Introduction
Non-alcoholic fatty liver disease (NAFLD) is
increasingly diagnosed worldwide and is considered to be the most
common liver disorder in Western countries (1). It comprises a disease spectrum which
includes variable degrees of simple steatosis (fatty liver),
non-alcoholic steatohepatitis (NASH) and cirrhosis. Simple
steatosis is benign, whereas steatohepatitis (NASH) is
characterized by hepatocyte injury, inflammation and fibrosis
(2), which can lead to cirrhosis,
liver failure and hepatocellular carcinoma (3–6).
NAFLD affecting 10–24% of the general population in
various countries (7–9) is strongly associated with obesity,
type 2 diabetes (10), insulin
resistance, hypertension and dyslipidemia (11), and is now regarded as the liver
manifestation of the metabolic syndrome. The pathogenesis of
non-alcoholic steatohepatitis is not fully understood, but the
working hypothesis suggests that a first hit leads to the
development of steatosis and is followed by a second hit that
causes inflammation, ballooning and fibrosis (steatohepatitis)
(12).
Nuclear factor (NF)-κB, a nuclear transcription
factor, regulates the expression of various genes, including
interleukin (IL)-1β, tumor necrosis factor (TNF)-α, inducible NO
synthase (iNOS), and cyclooxygenase-2 (COX-2), that play critical
roles in apoptosis, tumorigenesis, various autoimmune diseases and
inflammation (13,14). Due to the ubiquitous role in the
pathogenesis of inflammatory gene expression, NF-κB is a current
target for treating various diseases (15). NF-κB is activated in response to
various inflammatory stimuli, including bacterial LPS, cytokines
and viral proteins (16). Under
normal conditions, NF-κB is present in the cytoplasm as an inactive
heterotrimer consisting of p50, p65 and IκBα subunits. Upon
activation, degradation of IκBα exposes a localization signal on
the p50/p65 heterodimer, leading to nuclear translocation and
binding to a specific sequence in several promoters, which in turn
results in gene transcription of proinflammatory genes.
Rubus (Rubus L.), a major genus of the
Rosaceae family, is widely distributed throughout the world. The
anti-inflammatory effects of Rubus coreanus (17), the antioxidant activities
(18,19), the hypoglycemic activity (20), the apoptotic activity of a crude
extract of Rubus crataegifolius roots (21) and the antimicrobial activity of
Rubus ulmifolius (22)
have been reported. Rubus aleaefolius Poir. (R.
aleaefolius) is one species within Rubus spread
throughout India, Southeast Asia, the Philippines, Japan, Taiwan
and mainland China, Guizhou, Hunan, Fujian, Guangdong, Guangxi and
Yunnan and other places. Its root or leaves are used as medicine,
while the related activity of the edible fruit, in the research
literature is rare in journals. R. aleaefolius is generally
used as a folk medicinal herb to treat various types of hepatitis
in the Anxi County of Fujian Province, China. Preliminary
investigations by our study group based on carbon tetrachloride
(CCl4)-induced acute liver injury model in rat in
vivo showed that the total alkaloids found in Rubus
aleaefolius Poir. (TARAP) ameliorated adipose degeneration
caused by CCl4 (23).
In the present study, we fed rats with a modified
high-fat diet (mHFD), and investigated whether TARAP has
anti-inflammatory effects on liver metabolism and steatosis in the
mHFD rat model, and investigated the possible mechanisms.
Materials and methods
Preparation and content assay of
TARAP
The preparation of TARAP was performed as previously
described (24). The roots of
R. aleaefolius were collected from Anxi of Fujian Province,
China, identified and authenticated by experts in our University,
and the alkaloids were extracted as follows. The herb powder (1 g)
was extracted using 50 ml chloroform:methanol:ammonia solution
(15:4:3) for 2 h in an ice bath, sonicated for 30 min, brought to
room temperature and filtered. The filtered solution was collected
and dessicated. The resultant residue was dissolved by 2 ml of 2%
sulfuric acid solution and filtered. The filter paper and residue
were re-washed with 2 ml of 2% sulfuric acid solution and with
buffer solution (pH 3.6). The buffer was then added to make a final
volume of 50 ml, and the solution was saved for future use. Acid
dye colorimetry was used to measure the total alkaloid content. The
total alkaloid content was 0.81 mg of alkaloid per gram of initial
herb powder.
Reagents
TRIzol kit, M-MLV first-strand cDNA synthesis kit
and TaqDNA polymerase were obtained from Invitrogen (Carlsbad, CA,
USA). Assay kits for alanine aminotransferase (ALT), aspartate
transaminase (AST), γ-glutamyltransferase (GGT), alkaline
phosphatase (ALP), triglyceride (TG), total cholesterol (TC),
high-density lipoprotein cholesterol (HDL-C) and low-density
lipoprotein cholesterol (LDL-C) activity were obtained from the
Jiancheng Institute of Biotechnology (Nanjing, China). All of the
other chemicals, unless otherwise stated, were obtained from Sigma
Chemicals Co. (St. Louis, MO, USA).
