Introduction
The incidence of asthma in numerous countries is
increasing and the cause of the disease is complex (1,2).
Leukotrienes (LTs) play an important pro-inflammatory role in both
early-and late-phase asthmatic responses (3). LTs constitute a class of potent
biological mediators of inflammation and anaphylaxis.
5-Lipoxygenase (5-LO) is an essential enzyme which catalyzes the
first committed steps in the biosynthetic pathway leading to the
production of LTs (4–6). The 5-LO gene is located on chromosome
10q11.2 (7). In several studies,
the addition of an Sp-1 binding motif (-GGGCGG-) or the deletion of
one or two Sp-1 binding motifs in the 5-LO core promoter have been
shown to be associated with reduced gene expression (8,9). Our
previous study revealed that the E254K polymorphism in the 5-LO
gene may be associated with bronchial asthma (BA) in Japanese
children (10). However, few
studies have investigated the role of polymorphisms of the 5-LO
gene in a Chinese population. Therefore, it is necessary to
identify such single-nucleotide polymorphisms (SNPs) in the 5-LO
gene and to research the correlation between SNPs in the 5-LO gene
and the incidence of BA in the Chinese population.
Patients and methods
Patients and control subjects
A total of 179 Chinese Han children with BA (112
males and 67 females; mean age, 2.73±10.27 years), 36 adults with
BA (20 males and 16 females; mean age, 51±28 years), 50 Chinese
Mongolian children with BA (32 males and 18 females; mean age,
2.86±10.14 years), 50 non-allergic control Han children (30 males
and 20 females; mean age, 2.82±10.18 years) and 162 Han adult
controls (90 males and 72 females; mean age, 19.7±2.3 years) were
enrolled. The diagnosis of BA was made according to the criteria of
the American Thoracic Society (11). The controls were healthy and did
not have a history of allergic diseases. All subjects were randomly
selected among patients in our hospital or our college. Informed
consent was obtained from all individuals or their parents.
Peripheral blood collection and
separation of cells
Peripheral blood (2 ml) was obtained from each
patient and treated with 0.2% NaCl to collect leukocytes. Genomic
DNA was extracted from the leukocytes with a DNA Extract kit
(Bioteke, Beijing, China) (12).
Allele-specifc (AS)-PCR primer
design
AS-PCR is selective PCR amplification of one allele
and is used to detect SNPs. Selective amplification is usually
achieved by designing a primer such that it will match/mismatch one
of the alleles at the 3′ end and the penultimate base is designed
to incorporate the SNP in order to improve the specificity of the
primer. We designed the following primers: P1, 5′-cgc tgc aca gag
ctg cct g (254Glu); P2, 5′-cgc tgc aca gag ctg cct a (254Lys); P3,
5′-cgc aat tcc tcc tct gat gt (co-reverse primer); PCR product,
301-bp. We also designed a pair of reference primers: P4, 5′-aga
ggc gaa gtt ctc caa ca; P5, 5′-aac agg gac gga gag tga tg; PCR
product, 600-bp. The primer for c.780 G>A (c.780G) was 5′-aga
agc tcc cgg tga cca tg-3′ and that for c.780 G>A (c.780A) was
5′-aga agc tcc cgg tga cca ta-3′.
AS-PCR system and PCR conditions
P3, P4 and P5 primers were added to both A and B
tubes (P1,P3,P4 and P5 primers in A tube; P2,P3,P4 and P5 primers
in B tube). A and B tubes were prepared and added to 2 μl (10 μM)
wild-type P1 and mutant P2 primers, respectively. Two tubes were
added per 1 μl (10 μM) a pair of reference primers, P4 and P5, as
well as 2 μl (10 μM) common reverse primer P3 were also used. The
reaction mixture consisted of 25 μl 2X PCR Mastermix (Bioteke,
Beijing, China), 1 μl genomic DNA (100-150 ng/μl) and 18 μl
sterilized water in a total volume of 50 μl. The PCR conditions
were as follows: 95°C for 5 min, then 95°C for 30 sec, 56°C for 30
sec and 72°C for 1 min for 35 cycles, the last cycle after 72°C was
extended for 10 min. The products were stored at 4°C.
