Introduction
Levamisole is a broad spectrum anthelmintic agent
which is primarily used for expelling Ascaris and hookworms.
In addition, it enhances the functions of phagocytes and T cells,
and strengthens immune function (1,2,3). As
a result, levamisole is also used as an immunoregulant for certain
immune-related diseases, in order to increase the resistance of
patients to bacterial and viral infections. Levamisole may be
applied in the treatment of nephritic syndrome (4), dermatological diseases (5), recurrent aphthous ulcers (6) and inflammatory bowel disease
(7). In addition, levamisole, in
combination with 5-fluorouracil (5-FU), reduces the clinical
recurrence and overall mortality rates among patients with stage
III colon cancer by 41 and 33%, respectively (8). However, in 1992, Hook et al
(9) revealed that the combination
of 5-FU and levamisole may lead to serious multifocal
leukoencephalopathy. Since then, cases of demyelinating
leukoencephalopathy induced by an overdose of levamisole have
become increasingly common (10–12).
In China, levamisole has been widely applied as an insect
repellent, despite the fact that a normal dose is sufficient to
induce demyelinating leukoencephalopathy in certain patients
(13,14). The awareness that a single
administration of levamisole, or its combined use with 5-FU, may
induce demyelinating leukoencephalopathy has increased; however,
diagnostic errors, missed diagnoses and delays in treatment
frequently occur in clinic, due to the medical history of the
patients being overlooked, in addition to the long-term latency of
the disease and various other disease factors. As a consequence, a
number of sequelae may develop. Magnetic resonance imaging (MRI) is
an important method of examination for demyelinating
leukoencephalopathy. However, while MRI effectively displays brain
lesions, the differentiation of demyelinating leukoencephalopathy
from other diseases remains challenging.
The current study analyzed the MRI data of 15
patients with levamisole-induced demyelinating leukoencephalopathy,
who received treatment at the First Affiliated Hospital of Xinxiang
Medical University (Weihui, China) between January 2002 and June
2011. The aim of the investigation was to further enhance the
awareness of demyelinating leukoencephalopathy, to reduce the
sequelae caused by diagnostic errors and missed diagnoses and to
increase the diagnostic level of MRI for this disease.
Patients and methods
Patient data
A total of 15 patients with demyelinating
leukoencephalopathy were accepted for this study. Of the patients,
five were males and 10 were females, and the age range was between
31 and 54 years (mean age, 45.5±8.67 years). All patients had a
history of levamisole use for worm expulsion, between two weeks and
two months prior to the disease onset, with a dose range of 50–150
mg. According to the typical clinical and diagnostic features of
imidazole-induced delayed cerebral diseases (15–18),
and the seven diagnostic criteria proposed by Zheng in 1995
(19), a history of anthelmintic
contact prior to the occurrence of the disease is the first
condition for disease diagnosis. All patients in the study met this
criterion; in addition, at least two types of ancillary examination
supported the diagnosis, such as cerebrospinal fluid (CSF)
examination, electroencephalography (EEG) and head MRI.
This study was conducted in accordance with the
Declaration of Helsinki, and approved by the ethics committee of
The First Affiliated Hospital of Xinxiang Medical University.
Written informed consent was obtained from all participants.
Clinical data
Of the 15 patients, three presented with a fever,
eight had limb weakness (accompanied by dizziness and a headache in
two cases, and a fever in three cases), one had apathia/was
emotionally disturbed, one had limb myasthenia and progressive
language disorders and two had impaired vision. Nine patients
underwent a CSF examination, and the results demonstrated that four
had mild inflammatory changes. The white blood cell counts that
were obtained ranged from 8×109 to 1.2×1011/l
(predominated by monocytes), but the levels of glucose and chloride
were essentially normal. Six patients did not exhibit any
abnormalities. The EEG results of the 15 patients revealed
high-amplitude and slow waves, which suggested moderate or severe
abnormalities. There was an absence of evident waveforms in the
bilateral visual evoked potentials of two patients; however, no
abnormalities in the upper limb somatosensory evoked potentials
were observed. One patient presented with negative P14, N20, P28,
and N35 waveforms (indicative of a central impairment), and a
prolonged latent period of P100 with a low wave amplitude in visual
evoked potentials.
