Introduction
Glaucoma is the second leading cause of visual
impairment and blindness worldwide (1). It is a complex and chronic
progressive optic nerve injury that is accompanied by high
intraocular pressure (IOP), optic disc depression, optic nerve
fiber atrophy and visual field defect. Primary open-angle glaucoma
(POAG) is the most common type of glaucoma in developed countries,
accounting for >50% of glaucoma cases (2). In China, the ratio of diagnosis with
POAG has increased from 8.18% (1980s) to 19.25% (2012) in total
glaucoma patients (3). A
population-based cross-sectional study in China demonstrated that
glaucoma was one of the major causes for blindness with POAG
constituting the major type (4).
In another study, it was found that the likelihood of developing
POAG in relatives of POAG patients was 7- to 10-fold higher than
that in the general population (5).
The pathogenesis of POAG is unknown. It is generally
considered to be a multi-factorial disease and not a single genetic
disorder with the single gene mutation. Epidemiological studies
have demonstrated that genetic and environmental factors may affect
its etiology, while genetic factors were mainly involved in the
onset of POAG (6). Genetic
linkage analysis of POAG pedigrees revealed that there were ≥20
genetic loci for POAG and only three causative genes were
identified, i.e., myocilin (MYOC), optineurin (OPTN)
and WD repeat domain 36 (WDR36) (7). Of the three genes, MYOC is
deemed a direct causative gene leading to glaucoma, accounting for
~1 to 4% of mutations for POAG although the exact roles of
OPTN and WDR36 remain to be determined (7).
In the present study, we clinically followed up a
Chinese POAG family over a period of five years. A pedigree
analysis was performed, followed by molecular biology and
bioinformatics analyses, which were used to investigate MYOC
in the family members.
Materials and methods
Clinical data collection for study
participants
This study was performed according to the tenets of
the Declaration of Helsinki for Research Involving Human Subjects
and was approved by the Ethics Committee of the First Affiliated
Hospital at Henan University of Science and Technology (Henan,
China). The glaucoma family had five generations of 29 members.
Twelve members from three generations participated in this study
and were numbered from 096001 to 096012. Informed consent was
obtained from the 12 family members and 100 healthy controls.
The 12 family members received ophthalmologic
examination including visual acuity, the anterior chamber, IOP
measurement by applanation tonometry (Goldmann), anterior chamber
angle evaluation by gonioscopy (Goldmann), fundus examination with
a 90-diopter VOLK lens, and Octopus perimeter examination. The
family members were clinically followed up over a period of five
years, from 2005 to 2010. Diagnosis of POAG was based on the
observation of at least two of the following abnormalities:
characteristic glaucomatous optic disc changes [vertical cup-disc
(c/d) ratio of ≥0.7, notching of the neutral rim, and disc
hemorrhage], characteristic glaucomatous visual field defects, and
high IOP (>21 mmHg) in the presence of a normal open anterior
chamber angle. Diagnosis was made after the other secondary
glaucoma was excluded, such as traumatic, uveitis, steroid-induced,
and neovascular glaucoma. The age of diagnosis of the patients with
POAG <35 years old was sub-classified as juvenile-onset
open-angle glaucoma (JOAG). Individuals with IOP >22 mmHg but
with no characteristic optic disc damage or visual field impairment
were defined as ocular hypertension (OHT). Unaffected individuals
had IOP in the normal range and optic nerves presented normal in
appearance. In total, 100 healthy individuals (42 males and 58
females, 59.8±20.5 years) were included in the control group. A
comprehensive eye examination was conducted for the healthy
controls to exclude glaucoma and other genetic diseases including
diabetes, blood hypertension, retinoblastoma, and high myopia.
Genomic DNA collection
Genomic DNA was extracted from the venous blood of
12 family members and 100 healthy controls. Peripheral blood of 5
ml was collected from all the participants. Genomic DNA was
extracted following the standard phenol/chloroform extraction
method.
