3 vs. 25.8 vs.
16.7%, P=0.005, for groups C, B and A, respectively) (Table II).
 | Table II.Comparison of pretreatment
clinicopathological characteristics according to EGFR TKI
responses. |
Table II.
Comparison of pretreatment
clinicopathological characteristics according to EGFR TKI
responses.
| Patient
characteristics | PR, n (%) | SD, n (%) | PD, n (%) | P-value |
|---|
| Total (n=116) | 32 (27.6) | 43 (37.1) | 41 (35.3) | |
| Age (years) | | | | |
| <65 | 20 (25.6) | 31 (39.8) | 27 (34.6) | 0.663 |
| ≥65 | 12 (31.6) | 12 (31.6) | 14 (36.8) | |
| Gender | | | | |
| Male | 16 (24.2) | 28 (42.4) | 22 (33.4) | 0.371 |
| Female | 16 (32.0) | 15 (30.0) | 19 (38.0) | |
| Histologic
type | | | | |
|
Adenocarcinoma | 25 (35.2) | 19 (26.8) | 27 (38.0) | 0.009 |
|
Non-adenocarcinoma | 7 (15.6) | 24 (53.3) | 14 (31.1) | |
| Performance
status | | | | |
| 0–1 | 26 (32.9) | 30 (38.0) | 23 (29.1) | 0.07 |
| 2 | 6 (16.2) | 13 (35.2) | 18 (48.6) | |
