Introduction
The erbB [human epidermal growth factor receptor
(HER)] receptors are expressed in a variety of tissues of
epithelial, mesenchymal and neuronal origin, in which they play
fundamental roles in development, differentiation and proliferation
(1). The deregulated or
dysfunctional expression of the erbB receptors, in particular
epidermal growth factor receptor (EGFR/erbB1/HER1) and erbB2
(HER2), has been implicated in the development and malignancy of
numerous human cancers (2). The
overexpression of HER2 receptors, which results from HER2
gene amplification, leads to the activation of specific receptor
homodimers in the absence of a cognate soluble ligand. HER2
homodimerization and heterodimerization with the other EGFR family
members (EGFR, HER3 and HER4) stimulate tyrosine kinase
phosphorylation and activation of intracellular signal transduction
pathways, in particular the phosphatidylino-sitol 3-kinase (PI3K)
and mitogen-activated protein kinase (MAPK) pathways, promoting
potent proliferation and cell growth (3). In breast cancers, approximately 20%
of tumors show HER2 overexpression, which is clinically associated
with poor prognosis, decreased survival and shorter time to relapse
at all stages (4,5). In addition, patients with ovarian
cancer have also been shown to overexpress HER2 (3), although the prognostic significance
and incidence remain unclear. In one phase II study, 95/837 (11.4%)
patients with recurrent ovarian or primary peritoneal cancer were
found to overexpress HER2 protein as defined by
immunohistochemistry (IHC) 2+/3+ (6). In a separate study, HER2 gene
amplification as determined by fluorescence in situ
hybridization was observed in 6/33 (18.2%) of ovarian mucinous
carcinomas (7).
The high incidence of HER2 overexpression in cancer
cells and the prognostic value of HER2 in breast cancer led to the
development of trastuzumab (Herceptin®, Genentech, South
San Francisco, CA) for the treatment of HER2-positive breast cancer
in the metastatic (8,9) and adjuvant settings (10–13).
Recently, trastuzumab plus chemotherapy was approved for the
treatment of metastatic gastric and gastroesophageal cancers
(14). Trastuzumab is a humanized
immunoglobulin G1 monoclonal antibody that binds the HER2
extracellular domain (ECD) and inhibits growth-promoting downstream
signaling (15). The efficacy of
trastuzumab in the adjuvant and metastatic settings has clearly
been established. In the adjuvant setting, the addition of
trastuzumab to chemotherapy regimens has led to significant
improvements in disease-free survival and overall survival (OS)
(10–13). In metastatic breast cancer (MBC),
first-line trastuzumab plus chemotherapy has been shown to improve
response rates, time to progression, and OS (8), while second- or third-line
trastuzumab monotherapy has been shown to produce durable objective
responses for women whose tumors progress on chemotherapy (9). However, many HER2-positive breast
cancers do not respond to or progress on trastuzumab treatment,
suggesting that these patients may have intrinsic or developed
acquired resistance to trastuzumab, respectively.
A wide range of preclinical cancer models have been
developed to address resistance mechanisms that range from
insulin-like growth factor 1 (IGF-1)-induced loss of
p27Kip1, loss of phosphatase and tensin homolog (PTEN)
function, enhanced expression of EGFR ligands, masking of the
trastuzumab-binding epitope by mucin (MUC)4, induction of tumor
initiating cells by Y-box-binding protein 1, and enhanced Src
activity (16–21). However, the analysis of large-scale
clinical samples and rational translational research are required
to establish a causal link between these proposed mechanisms and
resistance to trastuzumab.
Preclinical ovarian carcinoma cell lines exhibit
HER2 gene amplification and accompanying HER2 receptor
overexpression. Many of these models have been used to test for the
antitumor efficacy of trastuzumab. Despite promising preclinical
results, however, translational activity in the clinical setting
with the use of trastuzumab to treat ovarian cancer has been
limited, with a low rate of objective responses in patients with
HER2 overexpression observed in a phase II study (6). The reason for the observed low
response rate in this tumor type is still unclear. Although many
mechanistic studies of resistance have been performed in
preclinical breast cancer models, obtaining data from a different
tumor type, such as HER2-overexpressing ovarian carcinoma, offers
an attractive alternative, enabling the outcomes to be compared and
contrasted, and added to the existing knowledge base. The
similarities between the HER2 oncogenic processes of HER2-positive
breast and ovarian cancers are numerous and may therefore allow the
results from one tumor type to be applied to another. New data from
ovarian cancer models may shed light on the causes of both acquired
and de novo trastuzumab resistance in breast cancer, and may
provide insight into the cause of the relative lack of
effectiveness of trastuzumab in patients with HER2-positive ovarian
cancer. Moreover, these studies may enable the identification of
pharmacodynamic biomarkers of resistance or new targets that could
potentially lead to new and potent antineoplastic agents. Lastly,
preclinical gene signatures resulting from microarray expression
profiling (MEP) may ultimately be used to identify patients who are
more likely to respond to or be resistant to trastuzumab therapy,
thereby informing treatment decisions.
In this study, we used the SKOV-3 human ovarian
carcinoma cell line to develop the first preclinical model of
ovarian carcinoma to study acquired or de novo resistance to
trastuzumab. The SKOV-3 human ovarian carcinoma cell line is one of
a few distinct ovarian preclinical models with HER2 gene
amplification, p185HER2 overexpression and sensitivity
to the effects of trastuzumab (3).
A trastuzumab-resistant variant was developed by in vivo
serial passaging of trastuzumab-treated subcutaneous SKOV-3 tumors
in athymic nu/nu (nude) mice with ultimate selection
of SKOV-3 Herceptin-resistant (HR) ‘breakthrough’ variants. We
characterized the SKOV-3 HR cell line by assessing the
antiproliferative and antitumor effects of trastuzumab on the cells
both in vitro and in vivo, by examining HER2
expression and number of trastuzumab binding sites, and by
determining the trastuzumab binding affinity for these cells,
comparing these results with those obtained in the parental SKOV-3
cell line. In addition, we performed MEP of the SKOV-3 HR and
parental cell lines to identify any differential gene expression
between the two cell lines that could potentially contribute to the
mechanism of resistance to trastuzumab therapy that the SKOV-3 HR
cell line exhibits.