Primer design and synthesis
According to the MYOC sequence, published by
the USA National Center for Biotechnology Information, primers were
designed to target the third Exon using Primer3 software (Table I) and produced by Beijing SBS
Genetech Co., Ltd. (Beijing, China).
 | Table IPrimers of the third exon in the
MYOC gene. |
Table I
Primers of the third exon in the
MYOC gene.
| Exon | Primer | Primer sequence
(5′–3′) | Product size
(bp) | Optimal annealing
temperature (°C) |
|---|
| 3 | MYOC-3-1F |
CTTCCGCATGATCATTGT | 352 | 58 |
| MYOC-3-1R |
CTTCCGCATGATCATTGT | | |
| 3 | MYOC-3-2F |
ATACTGCCTAGGCCACTGGAA | 440 | 58 |
| MYOC-3-2R |
CCGCTATAAGTACAGCAGCATGAT | | |
| 3 | MYOC-3-3F |
GCCTTCATCATCTGTGGCAC | 342 | 58 |
| MYOC-3-3R |
CAGGCAGCTTTGACTGCTTT | | |
Polymerase chain reaction (PCR)
The reaction (20 μl) mix contained 2
μl 10X PCR reaction buffer (20 mM Mg2+), 0.5
μl dNTPs (10 mmol/l), 0.5 μl forward and reverse
primers (each 10 pmol/μl), 0.5 μl TaqDNA
polymerize (2 U/μl), 2 μl template genome DNA (5–50
ng/μl) and 14 μl double-distilled water. The reaction
conditions used were : i) 94°C pre-degeneration for 5 min; ii) 94°C
degeneration for 30 sec; annealing for 30–60 sec; extension at 72°C
for 30–60 sec; total circulation of 30- to 36-fold; iii) extension
at 72°C for 10 min, and 1% agarose gel electrophoresis to detect
the amplification effect.
Purification, sequencing and
comparison
The amplified PCR products and corresponding primers
were purified and sequenced. An automatic fluorescence DNA
sequencer (ABI PRISM 373A; Perkin-Elmer, Foster City, CA, USA) was
used to sequence the purified PCR products in the forward and
reverse directions. The sequencing results were compared with the
published DNA sequence of MYOC (GenBank NM_000261) to screen
mutations.
Restriction fragment length polymorphism
(RFLP) analysis
To confirm the variations found in the sequencing,
restriction endonuclease CviKI-1 (New England Biolabs, Ipswich, MA,
USA) was used for all the participants. The reaction was performed
in a 10 μl volume containing 9.4 μl PCR product, 0.1
μl BSA (100 μg/ml), and 0.5 μl enzyme (10
