Introduction
Inflammatory bowel diseases (IBDs) such as
ulcerative colitis and Crohn’s disease are known as refractory and
recurrent diseases of the gastrointestinal tract. Although the
features of IBDs suggest a number of possible causes, including
genetic, infectious and immunological factors, the precise
pathogenesis of IBDs remains unknown (1–3).
Recent evidence suggests that oxidative stress caused by reactive
oxygen species (ROS) is an important factor involved in the onset
and development of intestinal inflammation. Furthermore, it has
also been demonstrated that disruptions in the antioxidant defense
system are involved in the pathophysiology of IBDs (4,5).
Therefore, it is important to investigate the oxidative
stress-related pathogenesis of IBDs.
Metallothioneins (MT) are a family of low molecular
weight proteins containing multiple cysteine residues that
coordinate multiple zinc and copper atoms, enabling a high affinity
for monovalent and divalent heavy metal atoms (6). The presence of MTs has been
confirmed in mammals, birds, fish, amphibians, reptiles and
invertebrates, as well as in a wide range of plants, and eukaryotic
and prokaryotic microorganisms (7). In mice, 4 MT isoforms have been
identified: MT-I and MT-II are ubiquitously expressed, MT-III is
expressed predominantly in the brain and MT-IV is expressed in
stratified squamous epithelial cells (8,9).
In humans, MTs are encoded by at least 10 identified functional
genes, and the encoded proteins are conventionally subdivided into
4 groups: MT-I, MT-II, MT-III and MT-IV (10). As MTs have a high affinity for
metals, as noted above, MTs have free radical-scavenging potential,
with MT-I/II exerting particularly potent antioxidant effects in
comparison with the other isoforms.
As regards the role of MTs in various
pathophysiological conditions causing inflammation, MTs have been
reported to play a protective role in various animal models,
including lipopolysaccharide (LPS)-induced lung injury (11), rheumatoid arthritis (12), multiple sclerosis (13), coagulatory disturbance (14), ethanol-induced gastroduodenal
mucosal injury (15) and
Helicobacter pylori-induced gastritis (16). MT-deficient mice are more
susceptible to LPS-induced lethal shock following D-galactosamine
(D-GalN) sensitization through the reduction of α1-acid
glycoprotein (17). By contrast,
there are conflicting data in a tumor necrosis factor (TNF)-induced
lethal shock model suggesting a reduction in mortality in
MT-deficient mice, as compared to wild-type mice (18). Furthermore, MT-overexpressing mice
are also more sensitive to the lethal effects of TNF compared to
wild-type mice (19).
Although MTs are known to a play protective role in
intestinal inflammation, studies on experimental colitis models
using MT-deficient mice have revealed that MTs do not protect
against the development of colitis. Tran et al (20) demonstrated that MT-deficient mice
had significantly lower disease activity index (DAI) than wild-type
mice in a dextran sodium sulfate (DSS)-induced colitis model, a
widely accepted model of human IBD. Oz et al (19) also reported that there were no
differences in histological damage following treatment with DSS
among wild-type, MT-deficient and MT transgenic mice. Hence, the
function of MTs in intestinal inflammation remains uncertain.
In the present study, we examined the role of MTs in
intestinal inflammation using a mouse model of DSS-induced colitis,
which is recognized as a useful experimental model of intestinal
inflammation and is considered to be similar to human IBD (21–24).
Materials and methods
Animals
We used 7-week-old male MT-I/II double knockout (MT
null) mice (25) and age-matched
C57BL/6 wild-type mice. MT-I/II knockout mice were kindly provided
by Dr Hirohisa Takano (Center for Environmental Health Sciences,
National Institute for Environmental Studies, and Kyoto University,
Graduate School of Engineering, Kyoto, Japan). C57BL/6 mice were
purchased from Shimizu Laboratory Supplies Co., Ltd. (Kyoto,
Japan). MT-I/II knockout mice were routinely bred in the
vivarium of Kyoto Prefectural University of Medicine. Mice
were housed in cages under a 12-h light/dark cycle, and a
controlled temperature of 22°C and negative atmospheric pressure.
Mice were maintained in a specific pathogen-free environment and
provided with tap water and food ad libitum. Mice were fed a
rodent diet (CE-2; Nihon CLEA, Tokyo, Japan) during the experiment.
All experimental procedures were carried out in accordance with the
National Institutes of Health (NIH) guidelines for the Care and Use
of Laboratory Animals. Experimental protocols were approved by the
Animal Care and Use Committee of Kyoto Prefectural University of
Medicine (Kyoto, Japan).
Induction of colitis by DSS
Male wild-type mice and MT-I/II knockout mice (7
weeks old) were randomized into control and experimental groups
(6–8 mice in each group). Experimental acute colitis was induced by
treatment with 2.0% DSS (molecular weight 1,000–9,000, Lot SDR4219;
Wako Pure Chemical Industries, Osaka, Japan) in drinking water for
7 days, as reported previously (26,27). Mice were sacrificed under
anesthesia 7 days after DSS treatment, and the colons were removed
for macroscopic and histological examination. Colonic specimens
were also obtained for biochemical assay and RNA isolation.
Evaluation of the severity of
colitis
DAI, colon length and histology were analyzed. DAI
was determined by scoring changes in animal weight, occult blood
positivity, gross bleeding and stool consistency, as described
previously (26,28,29). We used 5 grades of weight loss (0,
no loss or weight gain; 1, 1–5% loss; 2, 5–10% loss; 3, 10–20%
loss; 4, >20% loss), 3 grades of stool consistency (0, normal;
2, loose; and 4, diarrhea), and 3 grades of occult blood (0,
negative; 2, occult blood-positive; and 4, gross bleeding) based on
previous studies (26,27). After determining DAI, the mice
were sacrificed, and the entire colon was removed from the cecum to
the anus, and colon length was measured as an indirect marker of
inflammation.
Immediately after dissection, the distal colon was
fixed in 10% buffered formalin for histological analysis. Sections
(4-μm-thick) were prepared and stained with hematoxylin and eosin
(H&E). The slides were then examined and scored in a blinded
manner using a previously published grading system (30,31). Briefly, a combined score of
inflammatory cell infiltration and tissue damage was determined as
follows: as regards cell infiltration: score 0, occasional
inflammatory cells in the lamina propria; 1, increased inflammatory
infiltrate in the lamina propria predominantly at the base of
crypts; 2, confluence of inflammatory infiltrate extending into the
mucosa; and 3, transmural extension of inflammatory infiltrate. As
regards tissue damage: score 0, no mucosal damage; 1, partial (up
to 50%) loss of crypts in large areas; 2, partial to total 50–100%
loss of crypts in large areas, epithelium intact; and 3, total loss
of crypts in large areas and epithelium lost. The total
histological score represents the sum of the cell infiltration and
tissue damage scores, and thus ranges from 0 to 6.
Measurement of myeloperoxidase (MPO)
activity
MPO activity in the colonic mucosa, an index of
polymorphonuclear leukocyte accumulation, was determined using a
modification of the method described by Grisham et al
(32). A total of 2 ml of mucosal
homogenate was centrifuged at 20,000 × g for 15 min at 4°C to
pellet the insoluble cellular debris. The pellet was then
re-homogenized in an equivalent volume of 0.5%
hexadecyltrimethylammonium bromide. Samples were centrifuged at
20,000 × g for 15 min at 4°C, and the supernatants were saved. MPO
activity was assessed by measuring the
H2O2-dependent oxidation of
3,3′,5,5′-tetramethylbenzidine. One unit of enzyme activity was
defined as the amount of MPO that caused the absorbance to change
by 1.0/min at 655 nm and 25°C.