Animal model and treatment protocols
Male 8-week-old Sprague-Dawley (SD) rats (Slike Co.,
Ltd., Shanghai, China), weighing 180–200 g, were housed five per
cage in an environmentally controlled room at a temperature of
22±1°C and relative humidity 40–60%. Air ventilation was carried
out 12–18 times/h and a 12:12 h artificial light/dark cycle of
150–300 lux was maintained. Food and water was provided ad
libitum. The animal studies were approved by the Fujian
Institute of Traditional Chinese Medicine Animal Ethics Committee
(Fuzhou, China). The experimental procedures were carried out in
accordance with the Guidelines for Animal Experimentation of Fujian
University of Traditional Chinese Medicine (Fuzhou, China).
For the establishment of the aminal model, 60 rats
were randomly divided into 5 groups (12 rats/group). One group of
rats was used for the control. The remaining rats were fed a
modified high-fat diet (mHFD) ad libitum for 8 weeks. The
mHFD recipe we used to conform to NAFLD models of SD rat: 87.3% of
basal fodder, 10% of lard, 2% of cholesterol and 0.7% of swine bile
salt. After an 8-week feeding period, the rats fed with the mHFD
were randomly divided into 4 groups: the model group (model),
polyene phosphatidylcholine-treated group (PP), TARAP-treated
groups (high- and low-dose) (12 rats/group). PP (76 mg/kg body
weight/day), TARAP high-dose (1.44 g/kg body weight/day) and TARAP
low-dose (0.72 g/kg body weight/day) were administered as
previously described (24). The
model group and the control group received distilled water. The
body weights and food uptake were recorded weekly. All rats were
sacrificed by decapitation after 4 h of food deprivation. Blood
samples were collected for assays of ALT, AST, GGT, ALP, TG, TC,
HDL-C and LDL-C. Livers were rapidly dissected. A part of each
liver was cut and fixed in formaldehyde saline (4%) solution for
histological analysis; the rest was snap frozen in liquid nitrogen
and stored at −70°C until use.
Histological examination
The liver tissue was immediately fixed in 10%
buffered formalin for pathological analysis. Formalin-fixed liver
tissue was paraffin-embedded and then sections of 4–5 mm were
prepared and subsequently stained with hematoxylin and eosin
(H&E). Histological evaluation was performed twice by a
pathologist unaware of the treatments on two separate occasions. A
semiquantitative scoring system was used to assess the severity of
the hepatic steatosis and inflammatory cell infiltration in 10
microscopic fields. In brief, the following criteria were used for
scoring hepatic steatosis: grade 0 (−), no fat; grade 1 (+), fatty
hepatocytes occupying 33% of the hepatic parenchyma; grade 2 (++),
fatty hepatocytes occupying 33–66% of the hepatic parenchyma; grade
3 (+++), fatty hepatocytes occupying >66% of the hepatic
parenchyma.
Biochemical assays
Serum was separated by centrifugation at 4°C and
analyzed immediately or stored at -20°C. Serum ALT, AST, GGT, ALP,
TG, TC, HDL-C and LDL-C levels were determined by
spectrophotometry.
Reverse transcription-polymerase chain
reaction (RT-PCR) analysis
Total RNA was isolated from liver with TRIzol and
reverse transcribed by a two-step method using the SuperScript
First-Strand Synthesis system. Following RT, the product was stored
at 20°C. The cDNA product (1 μl) was added to prepare the PCR
reaction mixture containing 200 μM of dNTPs, 1.5 mM
MgCl2 and 1.25 units Platinum Taq polymerase
(Invitrogen) and 6 pmol of gene-specific primer sets for COX-2 or
NF-κB or TNF-α or IL-6 or GAPDH. In each case, amplification was
performed with a thermal cycler (GeneAmp PCR System 9600; Applied
Biosystems, Foster City, CA, USA), using the following cycling
parameters: denaturing at 95°C for 5 min; amplification cycle at
95°C for 30 sec, annealing temperature for 30 sec and 72°C for 1
min; and final extension at 72°C for 10 min. The expression of
hepatic genes COX-2 and NF-κB or TNF-α or IL-6 were analyzed. Six
to eight samples were chosen randomly from each group and the assay
of every gene in each sample was replicated three times. The rat
GAPDH gene was amplified as a loading control. The PCR products
were separated by electrophoresis on a 1.5% agarose gel. The genes
and their forward and reverse primers are listed as follows: COX-2
forward, 5′-AC ACT CTA TCA CTG GCA CCC-3′ and reverse, 5′-GA AGG
GAC ACC CCT TCA CAT-3′ (580 bp); NF-κB forward, 5′-GCA TTC TGA CCT
TGC CTA T-3′ and reverse, 5′-ATC CTT CCC AAA CTC CAC C-3′ (377 bp);
GAPDH forward, 5′-AGA TCC ACA ACG GAT-3′ and reverse, 5′-TCC CTC
AAG ATT GTC AGC AA-3′ (308 bp); TNF forward, 5′-CAG CAG ATG GGC TGT
ACC TT-3′ and reverse, 5′-AAG TAG ACC TGC CCG GAC TC-3′ (301 bp);
IL-6 forward, 5′-CAA CTC CAT CTG CCC TTC-3′ and reverse, 5′-TGG TCT
TGG TCC TTA GCC-3′ (596 bp).