Detection of SNPs in the 5-LO gene by
sequencing
The 14 exons of the 5-LO gene were amplified using
the PCR technique and sequenced using an ABI 3100 DNA
auto-sequencer (Applied Biosystems, Carlsbad, CA, USA) in
individuals with BA (n=30) and controls (n=30). For further study,
the E254K and c.780 G>A substitution was detected by AS-PCR in
all individuals with BA (n=215) and controls (n=212) and the other
two silent polymorphisms (c.270 G>A, c.1728 A>G) were
detected in 65 individuals with BA and 56 controls. The primer
details for the PCR used in the detection of these polymorphisms
are shown in Table I.
 | Table IPrimer details for PCR to detect 4
polymorphisms by sequencing. |
Table I
Primer details for PCR to detect 4
polymorphisms by sequencing.
| Primer | Sequence | Amplified product
size (bp) | Annealing temperature
(°C) |
|---|
| c.270 G>A FP | 5′ CTC CAG AAC AAA
GGC TCA GG 3′ | 360 | 56 |
| c.270 G>A RP | 5′ CCT GCA CAG CAG
TGT CAT TC 3′ | | |
| c.760 G>A (E254K)
FP | 5′ GCA GGG ACT CTG
CTC TTA GG 3′ | 473 | 56 |
| c.760 G>A (E254K)
RP | 5′ CGC AAT TCC TCC
TCT GAT GT 3′ | | |
| c.780 G>A FP | 5′ GCA GGG ACT CTG
CTC TTA GG 3′ | 473 | 56 |
| c.780 G>A RP | 5′ CGC AAT TCC TCC
TCT GAT GT 3′ | | |
| c.1728 A>G FP | 5′ GAA AGA GGA TGG
ACG GAC TG 3′ | 295 | 55 |
| c.1728 A>G RP | 5′ CTC GTT TTC CTG
GAA CTG GC 3′ | | |
Statistical analyses
Allele and genotype frequencies were calculated for
each locus and tested for Hardy-Weinberg equilibrium. The
distribution of the genotype of E254K in the 5-LO gene was analyzed
by Fisher’s exact test. P<0.05 was considered to indicate a
statistically significant result. The significance of the
differences in clinical characteristics was analyzed using the
two-sample t-test.
Results
Polymorphisms in the 5-LO gene
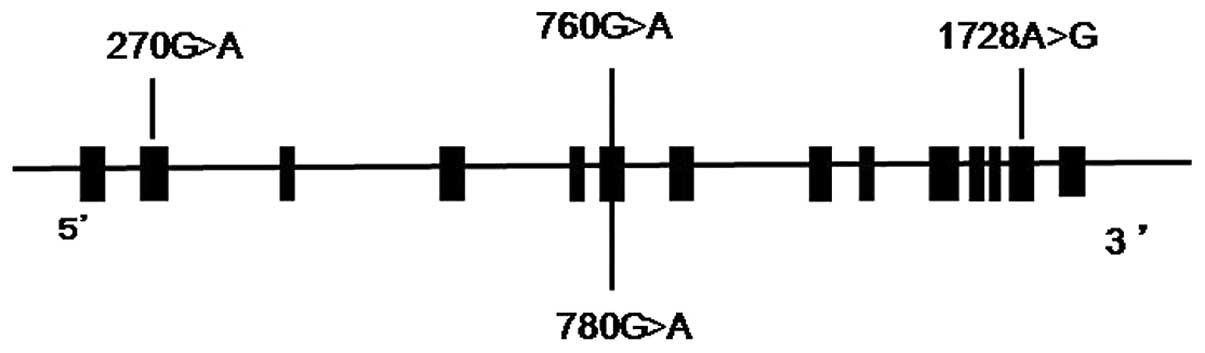
We identified 4 SNPs in the 5-LO gene in individuals
with BA (Fig. 1). Three SNPs were
silent polymorphisms [c.270 G>A (exon 2), c.780 G>A (exon 6)
and c.1728 A>G (exon 13)]. There were no differences in the
frequencies of these three SNPs between individuals with BA and
controls (Table II).
