Introduction
Diabetic nephropathy (DN) is one of the main chronic
complications of diabetes mellitus (DM) associated with
capillaries, often leading to chronic renal failure and end-stage
renal disease (1). Although the
precise pathogenesis of DN has not been elucidated, a number of
studies have demonstrated that glomerular hyperfiltration and
mesangial expansion are the two characteristic pathophysiological
changes occurring during the early stages of DN (2,3).
Mesangial cells (MCs), located within glomerular
capillary loops, play an important role in the regulation of
glomerular hemodynamics due to their contractile function (4). There is increasing evidence
indicating that hyperglycemia directly stimulates MCs, which
subsequently results in mesangial contractile dysfunction and
glomerular hyperfiltration (5,6).
The hypocontractility of MCs is closely related to the reduced
calcium influx (7,8).
Canonical transient receptor potential cation
channels (TRPCs), as members of the transient receptor potential
(TRP) superfamily, are Ca2+ permeable cation channels
widely expressed in a series of tissues and cells (9,10).
The TRPC family includes seven related members, designated as
TRPC1–7 (11). Among these, TRPC6
is closely associated with kidney disease (12,13). Möller et al (14) found that TRPC6 exists throughout
the glomerulus in kidney tissues, particularly in MCs.
Additionally, it has been dcemonstrated that hyperglycemia
downregulates the expression of TRPC6 protein, which results in a
decrease in intracellular calcium, leading to impaired MC
contractile response and glomerular hyperfiltration (15). However, the underlying molecular
mechanisms of TRPC6 protein downregulation in MCs have not yet been
fully elucidated.
Hyperglycemia can also cause the abnormal
proliferation of MCs in DN, leading to glomerular hypertrophy and
fibrosis (16). Clinical and
animal experiments have demonstrated an increase in the production
of reactive oxygen species (ROS) in MCs, as the direct result of
chronic exposure to high glucose (HG) (17,18). The overproduction of ROS can
transduce and amplify glucose signaling, playing a key role in MC
proliferation (19). ROS can also
modulate Ca2+ channels by activating various signaling
cascades (20).
In the kidneys, particularly in MCs, nicotinamide
adenine dinucleotide phosphate (NADPH) oxidase is the predominant
source of ROS (21). The
phagocyte NADPH oxidase consists of two membrane-associated
subunits, p22phox and gp91phox, and at least
four cytoplasmic components, p47phox,
p67phox, p40phox and the small GTPase, Rac.
NADPH oxidase in the resting state becomes activated upon
interaction between the catalytic core (membrane-associated
subunits) and the cytosolic regulatory subunits (22). There are six homologues of
phagocytic gp91phox proteins expressed by distinct
non-phagocytic cells (23). It
has been reported that NADPH oxidase 4 (Nox4) is the key subunit of
NADPH oxidase expressed in MCs, and Nox4-derived ROS is the major
contributor to renal morphological changes and functional
abnormalities in DN (24).
Akt, also known as protein kinase B (PKB), belonging
to serine/threonine kinase family members, is one of the downstream
effectors of phosphoinositide 3-kinase (PI3K) which participates in
numerous signaling pathways involved in diverse physiological
processes, including glucose metabolism, protein synthesis, cell
proliferation, cell apoptosis (25,26). Kim et al reported that Akt
was activated in renal damage in streptozotocin-induced diabetic
mice (27). It has also been
demonstrated that Akt is an important mediator of MC proliferation,
and can be regulated by ROS (28).
Akt is not only the downstream signaling molecule of
PI3K, but also the major upstream element in the activation of
nuclear factor-κB (NF-κB). Akt promotes the transcriptional
activity of NF-κB through a variety of mechanisms and the signaling
cascade eventually leads to cell proliferation and migration
(29). A number of studies have
demonstrated that NF-κB is activated in MCs by hyperglycemia, and
plays a crucial role in the progression of DN (30,31). In addition, a recent study found
that ROS is the important messenger in the NF-κB signaling pathway
(32). Of note, the promoter
region of TRPC6 has NF-κB binding sites (33). Therefore, we hypothesized that the
redox-sensitive NF-κB participates in the downregulation of TRPC6
in DN. Thus, we aimed to explore whether NADPH oxidase-derived ROS
is involved in HG-induced cell proliferation and the downregulation
of TRPC6 in MCs through the Akt/NF-κB pathway.
Astragaloside IV (AS-IV,
3-O-β-D-xylopyranosyl-6-O-β-D-glucopyranosylcycloastragenol), a
purified small molecular saponin, is one of the main active
ingredients of Radix Astragali, which has been reported to possess
comprehensive biological properties, including antioxidant,
anti-inflammatory, immunoregulatory and anti-aging properties and
to improve intellectual development (34). A recent studies indicated that
AS-IV ameliorates proteinuria in rats with adriamycin nephropathy
(35). It has also been reported
that AS-IV significantly inhibits renal oxidative stress and
apoptosis in STZ-induced diabetic rats (36). However, the protective effects and
the precise mechanisms of action of AS-IV on oxidative
stress-induced injury in MCs under HG conditions have not yet been
fully elucidated.
The present study aimed to investigate the effects
of AS-IV on HG-induced MC proliferation and the downregulation of
TRPC6 through a mechanism associated with the inhibition of NADPH
oxidase-mediated ROS production, Akt and NF-κB activation, in an
attempt to provide a novel therapeutic approach for the treatment
of DN.
Materials and methods
Reagents
Dulbecco’s modified Eagle’s medium (DMEM) and fetal
bovine serum (FBS) were purchased from HyClone Laboratories, Inc.
(Logan, UT, USA). Penicillin/streptomycin solution, 0.05%
Trypsin-EDTA, phoshate-buffered saline (PBS), and dimethyl
sulfoxide (DMSO) were purchased from Invitrogen Life Technologies
(Carlsbad, CA, USA). 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl
tetrazolium bromide (MTT), α-mannitol (MA), diphenyleneiodonium
(DPI), tempol, LY294002, sulfasalazine (Sul), Fluo3/AM ester, and
2′,7′-dichlorofluorescein diacetate (DCFH-DA) were obtained from
Sigma (St. Louis, MO, USA). The cell NADPH oxidase colorimetric
assay kit was purchased from GenMed Scientifics Inc. (Boston, MA,
USA). Antibodies were obtained from the following sources:
anti-Nox4 polyclonal antibody (pAb), anti-phospho-Akt pAb and
anti-Akt pAb were from Santa Cruz Biotechnology, Inc. (Santa Cruz,
CA, USA); anti-phospho-IκBα monoclonal antibody (mAb) and anti-IκBα
mAb were from Cell Signaling Technology, Inc. (Beverly, MA, USA);
anti-TRPC6 mAb was from Abcam (Cambridge, UK); anti-β-actin mAb,
horseradish peroxidase (HRP)-conjugated AffiniPure goat anti-mouse
IgG and anti-rabbit IgG were purchased from Zhongshan Golden Bridge
Biotechnology Co., Ltd. (Beijing, China).
AS-IV was purchased from Nanjing Zelang Medical
Technology Co., Ltd. (Nanjing, China; purity >98%, HPLC). The
chemical structure of AS-IV
(C41H68O14; molecular weight, 784)
is depicted in Fig. 1. AS-IV was
dissolved in DMSO to the concentration of 25 μmol/ml as a stock
solution. The stock solution was diluted with DMEM into AS-IV
solutions according to the respective group when used and the final
DMSO concentration did not exceed 0.5% (v/v).
MC culture
The human mesangial cell line (HMC) was obtained
from the Modern Analysis and Testing Center of Central South
University (Changsha, China), and maintained in normal DMEM (5.6 mM
glucose) supplemented with 10% FBS (v/v), 100 μ/ml penicillin and
100 μg/ml streptomycin at 37°C in an atmosphere containing 5%
CO2. HG treatment was performed by culturing the cells
in DMEM containing 25 mM glucose for 48 h.