Immunohistochemical assay
The paraffin-embedded liver samples were sectioned.
Sections were subjected to antigen retrieval and blocking of
endogenous peroxidase activity. For immunohistochemical staining,
slides were incubated with rabbit polyclonal antibodies against
COX-2, TNF-α, IL-6 or NF-κB (all in a 1:200 dilution; Santa Cruz
Biotechnology, Inc.). After washing with PBS, slides were incubated
with biotinylated secondary antibody followed by conjugated
horseradish peroxidase (HRP)-labeled streptavidin (Dako), and then
washed with PBS. The slides were then incubated with
diaminobenzidine (DAB) as the chromogen, followed by
counterstaining with diluted Harris hematoxylin (both from Sigma).
After staining, 5 high-power fields (x400) were randomly selected
in each slide, and the average proportion of positive cells in each
field was counted using the true color multi-functional cell image
analysis management system (Image-Pro Plus; Media Cybernetics,
USA). To rule out any non-specific staining, PBS was used to
replace the primary antibody as a negative control.
Statistical analyses
All data are the means of three determinations, and
data were analyzed using the SPSS package for Windows (version
11.5; SPSS, Inc., Chicago, IL, USA). Statistical analysis of the
data was performed with the Student’s t-test and ANOVA. Differences
of P<0.05 were considered statistically significant.
Results
Health condition of the rats
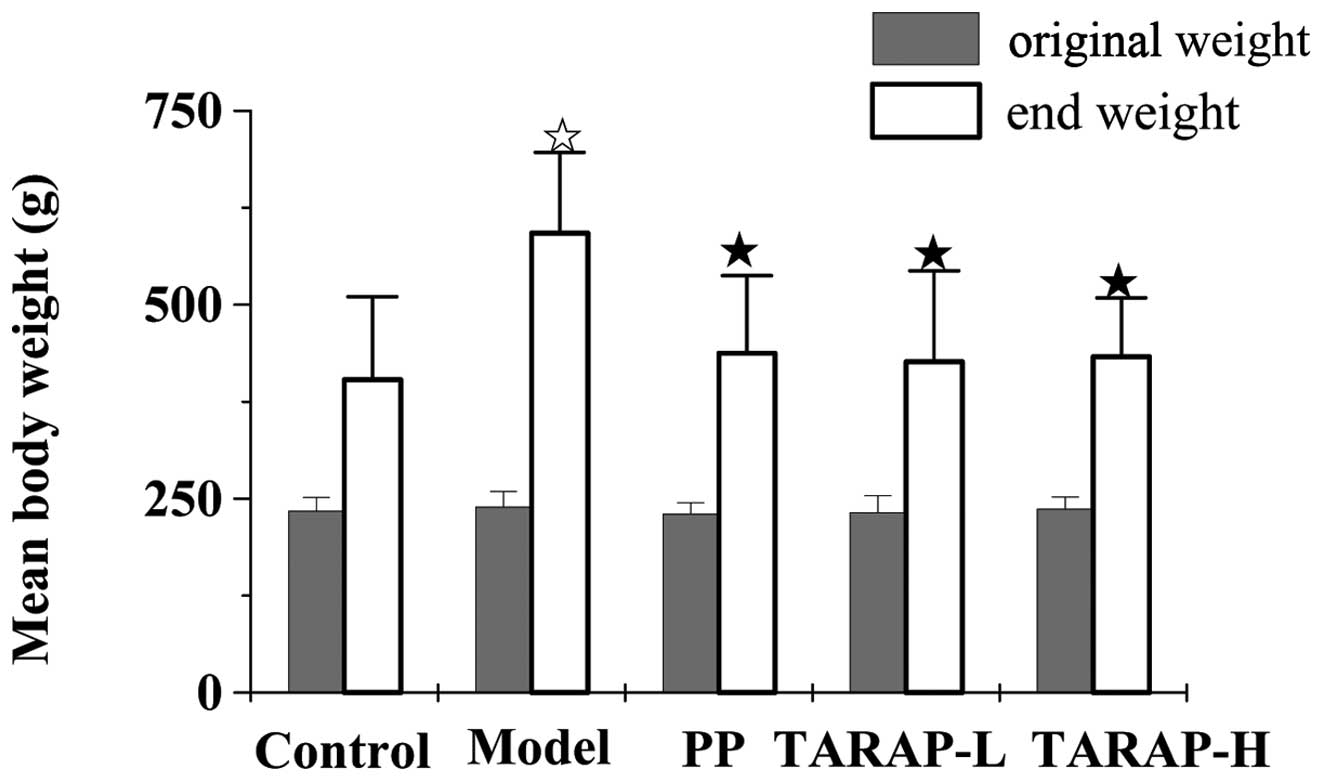
To determine the potential toxicity and safety of
TARAP to rats, the condition of the rats and the changes in body
weight were assayed. As shown in Fig.
1, the rats fed the mHFD for 8 weeks gained significantly more
weight than rats fed a normal diet. TARAP supplementation to the
high-fat diet decreased the body weight. In addition, the body
weight in the rats administered TARAP had no difference with the
control group. These results showed that TARAP is safe for
administration to SD rats.