 | Table IIAllele and genotype frequencies of
5-LO silent SNPs in a Chinese Han population. |
Table II
Allele and genotype frequencies of
5-LO silent SNPs in a Chinese Han population.
| Frequency of 5-LO
SNPs
| |
|---|
| Allele/genotype | Non-allergic
controls | Bronchial asthma
patients | P-value |
|---|
| Allele | | | |
| c.270 G>A | n=56 | n=65 | |
| G | 101 (0.902) | 118 (0.908) | >0.05 |
| A | 11 (0.098) | 12 (0.092) | |
| c.780 G>A | n=212 | n=215 | |
| G | 413 (0.974) | 417 (0.970) | |
| A | 11 (0.026) | 13 (0.030) | >0.05 |
| c.1728 A>G | n=56 | n=65 | |
| A | 105 (0.938) | 126 (0.969) | >0.05 |
| G | 7 (0.058) | 4 (0.033) | |
| Genotype | | | |
| c.270 G>A | n=56 | n=65 | |
| GG | 45 (0.804) | 53 (0.815) | >0.05 |
| GA | 11 (0.196) | 12 (0.185) | |
| AA | 0 | 0 | |
| c.780 G>A | n=212 | n=215 | |
| GG | 201 (0.948) | 202 (0.940) | |
| GA | 11 (0.052) | 12 (0.056) | |
| AA | 0 (0) | 1 (0.005) | >0.05 |
| c.1728 A>G | n=56 | n=65 | |
| AA | 46 (0.821) | 54 (0.831) | >0.05 |
| AG | 10 (0.179) | 11 (0.169) | |
| GG | 0 | 0 | |
One SNP was a missense polymorphism and the amino
acid at 254 was changed from Glu (E) to Lys (K) (rs2228065). We
determined the prevalence of c.760 G>A (E254K) in the 5-LO genes
of individuals with BA and controls. This SNP was found in 15 (8
males and 7 females; mean age, 2.19±4.81 years; 0.070) out of the
215 individuals with BA and 6 (0.028) of the 212 controls. The
mutant allele frequency was 0.035 in 215 individuals with BA and
0.014 in the 212 controls (Table
III). There was a significant difference in the E254K frequency
between individuals with BA and controls (Fisher’s exact test,
P<0.05). We detected the E254K polymorphism in 50 Chinese
Mongolian BA patients by AS-PCR and it was present in 3
patients.
 | Table IIIAllele and genotype frequencies of
5-LO missense SNP in a Chinese Han population. |
Table III
Allele and genotype frequencies of
5-LO missense SNP in a Chinese Han population.