Cell proliferation assay
Cells were seeded at a density of 1×104
cells/well in 96-well plates. When the cell confluence reached at
70–80%, the growth medium was replaced with DMEM containing 5.6 mM
glucose and 0.5% FBS. After 24 h, the quiescent cells were treated
with the indicated concentrations of glucose together with various
concentrations of AS-IV (5, 10, 25, 50 and 100 μM) or 0.5% DMSO
(vehicle control) for 48 h. The osmotic control medium was made by
supplementing normal DMEM with 24.5 mM MA. Following incubation
with the above-mentioned compounds, cell proliferation was
determined by MTT assay (37).
The absorbance was measured at 490 nm using a SpectraMax 190
Microplate Reader (Molecular Devices, Sunnyvale, CA, USA).
Measurement of total protein to cell
count ratio
The ratio of the total protein content to the cell
number is a well-established measure of cellular hypertrophy
(38). The MCs were seeded into
6-well plates and were synchronized into quiescence in DMEM
containing a normal glucose concentration and 0.5% FBS for 24 h.
The cells were then stimulated with HG and treated with various
concentrations of AS-IV (5, 10, 25, 50 and 100 μM) or 0.5% DMSO
(vehicle control) for 48 h. Following incubation, the cells were
washed twice with PBS and trypsinized. A small aliquot of the cells
was used for cell counting by a hemocytometer. The remaining cells
were lysed in RIPA buffer (Beyotime, Haimen, China), and the total
protein content was measured by a protein quantitative reagent
kit-BCA method (Beyotime). The total protein content to the cell
count ratio was expressed as microgram protein per 104
cells.
Detection of intracellular ROS
generation
The generation of ROS was detected using the
membrane permeable indicator, DCFH-DA. The cells were seeded in
24-well plates at a density of 1×105 cells/well. After
being synchronized, the cells were cultured in DMEM containing 5.6
or 25 mM glucose with or without various concentrations of AS-IV
(25, 50 and 100 μM) or tempol (100 μM) for 48 h, were then loaded
with 10 μM DCFH-DA in serum-free DMEM containing 5.6 or 25 mM
glucose at 37°C for 30 min in the dark, and the cell culture plate
was shaken every 5 min and washed three times with PBS in order to
remove residual probes. Subsequently, intracellular ROS production
were observed under a fluorescence microscope (Nikon, Tokyo, Japan;
excitation at 488 nm, emission at 525 nm). The mean fluorescence
intensity for each group of cells was determined using the
Image-Pro Plus 6.0 analysis system (MediaCybernetics, Rockville,
MD, USA).
NADPH oxidase activity assay
NADPH oxidase activity in the MCs was measured using
the cell NADPH oxidase colorimetric assay kit (GenMed Scientifics
Inc.) according to the manufacturer’s instructions. Briefly, the
cells grown in DMEM containing 5.6 or 25 mM glucose in the presence
or absence of AS-IV (25, 50 and 100 μM) or DPI (10 μM) for 48 h
were washed twice in PBS and scraped from the plate followed by
centrifugation at 12,000 g, 4°C, for 3 min, and suspended in PBS,
followed by incubation with 250 μM NADPH. NADPH consumption was
monitored by a decrease in absorbance at 340 nm for 5 min using a
SpectraMax 190 Microplate Reader (Molecular Devices). NADPH oxidase
activity was defined as picomoles per liter of substrate per minute
per milligram of protein.
Western blot analysis
The cells were lysed with cold lysis buffer
containing protease inhibitors. Equal amounts of protein extracts
were fractionated by SDS-PAGE and then transferred onto
polyvinylidene difluoride (PVDF) membranes (Millipore Corp.,
Bedford, MA, USA). After being blocked with 5% non-fat milk in
Tris-buffered saline with Tween-20 (TBST, pH 7.6) for 1 h at room
temperature, the membranes were incubated with the indicated
primary antibodies (Nox4, 1:200; Akt, 1:500; phospho-Akt, 1:500;
IκBα, 1:1,000; phospho-IκBα, 1:200; TRPC6, 1:1,000 and β-actin,
1:1,000) overnight at 4°C. The membranes membranes were rinsed
three times with TBST and incubated with the respective secondary
antibodies (1:10,000 dilutions of each antibody) for 1 h at room
temperature. The protein bands were visualized with SuperSignal
West Femto Maximum Sensitivity Substrate (Thermo Fisher Scientific
Inc., Rockford, IL, USA) and captured using a Bioshine ChemiQ 4600
Mini Chemiluminescence imaging system (Ouxiang, Shanghai, China).
The optical density of each band was quantified using ImageJ
software (NIH, Bethesda, MD, USA) and normalized to the intensity
of β-actin.
Fluorescence measurement of
[Ca2+]i
Measurements of the intracellular free calcium
concentration were performed using Fluo-3/AM
fluorospectrophotometry. The MCs, grown on 24-well plates, were
loaded with Fluo-3 by incubation for 40 min at 37°C in the dark in
HEPES buffer solution containing 5 μM Fluo-3/AM followed by washing
three times with the same buffer. The cells were then incubated
with Fluo-3-free HEPES buffer for an additional 20 min. Afer being
trypsinized and collected, the fluorescence intensity (F) was
monitored using an F-4600 fluorescence spectrophotometer (Hitachi,
Tokyo, Japan; excitation at 488 nm, emission at 530 nm). The
maximal density (Fmax) was measured using 0.1% Triton
X-100, and the minimum (Fmin) was measured using 20 mM
EGTA. The concentration of calcium was calculated using the
following formula: [Ca2+]i = Kd (F
- Fmin)/(Fmax - F), where Kd
represents the dissociation constant of Fluo-3 and calcium, and its
value is 450 nM.
Statistical analysis
Data are presented as the means ± standard deviation
(SD). Each experiment was repeated at least three times
independently. Statistical analysis was performed using SPSS 13.0
for Windows (SPSS, Inc., Chicago, IL, USA). Statistical differences
between two groups were analyzed by the unpaired Student’s t-test
and differences among multiple groups were analyzed by one-way
ANOVA. In all cases, values of P<0.05 were considered to
indicate statistically significant differences.