Materials and methods
Cells and antibodies
SKOV-3 cells were purchased from the American Type
Culture Collection (Manassas, VA, USA). MDA-MB-435 cells were
obtained from Dr Janet Price (MD Anderson Cancer Center, Houston,
TX, USA). Cells were cultured in appropriate medium with 10%
heat-inactivated fetal bovine serum (FBS) and 2 mM L-glutamine. All
cell culture materials were purchased from Invitrogen/Gibco (Grand
Island, NY, USA). Trastuzumab was obtained from F. Hoffmann-La
Roche Ltd. (Basel, Switzerland) as a lyophilized powder and
resuspended with sterile bacteriostatic water. The suspension was
further diluted with sterile water or saline for all the
experiments. Cell lysates for immunoblot experiments were prepared
with lysis buffer containing 1% NP-40, 50 mM Tris (pH 7.5), 150 mM
NaCl, 1 mM EDTA, 1 mM Na3VO4, and 1 mM NaF.
Proteins (≥10 μg) were resolved by sodium dodecyl sulfate
polyacrylamide gel electrophoresis (SDS-PAGE) followed by transfer
to a nitrocellulose membrane. The membrane was then immunoprobed
for HER2 protein (Neomarkers/Labvision, Fremont, CA, USA),
phosphorylated extracellular regulated kinases 1/2 (pERK1/2), AKT
(Cell Signaling Technology, Danvers, MA, USA) or β-actin
(Sigma-Aldrich, St. Louis, MO, USA). Primary antibodies were
diluted appropriately with 5% non-fat milk and 3% bovine serum
albumin (BSA) in phosphate-buffered saline-Tween. The membrane was
then incubated with horseradish peroxidase-linked secondary
antibodies, and the proteins were detected using enhanced
chemiluminescence (Amersham Biosciences, Little Chalfont, UK).
SKOV-3 HR model generation and in vivo
studies
Female athymic, nu/nu (nude) mice were
purchased from Charles River Laboratories (Wilmington, MA, USA).
Animal experiments were performed in accordance with protocols
approved by the Institutional Animal Care and Use Committee and in
accordance with local regulations. Mice with established,
subcutaneous, palpable SKOV-3 tumors (≥100 mm3) were
treated once weekly with trastuzumab administered intraperitoneally
(loading dose, 4 mg/kg; maintenance dose, 2 mg/kg). Tumors that
exhibited growth inhibition but later gradually progressed
(so-called breakthrough tumors) were extracted, homogenized and
implanted into a new set of naive, athymic nu/nu
mice. This procedure, which consisted of monitoring tumor growth,
treating with trastuzumab and re-implanting breakthrough tumors,
was repeated for four rounds. Selected breakthrough tumors were
subsequently named SKOV-3 HR and cultured in vitro for
further characterization. Mouse fibroblasts and other murine cells
with finite replicative capacity died out during in vitro
cell culture, and only viable highly proliferative human tumor
cells remained. SKOV-3 HR cells were further selected by
three-dimensional growth in soft agar containing exogenous
trastuzumab (10–30 μg/ml). Afterwards, the cells were used to
further characterize and profile responses to trastuzumab treatment
in vitro and in vivo.
Cellular antiproliferation assay
SKOV-3 and SKOV-3 HR cells were plated at
1×105 per well in 96-well plates in complete culture
medium. The following day, trastuzumab was added to the culture
medium at 10 or 20 μg/ml with reduced (0.5%) FBS. The plates were
allowed to incubate for 72 h after the addition of trastuzumab. All
treatments were performed in triplicate. 3H-thymidine
(New England Nuclear/Perkin Elmer, Boston, MA, USA) at 0.5 μCi/well
was added to all wells during the last 4 h before cell harvest.
Cells were washed, trypsinized and harvested using a PHD glass
filter scintillation harvester (Cambridge Technologies, Watertown,
MA, USA). Filters containing 3H-thymidine-labeled cells
were transferred into plastic scintillation vials and counted on a
β-radiation counter (Beckman Coulter, Fullerton, CA, USA).
Fluorescence-activated cell sorting
(FACS) analysis
SKOV-3, SKOV-3 HR and MDA-MB-435 cells were
incubated with 10 μg/ml of a mouse monoclonal antibody directed
against the ECD of human HER2 (Ab-2, Neomarkers/Labvision). After
the cells were washed to remove unbound primary antibody, they were
treated with a 1:30 dilution of a goat antimouse IgG-fluorescein
isothiocyanate (FITC) secondary antibody (Chemicon International,
Temecula, CA, USA). Cells were analyzed using a BD-FACStar™
instrument (Becton-Dickinson, Palo Alto, CA, USA) gating on a
forward and side scatter, and the relative fluorescence was
measured by excitation at 488 nm. The percentage of cells stained
with anti-HER2 and the intensity of the staining was determined by
the BD CellQuest™ program (Becton-Dickinson).
Scatchard plot analysis
Antibody radiolabeling with an isotopic tag has
previously been described (22).
Briefly, 4 ng of 125I-trastuzumab (4×105 CPM)
in a total volume of 100 μl of Earle’s balanced salt solution
containing 20 mM N-2-hydroxyethylpiperazine-N’-2-ethanesulfonic
acid, pH 7.5, 2.5 mg/ml BSA and an equal amount of unlabeled
trastuzumab were incubated at 20°C for 2 h with SKOV-3 and SKOV-3
HR cells grown in 1-cm2 48-well plates. At the end of
the incubation period, the unbound radioactivity was removed by
washing cell monolayers three times with ice-cold binding buffer.
The cells were solubilized by adding 0.5 ml of 1 M NaOH and heated
at 65°C for 30 min. Non-specific binding, which was 3–5% of the
total binding, was determined by incubating the cells with labeled
antibody in the presence of 100-fold excess unlabeled antibody. The
non-specific binding was subtracted from the total binding to
determine the specific binding.