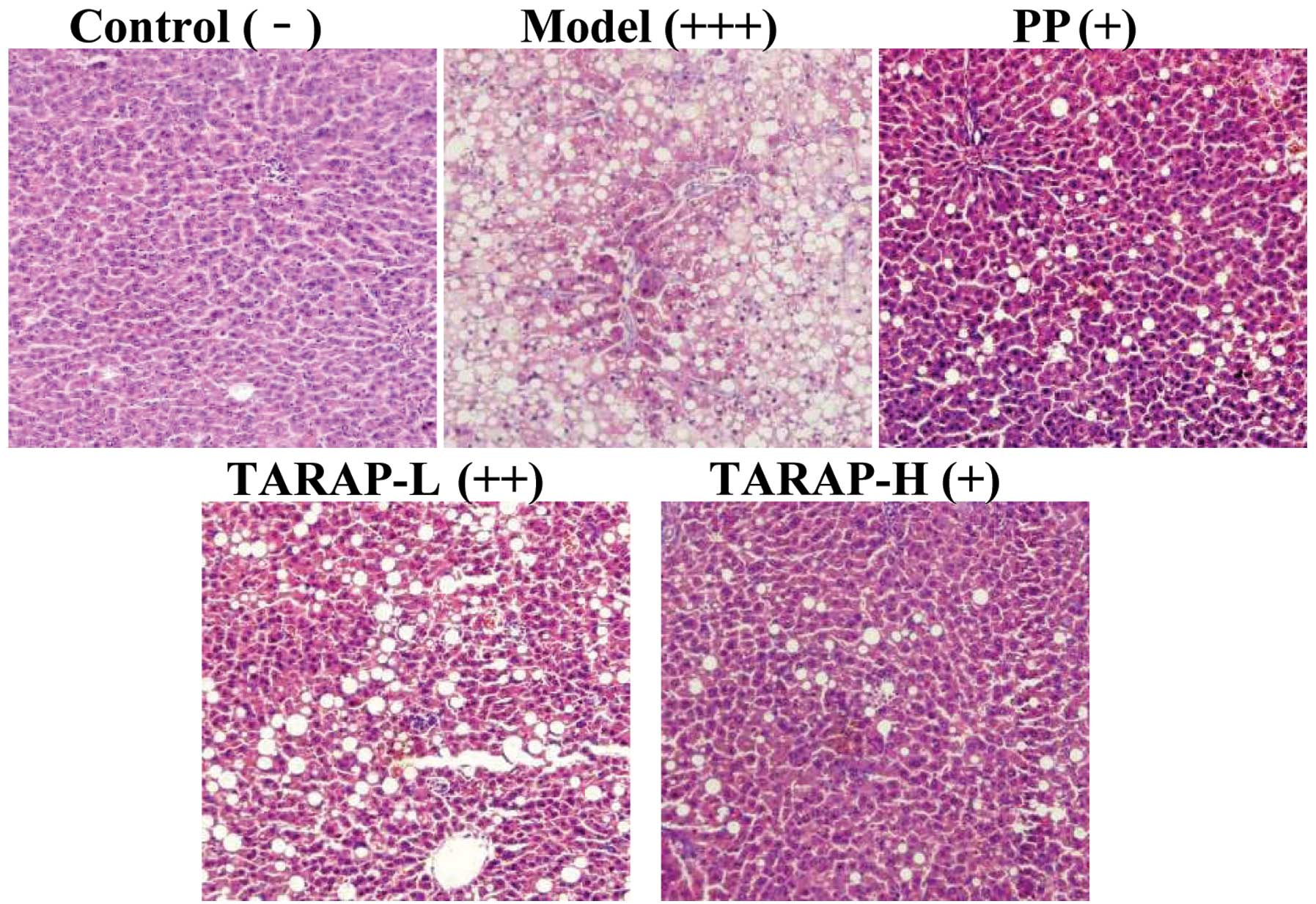
Effect of TARAP on liver histology
Histological evaluation is regarded as the ‘gold
standard’ approach with which to evaluate the presence and severity
of NAFLD (25). Thus, we
evaluated liver sections histologically to assess the extent to
which TARAP attenuated the development of hepatic steatosis.
Representative photomicrographs of the liver histology (Fig. 2) are shown. Rats fed a control
diet had normal liver histology. However, rats fed the mHFD showed
a high accumulation of fat and developed steatohepatitis, which was
characterized by hepatocyte ballooning, scattered lobular
inflammatory cell infiltration and inflammatory foci. Treatment
with PP or TARAP clearly improved hepatic steatosis. Histological
grading of liver sections confirmed that TARAP significantly
ameliorated both hepatic steatosis and necroinflammation.
Effect of TARAP on the secretion of serum
ALT, AST, GGT, ALP, TG, TC, HDL-C and LDL-C
As shown in Fig.
3, the mHFD significantly increased serum ALT, AST, GGT, ALP,
TC, TG and LDL-C levels (P<0.05) and reduced HDL-C levels
(P>0.05) in the rats in the model group, when compared with the
normal control group. However, the addition of PP or TARAP clearly
neutralized the effect of the high-fat diet on the expression of
these liver enzymes and blood lipids. The treatment of PP or TARAP
reduced the levels of serum ALT, AST, GGT and ALP, whose levels
were similar to the normal group. In addition, PP or TARAP
treatment significantly suppressed the increased TC, TG and LDL-C
levels induced by the mHFD (P<0.05), and upregulated the HDL-C
levels, but without significance.