Results
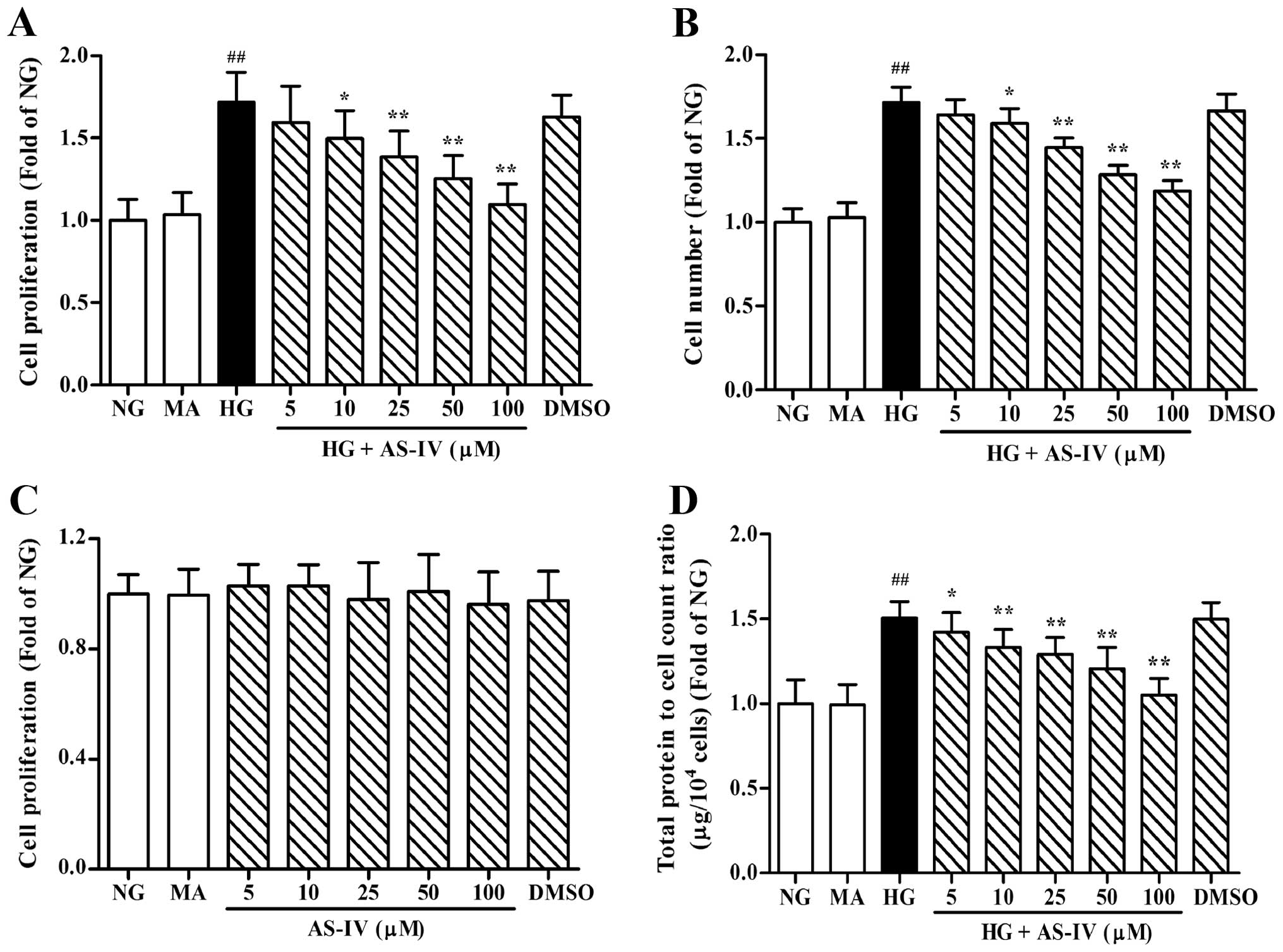
Effects of AS-IV on HG-induced MC
proliferation and hypertrophy
To determine the effects of AS-IV on MC
proliferation, the MTT assay and cell counting were employed. As
shown in Fig. 2A and B, HG
significantly stimulated the growth of the MCs in comparison to
normal glucose (NG) conditions (P<0.01). The administration of
AS-IV at the concentration range of 5–100 μM led to a significant
inhibition of cell growth induced by HG. The vehicle control
treated with DMEM containing HG and 0.5% DMSO also showed a
significant increase in the proliferation of MCs. In addition,
AS-IV at the examined concentrations had no effect on the viability
of MCs under NG conditions, which suggested that the inhibitory
effect of AS-IV upon cultured MCs was not due to its cytotoxicity
(Fig. 2C). HG also markedly
stimulated cell hypertrophy, defined as the protein content of MCs
per unit cell number, which was abolished by the AS-IV
administration in a dose dependent manner (Fig. 2D). Unlike HG, the addition of 24.5
mM MA to the medium did not exert an obvious effect on the
proliferation and hypertrophy of MCs as compared with control,
suggesting that the HG-triggered MC proliferation and hypertrophy
were not the results of high osmolality within the medium.
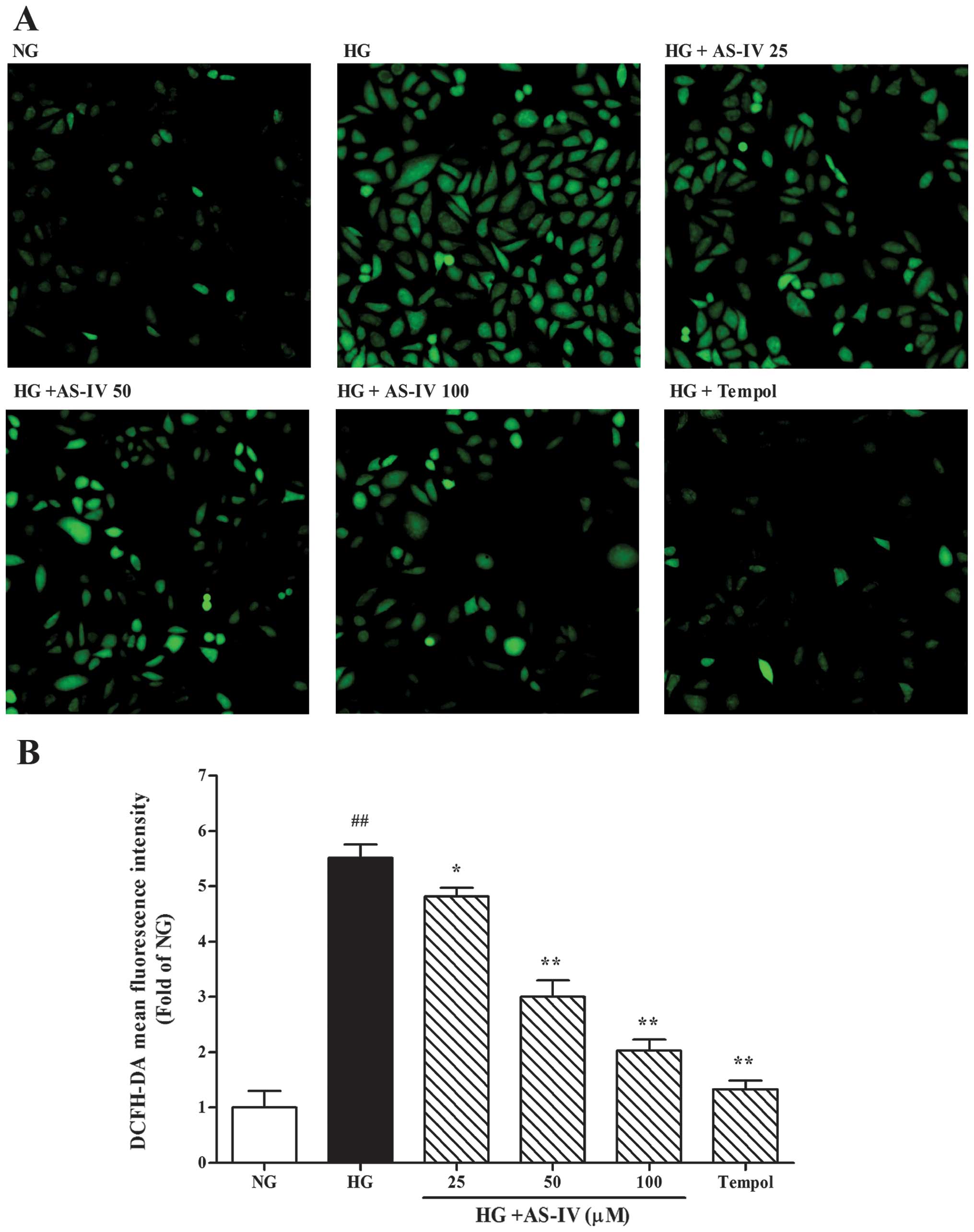
Effect of AS-IV on HG-stimulated ROS
generation in MCs
We examined the effects of AS-IV on HG-induced
intracellular ROS generation by DCFH-DA fluorescent probe assay
using fluorescence microscopy. As demonstrated in Fig. 3, the MCs cultured under HG
conditions for 48 h showed a significant increase in ROS generation
compared to the cells cultured under NG conditions (P<0.01). To
assess the antioxidative effects of AS-IV, we used tempol, a
classic antioxidant, as a positive control. The effects of
HG-induced ROS generation in the MCs were notably decreased by
treatment with AS-IV (25, 50 and 100 μM) or tempol (100 μM), and
AS-IV exerted its antioxidant effects in a dose-dependent
manner.