U/μl). After incubating the reaction overnight at 37°C, the
entire digest was run on a 1% agarose gel (with EB) at 100 V for 40
min and visualized under ultraviolet light.
Bioinformatics analysis
Garnier-Osguthorpe-Robson (GOR) software was used to
predict the effect of the mutation on the secondary structure of
MYOC. It infers the secondary structure of a sequence by
calculating the probability for each of the four structure classes
(helix, sheet, turn and loop) based on the central residue and its
neighbors from the calculated matrices.
Results
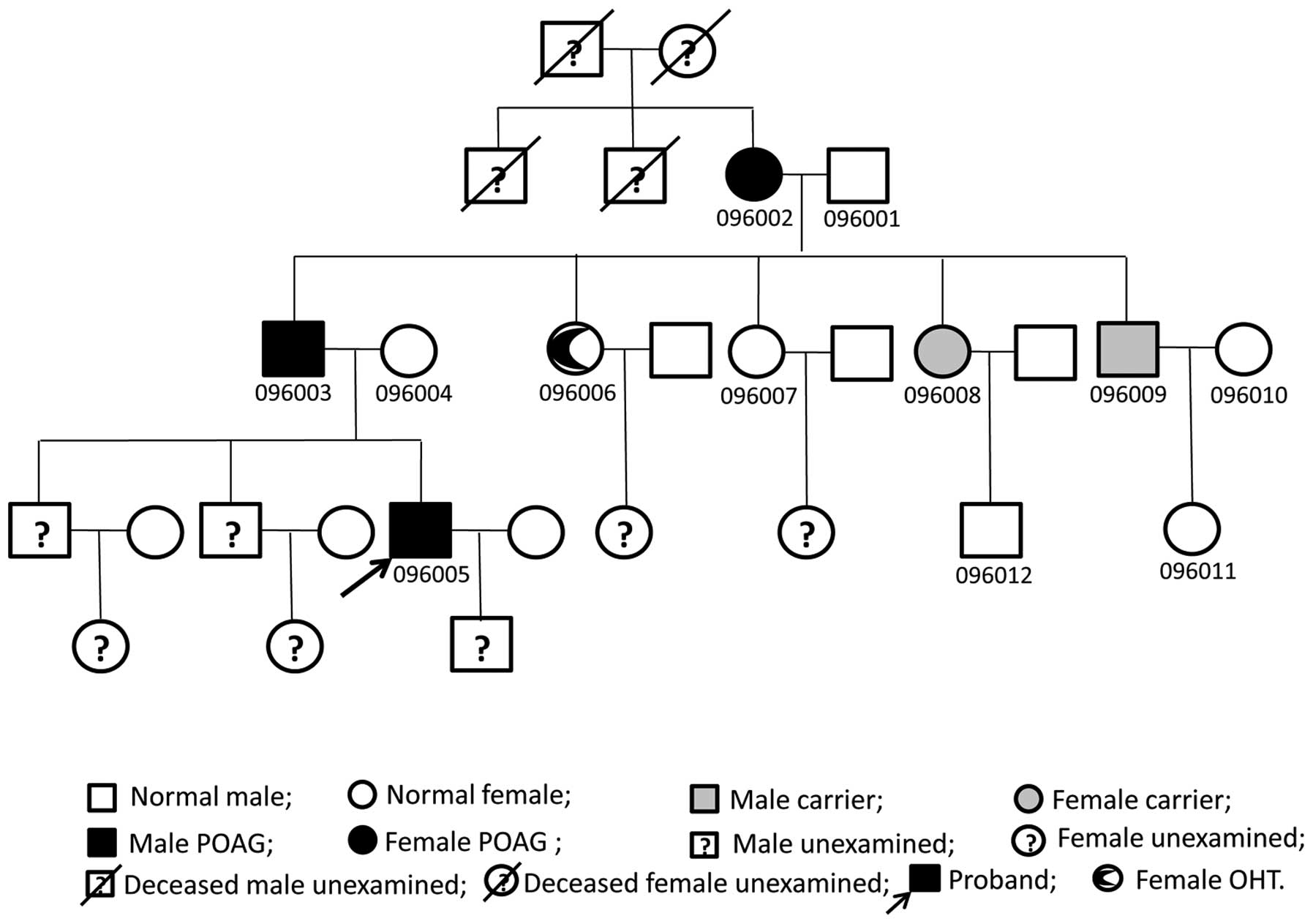
Pedigree structure of the POAG
family
The family comprised 29 members of five generations.
Twelve members from three generations participated in the study,
and POAG was diagnosed in three members from each generation. POAG
affected males and females in this family. The disease was passed
vertically from generation to generation and was not
gender-specific, indicating autosomal dominant genetic
characteristics (Fig. 1).
Clinical manifestations
Three members (096002, 096003 and 096005) were
diagnosed with POAG, and another one (096006) was diagnosed with
OHT in this family (Table II).
The proband of the family was diagnosed at 25 years of age, which
was consistent with the diagnosis of JOAG. Visual acuity, IOP,
optic nerve, and visual field were affected most severely among the
three patients. The other two POAG patients were all diagnosed at
>50 years of age and the exact age of onset was not known. The
member who was diagnosed with OHT was 55 years old at the last
visit. She had a maximum IOP 21/24 and a borderline C/D ratio
(0.6/0.6) without the visual field defect. Trabeculectomy was
performed on three POAG patients. Post-operative IOP is shown in
Table II and two of the patients
(096003 and 096005) required anti-glaucoma eye drops even though
the filtering blebs were functional (Fig. 2). The remaining family members
were treated as normal according to the diagnostic criteria.
 | Table IIClinical parameters of individuals in
this pedigree. |
Table II
Clinical parameters of individuals in
this pedigree.