RNA analysis
The mRNA expression levels of MT-I/II, TNF-α,
interferon (IFN)-γ, interleukin (IL)-17 and β-actin (internal
standard) were determined by real-time-PCR. Samples for mRNA
isolation were removed from colonic tissue. Total RNA was isolated
with the acid guanidinium phenol chloroform (AGPC) method using
Isogen reagent (Nippon Gene, Toyama, Japan). RNA was stored at 70°C
until it was used for reverse-transcription polymerase chain
reaction (RT-PCR). A total of 1 μg of extracted RNA was
reverse-transcribed into first-strand complementary DNA (cDNA)
using the High Capacity cDNA Reverse Transcription kit (Applied
Biosystems, Foster City, CA). Real-time PCR for CINC-1 and β-actin
was carried out with the 7300 Real-Time PCR system (Applied
Biosystems) using the DNA-binding dye SYBR®-Green for
the detection of PCR products. Primers had the following sequences:
MT-I sense, 5′-GCTGTGCCTGATGTGACGAA-3′ and antisense,
5′-AGGAAGACGCTGGGTTGGT-3′; MT-II sense, 5′-TGCGC TCGACCCAATACTC-3′
and antisense, 5′-TCTAGGAGCG TGATGGAGAGAAG-3′; TNF-α sense,
5′-ATCCGCGACGT GGAACTG-3′ and antisense, 5′-ACCGCCTGGAGTTCTG
GAA-3′; IFN-γ sense, 5′-CCTGCGGCCTAGCTCTGA-3′ and antisense,
5′-CCATGAGGAAGAGCTGCAAAG-3′; IL-17 sense,
5′-TCATCTGTGTCTCTGATGCTGTTG-3′ and antisense,
5′-TCGCTGCTGCCTTCACTGT-3′; and β-actin sense,
5′-GAGCAAACATCCCCCAAAGTT-3′ and antisense, 5′-GCC
GTGGATACTTGGAGTGACT-3′. Relative quantities of gene expression with
real-time PCR data were calculated relative to β-actin.
Determination of colonic concentrations
of TNF-α, IFN-γ and IL-17
We determined the concentrations of TNF-α, IFN-γ and
IL-17 in the supernatant of colonic mucosal homogenates using an
enzyme-linked immunosorbent assay (ELISA) kit for TNF-α, IFN-γ and
IL-17 (R&D Systems, Minneapolis, MN) according to the
manufacturer’s instructions. Subtractive readings at 550 nm from
the readings at 450 nm were converted to pg/ml using values
obtained from standard curves generated with varying concentrations
of recombinant TNF-α, IFN-γ and IL-17.
Immunohistochemical and
immunofluorescence staining of MT and F4/80 in colonic mucosa
After 24-h fixation in formalin, samples were
embedded in paraffin, after which 4-μm-thick sections were cut
using a microtome cryostat and mounted on MAS-coated slides. We
performed antigen retrieval using proteinase K solution, and the
sections were rinsed with distilled water for 5 min, followed by
incubation with 3% hydrogen peroxide in methanol for 30 min to
block endogenous peroxidase activity. After incubation, the
sections were washed in phosphate-buffered saline (PBS)-Tween-20
for 5 min. Non-specific binding was blocked by incubating the
slides with Dako Cytomation protein block (Dako, Tokyo, Japan) for
30 min at room temperature. The sections were then incubated with
primary antibody against MTs (Dako, Glostrup, Denmark) diluted at
1:50 and F4/80 (Novus Biologicals, Inc., Littleton, CO) diluted at
1:200 with antibody dilution (Dako) overnight at 4°C. The sections
were washed 3 times in PBS-Tween-20 for 5 min and incubated with
secondary antibody [Histofine Simple Stain mouse MAX PO (rabbit);
Nichirei Biosciences, Inc., Tokyo, Japan] for 30 min at room
temperature. Unbound antibodies were removed by 3 washes in PBS for
5 min, and bound antibodies were visualized using diaminobenzidine
as a chromogen substrate reagent. Negative controls for
non-specific binding incubated with secondary antibodies were
confirmed to produce no signal. All sections were counterstained
with hematoxylin. Sections were finally dehydrated, cleared and
coverslipped.
For the immunofluorescence staining of MTs and F4/80
(diluted at 1:200; Novus Biologicals, Inc.), bound antibodies were
visualized using the secondary antibodies, anti-rabbit Alexa 594
(Molecular Probes, Inc., Eugene, OR) and anti-rat Alexa 488
(Molecular Probes), respectively, diluted at 1:1,000. Negative
controls used for non-specific binding and incubated with secondary
antibodies were confirmed to produce no signal. Fluorescence
staining was observed under an inverted fluorescence microscope
(IX70-23FL/DIC-SP; Olympus, Tokyo, Japan).
F4/80-positive cells in colonic
mucosa
We evaluated the number of F4/80-positive cells in
the intestinal mucosa of wild-type and MT-I/II knockout mice
following DSS-induced colitis. The number of positively stained
cells in the intestinal mucosa was counted in 50 high-power fields
per section under a microscope, as described previously (33).
Isolation of resident peritoneal
macrophages and determination of cytokine production
Resident peritoneal macrophages were obtained by
peritoneal lavage using 10 ml of PBS from both wild-type and
MT-I/II knockout mice. Cells were plated at 5×105
cells/well (24-well plate) and incubated for 2 h at 37°C in
RPMI-1640 medium with 5% fetal bovine serum (FBS), 2 mM glutamine,
50 mg/ml streptomycin and 50 U/ml penicillin (all obtained from
Life Technologies Corp.). The cells were washed twice with PBS to
remove non-adherent cells and cultured in RPMI-1640 for 24 h at
37°C. Subsequently, the cells were stimulated with LPS
(Escherichia coli 055:B5; Sigma-Aldrich Corp., St. Louis,
MO) at 10 ng/ml. After a 12-h incubation with LPS, cell
supernatants were collected and stored at −70°C. TNF-α, IFN-γ and
IL-17 levels in culture supernatants were determined using an ELISA
kit according to the manufacturer’s instructions (R&D
Systems).
Statistical analysis
The results in this study are presented as the means
± standard error of the mean (SEM). Overall differences between the
groups were determined by one-way analysis of variance (ANOVA). If
the results from one-way ANOVA were significant, the differences
between individual groups were analyzed by Bonferroni’s multiple
comparisons test. P-values <0.05 were considered to indicate
statistically significant differences. All analyses were performed
using GraphPad Prism 5 software (GraphPad Software, San Diego, CA)
on a Windows-based computer.