HER2 DNA sequencing and reverse
transcriptase-polymerase chain reaction
Primary DNA sequencing and reverse transcriptase
polymerase chain reaction (RT-PCR) have previously been described
(23). Briefly,
poly(A)+ RNA was directly isolated and converted to
single-strand cDNA as a template for PCR amplification from cells
growing in culture. A total of six PCR reactions designed to
generate sufficient overlapping HER2 DNA fragments were performed
for each cell line. PCR was run for 40 cycles using high fidelity
pfx DNA polymerase. A fraction of the PCR-amplified products
was resolved on a 1% agarose gel and stained with ethidium bromide.
The remaining PCR products were gel-purified for DNA sequencing.
Six PCR amplicons were used to cover the entire HER2 coding
region.
MEP and genomic analysis
MEP using Affymetrix gene chips (Santa Clara, CA,
USA) has previously been described (24). SKOV-3 and SKOV-3 HR cells were
incubated with or without trastuzumab (50 μg/ml) for 72 h. Cells
were harvested, total RNA was isolated and purified, cDNA was
generated and mRNA was scanned onto Affymetrix gene chips. In
vitro transcription and hybridization were performed with human
genome U133 set consisting of two GeneChip arrays (Affymetrix) with
45,000 probe sets. These probe sets represent more than 39,000
transcripts derived from approximately 33,000 well-substantiated
human genes. After uploading the results to the Affymetrix
laboratory information management system, signal intensities were
retrieved from the database for analysis. Normalization was
performed by sample via median scaling to the median of all chips
in the study. Normalization was not performed on the gene level.
Differentially expressed genes with greater than a two-fold
(p≤0.05) difference in fluorescence expression were filtered and
isolated from background. T-tests were performed for two sets of
comparisons. First, the treated population was compared with the
control population in the parental background to determine the
effects of trastuzumab treatment. Probe sets were selected that
reached a p-value cut-off of <0.05 and at least a two-fold
change in either the positive or negative direction. Probe sets
with a maximum expression in either of the two conditions of <25
were omitted for low expression. The second comparison was between
the acquisition of probe sets from parental and resistant
background samples. The list of genes that were identified was
annotated using the Affymetrix NetAffx™ system at the probe set
level. Biological functions were assigned to each gene network
using the Ingenuity Pathways Knowledge Base (Mountain View, CA,
USA). The Ingenuity Pathways program cross-references the entered
probe sets from the NetAffx system and assigns a network of genes
with biological links or interactions based on data obtained from
published literature (25).
Fisher’s exact test is used to calculate a p-value that determines
the probability that the biological function assigned to that
network can be explained by chance alone.
MUC1 expression by
immunohistochemistry
SKOV-3 and SKOV-3 HR cells and tumors were fixed in
10% neutral buffered formalin. The expression of glycosylated MUC1
on the cell surface was determined by incubating the cells with the
NeoMarkers MUC1 peroxidase-conjugated antibody (cat. no. RB-9222-P;
Fremont, CA, USA) or the human MUC1 Ab-5 alkaline
phosphatase-conjugated antibody, which recognizes amino acids
239-255 on the MUC1 cytoplasmic domain. Antigen unmasking was
performed on the samples prior to the addition of the primary
antibodies. Samples were incubated with the primary antibody for
approximately 18–24 h at 4°C. After repeated washing (≥3 times) in
wash buffer, samples were incubated with secondary antibodies for
30 min at room temperature. Staining was performed with fast red
chromogen until the fixed cells turned dark red-brown in color.
After washing in distilled water and dehydrating in ethanol,
cover-slips were mounted on the slides. Photographs of slides were
taken with a light microscope using x40 magnification.
Results
Selection of SKOV-3 HR tumors and lack of
antiproliferative and antitumor response to trastuzumab
The development of the SKOV-3 HR model involved the
induction of acquired resistance over time in the in vivo
setting through continuous exposure to trastuzumab. Initially,
trastuzumab-sensitive SKOV-3 cells were implanted subcutaneously in
athymic nu/nu mice, which were administered a
therapeutic dose of trastuzumab once a week when tumors were firmly
established (26). Tumor growth
from individual animals was monitored for more than 120 days post
implant. When tumor progression occurred, breakthrough tumors that
reached ≥1000 mm3 were extracted, processed, divided
equally and implanted into a new set of naive mice (Fig. 1A). In the second round, upon tumor
establishment, trastuzumab treatment was again initiated and tumor
responses were followed. Three or four rounds of in vivo
serial passaging of breakthrough tumors were carried out to ensure
there was a lack of, or negligible, response to trastuzumab before
these tumors were cultured in vitro and characterized.
 | Figure 1(A) SKOV-3 cells (5×106
cells/mouse) were implanted subcutaneously in the right hind flank
of ten female athymic nu/nu mice with BD Matrigel™.
For each mouse, treatments started when tumor volume reached
>100 mm3. Trastuzumab (4 mg/kg loading dose for one
week and 2 mg/kg thereafter) was administered intraperitoneally
weekly for the entire duration of the study (>120 days
post-implantation). *Mouse number 6 showed initial
inhibition of tumor growth but later lost this response. The tumor
from this mouse was passaged into ten naive nu/nu
mice in the second round for further selection. (B) SKOV-3 and
SKOV-3 HR cells (5×104/well) were seeded in triplicate
in 96-well plates, and the next day trastuzumab was added at the
indicated doses (white bars, culture medium; gray bars, 10 μg/ml
trastuzumab; black bars, 20 μg/ml trastuzumab). Plates were allowed
to incubate at 37°C, 5% CO2 for an additional 2 h.
3H-thymidine was added 18 h prior to harvest.