| Pedigree ID
no. | Gender/age
(year) | Diagnosed at age
(year) | BCVA OD/OS
(2005) | BCVA OD/OS
(2010) | Maximum IOP OD/OS
mmHg | IOP (2005) OD/OS
mmHg | IOP (2010) OD/OS
mmHg | Optic Disc (C/D)
OD/OS (2010) | VF Loss OD/OS | Therapy OD/OS | Diagnosis 2005 | Diagnosis 2010 | S341P |
|---|
| 096001 | M/82 | | 0.6/0.5 | 0.4/0.25 | 17/17 | 17/17 | 16/16 | 0.3/0.4 | No/No | | Normal | Normal | No |
| 096002 | F/82 | 65 | 0.5/0.5 | 0.3/0.2 | 17/22 | 17/18 | 17/22 | 0.4/0.5 | Yes/Yes | S/S | POAG | POAG | Yes |
| 096003 | M/61 | 55 | 1.0/0.8 | 0.6/0.6 | 52/50 | 45/40 | 30/30 | 0.6/0.7 | Yes/Yes | s/s | POAG | POAG | Yes |
| 096004 | F/60 | | 1.0/1.0 | 1.0/0.8 | 17/17 | 17/17 | 17/17 | 0.3/0.4 | No/No | | Normal | Normal | No |
| 096005 | M/32 | 25 | 1.2/CF | 1.0/CF | 48/NA | 48/NA | 17/33 | 0.8/0.9 | Yes/Yes | S/S | POAG | POAG | Yes |
| 099006 | F/55 | 50 | 1.0/1.0 | 1.0/1.0 | 21/24 | 17/20 | 21/24 | 0.6/0.6 | No/No | NMT | OHT | OHT | Yes |
| 096007 | F/49 | | 1.2/1.2 | 1.0/1.0 | 17/16 | 17/17 | 17/16 | 0.3/0.3 | No/No | | Normal | Normal | No |
| 096008 | F/44 | | 1.0/1.2 | 1.0/1.0 | 17/18 | 17/18 | 17/18 | 0.5/0.4 | No/No | | Normal | carrier | Yes |
| 096009 | M/41 | | 1.0/1.2 | 1.0/1.2 | 17/21 | 17/21 | 17/21 | 0.4/0.4 | No/No | | Normal | carrier | Yes |
| 096010 | F/40 | | 1.0/1.2 | 1.0/1.2 | 17/17 | 16/16 | 16/16 | 0.3/0.3 | No/No | | Normal | Normal | No |
| 096011 | F/14 | | 1.0/1.0 | 1.0/1.2 | 16/16 | 16/16 | 16/16 | 0.3/0.3 | No/No | | Normal | Normal | No |
| 096012 | M/20 | | 1.0/1.0 | 1.0/1.0 | 17/17 | NA | NA | 0.3/0.3 | No/No | | Normal | Normal | No |
Sequencing and comparative analysis
The third exon of MYOC was PCR amplified and
received direct sequencing in the 12 family members. We found
c.1021 T→C heterozygous base change (Fig. 3) in members 096002, 096003,
096005, 096006, 096008 and 096009. The codon changed from TCC to
CCC, thus amino acids at position 341 were altered. Consequently,
the corresponding myocilin protein was altered from serine (Ser)
into proline (Pro) (p.S341P). This mutation was not identified in
members 096001, 096004, 096007, 096010, 096011 and 096012, or the
100 normal controls.
Restriction fragment length
polymorphism
Based on the results of MYOC sequencing, we
designed the relevant restriction endonuclease sites. We found that
CviKI-1 can recognize c.DNA1020 on base G and c.DNA1021 on
base T in the normal MYOC. The c.1021T→C (p.S341P) mutation
caused loss of the restriction enzyme sites of CviKI-1 in
the corresponding DNA sequence. In Table III and Fig. 4, we show the restriction
endonuclease recognition sequence, enzyme loci and restriction
fragment size. Loss of CviKI-1 sites was found in three POAG
patients and one OHT patient, which was also identified in two
members (096008 and 096009) who were not clinically diagnosed with
POAG (Table II). The homologous
chromosomes of family members (096002, 096003, 096005, 096006,
096008 and 096009) who had the mutation were cleaved into four
fragments (257, 209, 203 and 48 bp). The homologous chromosomes of
other members (096001, 096004, 096007, 096010 and 096011) were
cleaved into three fragments (209, 203 and 48 bp) (Fig. 5). The three members (096006,
096008 and 096009) without POAG diagnosis at the last visit were
considered to be gene mutation carriers.