| Smoking
history | | | | |
| None | 17 (30.9) | 18 (32.7) | 20 (36.4) | 0.673 |
| Current +
former | 15 (25.0) | 24 (40.0) | 21 (35.0) | |
| No. of prior
regimens | | | | |
| ≤1 | 13 (35.1) | 13 (35.1) | 11 (29.8) | 0.436 |
| ≥2 | 19 (24.1) | 30 (38.0) | 30 (38.0) | |
| Serum CEA level
(ng/ml) | | | | |
| <5 | 11 (21.1) | 16 (30.8) | 25 (48.1) | 0.034 |
| ≥5 | 21 (32.8) | 27 (42.2) | 16 (25.0) | |
| Serum CYFRA 21-1
level (ng/ml) | | | | |
| <3.3 | 18 (32.7) | 23 (41.8) | 14 (25.5) | 0.104 |
| ≥3.3 | 14 (23.0) | 20 (32.8) | 27 (44.2) | |
| Combination of CEA
and CYFRA 21-1 | | | | |
| Group C | 11 (42.3) | 12 (46.2) | 3 (11.5) | 0.005 |
| Group B | 17 (25.8) | 26 (39.4) | 23 (34.8) | |
| Group A | 4 (16.7) | 5 (20.8) | 15 (62.5) | |
| EGFR mutation
(n=84) | | | | |
| Negative | 7 (15.6) | 12 (26.7) | 26 (57.8) | <0.001 |
| Positive | 18 (52.9) | 10 (29.4) | 6 (17.6) | |
Association of serum markers with