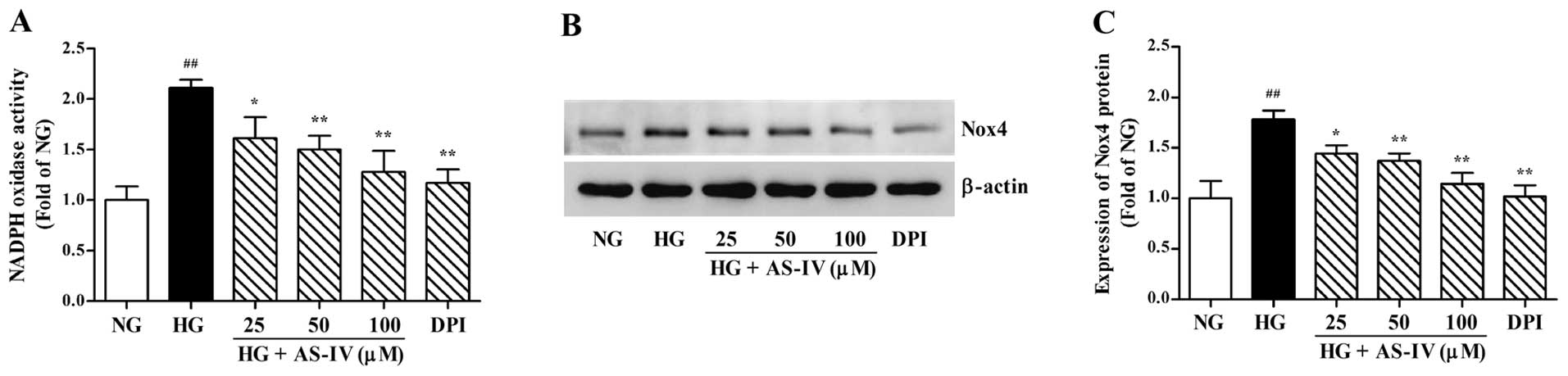
Effect of AS-IV on HG-mediated NADPH
oxidase activation in MCs
Since NADPH oxidase is the most important source of
intracellular ROS, we used the NADPH oxidase inhibitor, DPI, as a
positive control to assess the effects of AS-IV on HG-induced NADPH
oxidase activity. As shown in Fig.
4A, HG resulted in a significant increase in NADPH activity
that was markedly attenuated by AS-IV (25, 50 and 100 μM) or DPI
(10 μM). As Nox4 is the key membrane subunit of NADPH oxidase
expressed in MCs, we further examined whether AS-IV blocks the
expression level of Nox4 protein. The protein level of the Nox4
subunit was notably upregulated under HG conditions compared to the
control (P<0.01). Treatment with AS-IV (25, 50 and 100 μM) or
DPI (10 μM) for 48 h markedly downregulated the protein expression
level of Nox4 compared to the HG-treated group (Fig. 4B and C).
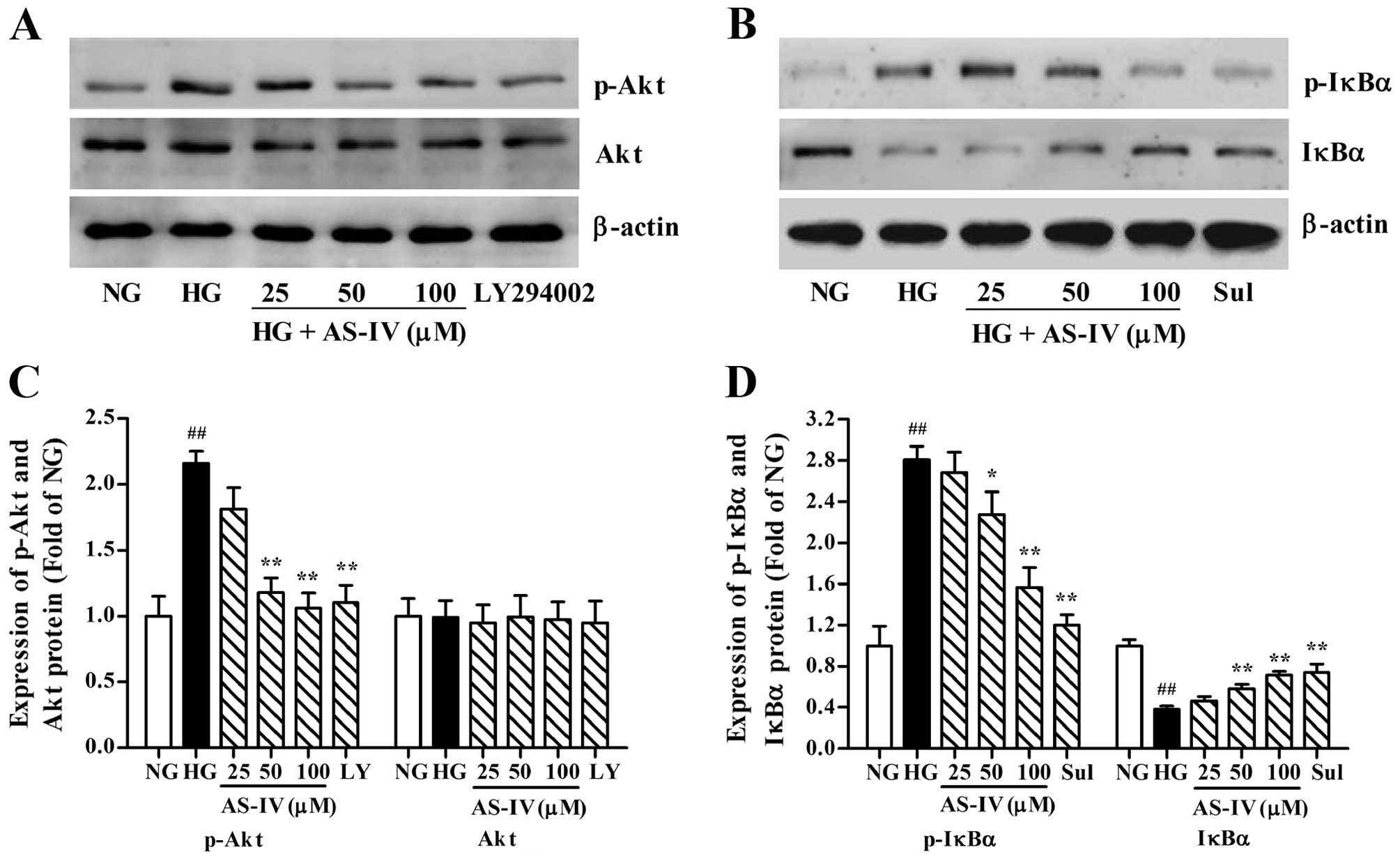
Inhibitory effects of AS-IV on HG-induced
phosphorylation of Akt and IκBα in MCs
Given the role that Akt and NF-κB signaling plays in
MC growth and proliferation, we examined the effects of AS-IV on
Akt and NF-κB activation. The phosphorylation level of Akt and IκBα
and the protein expression of total Akt and total IκBα were
detected by western blot analysis. It was observed that HG induced
Akt and NF-κB activation, as manifested by the fact that the
relative amount of phosphorylated Akt and IκBα and the degradation
of IκBα were significantly higher compared to the control cells
(Fig. 5). However, treatment with
AS-IV or the PI3K inhibitor, LY294002 (10 μM), effectively
abrogated the HG-induced Akt phosphorylation in the MCs, and the
phosphorylation and degradation level of IκBα was also markedly
inhibited by AS-IV or the IκBα inhibitor, Sul (0.5 mM). AS-IV did
not affect the protein expression level of total Akt in the
HG-cultured cells. These results suggest that AS-IV inhibits
HG-induced Akt and NF-κB activation in MCs.
Regulatory effect of AS-IV on the NADPH
oxidase/ROS/Akt/ NF-κB signaling pathway in MCs under HG
conditions
To further investigate the signaling cascade
involved in HG-induced NADPH oxidation, Akt and NF-κB activation,
specific inhibitors of several signaling molecules were used.