| Frequency of 5-LO SNP
| |
|---|
| Allele/genotype | Non-allergic controls
(n=212) | Bronchial asthma
patients (n=215) | P-value |
|---|
| c.760 G>A | | | |
| Allele | | | |
| G | 418 (0.986) | 415 (0.965) | |
| A | 6 (0.014) | 15 (0.035) | <0.05 |
| Genotype | | | |
| GG | 206 (0.972) | 200 (0.930) | |
| GA | 6 (0.028) | 15 (0.070) | <0.05 |
| AA | 0 | 0 (0) | |
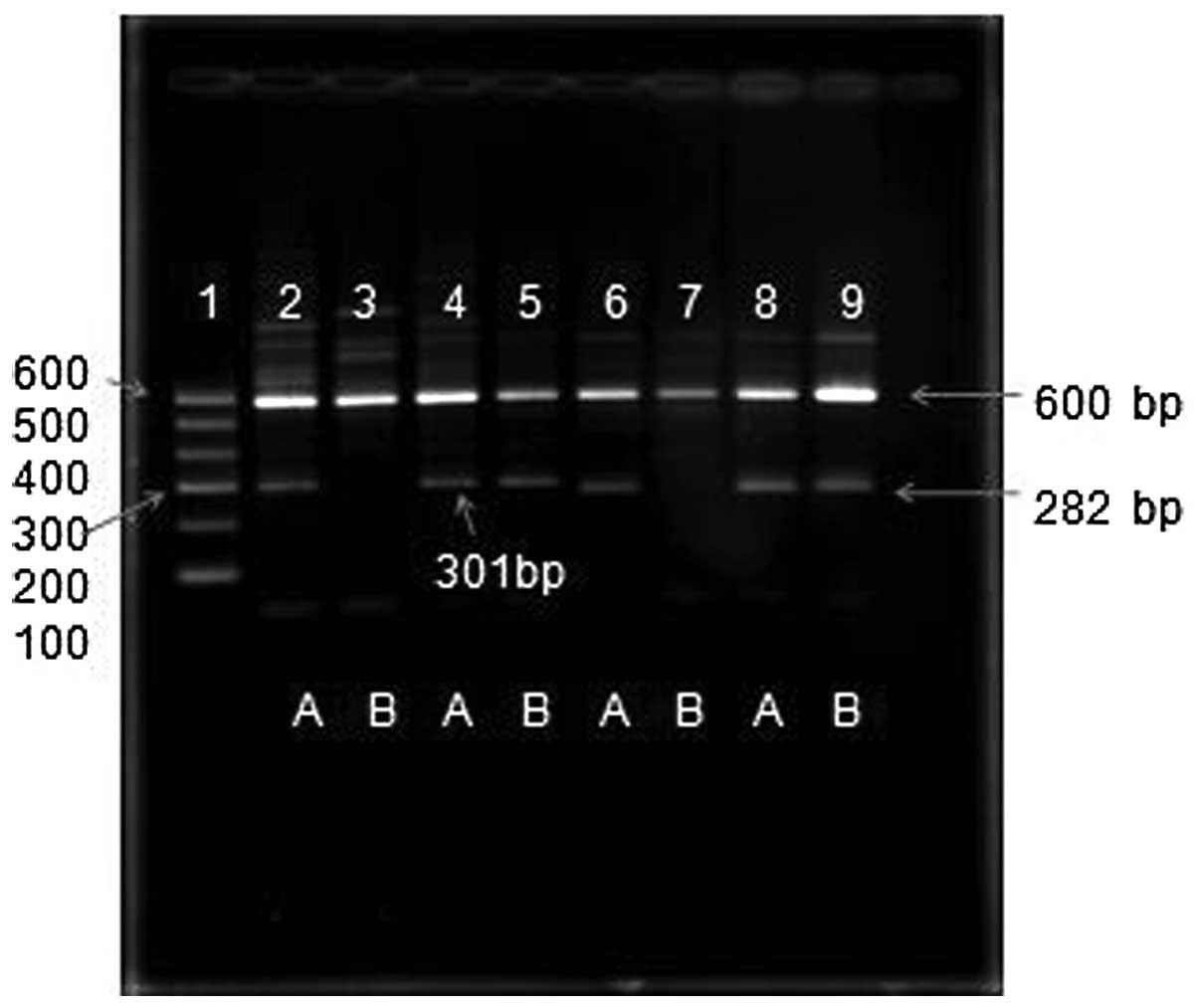
Agarose gel electrophoresis
DNA marker I (600, 500, 400, 300, 200 and 100 bp; 5
μl) and 10 μl AS-PCR products were analyzed by 2% agarose gel
electrophoresis. With our designed primers for AS-PCR, it was
possible to accurately and clearly detect the 5-LO gene rs2228065
(E254K) and rs116961353 (c.780G>A) polymorphisms, and the
genotype was directly distinguished (Fig. 2). Compared with the sequencing and
restriction enzyme method, this method is economical, quick and
simple.