survival
Patients with a h-CEA level had significantly better
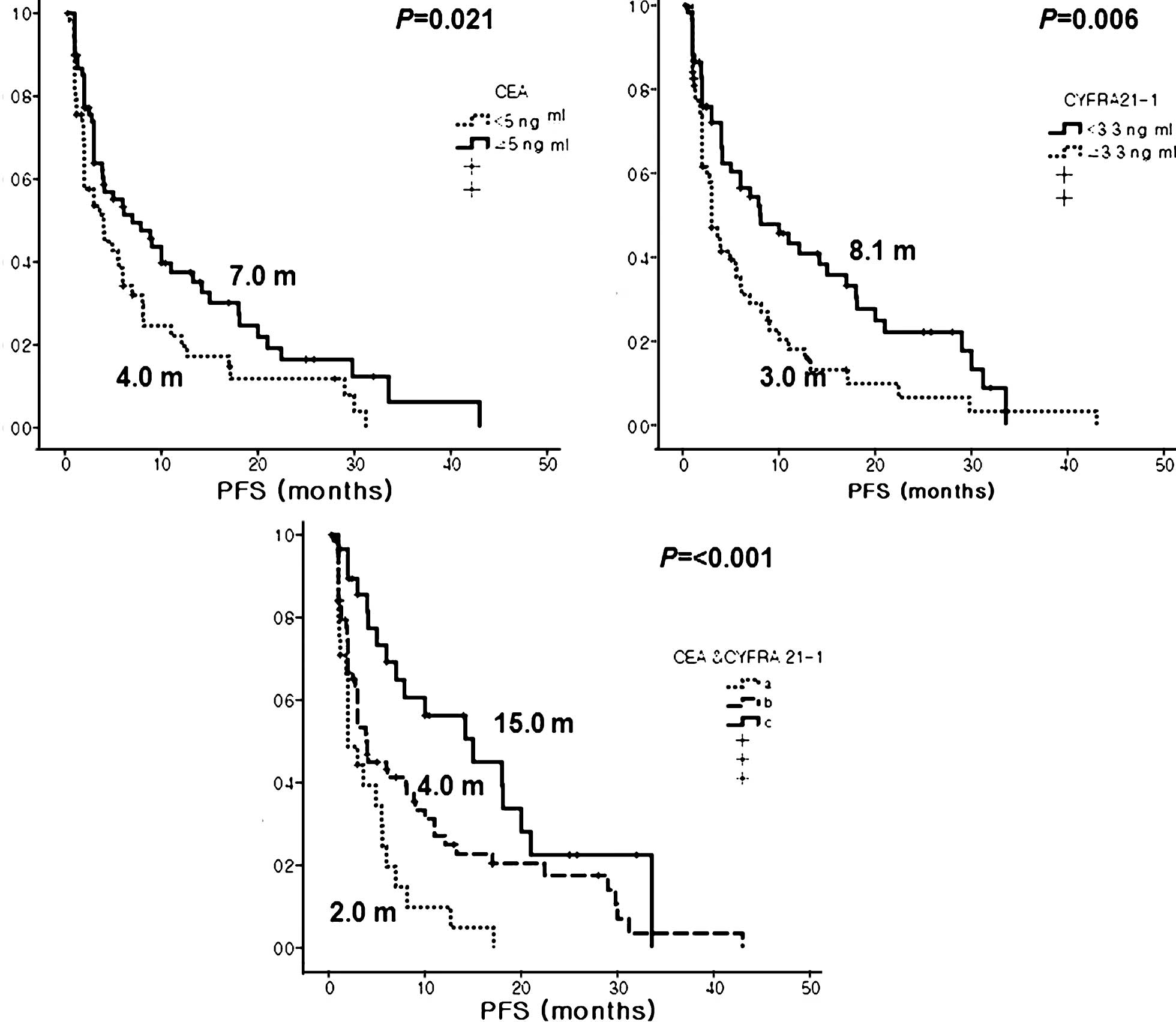
PFS than those with a l-CEA level (7.0 vs. 4.0 months, P=0.021). In
contrast, patients with a l-CYFRA 21-1 level also had significantly
better PFS than those with h-CYFRA 21-1 (8.1 vs. 3.0 months,
P=0.006). When subgrouped by combined CEA and CYFRA 21-1 levels,
the three groups showed significantly different PFS, and group C
showed the longest PFS among the three groups (15.0 vs. 4.0 vs. 2.0
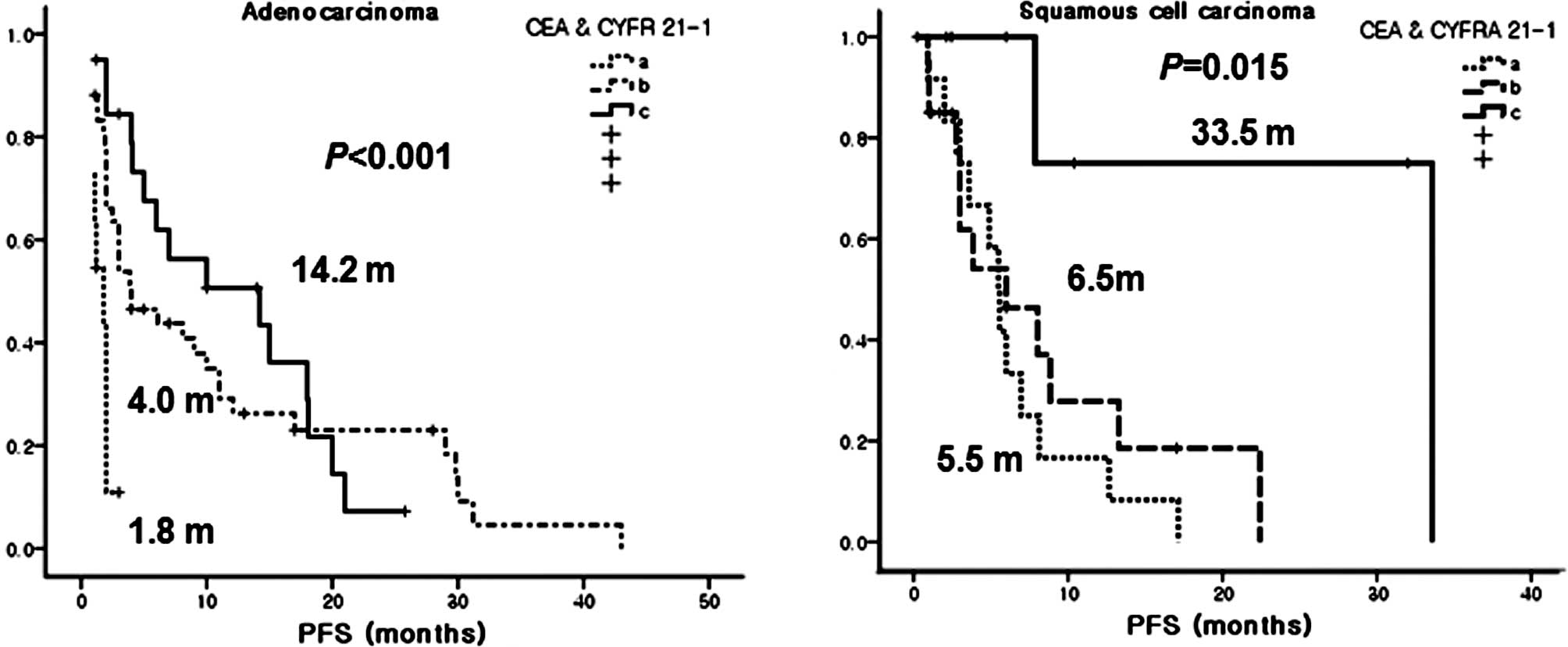
months, P<0.001, for groups C, B and A, respectively) (Fig. 1). Particularly, group C had the
longest PFS among the patients with squamous cell carcinoma
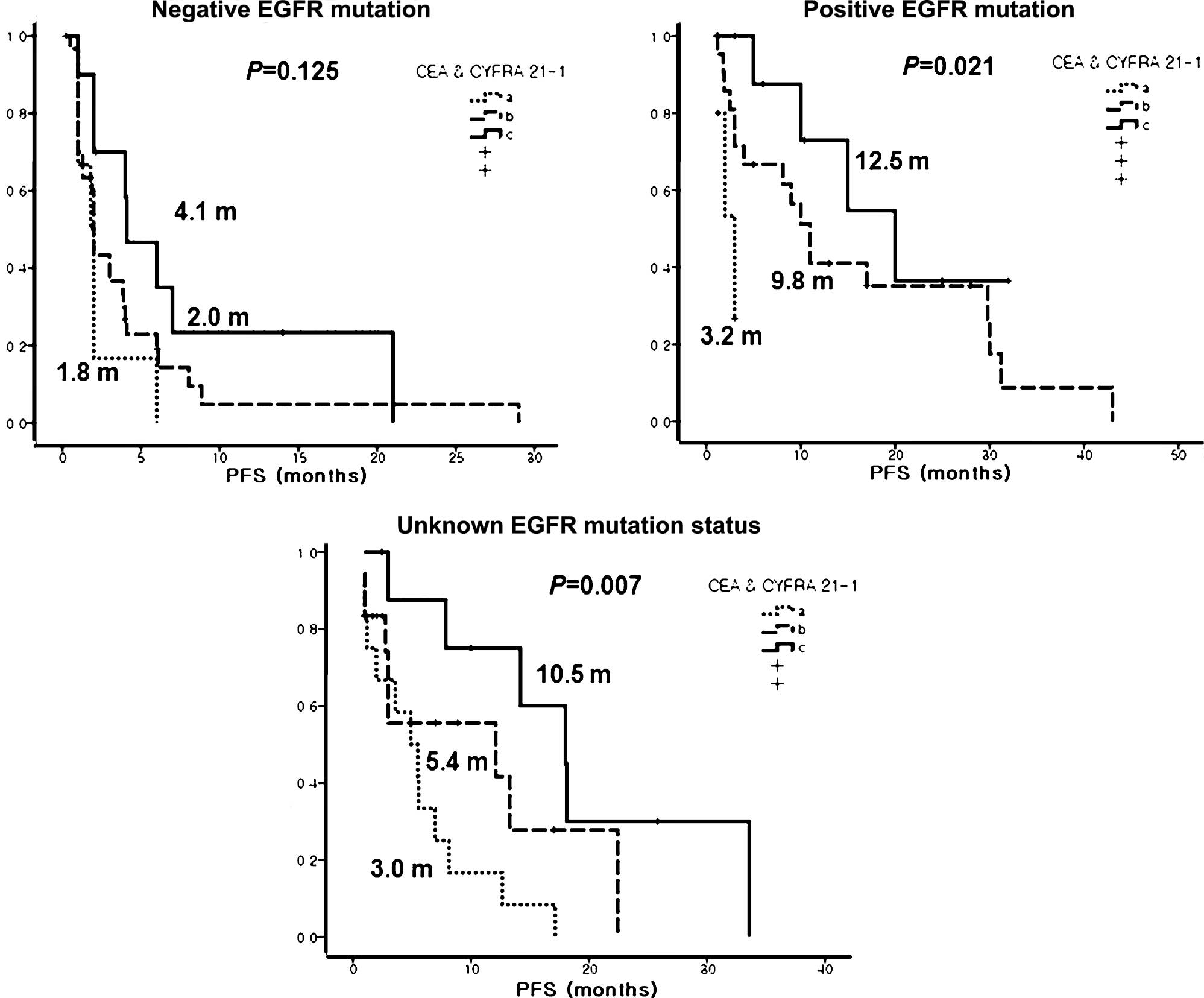
(Fig. 2). In addition, a h-CEA and
a l-CYFRA 21-1 level was a significant prognostic marker, not only
in patients with EGFR-mutant tumors, but in patients with an
unknown EGFR mutation status (Fig.