Treatment with NADPH oxidase inhibitor (DPI, 10 μM) or ROS
inhibitor (tempol, 100 μM) blocked Akt and NF-κB activation in the
MCs induced by HG (Fig. 6),
indicating that NADPH oxidase activation induced by HG occurs
upstream of Akt and NF-κB in MCs. The HG-induced Akt
phosphorylation was abolished by treatment with PI3K inhibitor
(LY294002, 10 μM), whereas Sul (0.5 mM), an IκBα inhibitor, failed
to inhibit Akt phosphorylation (Fig.
6A and C). IκBα phosphorylation and degradation were also
inhibited in the presence of Sul, LY294002 and DPI or tempol
(Fig. 6B and D). These data
demonstrate that the PI3K/Akt pathway is upstream of NF-κB in MCs
cultured under hyperglycemic conditions.
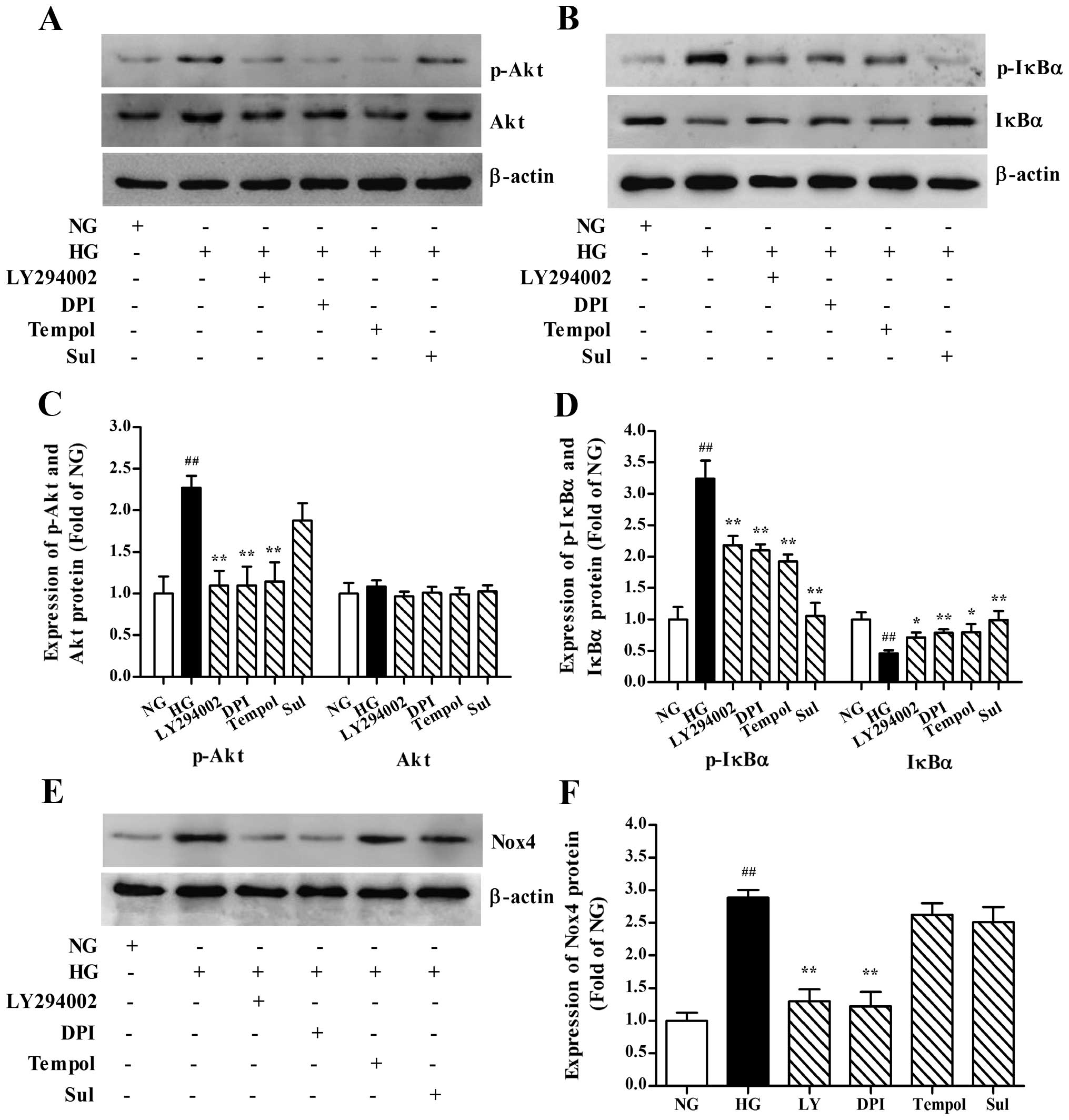 | Figure 6High glucose (HG)-potentiated
nicotinamide adenine dinucleotide phosphate (NADPH) oxidase
activation is affected by the phosphoinositide 3-kinase (PI3K)/Akt
pathway, but not nuclear factor-κB (NF-κB) in mesangial cells
(MCs). (A, B and E) Cells were incubated with HG in presence or
absence of various inhibitors [LY294002, 10 μM; diphenyleneiodonium
(DPI), 10 μM; tempol, 100 μM; sulfasalazine (Sul), 0.5 mM)] for 48
h. The phosphorylation level of Akt and IκBα, and the expression
level of Nox4, total Akt and total IκBα protein were determined by
western blot analysis. β-actin was used as a loading control. (C, D
and F) Representative quantitative analysis for results of (C) Akt,
(D) IκBα, and (F) Nox4 were expressed as fold change from the
control. ##P<0.01 compared with normal glucose (NG);
*P<0.05, **P<0.01 compared with HG.
Data in the bar graphs represent the means ± standard deviation
(SD) of three independent experiments. |
We also examined whether NADPH oxidase activation in
MCs is dependent on Akt or NF-κB activation. The expression level
of the Nox4 subunit was evaluated in the HG-exposed cells treated
with or without LY294002, DPI, tempol or Sul. LY294002 and DPI
effectively inhibited the HG-induced increase in the expression of
Nox4. However, no significant change in the expression level of
Nox4 was observed in the MCs treated with tempol and Sul (Fig. 6E and F). Taken together, the above
results suggest that NADPH oxidase activation and the PI3K/Akt
pathway may function in parallel or may interplay with each other,
which are upstream of NF-κB in HG-stimulated MCs.
Inhibitory effects of AS-IV on the
HG-induced downregulation of TRPC6 and the reduction in calcium
influx in MCs
TRPC6 is known as a Ca2+-conductive
cation channel and regulates the contractile function of MCs; it
plays a pivotal role during the early stages of HG-induced damage
to MCs (14,15). Thus, in this study, we examined
the effects of AS-IV on the expression level of TRPC6 protein and
the concentration of intracellular free calcium in MCs cultured
under HG conditions. As illustrated in Fig. 7A and C, incubation of the MCs with
HG for 48 h markedly decreased the expression level of TRPC6
protein compared with the cells cultured under NG conditions. The
downregulation of TRPC6 induced by HG was markedly abrogated by
treatment with AS-IV at a concentration of 25 to 100 μM and the
TRPC6 agonist, a diacylglycerol analog,
1-oleoyl-2-acetyl-sn-glycerol (OAG, 100 μM). Furthermore, in the
presence of inhibitors of signaling molecules, such as LY294002,
DPI, tempol and Sul, the HG-induced TRPC6 downregulation was
markedly abolished (Fig. 7B and
D). Ultimately, we detected the intracellular free calcium
concentration in MCs using Fluo-3/AM fluorospectrophotometry. As
shown in Fig. 7E, the HG-induced
reduction in calcium influx in the MCs was also greatly ameliorated
by AS-IV (25, 50 and 100 μM) or OAG (100 μM). These results suggest
that AS-IV protects MCs against contractile dysfunction under HG
conditions by upregulating the TRPC6 protein expression and
increasing Ca2+ influx through the NADPH
oxidase/ROS/Akt/NF-κB signaling pathway.