Associations of E254K with clinical
characteristics
We compared the clinical characteristics with the
results of a routine blood test by automatic blood analyser (Sysmex
1800i;. Sysmex Corp., Kobe, Japan) among the controls and patients
with BA with and without E254K (Table
IV). The percentages of lymphocytes and monocytes and the
levels of platelets were significantly higher in individuals with
BA (without or with E254K) than in controls (P<0.05). However,
the percentage and number of neutrophils were significantly lower
in individuals with BA (without or with E254K) than in the controls
(P<0.05). No significant difference was identified between the
clinical features in BA patients with E254K compared with those
without E254K. The percentage of eosinophils (EO%) was
significantly lower in individuals with BA and E254K than in
controls (P=0.04). There was no significant difference in age or
gender between BA patients with and without E254K (P>0.05).
 | Table IVClinical characteristics of the
controls, BA patients with E254K and BA patients without E254K. |
Table IV
Clinical characteristics of the
controls, BA patients with E254K and BA patients without E254K.
| Clinical
characteristics | Normal range | Controls (n=50)
mean ± SD | BA with E254K
(n=14) mean ± SD | BA without E254K
(n=114) mean ± SD | P-value (t-test)
|
|---|
| P1 | P2 | P3 |
|---|
| WBC
(109/l) | 4–10 | 8.07±1.6 | 7.86±3.49 | 8.79±3.7 | 0.852 | 0.445 | 0.496 |
| LYMPH% | 20–40 | 30.94±15.72 | 61.72±15.71 | 54.81±18.72 | 2.89E-6 | 1.79E-6 | 0.314 |
| MONO% | 3–8 | 5.62±1.6 | 8.88±1.81 | 9.2±3.54 | 2.82E-4 | 1.29E-4 | 0.807 |
| NEUT% | 50–75 | 60.34±8.56 | 28.15±16.25 | 33.97±19.72 | 4.01E-6 | 6.50E-7 | 0.42 |
| EO% | 1–3 | 1.78±0.55 | 1.07±0.98 | 2.09±3.16 | 0.040 | 0.698 | 0.369 |
| BASO% | 0–1 | 0.25±0.16 | 0.26±0.2 | 0.31±0.3 | 0.888 | 0.450 | 0.666 |
| LYMPH#
(109/1) | 0.8–4 | 3.38±1.26 | 4.95±2.89 | 4.85±3.03 | 0.091 | 0.059 | 0.931 |
| MONO#
(109/1) | 0.12–1.8 | 0.61±0.38 | 0.66±0.38 | 0.80±0.44 | 0.742 | 0.094 | 0.365 |
| NEUT#
(109/1) | 2–7.5 | 4.86±2.35 | 2.16±1.35 | 2.92±2.14 | 0.009 | 0.001 | 0.325 |
| EO#
(109/1) | 0–0.45 | 0.11±0.10 | 0.08±0.07 | 0.18±0.27 | 0.426 | 0.351 | 0.310 |
| PLT
(109/l) | 100–300 | 238.63±65.08 | 333.38±129.46 | 320.35±106.32 | 0.032 | 0.004 | 0.744 |
Discussion
Allergic diseases, including asthma and atopic
dermatitis, are complex genetic disorders that do not conform to a
simple Mendelian pattern of inheritance (13). The etiology is extremely complex
and the incidence of asthma is correlated with environmental and
genetic factors. Clinically similar asthma symptoms may be caused
by different mechanisms (14). In
this study, we studied polymorphisms of the 5-LO gene and attempted
to investigate the association of 5-LO gene polymorphisms with BA
in a Chinese Han population. We compared the difference in the 5-LO
gene polymorphism frequency between asthma patients and normal
controls and evaluated the correlation of the SNPs with asthma
onset, age, gender, severity of asthma and other clinical
characteristics of BA. We found a missense SNP and three silent
SNPs in the 5-LO gene in BA patients.