3). Finally, univariate analysis revealed several significant
factors for PFS including good ECOG PS (6.1 vs. 3.0 months,
P=0.016) and positive EGFR mutation status (11.0 vs. 2.0 months,
P<0.001) (Table III).
 | Table III.Univariate predictions of
survival. |
Table III.
Univariate predictions of
survival.
| PFS
| OS
|
|---|
| Category | Median
(months) | 95% CI | P-value | Median
(months) | 95% CI | P-value |
|---|
| Total | 5.0 | 3.3–6.7 | | 16.0 | 8.7–23.3 | |
| Age (years) | | | | | | |
| <65 | 4.1 | 2.0–6.2 | 0.982 | 15.1 | 10.5–19.7 | 0.843 |
| ≥65 | 5.6 | 2.8–8.4 | | 22.0 | 5.5-38-5 | |
| Gender | | | | | | |
| Male | 4.1 | 2.7–5.6 | 0.985 | 15.1 | 9.3–20.9 | 0.902 |
| Female | 6.0 | 2.5–9.50 | | 18.1 | 5.7–30.6 | |
| Histologic
type | | | | | | |
|
Adenocarcinoma | 5.6 | 3.3–7.8 | 0.942 | 18.1 | 0.0–36.3 | 0.716 |
|
Non-adenocarcinoma | 4.1 | 1.2–7.0 | | 16.0 | 8.3–23.7 | |
| Performance
status | | | | | | |
| 0–1 | 6.1 | 3.2–9.0 | 0.016 | 29.6 | 19.9–39.4 | <0.001 |
| 2 | 3.0 | 1.0–5.0 | | 6.1 | 1.7–10.6 | |
| Smoking
history | | | | | | |
| None | 5.0 | 2.8–7.2 | 0.331 | 16.0 | 7.0–25.0 | 0.780 |
| Current +
former | 4.9 | 3.1–6.7 | | 14.1 | 1.7–26.5 | |
| No. of prior
regimens | | | | | | |
| 0–1 | 8.1 | 3.2–13.0 | 0.176 | 29.6 | 3.3–55.3 | 0.447 |
| ≥ 2 | 4.0 | 2.0–6.0 | | 15.0 | 9.1–20.9 | |
| TKI | | | | | | |
| Gefitinib | 5.6 | 3.6–7.5 | 0.679 | 16.0 | 8.9–23.1 | 0.935 |
| Erlotinib | 3.9 | 1.5–6.3 | | 24.0 | 13.2–34.9 | |
| Serum CEA level
(ng/ml) | | | | | | |