 | Table IIIThe CviKI-1 endonuclease recognition
sequence, number of restriction sites and size of fragment on RFLP
analysis. |
Table III
The CviKI-1 endonuclease recognition
sequence, number of restriction sites and size of fragment on RFLP
analysis.
| MYOC exon
(mutation) | Endonuclease | Recognition
sequence | Number of
restriction | Size of fragment
(bp) |
|---|
| Exon 3
(c.1021T→C) | CviKI-1 |
5′…GAG▾CCC---3′
3′…CTC▾GGG---5′ | 1 | 257
(209/203/48) |
Protein secondary structure
prediction
The MYOC c.1021T→C base change caused amino
acid switch from Ser to Pro. We found that changes at sites 333,
337 and 342. The coil structure of amino acids 333 was replaced
with the β-sheet structure, and the β-sheet structure of amino
acids 337 and 342 were replaced with the coil structure (Fig. 6). The base change was at the
junction of the coil structure and the lamellar structure.
Homology analysis of protein
The homology analysis was applied to compare
οlfactomedin-like domains of amino acid coding of human (Q99972)
with other mammals (rat (Q9R1J4), mouse (O70624), cattle (Q9XTA3),
dog (Q2PT31), rabbit (Q866N2), cynomolgus (Q863A3), cat (Q594P2),
and zebrafish (Q5F0G5)). We found that amino acid 341 was conserved
as Ser (Fig. 7).
Discussion
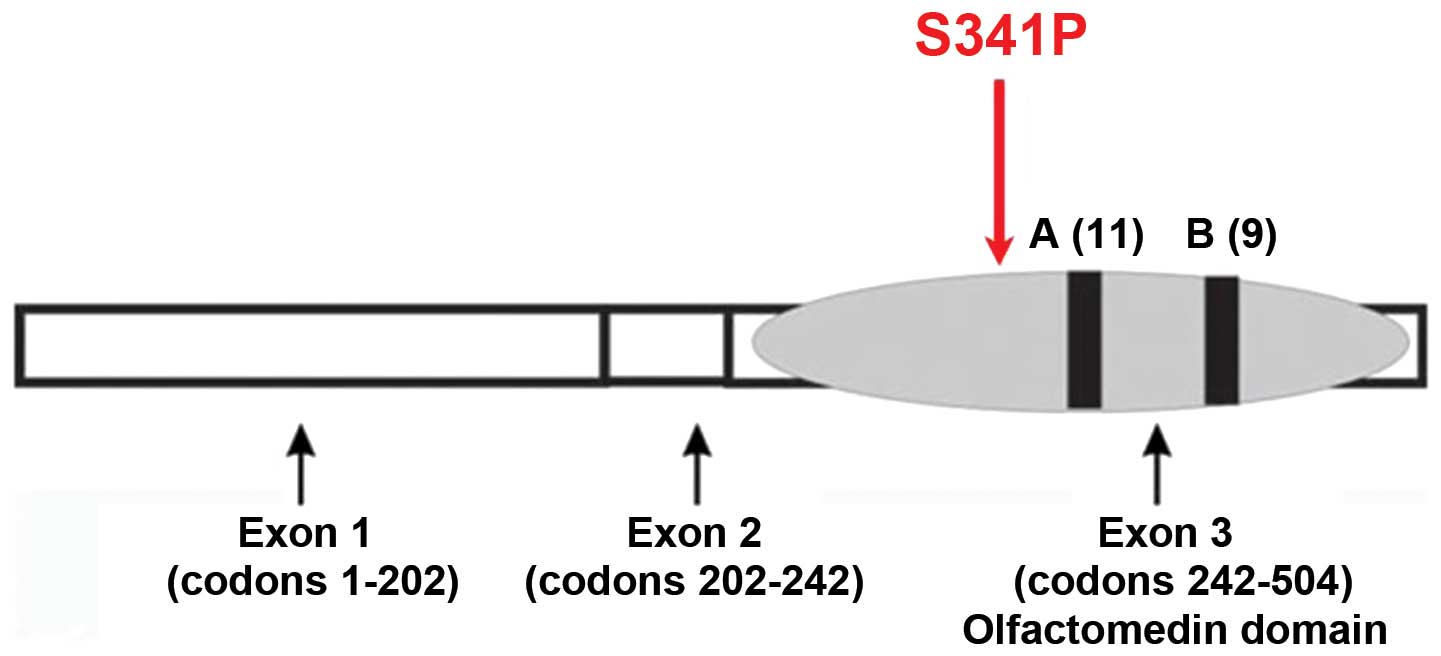
MYOC, also known as the TIGR
(trabecular meshwork inducible glucocorticoid response) gene, is
the first causative gene identified in JOAG. It contains three
exons, which are 604, 126 and 782 base pairs respectively. The
product, myocilin protein, is composed of 504 amino acids (8). The N-terminus of myocilin protein
has a signal peptide sequence, a myosin-like domain, and a zinc
finger-like domain. The C-terminal of myocilin protein has an
olfactory factor (Olfactomedin) domain. The MYOC promoter
region contains a number of important gene regulation motifs
(9). The POAG pathogenesis was
associated with at ≥70 MYOC mutation sites (10), with ~90% in the third exon of the
οlfactomedin homologous region (11). This domain is the active site of
protein function as that οlfactomedin homologous has an important
role for the function of myocilin protein expression.
In the present study, the MYOC mutation was
located in οlfactomedin homology and this mutation was not found in
normal conditions (Fig. 8). This
mutation was a heterozygosity mutation and switched Ser to Pro. The
two amino acids possess different physical and chemical properties.
Ser is a mixed hydrophilic amino acid with a molecular weight of
105.09 and an isoelectric point of 5.68. The relative distance of