MRI
All patients were subjected to MRI [including spin
echo T1-weighted imaging (SE T1WI), T2 fluid-attenuated inversion
recovery (FLAIR), heavy T1-weighted imaging (T2WI) and
diffusion-weighted imaging (DWI)], using the Signa 1.5T MRI system
(GE Healthcare, Waukesha, WI, USA). The scanning parameters were as
follows: SE T1WI: TR: 620 msec, TE: 12 msec, FOV: 24×24 cm, matrix:
200×256, thk5mm/sp1.5mm, NEX: 2. T2 FLAIR: TR: 8,000 msec, TE: 136
msec, TI: 2,000 msec, FOV: 24×24 cm, matrix: 512×512,
thk5mm/sp1.5mm, NEX: 2. DWI/T2WI: SE/EPI, TR: 3,600 msec, TE: 70.3
msec, FOV: 24×24 cm, matrix: 256×256, thk5mm/sp1.5mm, NEX: 2. In
addition, one patient underwent a contrast-enhanced scan, using
gadopentetate dimeglumine as a contrast medium.
Results
MRI
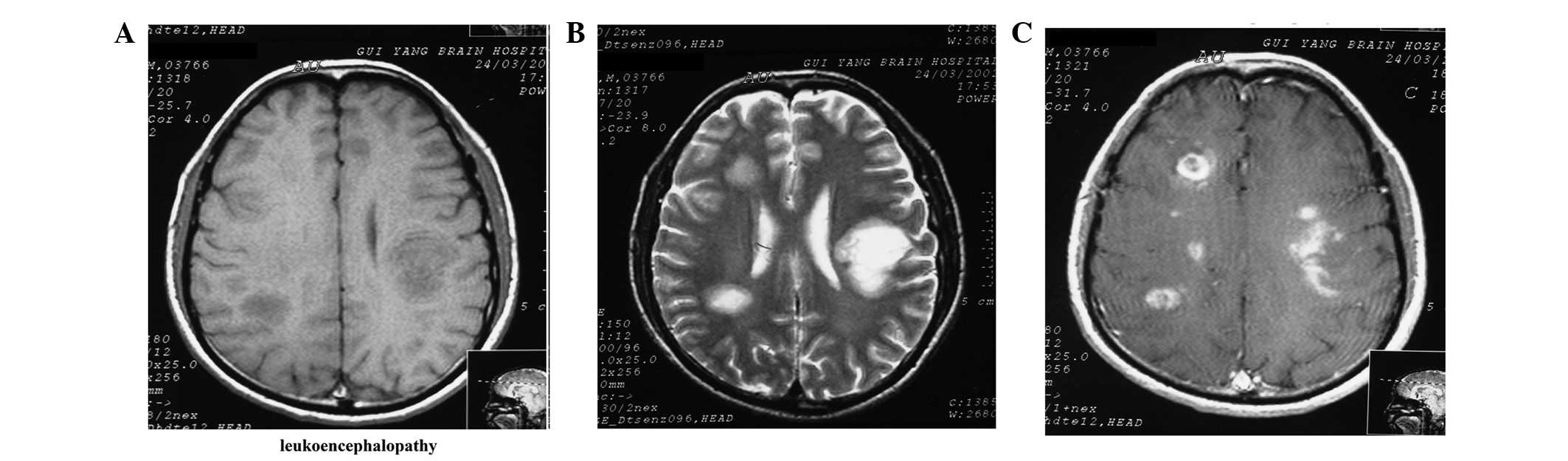
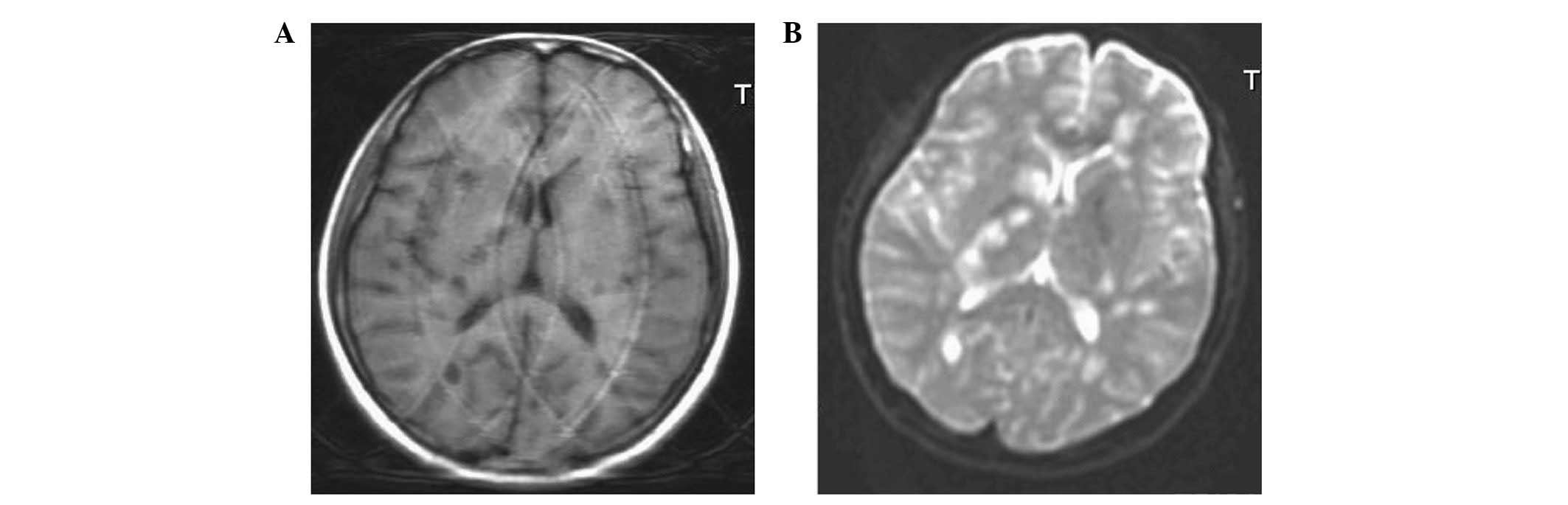
The MRI results demonstrated a 100% abnormality rate
in the patients. The majority of the foci were located at the
bilateral centrum ovale, peri-lateral ventricles and basal ganglia,
while the remaining foci were observed in the white matter areas of
the frontal, apical, occipital, and temporal lobes, as well as in
the brain stem and cerebellum (Figs.
1–3). All patients (100%) were
revealed to have foci present at the bilateral centrum ovale and
peri-lateral ventricles, while 10 patients (66.7%) exhibited foci
at the basal ganglia, four (26.7%) at the temporal lobe, one (6.7%)
at the frontal lobe, one (6.7%) at the apical lobe, one (6.7%) at
the occipital lobe and one (6.7%) in the brain stem and
cerebellum.
The foci observed in the MRI results were of
different sizes, with large foci present in round sheets, and small
foci appearing patchy, with indistinct boundaries (Figs. 1–3). Irregular patchy foci were observed in