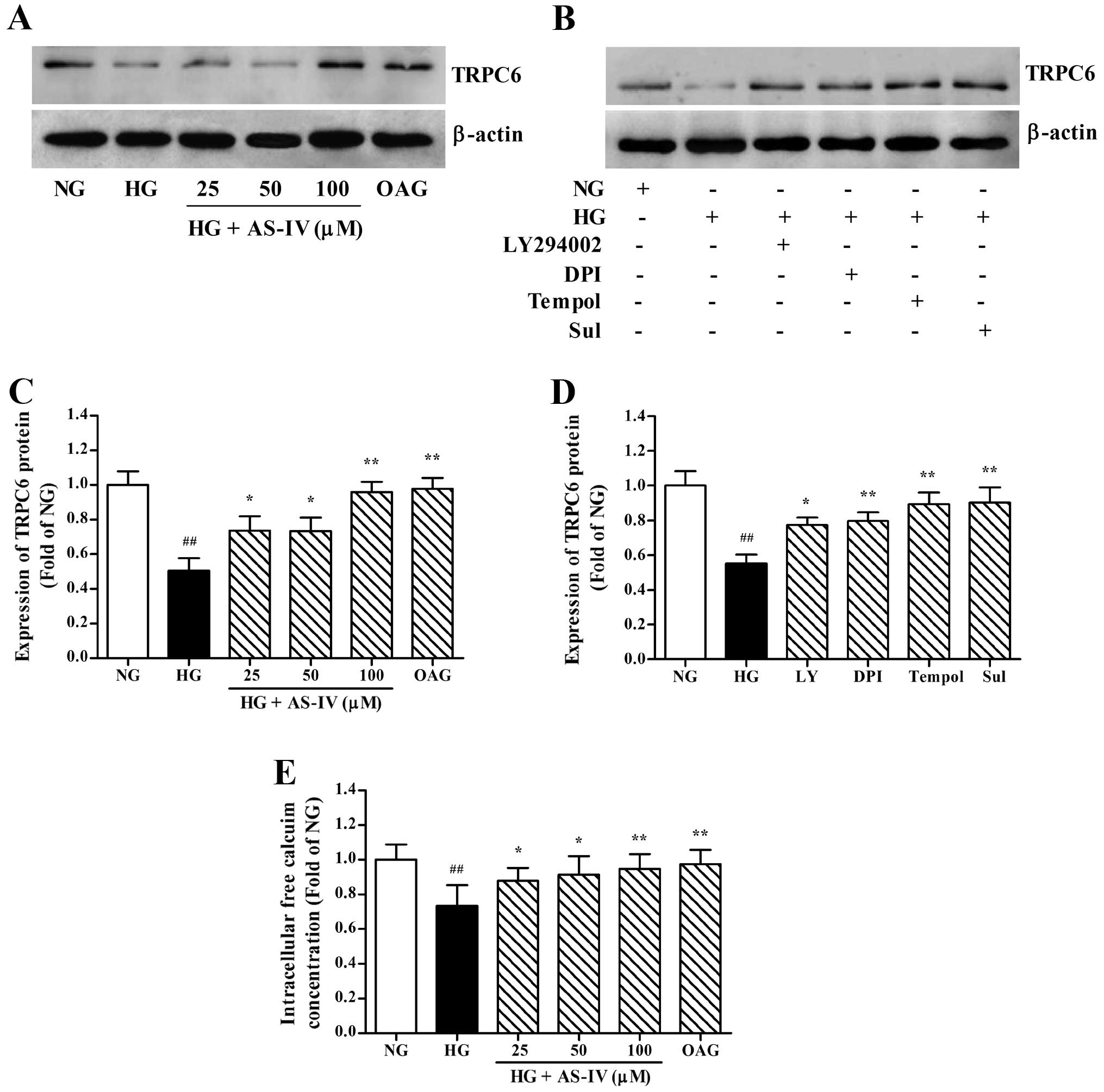 | Figure 7Treatment with astragaloside IV
(AS-IV) inhibits the downregulation of canonical transient receptor
potential cation channel 6 (TRPC6) protein in mesangial cells (MCs)
and the reduction in calcium influx under high glucose (HG)
conditions. (A) MCs were stimulated with HG for 48 h with or
without AS-IV at various concentration (25, 50, and 100 μM) or
1-oleoyl-2-acetyl-sn-glycerol (OAG) (100 μM). (B) MCs were cultured
in normal glucose (NG) or HG with or without different inhibitors
of signaling molecules [LY294002, 10 μM; diphenyleneiodonium (DPI),
10 μM; tempol, 100 μM; sulfasalazine (Sul), 0.5 mM] for 48 h. The
protein level of TRPC6 was detected by western blot analysis, and
β-actin was used as a loading control. (C and D) Summary data from
the experiments indicated in (A) and (B), respectively. (E) MCs
were loaded with the fluoroprobe Fluo-3/AM, and the fluorescence
intensity of intracellular calcium was detected by
fluorospectrophotometry. The Ca2+ concentration was
expressed as fold of NG. ##P<0.01 compared with NG;
*P<0.05, **P<0.01 compared with HG.
Results represent the means ± standard deviation (SD) of three
independent experiments. |
Discussion
Radix Astragali, the dried root of Astragalus
membranaceus (Fisch.) Bunge, has long been used in
traditional Chinese medicine for the treatment of cardiovascular
diseases and diabetes (39).
Recently, investigations into its active ingredients have attracted
much attention due to the unique pharmacological properties of many
of its constituents (34). AS-IV
is a novel saponin extracted from Radix Astragali, and it has been
reported to ameliorate podocyte apoptosis by attenuating ROS
production and to prevent acute kidney injury by inhibiting
oxidative stress (40,41). In a previous study of ours, we
suggested that AS-IV significantly reduced
H2O2-induced ROS overproduction in MCs
(42). In order to further
demonstrate that treatment with AS-IV can suppress oxidative
stress-induced injury in DN, the present study was designed to
examine the protective effects of AS-IV on the morphological and
functional abnormalities of MCs cultured under hyperglycemic
conditions.
Hyperglycemia, a common condition occurring in
diabetes, markedly increases the production of ROS in MCs (19). The redox imbalance between the
production of ROS and the compensatory response from the endogenous
antioxidant network results in oxidative stress. The interaction of
the excessive ROS generation with biomolecules, such as lipids,
proteins and DNA, can activate a series of cell signaling pathways,
leading to severe kidney injury and dysfunction (43). The most prominent effect is MC
proliferation, which often leads to glomerulosclerosis (GS), renal
fibrosis or even end-stage renal failure (44). NADPH oxidase, a multicomponent
enzyme, is the major source of ROS production in renal cells
(23). NADPH oxidase was
originally found in neutrophils (22). In many non-phagocytic cells, the
Nox family is a homologue of gp91phox, which is the
catalytic subunit of NADPH oxidase, including several types, such
as Nox1, Nox2, Nox3, Nox4 and Nox5 (45). Of these, the Nox4 isoform is
mainly found in MCs (46). The
activity of NADPH oxidase and the expression level of Nox4 protein
are both markedly increased in MCs under HG conditions. In this
study, we found that treatment with AS-IV markedly supressed
HG-induced intracellular ROS generation, as well as MC
proliferation and hypertrophy. Consistently, our experiments
demonstrated that AS-IV markedly attenuated the HG-stimulated NADPH
oxidase activation and the overexpression of Nox4 in MCs. Our
results indicated that there was no obvious direct cytotoxic effect
of AS-IV on MCs. These results provide evidence that AS-IV may
exert an inhibitory effect on HG-induced MC proliferation and
hypertrophy by downregulating Nox4-derived ROS generation.