Our study showed that the c.760 G>A (E254K)
polymorphism in the 5-LO gene was associated with BA in a Chinese
Han population. There was a significant difference in the E254K
frequency between individuals with BA and controls (Fisher’s exact
test, P<0.05). Three other silent SNPs, rs2228064 (c.270
G>A), rs116961353 (c.780G>A) and rs2229136 (c.1728 A>G),
described previously (15), were
also identified, but there were no significant differences in the
frequencies between individuals with BA and controls (P>0.05).
We also detected the E254K polymorphism in Chinese Mongolian BA
patients by AS-PCR; there were 3 heterozygotes out of 50 patients.
Our results indicate that the 5-LO E254K polymorphism is present in
patients with BA in Japanese (in our previous research), Chinese
Han and Chinese Mongolian populations. The E254K allele and
genotype frequencies were significantly different in individuals
with BA compared with the control group. Our study further
confirmed that the 5-LO E254K polymorphism may be correlated with
BA.
To examine the functional effects of c.760 G>A
(E254K) in the 5-LO gene, we compared the clinical characteristics
with results from routine blood test reports performed in the last
three years in our hospital among the controls, BA with E254K and
BA without E254K. However, the clinical features (including the age
and gender) in our study were not significantly different between
individuals with BA with and without E254K (P>0.05). This result
may be due to the number of individuals with BA and E254K being too
low to obtain accurate statistical results.
The percentages of lymphocytes, monocytes and
platelets (109/l) levels were significantly higher in
individuals with BA (without or with E254K) than in controls
(P<0.05). Monocytes are the largest cells in normal blood. They
act as phagocytes in certain inflammatory diseases and are the
body’s second line of defense against infection (16). Lymphocytes are the primary
components of the body’s immune system and increase in number in
numerous viral infections and with tuberculosis (17). Inflammatory disorders may cause a
high platelet count in a similar way to infections. Our date
support the theory that inflammation is a cause of asthma
attack.
The percentage and number of neutrophils were
significantly lower in individuals with BA (without or with E254K)
than in controls (P<0.05). A decrease in neutrophil levels is
known as neutropenia. Although most bacterial infections stimulate
an increase in neutrophils, certain bacterial infections, including
typhoid fever and brucellosis, and numerous viral diseases,
including hepatitis, influenza, rubella, rubeola and mumps,
decrease the neutrophil count. Our data support the hypothesis that
viral infection is a cause of asthma attack.
The mean level of eosinophils showed a tendency to
increase in individuals with BA without E254K more than in
controls. However, the percent eosinophil levels were significantly
lower in individuals with BA and E254K than in controls (P=0.04).
Eosinophils are associated with antigen-antibody reactions
(18). The most common reasons for
an increase in the eosinophil count are allergic reactions such as
hay fever, asthma or drug hypersensitivity. Decreases in the
eosinophil count may be observed when a patient is receiving
corticosteroid drugs.
Pharmacogenetics is the study of how genetic
differences influence the variability of patients’ responses to
therapy (19). Therefore, we
intend to study the correlation between these four SNPs and
patients’ responses to therapy. With our designed primers for
AS-PCR, the detection of the 5-LO gene E254K and c.780G>A
polymorphisms is accurate and clear and the genotype may be
directly distinguished. Compared with the sequencing and
restriction enzyme method, our method was economic, quick and
simple.
In summary, our findings contribute to the
evaluation of one of the genetic risk factors for BA and we report
an accurate and simple method to quickly detect the 5-LO E254K
polymorphism. It is important to further study the correlation
between the response to 5-LO inhibitors with the E254K polymorphism
in the clinic. These results are important for clarifying the
different mechanisms of BA.
Abbreviations:
|
SNP,
|
single-nucleotide polymorphism;
|
|
5-LO,
|
5-lipoxygenase;
|
|
BA,
|
bronchial asthma;
|
|
LTs,
|
leukotrienes;
|
|
AS-PCR,
|
allele-specific PCR
|
Acknowledgements
This study was supported by the Inner
Mongolia Natural Science Research Grant (2009ZD07) of China.