| <5 | 4.0 | 1.7–6.3 | 0.021 | 14.0 | 2.2–25.8 | 0.505 |
| ≥5 | 7.0 | 2.5–11.5 | | 18.0 | 10.3–25.7 | |
| Serum CYFRA 21-1
level (ng/ml) | | | | | | |
| <3.3 | 8.1 | 2.9–13.3 | 0.006 | NR | | <0.001 |
| ≥3.3 | 3.0 | 2.0–4.0 | | 8.0 | 5.2–10.8 | |
| Combination of CEA
and CYFRA 21-1 (ng/ml) | | | | | | |
| CEA ≥5 and CYFRA
21-1 <3.3 | 15.0 | 5.7–24.3 | <0.001 | NR | | 0.002 |
| CEA <5 and
CYFRA 21-1 <3.3 or | 4.0 | 3.1–4.9 | | 14.1 | 5.0–23.2 | |
| CEA ≥5 and CYFRA
21-1 ≥3.3 | | | | | | |
| CEA <5 and
CYFRA 21-1 >3.3 | 2.0 | 0.9–3.1 | | 8.0 | 4.8–11.2 | |
| EGFR mutation
(n=84) | | | | | | |
| Negative | 2.0 | 1.4–2.7 | <0.001 | 7.1 | 3.9–10.3 | 0.038 |
| Positive | 11.0 | 5.3–16.7 | | 22.0 | 13.1–31.0 | |
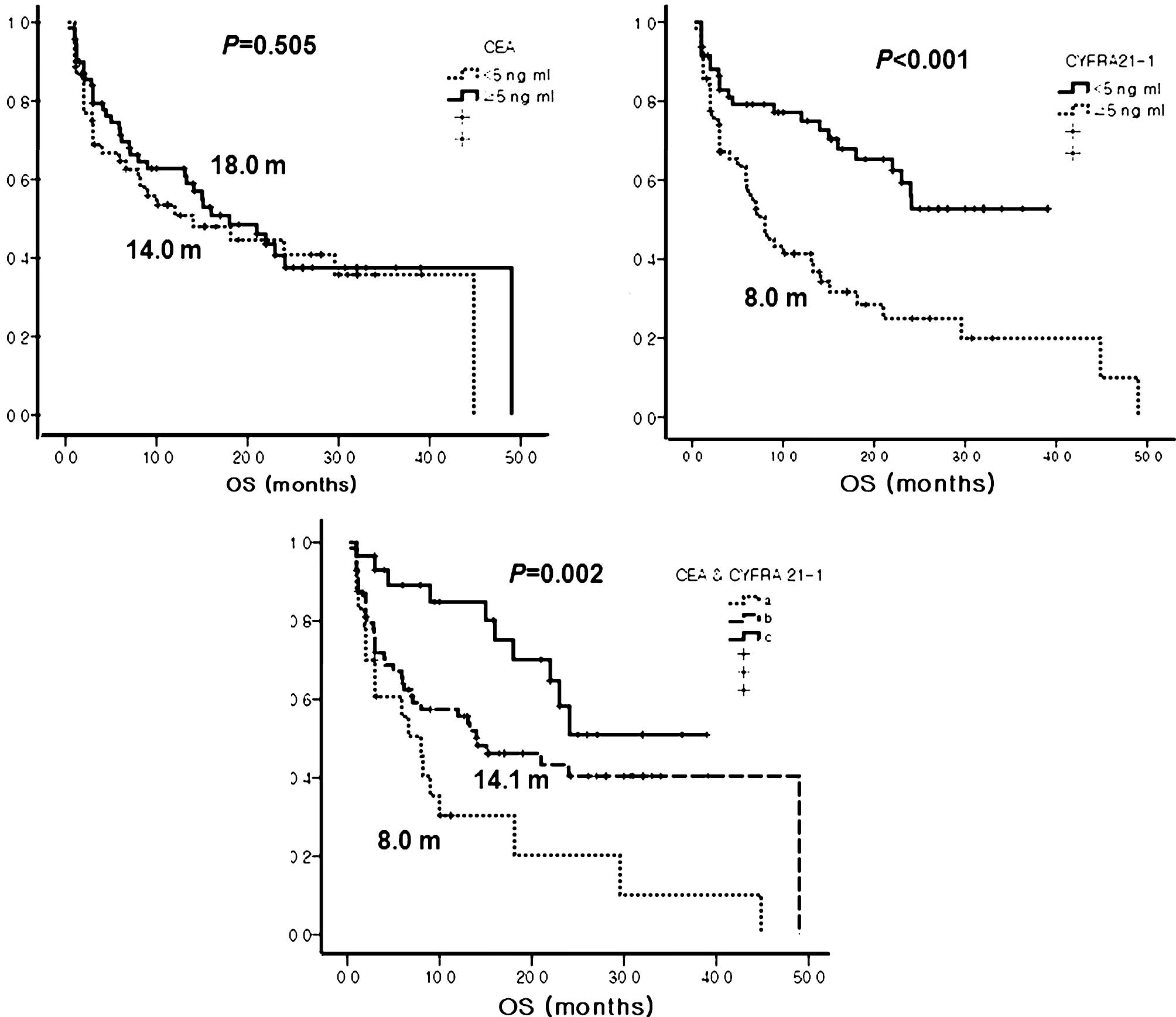
Patients with good ECOG PS and a positive EGFR
mutation status also had significantly longer OS than those who had
a poor ECOG PS and a negative EGFR mutation status (ECOG PS, 29.6
vs. 6.1 months, P<0.001; EGFR mutation status, 22.0 vs. 7.1
months, P=0.038, respectively). However, OS did not differ
according to pre-treatment CEA levels. Patients with a l-CYFRA 21-1
level had a longer OS than those with h-CYFRA 21-1 (not reached vs.
8.0 months, P<0.001). Patients in group C also had the longest
OS among the three groups (Table
IV and Fig. 4).
 | Table IV.Multivariate predictions of
survival. |
Table IV.
Multivariate predictions of
survival.
| PFS
| OS
|
|---|
| Category | Hazard ratio | 95% CI | P-value | Hazard ratio | 95% CI | P-value |
|---|
| Age (years) | | | | | | |
| <65 vs.