Results
Expression of MT in inflamed colonic
mucosa
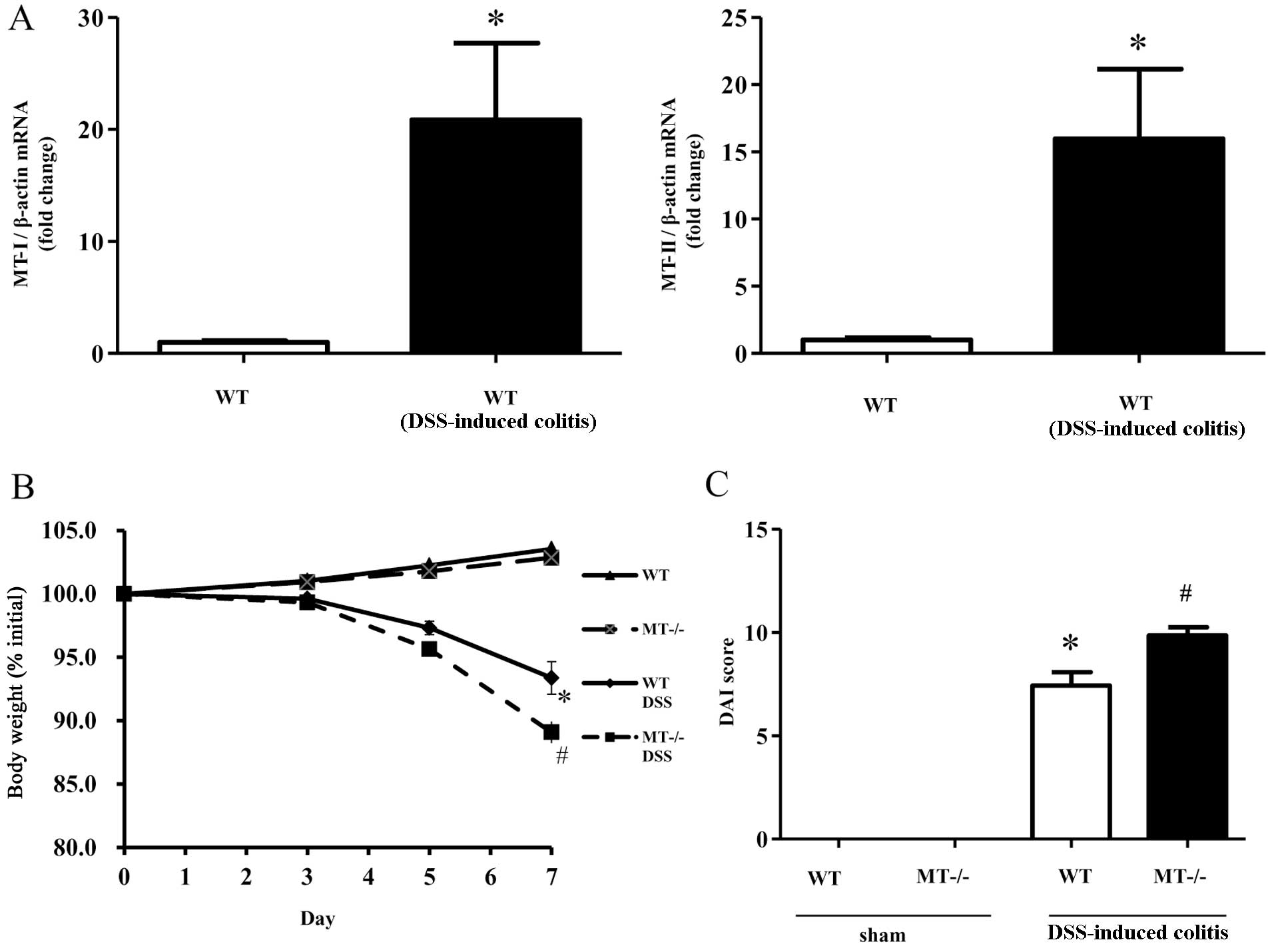
In order to investigate MT-I/II expression in the
inflamed mucosa following the administration of DSS, we examined
the expression of colonic MT-I/II mRNA using real-time PCR
analysis. The results confirmed that the expression of MT-I/II was
significantly higher in the inflamed colonic mucosa in the model of
DSS-induced colitis (Fig.
1A).
Body weight changes, DAI scores and colon
length following DSS administration
The intake of DSS solution was monitored throughout
the experiments and was found to be similar among the experimental
groups (data not shown). The mice exposed to 2.0% DSS developed
symptoms of acute colitis, with diarrhea accompanied by rectal
bleeding and body weight loss. Decreases in body weight were
significantly greater in the MT-I/II knockout mice in comparison to
the wild-type mice (Fig. 1B). DAI
scores in the DSS-treated mice, determined by weight loss, stool
consistency and blood in stool, were significantly increased in the
MT-I/II knockout mice compared to the wild-type mice (Fig. 1C).
Moreover, colon length is known to be reduced in DSS
colitis along with the exacerbation of intestinal inflammation. In
this study, MT-I/II knockout mice showed marked colon shortening 7
days after DSS administration (Fig.
1D). Colon length following DSS administration was
significantly decreased in the MT-I/II knockout mice compared to
the wild-type mice (Fig. 1E).
These data suggest that MTs play a crucial role in the development
of intestinal inflammation.
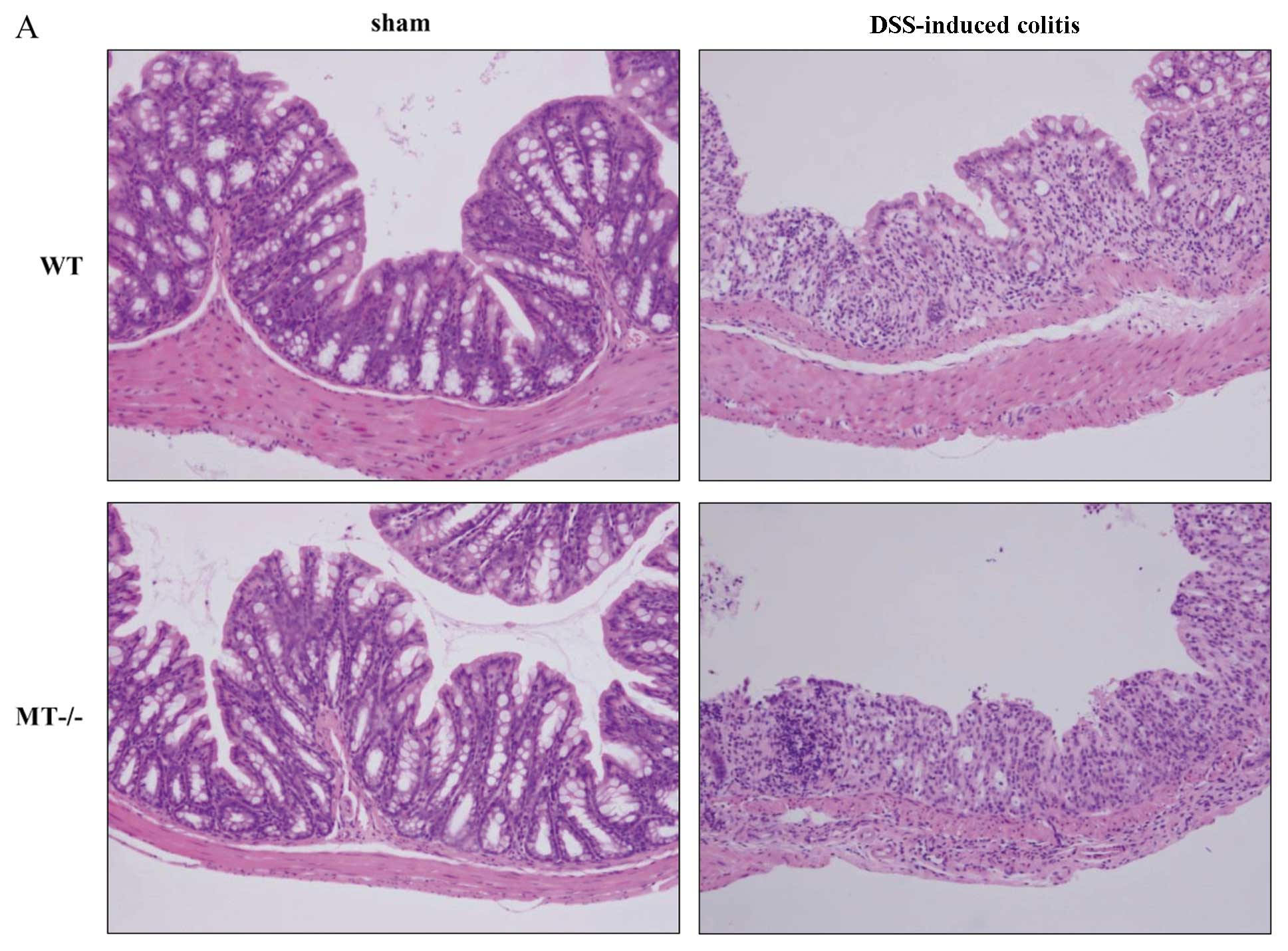
Histology and neutrophil accumulation
following DSS administration
The protective effects of MTs were also confirmed by
histological analysis. Fig. 2A
shows the typical histological appearance of wild-type and MT-I/II
knockout mice. In the wild-type mice, the administration of 2% DSS
for 7 days resulted in colonic ulceration associated with large
areas of epithelial crypt loss and inflammatory cell infiltration
throughout the mucosa. On the other hand, MT deficiency resulted in
larger erosions with a high degree of inflammatory cell
infiltration. The histological scores reflected these findings
(Fig. 2B).