six of the patients (40%), whereas the predominant round and
mass-like foci were observed in all patients (100%).
The foci were observed to be hypointense by T1WI,
hyperintense with clear boundaries by T2 FLAIR, hyperintense by
T2WI and predominantly hyperintense by DWI (Figs. 1–3). Three patients displayed mild edema
surrounding the foci.
One patient was demonstrated to have enhanced
demyelinating leukoencephalopathy, and axial enhancement revealed
the majority of the foci to be patch-, ring- and arc-like (Fig. 1C).
Treatment effectiveness
Hormone therapy was observed to be effective for all
the patients. The patients were discharged from the hospital
following improvement or recovery.
Discussion
Levamisole-induced cerebral disease is classified as
a delayed demyelinating leukoencephalopathy. The disease exhibits
many pathological similarities to acute disseminated
encephalomyelitis (ADEM) and multiple sclerosis (MS) (20). Demyelinating leukoencephalopathy is
characterized by a number of clinical features, and occurs within
two months of levamisole administration. The majority of cases have
an acute or subacute onset, which manifests as diffuse brain
injury, early mental symptoms and movement disorders. CSF
examinations of the patients appear normal, or with a marginally
raised white blood cell count, while EEG and MRI examinations
demonstrate diffuse slow waves and cerebral white matter
demyelination, respectively. Short-term cortical hormonal therapy
may achieve a successful outcome (21). In the present study, eight patients
presented with movement disorders as the primary symptom, and two
presented with headaches, dizziness or emotional disturbances.