Ser is 2,000; the irreplaceable nature is 0.64 and the number of
codons is six. By contrast, Pro is a highly hydrophilic amino acid
with a molecular weight of 115.13 and an isoelectric point of 6.30.
The relative distance of Pro is 1,720; the irreplaceable nature is
0.61 and the number of codons is four (12).
Our homology analysis showed that amino acid 341 is
Ser across various species. Prediction of the protein secondary
structure suggested that myocilin protein may misfold and become
hydrophobic with lower solubility, which blocks the trabecular
meshwork and affects the outflow of aqueous humor. It is known that
there are 13 MYOC mutations for POAG in China: P13L
(13), Q337Stop (13), S341P (14), P370L (10,15–17), C245Y (18,19), T353I (19,20), R91X(20), G252R (15,21), E300K (20,19), S313F (19), N450Y (23), Y471C (20,19), and T455K (22). These mutations were identified
only in Chinese patients with POAG, suggesting MYOC mutation
had ethnic and regional specificity. Although genes in the Chinese
POAG patients have been studied extensively (13,14,16–18,20–27), additional investigations are
necessary to provide evidence for the pathogenesis of POAG for
treatment and development of drugs (28–30).
Incomplete penetrance has been observed in most
families with MYOC mutations and the penetrances are age-dependent
and mutation-specific (10,15,31). The pedigree analysis revealed two
clinically healthy individuals and one OHT patient who harbored the
mutation of p.S341P and the affected haplotype. The penetrance of
this pedigree was that 66.67% (4/6) of the individuals carrying the
p.S341P mutation had developed POAG or OHT. With the exception of
the proband of the family, who was 25 years of age at diagnosis,
the remaining patients were >50 years of age at diagnosis and
their symptoms were less severe than those of the proband. This
observation suggests that unidentified factors (genetic or
environmental) may be associated with the POAG of this pedigree.
However, carriers should undergo ophthalmologic surveillance at
regular intervals.
The Ser341Pro mutation was detected in our study and
further investigation of the novel p.S341P mutation of MYOC
for POAG was necessary. The mutation spectrum of MYOC may be
expanded and a better diagnosis and treatment for POAG patients may
be achieved in the future.
Acknowledgments
We are grateful to all patients and their relatives
for their co-operation in this study. The study was supported by
the Beijing National Science Foundation (no. 07G0069).