Tissue-associated MPO activity (as an index of
polymorphonuclear leukocyte accumulation) in the colonic mucosa
increased from 0.130±0.079 mU/mg protein (basal concentration) to
0.903±0.114 mU/mg protein in the wild-type mice 7 days following
the administration of DSS (Fig.
2B). MPO activity increased in the wild-type mice following DSS
administration, and was significantly elevated in the DSS-treated
MT-I/II knockout mice (Fig.
2B).
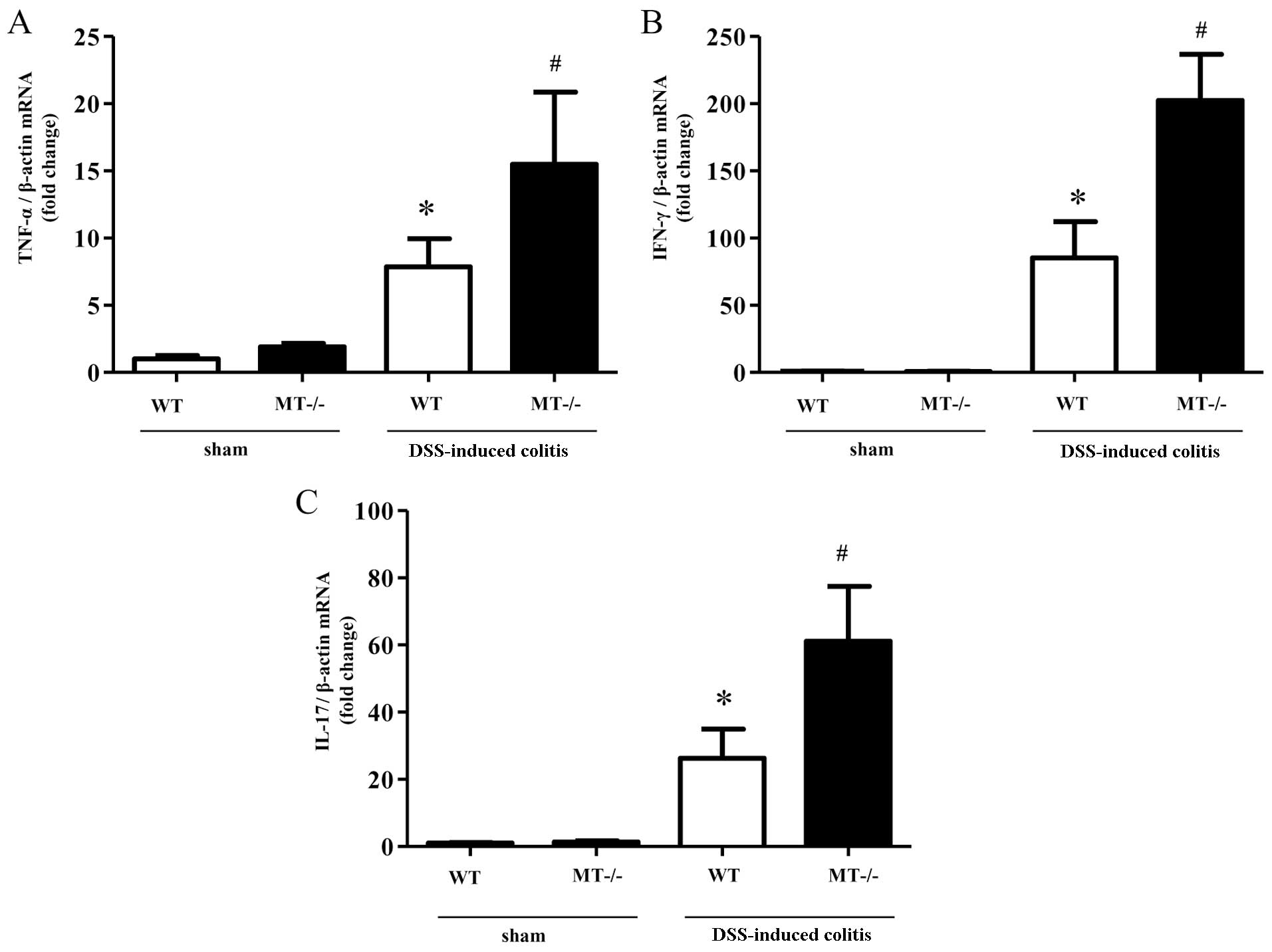
Colonic mRNA expression and mucosal
concentrations of various inflammatory cytokines following DSS
administration
The expression of inflammatory cytokines in the
colonic mucosa following DSS treatment was determined using
real-time PCR and ELISA. mRNA expression levels of inflammatory
cytokines (TNF-α, IFN-γ and IL-17) were significantly increased in
the colonic mucosal tissue of the MT-I/II knockout mice compared
with the wild-type mice following the administration of DSS
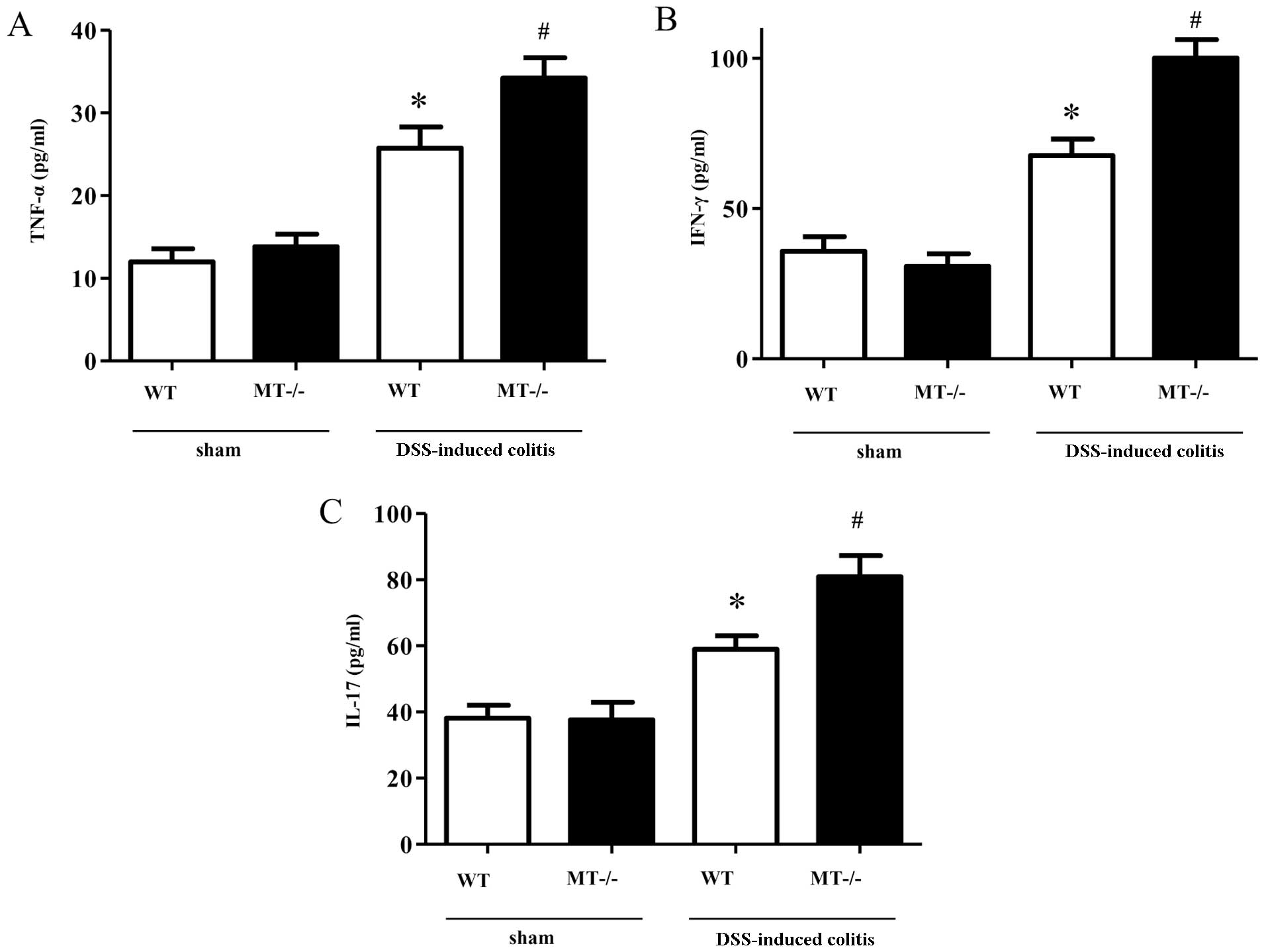
(Fig. 3). In agreement with these
results, the colonic mucosal concentrations of these inflammatory
cytokines (TNF-α, IFN-γ and IL-17) in the DSS-treated MT-I/II
knockout mice were significantly higher, as compared with the
DSS-treated wild-type mice (Fig.