References
|
1
|
Sanak M: Genetic Variance of
5-lipoxygenase Metabolic Pathway in Bronchial Asthma. International
Review of Asthma. 4:70–80. 2002.
|
|
2
|
Wong WS and Koh DS: Advances in
immunopharmacology of asthma. Biochem Pharmacol. 59:1323–1335.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen XS, Sheller JR, Johnson EN and Funk
CD: Role of leukotrienes revealed by targeted disruption of the
5-lipoxygenase gene. Nature. 372:179–182. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Samuelsson B: Leukotrienes: mediators of
immediate hypersensitivity reactions and inflammation. Science.
220:568–575. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Samuelsson B, Dahlén SE, Lindgren JA,
Rouzer CA and Serhan CN: Leukotrienes and lipoxins: structures,
biosynthesis, and biological effects. Science. 237:1171–1176. 1987.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lewis RA, Austen KF and Soberman RJ:
Leukotrienes and other products of the 5-lipoxygenase pathway.
Biochemistry and relation to pathobiology in human diseases. N EngI
J Med. 323:645–655. 1990. View Article : Google Scholar
|
|
7
|
Funk CD, Hoshiko S, Matsumoto T, Radmark O
and Samuelsson B: Characterization of the human 5-lipoxygenase
gene. Proc Natl Acad Sci USA. 86:2587–2591. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Drazen JM and Silverman ES: Genetic
determinants of 5-lipoxygenase transcription. Int Arch Allergy
Immunol. 118:275–278. 1999. View Article : Google Scholar
|
|
9
|
Silverman ES and Drazen JM: Genetic
variations in the 5-lipoxygenase core promoter. Description and
functional implications. Am J Respir Crit Care Med. 161:S77–S80.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bai C, Matsui E, Ohnishi H, et al: A novel
polymorphism, E254K, in the 5-lipoxygenase gene associated with
bronchial asthma. Int J Mol Med. 21:139–144. 2008.PubMed/NCBI
|
|
11
|
Proceedings of the ATS workshop on
refractory asthma. Am J Respir Crit Care Med. 162:2000.
|
|
12
|
Bai C, Zhou J, Rui Y, et al: Economical
and Effective Method for Extract Blood Genomic DNA. Chinese journal
of Laboratory Medicine and Clinic. 7:1795–1798. 2010.
|
|
13
|
Boyce JA, Broide D, Matsumoto K and
Bochner BS: Advances in mechanisms of asthma, allergy, and
immunology in 2008. J Allergy Clin Immunol. 123:569–574. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
De Caterina R and Zampolli A: From asthma
to atherosclerosis -5-lipoxygenase, leukotrienes, and inflammation.
New Eng J Med. 350:4–7. 2004.PubMed/NCBI
|
|
15
|
In KH, Silverman ES, Asano K, Beier D,
Fischer AR, Keith TP, et al: Mutations in the human 5-lipoxygenase
gene. Clin Rev Allergy Immunol. 17:59–69. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
van den Heuvel MM, Vanhee DD, Postmus PE,
et al: Functional and phenotypic differences of monocyte-derived
dendritic cells from allergic and nonallergic patients. Allergy
Clin Immunol. 101:90–95. 1998.PubMed/NCBI
|
|
17
|
Galli SJ: Complexity and reducdancy in the
pathogenesis of asthma: reassessing the roles of mast cells and T
cells. J Exp Med. 186:343–347. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Krouwels FH, Kerstens LC, van der Maarel
HW, Degenhart HJ and Neijens HJ: Density of eosinophils reflects
activity of disease in allergic asthmatic children. Clin Exp
Allergy. 25:1171–1178. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Berretta F, Butler RH, Diaz G, Sanarico N,
Arroyo J, Fraziano M, et al: Detailed analysis of the effects of
Glu/Lys beta69 human leukocyte antigen-DP polymorphism on
peptide-binding specificity. Tissue Antigens. 62:459–471. 2003.
View Article : Google Scholar : PubMed/NCBI
|