References
|
1
|
Hogewind BF, Gaplovska-Kysela K, Theelen
T, Cremers FP, Yam GH, Hoyng CB and Mukhopadhyay A: Identification
and functional characterization of a novel MYOC mutation in two
primary open angle glaucoma families from The Netherlands. Mol Vis.
13:1793–1801. 2007.PubMed/NCBI
|
|
2
|
Fan BJ, Leung YF, Wang N, Lam SC, Liu Y,
Tam OS and Pang CP: Genetic and environmental risk factors for
primary open-angle glaucoma. Chin Med J (Engl). 117:706–710.
2004.
|
|
3
|
Ge J: Glaucoma research progress and
development trend. Chin J Ophthalmol. 36:192–196. 2000.
|
|
4
|
Foster PJ, Oen FT, Machin D, Ng TP,
Devereux JG, Johnson GJ, et al: The prevalence of glaucoma in
Chinese residents of Singapore: a cross-sectional population survey
of the Tanjong Pagar district. Arch Ophthalmol. 118:1105–1111.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wolfs RC, Klaver CC, Ramrattan RS, van
Duijn CM, Hofman A and de Jong PT: Genetic risk of primary
open-angle glaucoma. Population-based familial aggregation study.
Arch Ophthalmol. 116:1640–1645. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Stone EM, Fingert JH, Alward WL, Nguyen
TD, Polansky JR, Sunden SL, et al: Identification of a gene that
causes primary open angle glaucoma. Science. 275:668–670. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Fan BJ, Wang DY, Lam DS and Pang CP: Gene
mapping for primary open angle glaucoma. Clin Biochem. 39:249–258.
2006. View Article : Google Scholar
|
|
8
|
Avisar I, Lusky M, Robinson A, Shohat M,
Dubois S, Raymond V and Gaton DD: The novel Y371D myocilin mutation
causes an aggressive form of juvenile open-angle glaucoma in a
Caucasian family from the Middle-East. Mol Vis. 15:1945–1950.
2009.PubMed/NCBI
|
|
9
|
Nguyen TD, Chen P, Huang WD, Chen H,
Johnson D and Polansky JR: Gene structure and properties of TIGR,
an olfactomedin-related glycoprotein cloned from
glucocorticoid-induced trabecular meshwork cells. J Biol Chem.
273:6341–6350. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hewitt AW, Mackey DA and Craig JE:
Myocilin allele-specific glaucoma phenotype database. Hum Mutat.
29:207–211. 2008. View Article : Google Scholar
|
|
11
|
Mohanty Kuldeep, Mishra Swetasmita, Dada
Rima and Dada Tanuj: Genetic Complexity of primary open-angle
glaucoma. JOCGP. 5:31–39. 2011. View Article : Google Scholar
|
|
12
|
Ma F, Wu Y and Xu X: Correlation analysis
of some physial chemistry properties among genetic codons and amino
acids. J Anhui Agric Univ. 30:439–445. 2003.
|
|
13
|
Xie X, Zhou X, Qu X, Wen J, Tian Y and
Zheng F: Two novel myocilin mutations in a Chinese family with
primary open-angle glaucoma. Mol Vis. 14:1666–1672. 2008.PubMed/NCBI
|
|
14
|
Qin L and Li J: Investigation on the
mutation of MYOC gene in two family pedigrees with primary
open-angle glaucoma in Shanxi. Yan Ke Xue Bao. 23:75–78. 2007.In
Chinese. PubMed/NCBI
|
|
15
|
Gong G, Kosoko-Lasaki O, Haynatzki GR and
Wilson MR: Genetic dissection of myocilin glaucoma. Hum Mol Genet.
13(Spec 1): R91–R102. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chen Y, Jiang D, Wan B, Yu L and Sun X:
Presymptomatic genetic diagnosis for consulters from a large
Chinese family with juvenile open angle glaucoma. Mol Vis.
12:360–366. 2006.PubMed/NCBI
|
|
17
|
Zhuo YH, Wei YT, Bai YJ, Duan S, Lin MK,
Saragovi HU and Ge J: Pro370Leu MYOC gene mutation in a large
Chinese family with juvenile-onset open angle glaucoma: correlation
between genotype and phenotype. Mol Vis. 14:1533–1539.