β-radiation emission counts per well were averaged and graphed as
radiation counts per min. (C) SKOV-3 and SKOV-3 HR cells
(5×106 cells/mouse) were implanted subcutaneously in the
right hind flank of female athymic nu/nu mice with
Matrigel (1:1 v/v). Treatments started at the indicated time when
group mean tumor volume reached approximately 100–200
mm3. Trastuzumab (4 mg/kg loading dose for one week and
2 mg/kg thereafter) and vehicle were administered intraperitoneally
weekly for 4.5 weeks. Circle, SKOV-3 cells treated with vehicle
(sterile saline); triangle, SKOV-3 cells treated with trastuzumab;
square, SKOV-3 HR cells treated with vehicle; diamond, SKOV-3 HR
cells treated with trastuzumab. CPM, counts per min; HR,
Herceptin-resistant; SEM, standard error of mean. |
First, the effects of exogenously added trastuzumab
on the proliferation of the breakthrough SKOV-3 HR cells were
determined in vitro. Trastuzumab was added to plates of
cultured SKOV-3 and SKOV-3 HR cells, and the incorporation of
3H-thymidine was measured as an indicator of
proliferation. β-radiation emissions revealed that the addition of
trastuzumab inhibited SKOV-3 proliferation compared with cells
incubated in culture medium alone (Fig. 1B). Even a low dose of trastuzumab
(10 μg/ml) over 72 h of incubation was enough to notice a
difference in proliferation compared with the control.
Interestingly, trastuzumab did not have an antiproliferative effect
on the SKOV-3 HR cells (Fig. 1B).
These proliferation experiments were performed at least twice and
similar outcomes were obtained each time.
To confirm the resistance of SKOV-3 HR cells to
trastuzumab, the effect of treatment with trastuzumab on these
cells was again examined in vivo. In a xenograft efficacy
experiment, the effects of trastuzumab therapy were compared side
by side in athymic nu/nu mice that were implanted
with SKOV-3 and SKOV-3 HR tumors. SKOV-3 HR tumors became palpable
more rapidly and grew faster than parental SKOV-3 tumors (Fig. 1C). Trastuzumab treatment for mice
implanted with SKOV-3 tumors was delayed for 11 days to ensure that
the starting tumor volumes (approximately 100–200 mm3)
were similar to SKOV-3 HR tumor volumes. Trastuzumab inhibited
SKOV-3 tumor growth by 50% (p=0.05) compared with SKOV-3
vehicle-treated controls over a 4.5-week treatment period. SKOV-3
HR tumor growth, in contrast, was not inhibited by trastuzumab that
was given at the same treatment regimen and schedule (tumor growth
inhibition = 8%, p=0.734).
HER2 receptor expression and downstream
signaling
The lack of trastuzumab activity in vitro and
in vivo against SKOV-3 HR cells may be due to the loss of
cell surface HER2 receptor expression. To address this possibility,
levels of cell surface HER2 expression on SKOV-3 and SKOV-3 HR
cells were compared. The MDA-MB-435 cell line, a human breast
carcinoma cell line that has low HER2 protein expression, served as
the negative control. SKOV-3, SKOV-3 HR and MDA-MB-435 cells were
incubated with labeled FITC-conjugated mouse monoclonal antibody
against the human HER2 receptor ECD. FACS analysis revealed that
SKOV-3 and SKOV-3 HR cells had similar levels of expression of cell
surface HER2 receptors (Fig. 2A),
suggesting that these SKOV-3 HR cells may exhibit aberrant
signaling downstream from HER2.
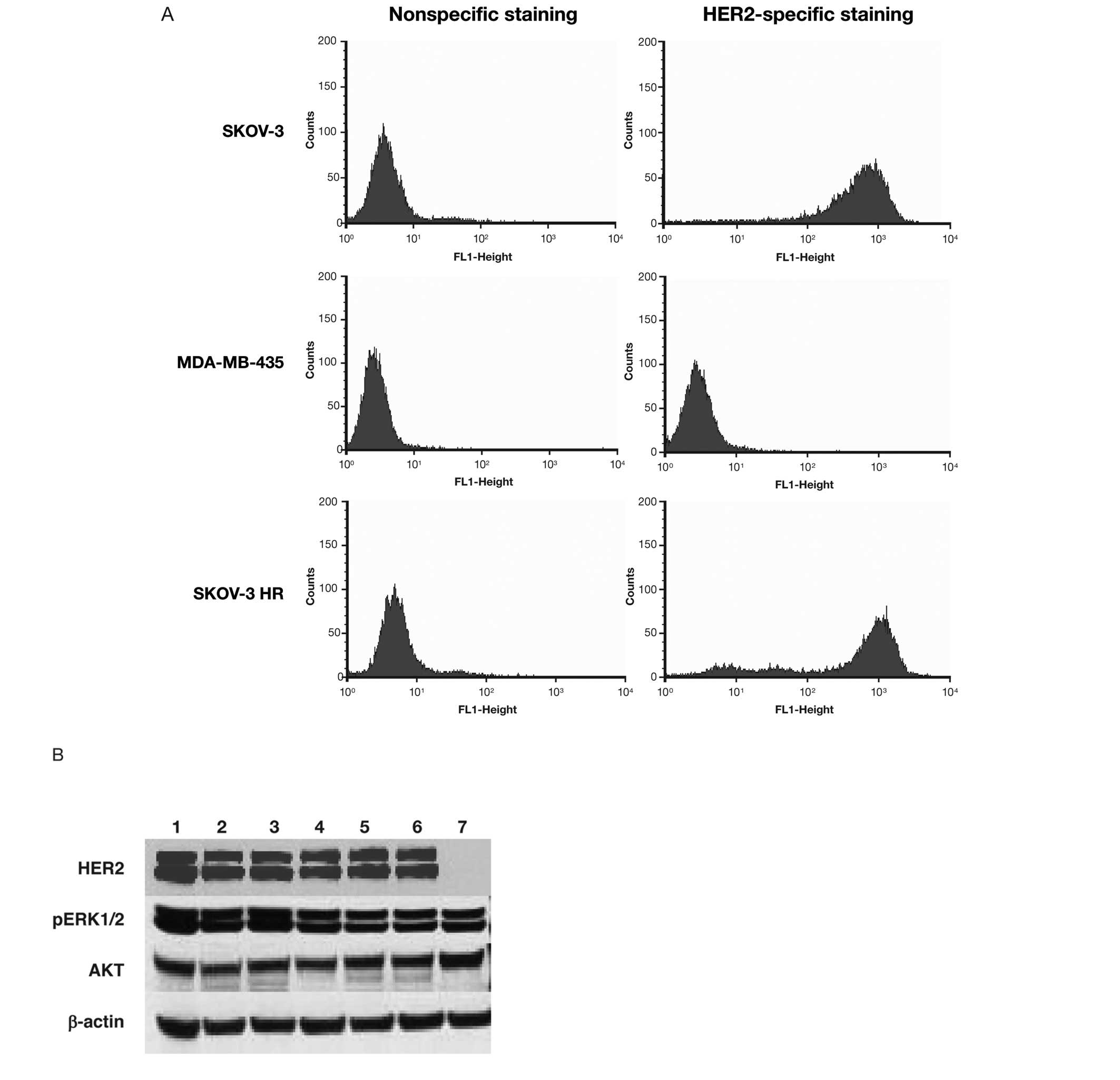 | Figure 2(A) Determination of HER2 cell
surface receptor expression was performed by FACS analysis. SKOV-3,
MDA-MB-435 and SKOV-3 HR cells were tested for HER2 receptors with
mouse monoclonal antibody against ECD followed by a secondary goat
antimouse FITC-antibody. Non-specific staining refers to
fluorescence uptake with the secondary antibody only, while
specific staining indicates fluorescence uptake with primary and
secondary antibodies. The mean channel fluorescence was similar in
testing all cell lines, indicating similar levels of cell surface
HER2 receptors on SKOV-3 and SKOV-3 HR cells. (B) Immunoblot of
SKOV-3, SKOV-3 HR and Hek293 (HER2-negative control). Multiple
samples of SKOV-3 (lanes 1–3) and SKOV-3 HR (lanes 4–6), and one
sample of Hek293 (lane 7) cell lysates were blotted for protein
expression levels of HER2, AKT, and p44/42 (mitogen-activated
protein kinase). β-actin served as the loading control. FACS,
fluorescence-activated cell sorting; ECD, extracellular domain;
FITC, fluorescein isothiocyanate; HER2, human epidermal growth