Based on these symptoms, it was possible to exclude a diagnosis of
brain parenchymal disease by computed tomography or MRI. Two
patients initially presented with impaired vision, and no obvious
waveforms were observed in their visual evoked potentials. It was
suggested that one of the patients had a central impairment. Based
on these symptoms, it was not possible to exclude the possibility
that the visual impairment was caused by a cerebral disease. It is
necessary to enhance the awareness of the significance of ancillary
examinations, in order to increase the diagnostic rate of
demyelinating leukoencephalopathy.
Clinically, levamisole-induced demyelinating
leukoencephalopathy improves or recovers following treatment;
however, its radiological manifestations, with regard to lesion
distribution, morphology, signals, enhancement effects and dynamic
changes, have been demonstrated to appear later than the clinical
signs of the disease (10,20,22).
Although MRI provides an important diagnostic basis for
demyelinating leukoencephalopathy, the differentiation of the
disease from ADEM and MS remains challenging. For multifocal
leukoencephalopathy, demyelinating lesions may be easily diagnosed
by MRI. However, it is still necessary to differentiate
demylelinating leukoencephalopathy from other diseases, as follows.
With regard to MS, differentiation may be achieved due to the fact
that the majority of the cases of demyelinating leukoencephalopathy
present with fever in the clinic, and the MRI results exhibit foci
that are larger than those of MS. In addition, the foci in
demyelinating leukoencephalopathy appear in round, as opposed to
rectangular, sheets. By contrast, the differentiation between
demyelinating leukoencephalopathy and ADEM is predominantly based
on the clinical symptoms of the disease and the outcome of the CSF
examination. However, in certain cases the diagnosis remains
difficult. In such cases, a repeated inquiry into the case history
is required. In addition to MS and ADEM, it may be necessary to
differentiate between demyelinating leukoencephalopathy and
metastatic tumors, since diffuse mass-like foci may be observed in
certain cases of demyelinating leukoencephalopathy. This
differentiation may be achieved through the observation of
peripheral edema, with an apparent space -occupying effect.
Peripheral edema is not observed in demyelinating
leukoencephalopathy. Furthermore, metastatic tumors exhibit
characteristic enhancement features, where the enhancements appear
node-like or uneven ring-shaped. By contrast, enhancements may be
absent in demyelinating leukoencephalopathy, or the enhancements
may appear stippled or arc-like.
Although the MRI manifestations of demyelinating
leukoencephalopathy are not specific, the outcomes of the MRI, in
combination with the focal morphology, location and enhancement
phenomena, may indicate a potential diagnosis. The diagnosis of
demyelinating leukoencephalopathy may then be confirmed if the
patient has a recent history of insect repellent use.
In conclusion, MRI effectively demonstrates cerebral
white matter demyelination. The appearance of multiple cerebral
demyelination lesions is indicative that demyelinating
leukoencephalopathy may be considered as a potential diagnosis.
Based on this consideration, it is necessary to establish the
medical history of the patient, prior to the disease occurrence.
Once the diagnosis of demyelinating leukoencephalopathy is
confirmed, a clinical treatment protocol may be devised.
Acknowledgements
The authors would like to thank the
teachers of Department of Neurology and Department of Radiology,
the Affiliated Hospital of Guiyang Medical University.
References
|
1.
|
Laurence DR, Barnett PN and Brown MJ:
Neoplastic disease and immunosuppression. Clinical Pharmacology.
8th edition. Churchill Livingstone; New York: pp. 5601997
|
|
2.
|
Bondy DC, Bondy PK, Feinstein AR, et al:
Antivirus drug. The Merck Manual of Diagnosis and Therapy. 14th
edition. Merck & Co Inc.; New Jersey: pp. 23–24. 1982
|
|
3.
|
Katzung BG: Basic and Clinical
Pharmacology. 6th edition. Prentice-Hall; International Inc: pp.