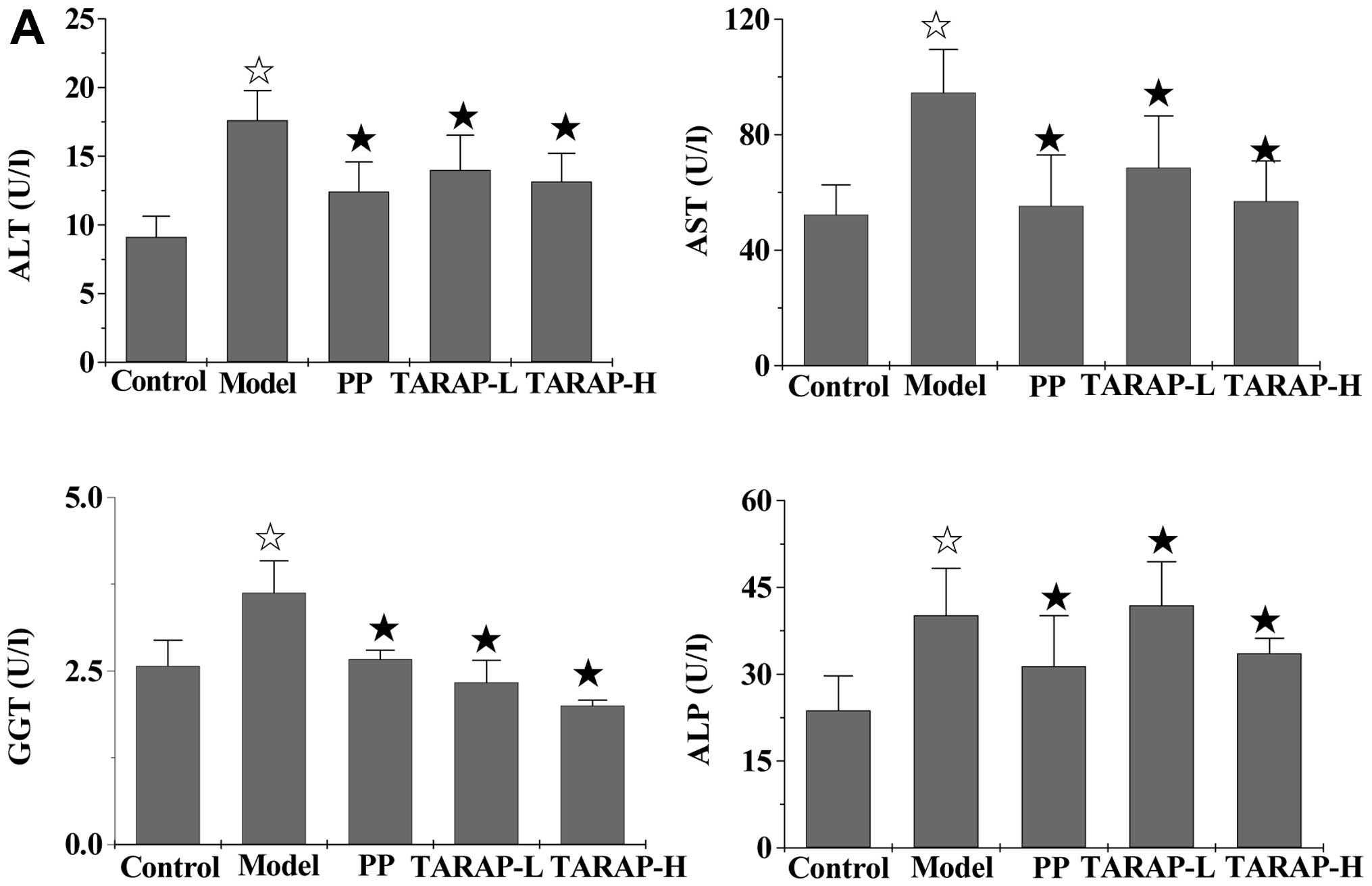 | Figure 3Effects of TARAP on mHFD-induced
increases in hepatic enzymes and serum lipids in the rat groups.