4).
Immunohistochemical and
immunofluorescence staining of MTs in inflamed colonic mucosa
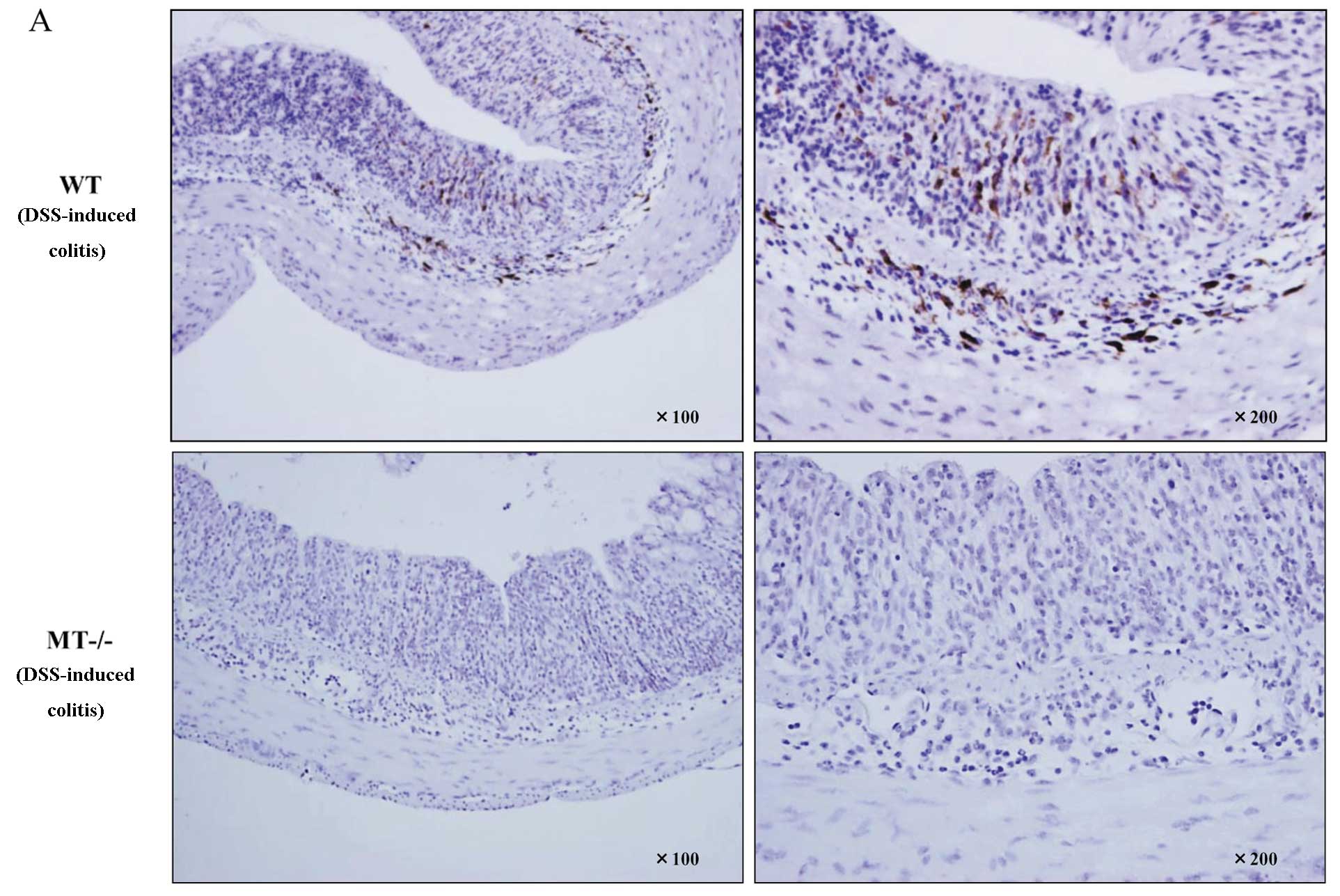
MTs are mainly localized in mononuclear cells in the
lamina propria and submucosal layer, and these MT-positive cells
were elevated in the DSS-treated wild-type mice. By contrast,
MT-positive cells were not detected in the colons of MT-I/II
knockout mice before and after DSS administration (Fig. 5A).
In addition, in order to investigate the detailed
localization of MT expression in the colonic mucosa, we conducted
immunofluorescence staining of the colonic mucosa. MT-positive
cells largely corresponded with F4/80-positive macrophages
(Fig. 5B).