550–551. 1995
|
|
4.
|
Sajid MS, Iqbal Z, Muhammad G and Iqbal
MU: Immunomodulatory effect of various anti-parasitics: a review.
Parasitology. 132:301–313. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Scheinfeld N, Rosenberg JD and Weinberg
JM: Levamisole in dermatology: a review. Am J Clin Dermataol.
5:97–104. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Sun A, Chiang CP, Chiou PS, Wang JT, Liu
BY and Wu YC: Immunomodulation by levamisole in patients with
recurrent aphthous ulcers or oral lichen planus. J Oral Pathol Med.
23:172–177. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Hawthorne AB and Hawkey CJ:
Immunosuppressive drugs in inflammatory bowel disease. A review of
their mechanisms of efficacy and place in therapy. Drugs.
38:267–288. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Moertel CG, Fleming TR, Macdonald JS, et
al: Levamisole and fluorouracil for adjuvant therapy of resected
colon carcinoma. N Engl J Med. 322:352–358. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Hook CC, Kimmel DW, Kvols LK, et al:
Multifocal inflammatory leukoencephalopathy with 5-fluorouracil and
levamisole. Ann Neurol. 31:262–267. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Liu HM, Hsieh WJ, Yang CC, Wu VC and Wu
KD: Leukoencephalopathy induced by levamisole alone for the
treatment of recurrent aphthous ulcers. Neurology. 67:1065–1067.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Lucia P, Pocek M, Passacantando A,
Sebastiani ML and De Martinis C: Multifocal leucoencephalopathy
induced by levamisole. Lancet. 348:14501996. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Kimmel DW and Schutt AJ: Multifocal
leukoencephalopathy: occurrence during 5-fluorouracil and
levamisole therapy and resolution after discontinuation of
chemotherapy. Mayo Clin Proc. 68:363–365. 1993. View Article : Google Scholar
|
|
13.
|
Zheng R and Zhang X: Analysis on exposure
of anthelmintic imidazoles-induced delayed encephalopathy in 202
cases. Chinese Journal of Pharmaco Epidemiology. 5:146–149.
1996.
|
|
14.
|
Li S and Li G: A study of clinic, CT and
MRI in levamisole induced demyelinating encephalopathy. Stroke and
Nervous Diseases. 7:167–169. 2000.
|
|
15.
|
Kimmel DW, Wijdicks EF and Rodriguez M:
Multifocal inflammatory leukoencephalopathy associated with
levamisole therapy. Neurology. 45:374–376. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
16.
|
Lucchinetti CF, Kimmel DW, Pavelko K and
Rodriguez M: 5-Fluorouracil and levamisole exacerbate demyelination
in susceptible mice infected with Theiler’s virus. Exp Neurol.
147:123–129. 1997.PubMed/NCBI
|
|
17.
|
Zuo RF, Shen GQ and Han DM: MRI diagnosis
of encephalopathy induced by levamisole. Journal of Xinxiang
Medical College. 5:518–519. 2010.(In Chinese).
|
|
18.
|
Chen TC, Hinton DR, Leichman L, Atkinson
RD, Apuzzo ML and Couldwell WT: Multifocal inflammatory
leukoencephalopathy associated with levamisole and 5-fluorouracil:
case report. Neurosurgery. 35:1138–1143. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Zheng RY: Differential diagnosis of
delayed encephalopathy induced by anthelmintic imidazoles. Zhonghua
Nei Ke Za Zhi. 34:468–471. 1995.(In Chinese).
|
|
20.
|
Xu N, Zhou W, Li S, Zhou G, Zhang N and
Liang J: Clinical and MRI characteristics of levamisole-induced
leukoencephalopathy in 16 patients. J Neuroimaging. 19:326–331.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Li S, Peng G and Lv B: A clinical and MRI
study of levamisole induced demyelinating encephalopathy. Chinese
Journal of Neurology. 32:335–337. 1999.
|
|
22.
|
Wu VC, Huang JW, Lien HC, et al:
Levamisole-induced multifocal inflammatory leukoencephalopathy:
clinical characteristics, outcome, and impact of treatment in 31
patients. Medicine (Baltimore). 85:203–213. 2006. View Article : Google Scholar
|