⋆P<0.05, compared with the control group;
★P<0.05, compared with the model group. PP, polyene
phosphatidylcholine; TARAP-L, low-dose TARAP group; TARAP-H,
high-dose TARAP group; ALT, alanine aminotransferase; AST,
aspartate transaminase; GGT, γ-glutamyltransferase; ALP, alkaline
phosphatase; TG, triglyceride; TC, total cholesterol; HDL-C,
high-density lipoprotein cholesterol; LDL-C, low-density
lipoprotein cholesterol. |
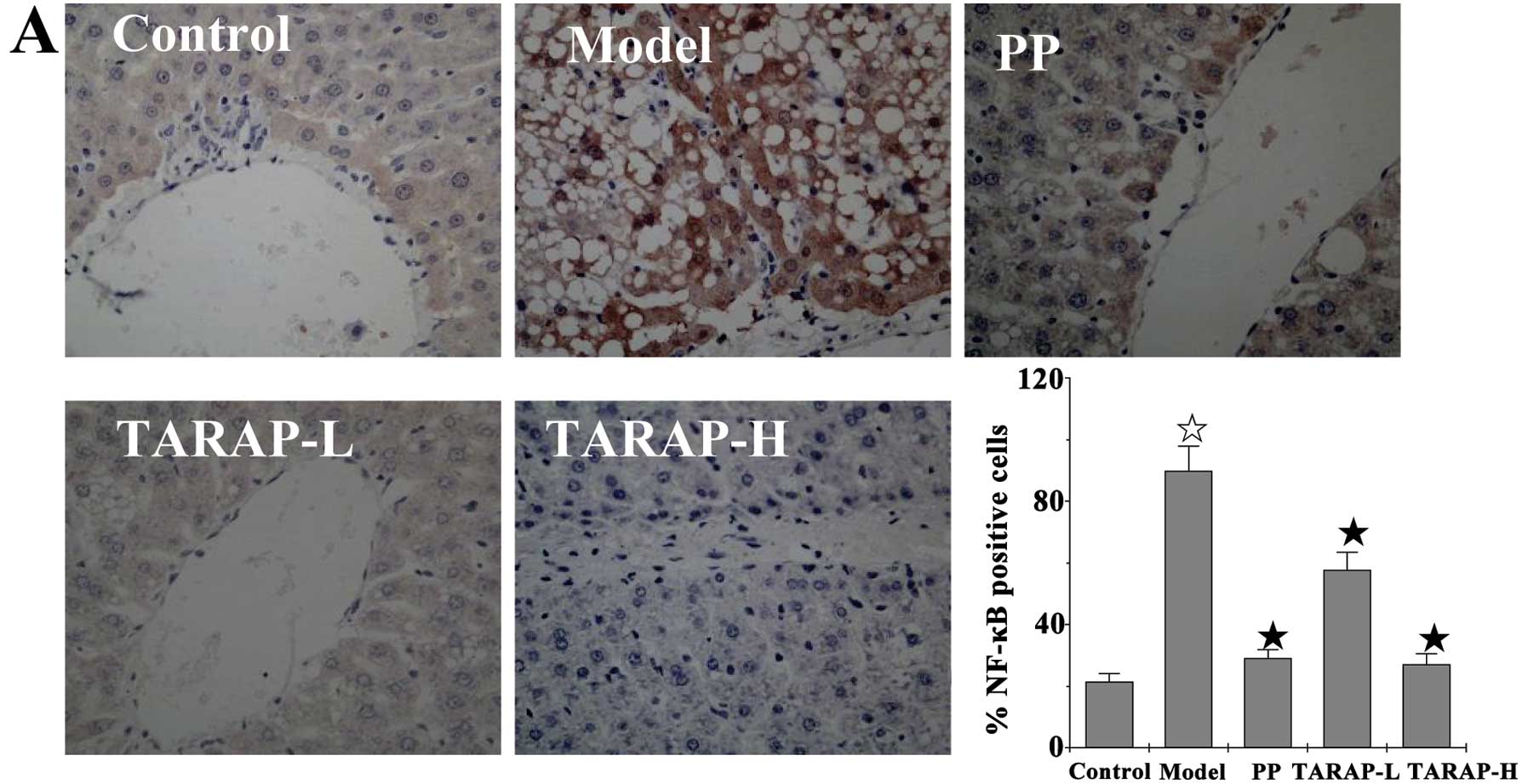
TARAP suppresses the expression of NF-κB,
TNF-α, COX-2 and IL-6 in the liver
The mRNA and protein expression of NF-κB, TNF-α,
COX-2 and IL-6 in the liver was determined by RT-PCR and
immunohistochemical analysis, respectively. The results of the
RT-PCR assay showed that PP or TARAP treatment markedly reduced
NF-κB, TNF-α, COX-2 and IL-6 mRNA expression (Fig. 4). Data from the
immunohistochemical assay showed that the protein expression
patterns of NF-κB, TNF-α, COX-2 and IL-6 emulated the changes in
the mRNA levels (Fig. 5). The
percentages of cells positive for NF-κB, TNF-α, COX-2 or IL-6 were
significant in the model group (89.88±8.09, 89.68±9.31, 67.2±2.02
and 85.98±7.54%) when compared with the control group (21.48±2.59,
31.2±3.08, 22.21±3.11 and 22.15±3.67%). Whereas these levels in the
TARAP low- or high-treated rats were 57.8±5.69, 50.31±5.66,
27.92±2.65, 59.38±4.21% and 27.19±3.39, 42.12±3.87, 31.67±2.58,
47.09±5.55%, respectively. Collectively, TARAP was found to
attenuate inflammation induced by the mHFD via regulation of the
NF-κB pathway.
Discussion
Non-alcoholic fatty liver disease (NAFLD) is one of
the most common etiologies of chronic liver disease worldwide
(8,26). Although the pathogenesis of NAFLD
remains undefined, various investigators have proposed a ‘two hit’
theory for the development of NASH. While the ‘first hit’ involves
insulin resistance and results in the accumulation of fat in the
liver, the ‘second hit’ includes oxidative stress resulting in
lipid peroxidation, hepatocellular degeneration, cell death,
hepatic stellate cell activation and fibrogenesis (27,28). The two-hit hypothesis of
steatohepatitis suggests that inflammation plays important roles in
the development of steatohepatitis.