factor receptor; HR, Herceptin-resistant; pERK1/2, phosphorylated
extracellular regulated kinases 1/2. |
Downstream signaling components were then evaluated
for alterations in their protein expression. Immunoblotting was
used to determine the total protein levels of p44/42 (pErk1/2) and
AKT in cell lysates from SKOV-3, SKOV-3 HR and Hek293 cells
(Fig. 2B), with Hek293 serving as
the HER2-negative control. The results revealed no significant
differences in the expression levels of AKT and pERK1/2 (p44/42)
between SKOV-3 and SKOV-3 HR cells.
Increased binding sites but decreased
binding affinity
A ligand receptor-binding assay and Scatchard plot
analysis were performed with radioactively tagged trastuzumab to
determine the binding affinity of trastuzumab for cognate HER2
receptors. Purified 125I-labeled trastuzumab was
exogenously added to SKOV-3 and SKOV-3 HR cells in culture and
later saturated with unlabeled or cold trastuzumab for competitive
binding. Scatchard plot analysis revealed that
125I-trastuzumab bound to more binding sites on the
surface of SKOV-3 HR cells (1.1×106 sites/cell) than of
SKOV-3 cells (0.335×106 sites/cell). However, the
binding affinity was decreased by approximately two- to three-fold
(7.6×10−10 vs. 3.1×10−10 M, respectively;
Fig. 3).
Sequencing of HER2 primary DNA
transcripts
As the number of trastuzumab binding sites is
increased on SKOV-3 HR cells, the decreased binding affinity of
trastuzumab for these cells could be due to an alteration or
mutation in the HER2 ECD. To address this, the HER2 primary
transcripts from both cell lines were compared. Six PCR amplicons
with overlapping sizes were used to cover the entire HER2 coding
region (Fig. 4A). All PCR products
were found at the expected sizes by agarose gel electrophoresis
(Fig. 4B). Overall, the PCR
amplified products from both cell lines were identical. Analyzing
the DNA sequence following RT-PCR revealed that the full-length
HER2 sequences obtained from SKOV-3 and SKOV-3 HR cells were
identical (Fig. 4C). Both
sequences aligned well with a HER2 sequence from a published
database (e.g., SWISSPROT). Therefore, the decreased binding
affinity of trastuzumab for HER2 receptors on the SKOV-3 HR cells
is likely not due to mutations in the HER2 gene.
Genes differentially expressed in SKOV-3
HR cells
Through in vivo induction of acquired
resistance with continuous trastuzumab treatment of tumor-bearing
mice, the SKOV-3 HR cells underwent specific genomic changes that
differentiated them from their parental counterparts. Performing
MEP using Affymetrix gene chips revealed genes that may have given
rise to the resistance phenotype or, as a result of the resistance
induction, were now distinctly expressed genes. Genes with
expression that differed by a greater than two-fold (p≤0.05)
magnitude of difference in fluorescence were filtered and isolated
from background. Fluorescence expression analysis revealed that a
larger number of genes were affected by trastuzumab in the parental
background than in the resistant background, suggesting that
trastuzumab induced or affected the functionality of the SKOV-3
cells. The gene chip fluorescence results were annotated using the
Affymetrix NetAffx system at the probe set level. Biological
functions were assigned to each gene network using the Ingenuity
Pathways Knowledge Base. The Ingenuity Pathways program identified
a set of genes with a known biological link or interaction with the
EGFR family. The basal fluorescence expression of these genes in
the two cell lines is presented in Fig. 5A. The expression of these genes in
the two cell lines after trastuzumab treatment is shown in Fig. 5B. This set of 33 genes was
differentially expressed between the two cell lines and had a
greater than two-fold change in expression level between the basal
state and after trastuzumab treatment. The genes that the Ingenuity
Pathways program assigned biological functions to have notable and
well-studied roles in tumorigenesis, angiogenesis, invasion and
metastases, apoptosis, proliferation and survival. Some of these
genes include angiopoietin 2, nestin,
ceruloplasmin, fibroblast growth factor receptor 3,
integrin α/β, Vav3 oncogene, matrix
metalloproteinase-7, keratin 19, CD24, ETS
transcription factor, intercellular adhesion molecule 2,
urokinase plasminogen activator receptor, epiregulin,
thrombomodulin, Id-1, heparin-binding growth
factor, IGF-binding protein 6 and MUC1.