≥65 | 1.23 | 0.67–2.28 | 0.506 | 1.18 | 0.64–2.29 | 0.633 |
| Gender | | | | | | |
| Female vs.
male | 2.86 | 0.75–10.89 | 0.124 | 1.24 | 0.27–6.75 | 0.808 |
| Histologic
type | | | | | | |
| Non-adeno vs.
adeno | 0.80 | 0.41–1.56 | 0.521 | 1.59 | 0.81–3.14 | 0.182 |
| Clinical stage | | | | | | |
| IV vs. III | 0.64 | 0.35–1.15 | 0.134 | 0.80 | 0.40–1.60 | 0.534 |
| Performance
status | | | | | | |
| 0–1 vs. 2 | 2.02 | 1.13–3.61 | 0.017 | 2.13 | 1.14–3.98 | 0.018 |
| Smoking
history | | | | | | |
| None vs. current
+ former | 1.48 | 0.39–5.56 | 0.57 | 1.38 | 0.25–7.61 | 0.706 |
| Serum CEA level
(ng/ml) | | | | | | |
| <5 vs. ≥5 | 0.41 | 0.24–0.78 | 0.007 | 0.55 | 0.25–1.21 | 0.554 |
| Serum CYFRA 21-1
level (ng/ml) | | | | | | |
| <3.3 vs.
≥3.3 | 1.93 | 1.09–3.44 | 0.025 | 2.76 | 1.38–5.53 | 0.004 |
| EGFR mutation
(n=84) | | | | | | |
| Negative vs.
positive | 0.22 | 0.11–0.42 | <0.001 | 0.53 | 0.28–1.004 | 0.051 |
Multivariate analysis using a Cox proportional
hazards model indicated that a good ECOG PS, positive EGFR mutation
status, high pre-treatment CEA levels, and low pre-treatment CYFRA
21-1 levels are independent predictive factors for PFS. Meanwhile,
predictive factors for OS included a good ECOG PS, positive EGFR
mutation status and l-CYFRA 21-1, but not h-CEA (Table IV).
Discussion
Detection of a mutation in the EGFR gene in NSCLC
patients treated with an EGFR TKI is the most important factor for
the prediction of a good response to these drugs (4). However, the detection of an EGFR
mutation may be difficult due to the limited amount of available
tissue (2–4). Therefore, a surrogate biomarker that
can improve the prediction of response to these targeted drugs is
needed.
CEA was first described by Gold and Freedman in 1965
as an antigen expressed by gastrointestinal carcinoma cells
(17). Although CEA was often
falsely elevated in smokers and in patients with restrictive or
obstructive pulmonary disease (18–20),
abnormally elevated CEA levels were reported in 30–70% of patients
with NSCLC and were most frequently observed in patients with
adenocarcinoma and advanced stage carcinoma (21). In addition, several studies have
shown that h-CEA is a potential marker of poor prognosis in NSCLC
regardless of treatment (7,21).
On the contrary, Okamoto et al (11) reported that patients treated with
EGFR TKIs with high pre-treatment levels of CEA had a longer
survival and a better response than those with l-CEA. They
attributed this to a possible anti-apoptotic signal of the mutant
EGFR pathway that may elevate the expression level of CEA protein.
Our data are similar to the data of Okamato et al (11). Shoji et al (22) reported that the rate of the EGFR
gene mutation significantly increased as the serum CEA level
increased (for serum CEA levels of <5, ≥5 but <20, and ≥20,
the rate of the EGFR gene mutation was 35, 55 and 87.5%,
respectively; P=0.040). However, our data showed that the status of
the EGFR mutation made no difference in the CEA levels. Based on
previous reports, the function of CEA has not been elucidated but
may include the following: i) CEA is a cell surface adhesion
protein and may play a role in cell-to-cell adhesion (23); ii) overexpression of CEA is thought
to play a role in tumorigenesis (24); iii) CEA has a dominant effect in
blocking differentiation, and it also cooperates with Myc and Bcl-2
in cellular transformation (25);
and iv) it can inhibit cell death induced by a loss of anchorage to
the extracellular matrix (anoikis) (26). Although these findings suggest that
CEA has anti-apoptotic effects in cancer cells, a direct
relationship between h-CEA and response to EGFR TKIs has not yet
been established.
CYFRA 21-1, a fragment of cytokeratin subunit 19,
was first identified in 1993 as a valuable marker in lung cancer
patients (27). CYFRA 21-1 was
found to be associated with TNM stage and ECOG PS, reflecting an
unfavorable prognosis for NSCLC patients regardless of treatment
(8,21,28–30).
In our study, patients with a poor ECOG PS had a higher CYFRA 21-1
level than patients with a good ECOG PS (6.4 vs. 3.0 ng/ml;
P=0.03). Patients with h-CYFRA 21-1 levels were more likely to have
a history of smoking; however, this association was not significant
(P=0.072). Previous studies have also reported that smoking has no
effect on serum CYFRA 21-1 levels (31,32).
Univariate and multivariate analyses demonstrated that CYFRA 21-1
levels higher than 3.3 ng/ ml had an independent negative impact on
PFS (HR=1.93, 95% CI 1.09–3.44; P=0.025) and OS (HR=2.76, 95% CI
1.38–5.53; P=0.0004). Therefore, CYFRA 21-1 is an independent
marker for poor prognosis in NSCLC patients receiving an EGFR TKI,
which is consistent with a previous study (12).