We succeeded in establishing an NAFLD rat model,
This diet-induced model has more physiological similarities to
human NASH than do models based on genetic defects or chemically
induced disease. Moreover, cellular ballooning, one of the most
important pathological features of NASH in humans, was observed in
the mHFD model. This study was designed to investigate the possible
beneficial effects of TARAP on NASH of rats fed an mHFD.
R. aleaefolius has been proven to have
hepatoprotective activity on carbon tetrachloride-induced acute
liver injury in rats in vivo. It can elevate the content of
SOD and decrease TNF-α (29).
These reports led us to hypothesize that TARAP may exert a
beneficial effect on inflammation in NASH induced by a high-fat
diet.
It is well known that cholesterol is a fatty
substance which is an important part of the membrane of cells in
the body of animals. The liver is capable of removing cholesterol
from the blood circulation as well as manufacturing cholesterol and
secreting cholesterol into the blood circulation. TC is a measure
of the total amount of all cholesterol in blood at a given time and
is the sum of HDL-C, LDL-C, very low-density lipoprotein
cholesterol (VLDL-C), and intermediate density cholesterol (IDL).
Triglycerides (TG) are the main lipids stored in the liver of NAFLD
patients. Paradoxically, some experimental evidence shows that
inhibition of TG synthesis improves liver steatosis but worsens
liver damage, indicating that accumulation of liver TG may also be
a protective mechanism to prevent liver damage in NAFLD (30).
As shown in Fig.
2B, the levels of TC and TG significantly increased in the
model group. When mHFD-fed rats received TARAP, the levels of TC
and TG evidently decreased. HDL-C removes excess cholesterol from
arteries and moves it to the liver for further processing or to be
eliminated from the body. A high serum HDL-C is beneficial.
Therefore HDL-C is called ‘good’ cholesterol. LDL-C contributes to
the build up of fat deposits in the arteries (atherosclerosis),
which can cause decreased blood flow and heart attack. Thus, it is
termed ‘bad’ cholesterol, and low levels are desirable. In the
present study, we found high LDL-C levels and low HDL-C levels in
the model rats, while PP and TARAP treatment significantly
decreased the levels of LDL-C (P<0.05), and increased HDL-C
differentially, but without significance (P>0.05). The results
reveal that TARAP modulates hyperlipidemia, and has protective
effects against NASH.
The inflammatory response is a complex process
involving many different signaling pathways. NF-κB transcription
factors regulate genes involved in many aspects of the inflammatory
response (31). It has been shown
that NF-κB activation is a key factor in the production of iNOS,
COX-2 and various cytokines (TNF-α, IL-6) in macrophages in
response to LPS (13,32). Since the expression of these
proinflammatory mediators is modulated by NF-κB, our findings
suggest that treatment of rats fed an mHFD with TARAP effectively
prevents NF-κB activation and the associated downregulation of
several proinflammatory genes such as TNF-α, IL-6 and COX-2.
In summary, we demonstrated that TARAP treatment
decreases the levels of ALT, AST, GGT, ALP, TC, TG, LDL-C in
mHFD-fed rats. This anti-inflammatory effect occurs through the
downregulation of COX-2, IL-6 and TNF-α expression via the
suppression of NF-κB activation. Thus, TARAP may be relevant for
the clinical use for treatment of inflammatory diseases, including
endotoxemia or sepsis.
Acknowledgements
The present study was supported by the Nature
Science Foundation of the Fujian Province of China (no. 2010J01191
and 2010J01194) and the Project Sponsored by the Medical
Originality Foundation of Fujian Province of China (no.
2009-CX-18).
References
|
1
|
de Alwis NM and Day CP: Non-alcoholic
fatty liver disease: the mist gradually clears. J Hepatol.
48:S104–S112. 2008.PubMed/NCBI
|
|
2
|
Angulo P, Keach JC, Batts KP and Lindor
KD: Independent predictors of liver fibrosis in patients with
nonalcoholic steatohepatitis. Hepatology. 30:1356–1362. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Matteoni CA, Younossi ZM, Gramlich T, et
al: Non-alcoholic fatty liver disease: a spectrum of clinical and
pathological severity. Gastroenterology. 116:1413–1419. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Teli MR, James OF, Burt AD, et al: The
natural history of non-alcoholic fatty liver: a follow-up study.
Hepatology. 22:1714–1719. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Day CP and James OFW: Steatohepatitis: a
tale of two ‘hits’? Gastroenterology. 114:842–845. 1998.
|
|
6
|
Cortez-Pinto H, de Moura MC and Day CP:
Non-alcoholic steatohepatitis: from cell biology to clinical
practice. J Hepatol. 44:197–208. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chen CH, Huang MH, Yang JC, et al:
Prevalence and risk factors of nonalcoholic fatty liver disease in
an adult population of Taiwan: metabolic significance of
nonalcoholic fatty liver disease in nonobese adults. J Clin
Gastroenterol. 40:745–752. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bellentani S, Saccoccio G, Masutti F, et
al: Prevalence of and risk factors for hepatic steatosis in
Northern Italy. Ann Intern Med. 132:112–117. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shen L, Fan JG, Shao Y, et al: Prevalence
of nonalcoholic fatty liver among administrative officers in
Shanghai: an epidemiological survey. World J Gastroenterol.