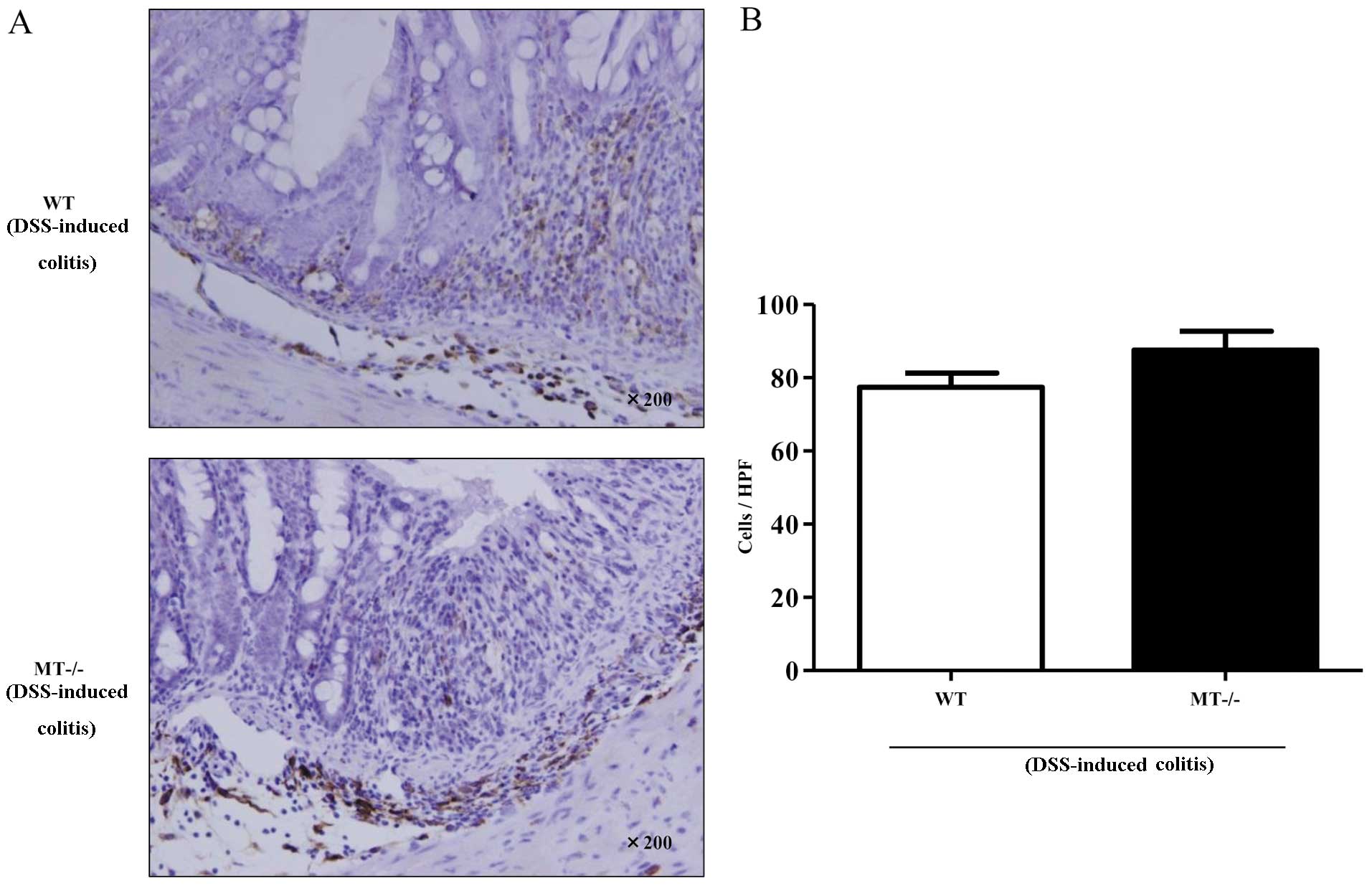
Number of F4/80-positive cells in
inflamed colonic mucosa
F4/80-positive cells were mainly localized in the
lamina propria and submucosal layer in wild-type and MT-I/II
knockout mice after DSS administration (Fig. 6A). Furthermore, we evaluated the
number of the F4/80-positive cells in the intestinal mucosa of
wild-type mice and MT-I/II knockout mice. No significant
differences were observed in the number of F4/80-positive cells
between the DSS-treated wild-type and MT-I/II knockout mice
(Fig. 6B).
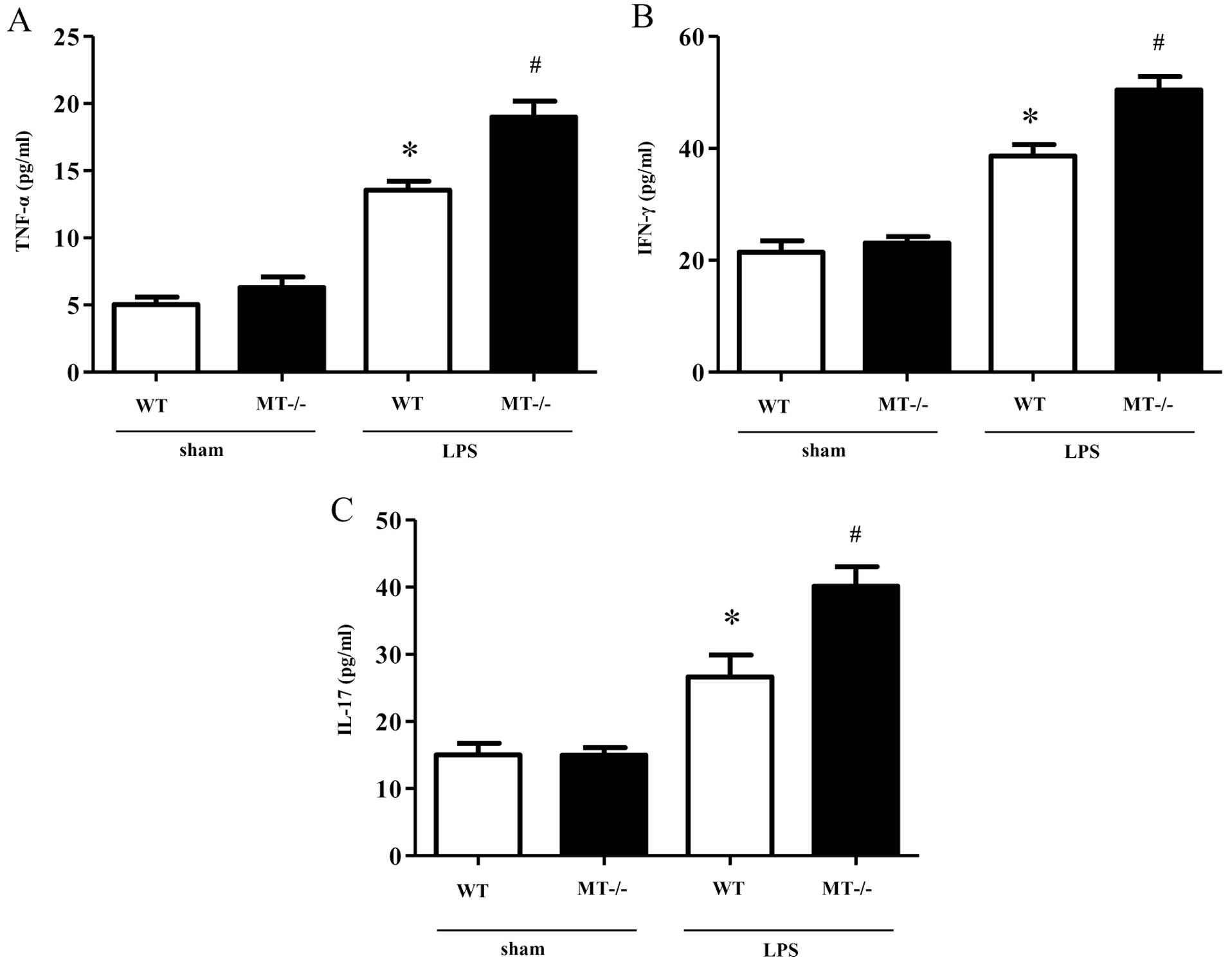
Production of inflammatory cytokines in
peritoneal macrophages following LPS treatment
In order to determine the production of inflammatory
cytokines (TNF-α, IFN-γ and IL-17) in murine peritoneal macrophages
following treatment with LPS, we measured the levels of these
cytokines in the supernatant of cultured cells using ELISA. The
concentrations of the inflammatory cytokines (TNF-α, IFN-γ and
IL-17) in the LPS-treated macrophages isolated from the MT-I/II
knockout mice were significantly higher as compared with the
LPS-treated macrophages derived from the wild-type mice (Fig. 7).