Since the activation of various cellular molecules,
such as transcription factors, cytokines, hormones and protein
kinases has been reported to contribute to the signal transduction
cascades of DN (47,48), we investigated the mechanisms
through which AS-IV prevents damage to human MCs induced by HG
stimulation in detail in order to explore the underlying molecular
mechanisms involved in the above-mentioned effects of AS-IV. Our
results revealed that following HG stimulation, the levels of
several cellular phosphorylated molecules were decreased by
treatment with AS-IV. Akt, one of the downstream effectors of PI3K,
is involved in cell proliferation and hypertrophy (26). It has been demonstrated that Akt
is activated in renal cells and is regulated by intracellular ROS
(28). In the present study, we
found that the phosphorylation level of Akt was increased by HG
stimulation in MCs. Akt phosphorylation was also markedly reduced
by treatment with AS-IV. Moreover, Akt plays a key role in
promoting the transcriptional activity of NF-κB (29). NF-κB is one of the most important
transcription factors, which can be activated by various stimuli in
DN, such as hyperglycemia, advanced glycation end products (AGEs),
angiogeninII (AngII), oxidative stress and the PI3K/Akt signaling
pathway (49). Akt promotes the
activation of NF-κB by activating the IκB kinase (IKK) to
accelerate the phosphorylation and degradation of IκB, thereby
promoting the translocation of NF-κB from the cytoplasm into the
nucleus and subsequently binding to specific sequences in DNA,
which in turn results in gene transcription. These signaling
cascades eventually lead to MC proliferation (31). Our results revealed that HG
enhanced the activation of NF-κB, and that the degradation and
phosphorylation levels of IκBα were marekdly decreased in the MCs
treated with AS-IV. These data strongly suggest that the Akt/NF-κB
signaling pathway is involved in the pathogenesis of DN.
In addition, we used specific inhibitors of
signaling molecules as a control to compare the effectiveness of
AS-IV in order to validate the upstream and downstream association
among HG-induced NADPH oxidase, Akt and NF-κB activation. The
phosphorylation levels of Akt and IκBα were markedly inhibited by
the NADPH oxidase inhibitor, DPI, or the ROS inhibitor, tempol. Of
note, the PI3K inhibitor, LY294002, also abolished the
HG-stimulated Nox4 expression, as well as IκBα phosphorylation and
degradation. However, the IκBα inhibitor, Sul, did not suppress
Nox4 expression and Akt activation induced by HG. Studies have
indicated that ROS, as an important stimulator of NF-κB activation,
mediate the activation of Akt in MCs and other cultured cells
(50). However, controversially,
there are also data reporting that the intracellular ROS level is
regulated by Akt (26). These
discrepancies suggest the existence of a cross-talk between NADPH
oxidase-derived ROS and Akt activation. Our results support this
assumption and indicate that both the activation of NADPH oxidase
and Akt may be required for HG-induced IκBα phosphorylation and
degradation in MCs.
The early distinctive pathological characteristics
of DN are not only MC proliferation, but also the hypocontractility
of MCs, which is induced by the decreased Ca2+ influx.
TRPC6 is Ca2+ permeable cation channel which plays a
pivotal role in regulating Ca2+ signaling in MCs,
proving a mechanism for impaired MC contraction in diabetes
(10). It has previously been
suggested that the abundance of TRPC6 protein in MCs is decreased
by ROS and PKC in diabetes (51).
Moreover, it has also been reported that NF-κB participates in the
regulation of TRPC6 expression (33). The results of the present study
revealed that exposure to HG resulted in the downregulation of
TRPC6 protein and a reduction in free calcium concentration in the
MCs, inducing the contractile dysfunction of MCs; the NADPH
oxidase/ROS/Akt/NF-κB signaling pathway may also be involved in
these effects, which were markedly supressed by treatment with
AS-IV.
In conclusion, the present study indicates that
hyperglycemia induces glomerular MC proliferation and the
downregulation of TRPC6 protein by promoting Nox4 upregulation, ROS
generation, Akt and NF-κB activation. Treatment with AS-IV inhibits
HG-induced MC proliferation and contractile dysfunction through the
NADPH oxidase/ROS/Akt/NF-κB signaling pathway. Therefore, we
suggest that AS-IV may be a valuable candidate for the prevention
and treatment of early DN. However, other relevant mechanisms
underlying the effects of AS-IV require further investigation.
Acknowledgements
This study was supported by grants from the National
Natural Science Foundation of China (no. 81173624), the Nature
Science Foundation of Anhui Province (no. 11040606M201) and
International scientific and Technological Cooperative Project of
Anhui province (no. 1230603007). The authors would like to thank Li
Gui and Dake Huang from the Synthetic Laboratory of Anhui Medical
University for their helpful technical assistance.
References
|
1
|
Parving HH: Diabetic nephropathy:
prevention and treatment. Kidney Int. 60:2041–2055. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bank N: Mechanisms of diabetic
hyperfiltration. Kidney Int. 40:792–807. 1991. View Article : Google Scholar
|
|
3
|
Wei P, Lane PH, Lane JT, Padanilam BJ and
Sansom SC: Glomerular structural and functional changes in a
high-fat diet mouse model of early-stage Type 2 diabetes.
Diabetologia. 47:1541–1549. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Schlöndorff D and Banas B: The mesangial
cell revisited: no cell is an island. J Am Soc Nephrol.
20:1179–1187. 2009.PubMed/NCBI
|
|
5
|
Menè P, Pugliese G, Pricci F, Di Mario U,
Cinotti GA and Pugliese F: High glucose level inhibits capacitative
Ca2+ influx in cultured rat mesangial cells by a protein
kinase C-dependent mechanism. Diabetologia. 40:521–527.
1997.PubMed/NCBI
|
|
6
|
Nutt LK and O’Neil RG: Effect of elevated
glucose on endothelin-induced store-operated and non-store-operated
calcium influx in renal mesangial cells. J Am Soc Nephrol.
11:1225–1235. 2000.PubMed/NCBI
|
|
7
|
Whiteside CI, Hurst RD and Stevanovic ZS:
Calcium signaling and contractile response of diabetic glomerular
mesangial cells. Kidney Int. 51(Suppl): S28–S33. 1995.PubMed/NCBI
|
|
8
|
Wu JP, Zhang W, Wu F, et al: Honokiol: an
effective inhibitor of high-glucose-induced upregulation of
inflammatory cytokine production in human renal mesangial cells.
Inflamm Res. 59:1073–1079. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hsu YJ, Hoenderop JG and Bindels RJ: TRP
channels in kidney disease. Biochim Biophys Acta. 1772:928–936.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Smyth JT, Hwang SY, Tomita T, DeHaven WI,
Mercer JC and Putney JW: Activation and regulation of
store-operated calcium entry. J Cell Mol Med. 14:2337–2349. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Nilius B and Owsianik G: The transient
receptor potential family of ion channels. Genome Biol. 12:2182011.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Dietrich A and Gudermann T: Trpc6. Handb
Exp Pharmacol. 125–141. 2007. View Article : Google Scholar
|
|
13
|
Ma R, Du J, Sours S and Ding M:
Store-operated Ca2+ channel in renal microcirculation
and glomeruli. Exp Biol Med (Maywood). 231:145–153. 2006.
|
|
14
|
Möller CC, Wei C, Altintas MM, et al:
Induction of TRPC6 channel in acquired forms of proteinuric kidney
disease. J Am Soc Nephrol. 18:29–36. 2007.PubMed/NCBI
|
|
15
|
Graham S, Ding M, Sours-Brothers S, Yorio
T, Ma JX and Ma R: Downregulation of TRPC6 protein expression by
high glucose, a possible mechanism for the impaired Ca2+
signaling in glomerular mesangial cells in diabetes. Am J Physiol
Renal Physiol. 293:F1381–F1390. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Danesh FR, Sadeghi MM, Amro N, et al:
3-Hydroxy-3-methylglutaryl CoA reductase inhibitors prevent high
glucose-induced proliferation of mesangial cells via modulation of
Rho GTPase/p21 signaling pathway: Implications for diabetic
nephropathy. Proc Natl Acad Sci USA. 99:8301–8305. 2002. View Article : Google Scholar
|
|
17
|
Banday AA, Fazili FR and Lokhandwala MF:
Oxidative stress causes renal dopamine D1 receptor dysfunction and
hypertension via mechanisms that involve nuclear factor-kappaB and
protein kinase C. J Am Soc Nephrol. 18:1446–1457. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Forbes JM, Coughlan MT and Cooper ME:
Oxidative stress as a major culprit in kidney disease in diabetes.