 | Figure 5MFI of genes that were identified
with differential expression by the Ingenuity Pathways program
under (A) basal or constitutive condition (without trastuzumab
treatment), and (B) with the addition of trastuzumab (50 μg/ml) for
72 h after signal intensities were retrieved from the Affymetrix
LIMS database and normalized using median scaling to the median of
all chips. Probesets were selected which reached a p-value cut-off
of <0.05 and a fold change of at least two-fold in either the
positive or negative direction. SKOV-3 cells, white bars; SKOV-3 HR
cells, black bars. ETS, E-twenty-six; FGFR, fibroblast growth
factor receptor; ICAM2, intercellular adhesion molecule 2; IGF,
insulin-like growth factor; IL-1, interleukin 1; HBGF,
heparin-binding growth factors; MFI, mean fluorescence intensity;
MMP7, matrix metalloproteinase-7; MUC1, mucin 1; uPAR, urokinase
plasminogen activator receptor. |
MUC1 is overexpressed in SKOV-3 HR
cells
The role of MUC1 in cancer pathogenesis has been
extensively studied, and MUC1 is known to interact with the EGFR
family (27). Since the
MUC1 gene was shown to be highly expressed in SKOV-3 HR
cells, MUC1 protein expression was confirmed by IHC. Examination of
the MUC1 cell surface and cytoplasmic expression revealed a
striking difference in MUC1 positivity between SKOV-3 and SKOV-3 HR
cells. Not only was MUC1 expression on the cell surface of the
SKOV-3 cells dramatically decreased compared with the SKOV-3 HR
cells, there were significantly fewer SKOV-3 cells that were
positive for MUC1 (Fig. 6A and B).
Almost all (≥90%) of the SKOV-3 HR cells were stained dark
red-brown for positive MUC1 expression compared with ≤20% of the
SKOV-3 cells. Moreover, the intensity of positive MUC1 staining on
SKOV-3 HR cells was greater compared with that of the parental
cells. The overexpression of MUC1 protein on SKOV-3 HR cells
correlated with the significantly higher gene expression obtained
from the microarrays. Examination of SKOV-3 and SKOV-3 HR in
vivo subcutaneous tumors for MUC1 expression revealed a pattern
similar to that observed with the cultured cells. MUC1 expression
by IHC in the SKOV-3 HR cells was more intense than the MUC1
expression on parental SKOV-3 tumors that were extracted at the
same time (Fig. 6C and D).
Discussion
The development and subsequent approval of
trastuzumab has led to the availability of the first antibody-based
molecular targeted therapy for MBC in patients whose tumors have
HER2 gene amplification and concomitant HER2 protein
overexpression. However, despite clinical responses observed with
both monotherapy and in combination regimens, there are still many
patients whose tumors progress on trastuzumab-based therapy,
indicating acquired resistance to therapy (28). Moreover, a subset of patients do
not have objective responses despite HER2 overexpression,
indicating that they may have some form of de novo or
intrinsic resistance. Thus, patients with HER2-positive breast
cancer who do not respond to trastuzumab or progress during
trastuzumab-based treatment may benefit from new therapeutics with
different targets, or different treatment regimens. Studies
exploring the mechanism of trastuzumab resistance in preclinical
cancer models as well as methods to reverse resistance, thereby
restoring sensitivity to and effectiveness of trastuzumab
treatment, are ongoing (16–21,29–31).
Preclinical studies, including our own, have demonstrated the
antitumor activity of trastuzumab in experiments using ovarian
carcinoma models with intrinsic HER2 overexpression. However, not
all HER2-overexpressing tumors respond to trastuzumab, the reasons
for which are unclear, and there is a continuous need to
investigate the mechanisms behind the lack of translational
efficacy. The knowledge we gain from studying models with
sensitivity to trastuzumab and investigating the subsequent loss of
that sensitivity is vital for future management of the disease and
the improvement of treatment options. Intrinsic and acquired
resistance, as well as genetic or epigenetic factors, may limit the
effectiveness of trastuzumab in ovarian cancers and other
HER2-overexpressing epithelial cancers (6,32).
Our group is the first to develop a preclinical
model of ovarian carcinoma, SKOV-3 HR, with induced (or acquired)
resistance to trastuzumab. The SKOV-3 HR model was developed from
the parental SKOV-3 cell line. SKOV-3 is used by many investigators
to demonstrate the in vitro and in vivo
antiproliferative and antitumor effects of trastuzumab, making it a
useful tool to investigate resistance mechanisms from a preclinical
perspective (3,33). We generated the SKOV-3 HR model by
in vivo serial passaging of SKOV-3 tumors that had lost
sensitivity to trastuzumab for three to four rounds. SKOV-3 HR is
the first trastuzumab-resistant ovarian model to be developed by
this method, which, as an in vivo model, may prove to be
more clinically relevant than an artificial in vitro tissue
culture environment.