We demonstrated that pre-treatment levels of CEA and
CYFRA 21-1 serve as prognostic and predictive markers in NSCLC
patients treated with gefitinib or erlotinib. Patients with a high
pre-treatment CEA level showed better responses and longer PFS, and
patients with a low pre-treatment CYFRA 21-1 level showed longer
PFS and OS. In addition, the prediction accuracy of the EGFR TKI
response and prognosis improved when all patients were divided into
three groups according to combined levels of CEA and CYFRA
21-1.
It is difficult to predict high efficacy of EGFR
TKIs when they are used in patients with non-adenocarcinoma
histology since the incidence of EGFR mutation is extremely rare in
these tumors (33). However, the
present study revealed that CEA and CYFRA 21-1 levels can also be
prognostic markers in patients with squamous cell carcinoma or
patients with unknown EGFR mutation status (Figs. 2 and 3).
In conclusion, pre-treatment serum levels of CEA and
CYFRA 21-1 are simple and easy to detect, and can serve as
predictive and prognostic factors for advanced NSCLC patients being
treated with EGFR TKIs, particularly in patients with squamous cell
carcinoma or patients with an unknown EGFR mutation status.
Acknowledgements
This study was supported by a faculty
research grant of Yonsei University College of Medicine for
6-2007-0194.
References
|
1.
|
Jemal A, Siegel R, Ward E, et al: Cancer
statistics, 2008. CA Cancer J Clin. 58:71–96. 2008. View Article : Google Scholar
|
|
2.
|
Shepherd FA, Rodrigues Pereira J, Ciuleanu
T, et al: Erlotinib in previously treated non-small-cell lung
cancer. N Engl J Med. 353:123–132. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Kim ES, Hirsh V, Mok T, et al: Gefitinib
versus docetaxel in previously treated non-small-cell lung cancer
(INTEREST): a randomised phase III trial. Lancet. 372:1809–1818.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Mok TS, Wu Y, Thongprasert S, et al:
Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N
Engl J Med. 361:947–957. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Maheswaran S, Sequist LV, Nagrath S, et
al: Detection of mutations in EGFR in circulating lung-cancer
cells. N Engl J Med. 359:366–377. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Molina R, Auge JM, Escudero JM, et al:
Mucins CA 125, CA 19.9, CA 15.3 and TAG-72.3 as tumor markers in
patients with lung cancer: comparison with CYFRA 21-1, CEA, SCC and
NSE. Tumour Biol. 29:371–380. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Okada M, Nishio W, Sakamoto T, et al:
Prognostic significance of perioperative serum carcinoembryonic
antigen in non-small cell lung cancer: analysis of 1,000
consecutive resections for clinical stage I disease. Ann Thorac
Surg. 78:216–221. 2004. View Article : Google Scholar
|
|
8.
|
Pujol JL, Boher JM, Grenier J and Quantin
X: CYFRA 21-1, neuron specific enolase and prognosis of non-small
cell lung cancer: prospective study in 621 patients. Lung Cancer.
31:221–231. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Holdenrieder S, von Pawel J, Dankelmann E,
et al: Nucleosomes and CYFRA 21-1 indicate tumor response after one
cycle of chemotherapy in recurrent non-small cell lung cancer. Lung
Cancer. 63:128–135. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Ardizzoni A, Cafferata MA, Tiseo M, et al:
Decline in serum carcinoembryonic antigen and cytokeratin 19
fragment during chemotherapy predicts objective response and
survival in patients with advanced non-small cell lung cancer.
Cancer. 107:2842–2849. 2006. View Article : Google Scholar
|
|
11.
|
Okamoto T, Nakamura T, Ikeda J, et al:
Serum carcinoembryonic antigen as a predictive marker for
sensitivity to gefitinib in advanced non-small cell lung cancer.
Eur J Cancer. 41:1286–1290. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Barlsi F, Tchouhadjian C, Doddoli C, Torre
JP, Astoul P and Kleisbauer JP: CYFRA 21-1 level predicts survival
in non-small cell lung cancer patients receiving gefitinib as
third-line therapy. Br J Cancer. 92:13–14. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Chiu C, Shih Y, Tsai C, Liou J, Chen Y and
Perng R: Serum tumor markers as predictors for survival in advanced
non-small cell lung cancer patients treated with gefitinib. Lung
Cancer. 57:213–221. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Travis WD, Colby TV, Corrin B, Shimosato Y
and Brambilla E: Histological Typing of Lung and Pleural Tumours.