2008.PubMed/NCBI
|
|
18
|
Fan BJ, Leung DY, Wang DY, Gobeil S,
Raymond V, Tam PO, et al: Novel myocilin mutation in a Chinese
family with juvenile-onset open-angle glaucoma. Arch Ophthalmol.
124:102–106. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Fan BJ, Wang DY, Fan DS, Tam PO, Lam DS,
Tham CC, Lam CY, Lau TC and Pang CP: SNPs and interaction analyses
of myocilin, optineurin, and aplipoprotein E in primary open angle
glaucoma patients. Mol Vis. 11:625–631. 2005.PubMed/NCBI
|
|
20
|
Pang CP, Leung YF, Fan B, Baum L, Tong WC,
Lee WS, et al: TIGR/MYOC gene sequence alterations in individuals
with and without primary open-angle glaucoma. Invest Ophthalmol Vis
Sci. 43:3231–3235. 2002.PubMed/NCBI
|
|
21
|
Vincent AL, Billingsley G, Buys Y, Levin
AV, Priston M, Trope G, Williams-Lyn D and Heon E: Digenic
Inheritance of early-onset glaucoma: CYP1B1, a potential modifier
gene. Am J Hum Genet. 70:448–460. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tian Q, Li FH, Zhao KX, Wang L, Shan XY,
Pang YY, et al: A novel mutation in the myocilin gene identified in
a Chinese primary open angle glaucoma family. Zhonghua Yi Xue Yi
Chuan Xue Za Zhi. 24:629–634. 2007.In Chinese. PubMed/NCBI
|
|
23
|
Lam DS, Leung YF, Chua JK, Baum L, Fan DS,
Choy KW and Pang CP: Truncations in the TIGR gene in individuals
with and without primary open-angle glaucoma. Invest Ophthalmol Vis
Sci. 41:1386–1391. 2000.PubMed/NCBI
|
|
24
|
Zhao X, Yang C, Tong Y, Zhang X, Xu L and
Li Y: Identification a novel MYOC gene mutation in a Chinese family
with juvenile-onset open angle glaucoma. Mol Vis. 16:1728–1735.
PubMed/NCBI
|
|
25
|
Wang DY, Fan BJ, Chua JK, Tam PO, Leung
CK, Lam DS and Pang CP: A genome-wide scan maps a novel
juvenile-onset primary open-angle glaucoma locus to 15q. Invest
Ophthalmol Vis Sci. 47:5315–5321. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Pang CP, Fan BJ, Canlas O, Wang DY, Dubois
S, Tam PO, et al: A genome-wide scan maps a novel juvenile-onset
primary open angle glaucoma locus to chromosome 5q. Mol Vis.
12:85–92. 2006.PubMed/NCBI
|
|
27
|
Lin Y, Liu T, Li J, Yang J, Du Q, Wang J,
et al: A genome-wide scan maps a novel autosomal dominant
juvenile-onset open-angle glaucoma locus to 2p15–16. Mol Vis.
14:739–744. 2008.PubMed/NCBI
|
|
28
|
Shimizu S, Lichter PR, Johnson AT, Zhou Z,
Higashi M, Gottfredsdottir M, et al: Age-dependent prevalence of
mutations at the GLC1A locus in primary open-angle glaucoma. Am J
Ophthalmol. 130:165–177. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Mardin CY, Velten I, Ozbey S,
Rautenstrauss B and Michels Rautenstrauss K: A GLC1A gene
Gln368Stop mutation in a patient with normal-tension open-angle
glaucoma. J Glaucoma. 8:154–156. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Baird PN, Richardson AJ, Craig JE,
Rochtchina E, Mackey DA and Mitchell P: The Q368STOP myocilin
mutation in a population-based cohort: the Blue Mountains Eye
Study. Am J Ophthalmol. 139:1125–1126. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Fingert JH, Stone EM, Sheffield VC and
Alward WL: Myocilin glaucoma. Surv Ophthalmol. 47:547–561. 2002.
View Article : Google Scholar : PubMed/NCBI
|