The results from our study with the SKOV-3 HR model
may be cross-referenced or translatable to other preclinical models
or clinical specimens, aiding in deciphering the mechanisms of
trastuzumab resistance and ultimately in developing strategies and
therapies to overcome such mechanisms. For instance, several
laboratories have developed trastuzumab-resistant breast cancer
cell lines that are sensitive to EGFR tyrosine kinase inhibitors
(34). Similarly, we observed that
the SKOV-3 HR tumors developed in this study also exhibit
sensitivity to the tyrosine kinase inhibitor erlotinib (data not
shown). In addition, evidence has emerged that trastuzumab
resistance can be circumvented or abrogated by targeting HER2
receptor heterodimerization and kinase activity, or disruption of
HER2 signaling with another anti-HER2 antibody, such as pertuzumab
(35,36).
The characterization of SKOV-3 HR cells revealed
constitutive expression of the HER2 protein similar to that
observed with trastuzumab-sensitive parental cells. Similar
findings of unaltered HER2 expression and downstream growth
signaling regulators before and after trastuzumab treatment have
been observed in other models of trastuzumab resistance, implying
that cancer cells can circumvent trastuzumab activity but continue
to depend on the activation of the HER2 oncogene (19,34).
HER2 overexpression and protein downregulation that result from
trastuzumab treatment are not predictors of response (37). Therefore, factors other than HER2
overexpression may be predictive of antitumor response to
trastuzumab in ovarian cancer (38). However, the fundamental reasons for
the clinical activity of trastuzumab in HER2-positive breast and
gastric cancers, but not in other HER2-positive cancers, such as
ovarian cancer, are not completely understood.
We demonstrate that trastuzumab is not effective
against SKOV-3 HR proliferation and in vivo tumor growth.
The reasons for these findings could be due to the
ineffectual/incomplete binding of trastuzumab to HER2 receptors, as
demonstrated by the receptor/ligand binding assay and Scatchard
plot analysis. We found that there was a three-fold or greater
increase in trastuzumab binding sites on SKOV-3 HR cells, but which
was accompanied by a decrease in binding affinity compared with the
parental line. This decreased trastuzumab binding affinity could
indicate an alteration in the trastuzumab recognition or binding
epitope on the HER2 ECD; however, DNA sequence analysis revealed
that the HER2 primary transcripts from both cell lines were
similar, with no obvious differences. Similar results regarding the
increase in binding sites and the diminished binding affinity were
noted in the characterization of JIMT-1, a trastuzumab-resistant
breast cancer cell line, by Nagy et al (19). Moreover, the JIMT-1 cells retained
HER2 amplification and protein overexpression and maintained
unaltered expression of downstream growth regulatory signaling
proteins, which was also observed with the SKOV-3 HR cells in this
study.
Determination of the genomic differences between
SKOV-3 and SKOV-3 HR may aid in elucidating the mechanisms of the
development of resistance. MEP identified a set of genes that were
differentially expressed at constitutive or basal levels between
parental SKOV-3 and SKOV-HR cells. A similar MEP was performed
after the addition of in vitro exogenous trastuzumab to
cells in culture. The Ingenuity Pathways program identified a
dataset of 33 genes that have direct network/links/interactions to
HER/EGFR biological processes. Functional analysis grouped these
genes by their role in biological processes, such as
angiogenesis/invasion, differentiation, proliferation, cell growth
and survival. These genes included MUC1, angiopoietin
2, CD24, Vav3 oncogene, nestin and ETS
transcription factor, whose differential and dysregulated
expression and subsequent biological interaction with the EGFR
family could support the development of SKOV-3 HR cells that are
phenotypically aggressive with acquired therapeutic resistance to
trastuzumab.
One of the genes that was overexpressed at least
five-fold at the genomic and protein level by the SKOV-3 HR cells
compared with the SKOV-3 cells is MUC1. MUC1 is an
O-glycosylated transmembrane protein that is aberrantly expressed
in a majority of breast, ovarian and many other epithelial
carcinomas (39). Cell surface
mucins interact with the EGFR family to regulate signaling events
related to cell growth, motility, differentiation, inflammation and
other higher-order functions. MUC1 protein overexpression in SKOV-3
HR cells was confirmed by the marked positive staining by IHC.
SKOV-3 HR tumors exhibit distinct MUC1 overexpression, which may
contribute to a more robust tumorigenicity and the overt aggressive
growth of the tumor cells. MUC1 expression is highly regulated and
can be inhibited by HER2 overexpression in transformed mammary
epithelial cells (40).
Conceivably, the long-term use of trastuzumab could have led to
HER2 inhibition, and, possibly in conjunction with the altered
expression of the genes that we identified (angiopoeitin 2,
increased nestin, increased Vav3 oncogene expression)
caused the upregulation of MUC1 mRNA and protein overexpression in
SKOV-3 HR tumors. Consequently, this course of events could have
led to the formation of an aggressive phenotype that confers
resistance to trastuzumab.
Cell surface mucins such as MUC1 form complex
multivalent tandem oligosaccharide repeats that form densely
arrayed, highly ordered structural layers that facilitate the
ingress and egress of molecules to the cell. These oligosaccharide
tandem repeats surround the tumor cell like a molecular ‘shield’ to
block toxic compounds, such as chemotherapeutic agents or
anticancer antibodies (41).
Therefore, similar to MUC4 in the JIMT-1 cells, overexpression of
MUC1 in SKOV-3 HR cells could present a steric barrier or
constraint that affects the binding of trastuzumab to HER2
receptors, thereby causing a decrease in binding affinity and a
subsequent lack of antiproliferative response. Furthermore, the
increased phosphorylation of MUC1 could potentiate downstream
signaling pathways, such as Ras/Raf/Erk (MAPK), leading to
uncontrolled growth and survival (27).
The data obtained from these studies provide further
evidence of the role of MUC1 in trastuzumab resistance. For
instance, Fessler et al showed that a cleaved form of the
MUC1 protein (MUC1*) is upregulated in trastuzumab-resistant breast
cancer cells and that the inhibition of MUC1* could
reverse resistance and restore trastuzumab sensitivity (30). Moreover, the expression of
MUC1* could be involved in so-called intrinsically
resistant or refractory HER2-positive cancers (IHC
1+/2+ HER2-positive breast cancer and ovarian
cancer) for which trastuzumab is less effective. Therefore, the
combination of trastuzumab and a MUC1 antagonist could be used to
reverse acquired and intrinsic resistance to trastuzumab, and may
be effective in deterring the onset of resistance in the adjuvant
setting.