Diabetes. 57:1446–1454. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ha H and Lee HB: Reactive oxygen species
as glucose signaling molecules in mesangial cells cultured under
high glucose. Kidney Int Suppl. 77:S19–S25. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hool LC and Corry B: Redox control of
calcium channels: from mechanisms to therapeutic opportunities.
Antioxid Redox Signal. 9:409–435. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gill PS and Wilcox CS: NADPH oxidases in
the kidney. Antioxid Redox Signal. 8:1597–1607. 2006. View Article : Google Scholar
|
|
22
|
Babior BM, Lambeth JD and Nauseef W: The
neutrophil NADPH oxidase. Arch Biochem Biophys. 397:342–344. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Nauseef WM: Biological roles for the NOX
family NADPH oxidases. J Biol Chem. 283:16961–16965. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Block K, Eid A, Griendling KK, Lee DY,
Wittrant Y and Gorin Y: Nox4 NAD(P)H oxidase mediates Src-dependent
tyrosine phosphorylation of PDK-1 in response to angiotensin II:
role in mesangial cell hypertrophy and fibronectin expression. J
Biol Chem. 283:24061–24076. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Koyasu S: The role of PI3K in immune
cells. Nat Immunol. 4:313–319. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shiojima I and Walsh K: Role of Akt
signaling in vascular homeostasis and angiogenesis. Circ Res.
90:1243–1250. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kim SY, Park KH, Gul R, Jang KY and Kim
UH: Role of kidney ADP-ribosyl cyclase in diabetic nephropathy. Am
J Physiol Renal Physiol. 296:F291–F297. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kim SY, Gul R, Rah SY, et al: Molecular
mechanism of ADP-ribosyl cyclase activation in angiotensin II
signaling in murine mesangial cells. Am J Physiol Renal Physiol.
294:F982–F989. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sousa LP, Carmo AF, Rezende BM, et al:
Cyclic AMP enhances resolution of allergic pleurisy by promoting
inflammatory cell apoptosis via inhibition of PI3K/Akt and
NF-kappaB. Biochem Pharmacol. 78:396–405. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li J and Bertram JF: Review:
Endothelial-myofibroblast transition, a new player in diabetic
renal fibrosis. Nephrology (Carlton). 15:507–512. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yang WS, Seo JW, Han NJ, et al: High
glucose-induced NF-kappaB activation occurs via tyrosine
phosphorylation of IkappaBalpha in human glomerular endothelial
cells: involvement of Syk tyrosine kinase. Am J Physiol Renal
Physiol. 294:F1065–F1075. 2008. View Article : Google Scholar
|
|
32
|
Nitti M, Furfaro AL, Patriarca S, et al:
Human mesangial cells resist glycoxidative stress through an
antioxidant response. Int J Mol Med. 27:213–219. 2011.PubMed/NCBI
|
|
33
|
Wang XW, Tan BZ, Sun M, Ho B and Ding JL:
Thioredoxin-like 6 protects retinal cell line from photooxidative
damage by upregulating NF-kappaB activity. Free Radic Biol Med.
45:336–344. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ren S, Zhang H, Mu Y, Sun M and Liu P:
Pharmacological effects of Astragaloside IV: a literature review. J
Tradit Chin Med. 33:413–416. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
You H, Lu Y, Gui D, Peng A, Chen J and Gu
Y: Aqueous extract of Astragali Radix ameliorates proteinuria in
adriamycin nephropathy rats through inhibition of oxidative stress
and endothelial nitric oxide synthase. J Ethnopharmacol.
134:176–182. 2011. View Article : Google Scholar
|
|
36
|
Motomura K, Fujiwara Y, Kiyota N, et al:
Astragalosides isolated from the root of astragalus radix inhibit
the formation of advanced glycation end products. J Agric Food
Chem. 57:7666–7672. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wang O, Cai K, Pang S, et al: Mechanisms
of glucose-induced expression of pancreatic-derived factor in
pancreatic beta-cells. Endocrinology. 149:672–680. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Jeong SI, Kim SJ, Kwon TH, Yu KY and Kim
SY: Schizandrin prevents damage of murine mesangial cells via
blocking NADPH oxidase-induced ROS signaling in high glucose. Food
Chem Toxicol. 50:1045–1053. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wu F and Chen X: A review of
pharmacological study on Astragalus membranaceus(Fisch.)
Bge. Zhong Yao Cai. 27:232–234. 2004.(In Chinese).
|
|
40
|
Gui D, Guo Y, Wang F, et al: Astragaloside
IV, a novel antioxidant, prevents glucose-induced podocyte
apoptosis in vitro and in vivo. PloS one. 7:e398242012. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Gui D, Huang J, Liu W, Guo Y, Xiao W and
Wang N: Astragaloside IV prevents acute kidney injury in two rodent
models by inhibiting oxidative stress and apoptosis pathways.
Apoptosis. 18:409–422. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Cao LL, Li WZ, Si XL, Sun L and Li WP:
Protective effect and mechanism of astragaloside IV on oxidative
stress injury of mesangial cells. Zhongguo Zhong Yao Za Zhi.
38:725–730. 2013.(In Chinese).
|
|
43
|
Ha H, Hwang IA, Park JH and Lee HB: Role
of reactive oxygen species in the pathogenesis of diabetic
nephropathy. Diabetes Res Clin Pract. 82(Suppl 1): S42–S45. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Hodgkinson AD, Bartlett T, Oates PJ,
Millward BA and Demaine AG: The response of antioxidant genes to
hyperglycemia is abnormal in patients with type 1 diabetes and
diabetic nephropathy. Diabetes. 52:846–851. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Babior BM: NADPH oxidase. Curr Opin
Immunol. 16:42–47. 2004. View Article : Google Scholar
|
|
46
|
Gorin Y, Ricono JM, Kim NH, Bhandari B,
Choudhury GG and Abboud HE: Nox4 mediates angiotensin II-induced
activation of Akt/protein kinase B in mesangial cells. Am J Physiol
Renal Physiol. 285:F219–F229. 2003.PubMed/NCBI
|
|
47
|
Devaraj S, Cheung AT, Jialal I, et al:
Evidence of increased inflammation and microcirculatory
abnormalities in patients with type 1 diabetes and their role in
microvascular complications. Diabetes. 56:2790–2796. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Gruden G, Setti G, Hayward A, et al:
Mechanical stretch induces monocyte chemoattractant activity via an
NF-kappaB-dependent monocyte chemoattractant protein-1-mediated
pathway in human mesangial cells: inhibition by rosiglitazone. J Am
Soc Nephrol. 16:688–696. 2005. View Article : Google Scholar
|
|
49
|
Iwamoto M, Mizuiri S, Arita M and Hemmi H:
Nuclear factor-kappaB activation in diabetic rat kidney: evidence
for involvement of P-selectin in diabetic nephropathy. Tohoku J Exp
Med. 206:163–171. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Bäumer AT, Ten Freyhaus H, Sauer H, et al:
Phosphatidylinositol 3-kinase-dependent membrane recruitment of
Rac-1 and p47phox is critical for alpha-platelet-derived
growth factor receptor-induced production of reactive oxygen
species. J Biol Chem. 283:7864–7876. 2008.PubMed/NCBI
|
|
51
|
Graham S, Gorin Y, Abboud HE, et al:
Abundance of TRPC6 protein in glomerular mesangial cells is
decreased by ROS and PKC in diabetes. Am J Physiol Cell Physiol.
301:C304–C315. 2011. View Article : Google Scholar : PubMed/NCBI
|