3rd edition. Springer-Verlag; Berlin: 1999, View Article : Google Scholar
|
|
15.
|
Therasse P, Arbuck SG, Eisenhauer EA, et
al: New guidelines to evaluate the response to treatment in solid
tumors. European Organization for Research and Treatment of Cancer,
National Cancer Institute of the United States, National Cancer
Institute of Canada. J Natl Cancer Inst. 92:205–216. 2000.
View Article : Google Scholar
|
|
16.
|
Han S, Kim T, Hwang PG, et al: Predictive
and prognostic impact of epidermal growth factor receptor mutation
in non-small-cell lung cancer patients treated with gefitinib. J
Clin Oncol. 23:2493–2501. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Gold P and Freedman SO: Demonstration of
tumor-specific antigens in human colonic carcinomata by
immunological tolerance and absorption techniques. J Exp Med.
121:439–462. 1965. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Fujishima T, Honda Y, Shijubo N, Takahashi
H and Abe S: Increased carcinoembryonic antigen concentrations in
sera and bronchoalveolar lavage fluids of patients with pulmonary
alveolar proteinosis. Respiration. 62:317–321. 1995. View Article : Google Scholar
|
|
19.
|
Rule AH, Straus E, Vandevoorde J and
Janowitz HD: Tumor-associated (CEA-reacting) antigen in patients
with inflammatory bowel disease. N Engl J Med. 287:24–26. 1972.
View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Okada M, Nishio W, Sakamoto T, et al:
Effect of histologic type and smoking status on interpretation of
serum carcinoembryonic antigen value in non-small cell lung
carcinoma. Ann Thorac Surg. 78:1004–1009. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Matsuoka K, Sumitomo S, Nakashima N,
Nakajima D and Misaki N: Prognostic value of carcinoembryonic
antigen and CYFRA21-1 in patients with pathological stage I
non-small cell lung cancer. Eur J Cardiothorac Surg. 32:435–439.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
22.
|
Shoji F, Yoshino I, Yano T, et al: Serum
carcinoembryonic antigen level is associated with epidermal growth
factor receptor mutations in recurrent lung adenocarcinomas.
Cancer. 110:2793–2798. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Benchimol S, Fuks A, Jothy S, Beauchemin
N, Shirota K and Stanners CP: Carcinoembryonic antigen, a human
tumor marker, functions as an intercellular adhesion molecule.
Cell. 57:327–334. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Hammarström S: The carcinoembryonic
antigen (CEA) family: structures, suggested functions and
expression in normal and malignant tissues. Semin Cancer Biol.
9:67–81. 1999.PubMed/NCBI
|
|
25.
|
Screaton RA, Penn LZ and Stanners CP:
Carcinoembryonic antigen, a human tumor marker, cooperates with Myc
and Bcl-2 in cellular transformation. J Cell Biol. 137:939–952.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
26.
|
Ordoez C, Screaton RA, Ilantzis C and
Stanners CP: Human carcinoembryonic antigen functions as a general
inhibitor of anoikis. Cancer Res. 60:3419–3424. 2000.PubMed/NCBI
|
|
27.
|
Stieber P, Bodenmller H, Banauch D, et al:
Cytokeratin 19 fragments: a new marker for non-small cell lung
cancer. Clin Biochem. 26:301–304. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
28.
|
Nisman B, Lafair J, Heching N, et al:
Evaluation of tissue polypeptide-specific antigen, CYFRA 21-1, and
carcinoembryonic antigen in nonsmall cell lung carcinoma: Does the
combined use of cytokeratin markers give any additional
information? Cancer. 82:1850–1859. 1998. View Article : Google Scholar
|
|
29.
|
Barlsi F, Gimenez C, Torre J, et al:
Prognostic value of combination of CYFRA 21-1, CEA and NSE in
patients with advanced non-small cell lung cancer. Respir Med.
98:357–362. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Pujol JL, Molinier O, Ebert W, et al:
CYFRA 21-1 is a prognostic determinant in non-small-cell lung
cancer: results of a meta-analysis in 2063 patients. Br J Cancer.
90:2097–2105. 2004.PubMed/NCBI
|
|
31.
|
Kao CH, Hsieh JF, Ho YJ, Tsai SC and Lee
JK: Cytokeratin fragment 19 (CYFRA 21-1) in healthy smokers.
Anticancer Res. 19:4545–4546. 1999.PubMed/NCBI
|
|
32.
|
Molina R, Filella X, Aug JM, et al: Tumor
markers (CEA, CA 125, CYFRA 21-1, SCC and NSE) in patients with
non-small cell lung cancer as an aid in histological diagnosis and
prognosis. Comparison with the main clinical and pathological
prognostic factors. Tumour Biol. 24:209–218. 2003. View Article : Google Scholar
|
|
33.
|
Shigematsu H, Lin L, Takahashi T, et al:
Clinical and biological features associated with epidermal growth
factor receptor gene mutations in lung cancers. J Natl Cancer Inst.
97:339–346. 2005. View Article : Google Scholar : PubMed/NCBI
|