Discussion
In the current study, intestinal injury was assessed
based on a variety of factors, including body weight, disease
activity score, colon length and histology. In each assessment,
colonic injury was found to be significantly aggravated in the
DSS-treated MT-I/II knockout mice, as compared with the DSS-treated
wild-type mice. Histological analysis also showed an enhanced
infiltration of inflammatory cells, particularly neutrophils and
mononuclear cells, as well as mucosal epithelial cell disruption in
the DSS-treated MT-I/II knockout mice. Enhanced intestinal injury
and neutrophil accumulation in MT-I/II knockout mice were also
confirmed by the measurement of histological scores using a
previously reported grading system (30,31) and MPO activity of the colon,
respectively.
A number of previous studies have described the role
of MTs in the inflammatory response to DSS. Tran et al
(20) first demonstrated that
MT-deficient mice had significantly lower DAI when compared with
wild-type mice in a model of DSS-induced colitis. However, the
results from our study are not in agreement with the histological
assessment made in the study by Tran et al. Another study by
Oz et al (19) reported no
differences in histological damage following treatment with DSS
among wild-type mice, MT-deficient mice and MT transgenic mice.
However, their study did not include the assessment of DAI. On the
other hand, our data suggest that MT deficiency results in disease
exacerbation in a mouse model of DSS-induced colitis, and that MTs
play a protective role in the development of intestinal
inflammation. While considering these results, we noted differences
in the manufacture of DSS and vivarium in these studies, and
it has been reported that these factors can lead to differences in
the severity of DSS-induced colitis (34,35).
As regards MT expression in intestinal inflammation,
there are conflicting reports on the role of MTs in intestinal
inflammation. Mulder et al (36) demonstrated that MT expression was
decreased in the colonic mucosa of patients with IBD. Kruidenier
et al (37) also reported
that patients with IBD had decreased MT expression levels in the
colon. By contrast, Lih-Brody et al (38) and Bruwer et al (39) reported that MT expression was
markedly elevated in the colonic mucosa of patients with IBD.
Similarly, MT-I/II expression was significantly higher in the
inflamed colonic mucosa in mice with DSS-induced colitis, as
compared with normal colonic mucosa in wild-type mice in the
present study.
In the present study, in order to investigate the
localization of MTs in the intestine, immunohistochemical staining
was performed with murine colonic samples using anti-MT-I/II
antibody. MT-I/II expression was mainly observed in mononuclear
cells in the lamina propria and submucosal layer of the murine
intestine. Further examination using immunofluorescence staining
revealed that these mononuclear cells were F4/80-positive
macrophages. Although the localization of MTs in the
gastrointestinal tract is not yet fully understood, Kruidenier
et al (37) demonstrated
that MTs are mainly expressed in the small intestinal epithelium,
not in cells in the lamina propria. By contrast, a previous study
by Al-Gindan et al (40)
demonstrated that MT expression is localized in submucosal cells in
the colonic mucosa in rats, similar to our results.
As regards the expression of MTs in macrophages,
there is an extensive amount of evidence suggesting that alveolar
macrophages show strong MT expression. Mehta et al (41) demonstrated that the reduction in
MT expression induced by zinc deficiency in alveolar macrophages
inhibits phagocytic function, indicating that MTs mediate innate
immune function. Pankhurst et al (42) also reported that MT-expressing
macrophages in the murine brain may play a crucial role in the
regulation of Th1/Th2 cytokine balance. In this study, the
production of Th1, Th2 and Th17 cytokines was significantly
elevated in the MT-I/II knockout mice, indicating that MTs possibly
regulate the production of these cytokines. It is known that TNF-α,
IFN-γ and IL-17 are mainly released by helper T cells (Th-1, Th-2
and Th-17) and macrophages (43,44), and large amounts of cytokine
expression lead to severe mucosal inflammation in ulcerative
colitis and Crohn’s disease (45). In the present study, we evaluated
the function of macrophages derived from MT-deficient mice, as the
number of infiltrating F4/80-positive macrophages in the intestinal
mucosa of mice treated with DSS did not differ between the
wild-type and MT-I/II knockout mice. We found that the production
of inflammatory cytokines (TNF-α, IFN-γ and IL-17) in the
supernatant of macrophages isolated from MT-I/II knockout mice
following LPS stimulation was significantly higher compared to
wild-type mice. Thus, MTs appear to play an important role in the
anti-inflammatory function of macrophages and MT-expressing
macrophages may play a protective role in the pathogenesis of
IBDs.
In conclusion, our results suggest that MTs play a
protective role against colonic mucosal inflammation in a mouse
model of DSS-induced colitis through their anti-inflammatory
function in macrophages, indicating that endogenous MTs play an
important role in the protection of the intestinal mucosa. In the
future, MTs may prove to be novel therapeutic target molecules in
IBDs.
Acknowledgements
The authors thank Dr Chiharu Tohyama (The University
of Tokyo) for providing the MT-I/II knockout mice.
References
|
1
|
Hanauer SB: Inflammatory bowel disease:
Epidemiology, pathogenesis, and therapeutic opportunities. Inflamm
Bowel Dis. 12(Suppl 1): S3–S9. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Xavier RJ and Podolsky DK: Unravelling the
pathogenesis of inflammatory bowel disease. Nature. 448:427–434.
2007. View Article : Google Scholar
|
|
3
|
Strober W, Fuss I and Mannon P: The
fundamental basis of inflammatory bowel disease. J Clin Invest.
117:514–521. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Naito Y, Takagi T and Yoshikawa T:
Molecular fingerprints of neutrophil-dependent oxidative stress in
inflammatory bowel disease. J Gastroenterol. 42:787–798. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Iborra M, Moret I, Rausell F, et al: Role
of oxidative stress and antioxidant enzymes in Crohn’s disease.
Biochem Soc Trans. 39:1102–1106. 2011.
|
|
6
|
Margoshes M and Vallee BL: Flame
photometry and spectrometry; principles and applications. Methods
Biochem Anal. 3:353–407. 1956. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kojima Y and Hunziker PE: Amino acid
analysis of metallothionein. Methods Enzymol. 205:419–421. 1991.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Minami T, Yoshita C, Tanaka M, Kubo K,
Okabe N and Okazaki Y: Separation of metallothionein isoforms of
mouse liver cytosol by capillary zone electrophoresis. Talanta.
46:347–354. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Coyle P, Philcox JC, Carey LC and Rofe AM:
Metallothionein: the multipurpose protein. Cell Mol Life Sci.
59:627–647. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Alvarez L, Gonzalez-Iglesias H, Garcia M,
Ghosh S, Sanz-Medel A and Coca-Prados M: The stoichiometric
transition from Zn6Cu1-metallothionein to
Zn7-metallothionein underlies the up-regulation of
metallothionein (MT) expression: quantitative analysis of MT-metal
load in eye cells. J Biol Chem. 287:28456–28469. 2012.PubMed/NCBI
|
|
11
|
Takano H, Inoue K, Yanagisawa R, et al:
Protective role of metallothionein in acute lung injury induced by
bacterial endotoxin. Thorax. 59:1057–1062. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ashino T, Arima Y, Shioda S, Iwakura Y,
Numazawa S and Yoshida T: Effect of interleukin-6 neutralization on
CYP3A11 and metallothionein-1/2 expressions in arthritic mouse
liver. Eur J Pharmacol. 558:199–207. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Pedersen DS, Fredericia PM, Pedersen MO,
et al: Metallic gold slows disease progression, reduces cell death
and induces astrogliosis while simultaneously increasing stem cell
responses in an EAE rat model of multiple sclerosis. Histochem Cell
Biol. 138:787–802. 2012. View Article : Google Scholar
|
|
14
|
Inoue K, Takano H, Shimada A, et al: Role
of metallothionein in coagulatory disturbance and systemic
inflammation induced by lipopolysaccharide in mice. FASEB J.