9:1106–1110. 2003.PubMed/NCBI
|
|
10
|
Angulo P: Nonalcoholic fatty liver
disease. N Engl J Med. 346:1221–1231. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bugianesi E, McCullough AJ and Marchesini
G: Insulin resistance: a metabolic pathway to chronic liver
disease. Hepatology. 42:987–1000. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
McCullough AJ: Update on non-alcoholic
fatty liver disease. J Clin Gastroenterol. 34:255–262. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lawrence T, Gilroy DW, Colville-Nash PR
and Willoughby DA: Possible new role for NF-κB in the resolution of
inflammation. Nat Med. 7:1291–1297. 2001.
|
|
14
|
Riehemann K, Behnke B and Schulze-Osthoff
K: Plant extracts from stinging nettle (Urtica dioica), an
antirheumatic remedy, inhibit the proinflammatory transcription
factor NF-κB. FEBS Lett. 442:89–94. 1999.PubMed/NCBI
|
|
15
|
Renard P and Raes M: The proinflammatory
transcription factor NF-kappaB: a potential target for novel
therapeutical strategies. Cell Biol Toxicol. 15:341–344. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Makarov SS: NF-κB as a therapeutic target
in chronic inflammation: recent advances. Mol Med Today. 6:441–448.
2000.
|
|
17
|
Ywsilada E, Ustun O, Sezik E, et al:
Inhibitory effect of Turkish folk remedies on inflammatory
cytokines: interleukin-1α, interleukin-1β and tumor necrosis factor
α. J Ethnopharmacol. 58:59–73. 1997.PubMed/NCBI
|
|
18
|
Costantino L, Albasini A, Rastelli G and
Benvenuti S: Activity of polyphenolic crude extracts as scavengers
of superoxide radicals and inhibitors of xanthine oxidase. Planta
Med. 58:342–344. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wang SY and Lin HS: Antioxidant activity
in fruits and leaves of blackberry, raspberry, and strawberry
varies with cultivar and development stage. J Agric Food Chem.
48:140–146. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Alonso R, Cadavid I and Calleja JM: A
preliminary study of hypoglycemic activity of Rubus
fruticosus. Planta Med. 7(Suppl): S102–S106. 1980. View Article : Google Scholar
|
|
21
|
Lee JH, Ham YA, Choi SH, et al: Activity
of crude extract of Rubus crataegifolius roots as a potent
apoptosis inducer and DNA topoisomerase I inhibitor. Arch Pharm
Res. 23:338–343. 2000.
|
|
22
|
Rauha JP, Remes S, Heinonen M, et al:
Antimicrobial effects of Finnish plant extracts containing
flavonoids and other phenolic compounds. Int J Food Microbiol.
56:3–12. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hong ZF, Li TJ and Zhao JY: Effect of
total alkaloids of Rubus aleaefolius Poir on gene
expressions of CYP2E2 and CYP3A1 in rats with acute liver injury.
Zhongguo Zhong Xi Yi Jie He Za Zhi. 29:711–715. 2009.(In
Chinese).
|
|
24
|
Lin JM, Zhao JY, Li TJ, et al:
Hepatoprotection in a rat model of acute liver damage through
inhibition of CY2E1 activity by total alkaloids extracted from
Rubus alceifolius Poir. Int J Toxicol. 30:237–243. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Brunt EM: Nonalcoholic steatohepatitis:
pathologic features and differential diagnosis. Semin Diagn Pathol.
22:330–338. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Bellentani S and Tiribelli C: The spectrum
of liver disease in the general population: lesson from the
Dionysos study. J Hepatol. 35:531–537. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Brunt EM: Pathology of nonalcoholic
steatohepatitis. Hepatol Res. 33:68–71. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Farrell GC and Larter CZ: Nonalcoholic
fatty liver disease: from steatosis to cirrhosis. Hepatology.
43(Suppl 1): S99–S112. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ye HZ, Hong ZF, Wang YH and Chen YH:
Effects of crude extract of Rubus Alcaefolius Poir on
experimental liver injury in rats. Chin Arch Tradit Chin Med.
23:829–831. 2005.
|
|
30
|
Yamaguchi K, Yang L, McCall S, et al:
Inhibiting triglyceride synthesis improves hepatic steatosis but
exacerbates liver damage and fibrosis in obese mice with
nonalcoholic steatohepatitis. Hepatology. 45:1366–1374. 2007.
View Article : Google Scholar
|
|
31
|
Bonizzi G and Karin M: The two NF-kappaB
activation pathways and their role in innate and adaptive immunity.
Trends Immunol. 25:280–288. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Baldwin AS Jr: The NF-kappaB and I-kappaB
proteins: new discoveries and insights. Annu Rev Immunol.
14:649–683. 1996. View Article : Google Scholar : PubMed/NCBI
|