20:533–535. 2006.PubMed/NCBI
|
|
15
|
Takano H, Satoh M, Shimada A, Sagai M,
Yoshikawa T and Tohyama C: Cytoprotection by metallothionein
against gastroduodenal mucosal injury caused by ethanol in mice.
Lab Invest. 80:371–377. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Mita M, Satoh M, Shimada A, et al:
Metallothionein is a crucial protective factor against
Helicobacter pylori-induced gastric erosive lesions in a
mouse model. Am J Physiol Gastrointest Liver Physiol.
294:G877–G884. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kimura T, Itoh N, Takehara M, et al:
Sensitivity of metallothionein-null mice to
LPS/D-galactosamine-induced lethality. Biochem Biophys Res Commun.
280:358–362. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Klosterhalfen B, Tons C, Hauptmann S, et
al: Influence of heat shock protein 70 and metallothionein
induction by zinc-bis-(DL-hydrogenaspartate) on the release of
inflammatory mediators in a porcine model of recurrent endotoxemia.
Biochem Pharmacol. 52:1201–1210. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Oz HS, Chen T, de Villiers WJ and McClain
CJ: Metallothionein overexpression does not protect against
inflammatory bowel disease in a murine colitis model. Med Sci
Monit. 11:BR69–BR73. 2005.PubMed/NCBI
|
|
20
|
Tran CD, Ball JM, Sundar S, Coyle P and
Howarth GS: The role of zinc and metallothionein in the dextran
sulfate sodium-induced colitis mouse model. Dig Dis Sci.
52:2113–2121. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Korenaga D, Takesue F, Kido K, et al:
Impaired antioxidant defense system of colonic tissue and cancer
development in dextran sulfate sodium-induced colitis in mice. J
Surg Res. 102:144–149. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Vowinkel T, Kalogeris TJ, Mori M,
Krieglstein CF and Granger DN: Impact of dextran sulfate sodium
load on the severity of inflammation in experimental colitis. Dig
Dis Sci. 49:556–564. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Melgar S, Karlsson A and Michaelsson E:
Acute colitis induced by dextran sulfate sodium progresses to
chronicity in C57BL/6 but not in BALB/c mice: correlation between
symptoms and inflammation. Am J Physiol Gastrointest Liver Physiol.
288:G1328–G1338. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Schepp-Berglind J, Atkinson C, Elvington
M, Qiao F, Mannon P and Tomlinson S: Complement-dependent injury
and protection in a murine model of acute dextran sulfate
sodium-induced colitis. J Immunol. 188:6309–6318. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Michalska AE and Choo KH: Targeting and
germ-line transmission of a null mutation at the metallothionein I
and II loci in mouse. Proc Natl Acad Sci USA. 90:8088–8092. 1993.
View Article : Google Scholar
|
|
26
|
Takagi T, Naito Y, Uchiyama K, et al:
Carbon monoxide liberated from carbon monoxide-releasing molecule
exerts an anti-inflammatory effect on dextran sulfate
sodium-induced colitis in mice. Dig Dis Sci. 56:1663–1671. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Naito Y, Katada K, Takagi T, et al:
Rosuvastatin, a new HMG-CoA reductase inhibitor, reduces the
colonic inflammatory response in dextran sulfate sodium-induced
colitis in mice. Int J Mol Med. 17:997–1004. 2006.PubMed/NCBI
|
|
28
|
Cooper HS, Murthy SN, Shah RS and
Sedergran DJ: Clinicopathologic study of dextran sulfate sodium
experimental murine colitis. Lab Invest. 69:238–249.
1993.PubMed/NCBI
|
|
29
|
Howarth GS, Xian CJ and Read LC:
Predisposition to colonic dysplasia is unaffected by continuous
administration of insulin-like growth factor-I for twenty weeks in
a rat model of chronic inflammatory bowel disease. Growth Factors.
18:119–133. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Smith P, Mangan NE, Walsh CM, et al:
Infection with a helminth parasite prevents experimental colitis
via a macrophage-mediated mechanism. J Immunol. 178:4557–4566.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Siegmund B, Lehr HA, Fantuzzi G and
Dinarello CA: IL-1 beta-converting enzyme (caspase-1) in intestinal
inflammation. Proc Natl Acad Sci USA. 98:13249–13254. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Grisham MB, Hernandez LA and Granger DN:
Xanthine oxidase and neutrophil infiltration in intestinal
ischemia. Am J Physiol. 251:G567–G574. 1986.PubMed/NCBI
|
|
33
|
Ban K and Kozar RA: Protective role of
p70S6K in intestinal ischemia/reperfusion injury in mice. PloS One.
7:e415842012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Bamba S, Andoh A, Ban H, et al: The
severity of dextran sodium sulfate-induced colitis can differ
between dextran sodium sulfate preparations of the same molecular
weight range. Dig Dis Sci. 57:327–334. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Smith P, Siddharth J, Pearson R, et al:
Host genetics and environmental factors regulate ecological
succession of the mouse colon tissue-associated microbiota. PloS
One. 7:e302732012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Mulder TP, Verspaget HW, Janssens AR, de
Bruin PA, Pena AS and Lamers CB: Decrease in two intestinal
copper/zinc containing proteins with antioxidant function in
inflammatory bowel disease. Gut. 32:1146–1150. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kruidenier L, Kuiper I, van Duijn W, et
al: Differential mucosal expression of three superoxide dismutase
isoforms in inflammatory bowel disease. J Pathol. 201:7–16. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Lih-Brody L, Powell SR, Collier KP, et al:
Increased oxidative stress and decreased antioxidant defenses in
mucosa of inflammatory bowel disease. Dig Dis Sci. 41:2078–2086.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Bruwer M, Schmid KW, Metz KA, Krieglstein
CF, Senninger N and Schurmann G: Increased expression of
metallothionein in inflammatory bowel disease. Inflamm Res.
50:289–293. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Al-Gindan Y, Shawarby M, Noto A and Taylor
CG: Intestinal inflammation in rats induces metallothionein in
colonic submucosa. J Clin Biochem Nutr. 44:131–141. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Mehta AJ, Joshi PC, Fan X, et al: Zinc
supplementation restores PU.1 and Nrf2 nuclear binding in alveolar
macrophages and improves redox balance and bacterial clearance in
the lungs of alcohol-fed rats. Alcohol Clin Exp Res. 35:1519–1528.
2011.PubMed/NCBI
|
|
42
|
Pankhurst MW, Bennett W, Kirkcaldie MT,
West AK and Chung RS: Increased circulating leukocyte numbers and
altered macrophage phenotype correlate with the altered immune
response to brain injury in metallothionein (MT)-I/II null mutant
mice. J Neuroinflammation. 8:1722011. View Article : Google Scholar
|
|
43
|
Otani K, Watanabe T, Tanigawa T, et al:
Anti-inflammatory effects of IL-17A on Helicobacter
pylori-induced gastritis. Biochem Biophys Res Commun.
382:252–258. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Yang Z, Ding J, Yang C, et al:
Immunomodulatory and anti-inflammatory properties of artesunate in
experimental colitis. Curr Med Chem. 19:4541–4551. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Kamada N, Hisamatsu T, Honda H, et al:
TL1A produced by lamina propria macrophages induces Th1 and Th17
immune responses in cooperation with IL-23 in patients with Crohn’s
disease. Inflamm Bowel Dis. 16:568–575. 2010.PubMed/NCBI
|