Zhang et al showed that increased c-SRC
activation mediated by the loss of PTEN or the overexpression of
EGFR or the type 1 IGF receptor (IGF1R) can confer therapeutic
resistance to trastuzumab in breast cancer cells, and the direct
dephosphorylation of c-SRC can restore trastuzumab sensitivity
(21). Of note, MUC1 is associated
with the c-SRC tyrosine kinase. c-SRC phosphorylates the MUC1
cytoplasmic domain and can inhibit the interaction between MUC1 and
glycogen synthase kinase 3-β. Moreover, the c-SRC-mediated
phosphorylation of MUC1 can increase the binding of MUC1 and
β-catenin (42). Thus, the
overexpression of MUC1 in the SKOV-3 HR cells and the interaction
with its substrate, c-SRC, may implicitly confer resistance to
trastuzumab.
Other mechanisms of resistance to trastuzumab that
were proposed from research in preclinical models include
amplification of HER/ErbB signaling, cross-talk with heterologous
receptor tyrosine kinases, accumulation of the constitutively
active truncated p95-HER2, alternate or amplification of survival
signaling pathways, such as the IGF1R and PI3K/AKT pathways,
respectively, and the alteration of downstream signaling, such as
the loss of PTEN. This diversity suggests that no one mechanism may
be responsible for causing the resistance of HER2-overexpressing
tumor cell to trastuzumab. Our data add to the knowledge that
continues to accumulate in this field, and strategies are being
developed to overcome resistance in trastuzumab-treated patients.
For instance, novel agents are being evaluated as single agents or
in combination, in patients with HER2-positive MBC that has
progressed on prior trastuzumab therapy. For example, pertuzumab in
combination with trastuzumab has demonstrated a response rate of
24.2% in patients in this setting (43). Trastuzumab emtansine (T-DM1),
composed of trastuzumab conjugated via a stable thioether linker to
DM1 (a potent microtubule inhibitor), has also demonstrated
activity in this setting. Phase II trials evaluating single-agent
T-DM1 have demonstrated response rates of 35.5 and 25.9% in
patients with HER2-positive MBC who had received prior trastuzumab
and chemotherapy (44,45). Lastly, lapatinib, a small molecule
HER2 and EGFR kinase inhibitor, has a mechanism of action distinct
from trastuzumab (46). Clinical
benefit in the trastuzumab-refractory HER2-positive MBC setting was
observed when lapatinib was administered in combination with
chemotherapy, resulting in a reduction in the risk of progression
of 51% in the lapatinib arm compared with the control arm (47).
Our study on the development and characterization of
the SKOV-3 HR trastuzumab-resistant ovarian carcinoma model add to
the increasing body of knowledge of resistance mechanisms observed
with many cancer therapies. Our proposed mechanism of trastuzumab
resistance supports therapies that target MUC1 along with HER2 to
achieve increased antitumor efficacy. Moreover, we identified a
gene signature that may be responsible for the onset of resistance
to trastuzumab and could provide the basis for elucidating the lack
of trastuzumab activity in patients with ovarian cancer. This gene
signature could correlate with clinical samples with acquired and
intrinsic resistance and could potentially provide the basis for
identifying patient populations that may or may not respond to
trastuzumab treatment. Although further research is required, the
inhibition of these genes and proteins could potentially result in
the reversal of resistance and lead to the discovery of more
effective therapies for breast, ovarian and other cancers.
Acknowledgements
We appreciate the technical expertise
of Dr Wei Chu, Mr. Christian Tovar, Ms. Violeta Adames, Ms.
Markella Kordoyanni and Ms. Urmi Bhatt.
References
|
1
|
Alroy I and Yarden Y: The ErbB signaling
network in embryogenesis and oncogenesis: signal diversification
through combinatorial ligand-receptor interactions. FEBS Lett.
410:83–86. 1997. View Article : Google Scholar
|
|
2
|
Olayioye MA, Neve RM, Lane HA and Hynes
NE: The ErbB signaling network: receptor heterodimerization in
development and cancer. EMBO J. 19:3159–3167. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Slamon DJ, Godolphin W, Jones LA, Holt JA,
Wong SG, Keith DE, Levin WJ, Stuart SG, Udove J, Ullrich A, et al:
Studies of the HER-2/neu proto-oncogene in human breast and ovarian
cancers. Science. 244:707–712. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ross JS, Slodkowska EA, Symmans WF,
Pusztai L, Ravdin PM and Hortobagyi GN: The HER-2 receptor and
breast cancer: ten years of targeted anti-HER-2 therapy and
personalized medicine. Oncologist. 14:320–368. 2009.PubMed/NCBI
|
|
5
|
Dawood S, Broglio K, Buzdar AU, Hortobagyi
GN and Giordano SH: Prognosis of women with metastatic breast
cancer by HER2 status and trastuzumab treatment: an
institutional-based review. J Clin Oncol. 28:92–98. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bookman MA, Darcy KM, Clarke-Pearson D,
Boothby RA and Horowitz IR: Evaluation of monoclonal anti-HER2
antibody, trastuzumab, in patients with recurrent or refractory
ovarian or primary peritoneal carcinoma with overexpression of
HER2: a phase II trial of the Gynecologic Oncology Group. J Clin
Oncol. 21:283–290. 2003. View Article : Google Scholar
|
|
7
|
McAlpine JN, Wiegand KC, Vang R, Ronnett
BM, Adamiak A, Köbel M, Kalloger SE, Swenerton KD, Huntsman DG,
Gilks CB and Miller DM: HER2 overexpression and amplification is
present in a subset of ovarian mucinous carcinomas and can be
targeted with trastuzumab therapy. BMC Cancer. 9:4332009.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Slamon DJ, Leyland-Jones B, Shak S, Fuchs
H, Paton V, Bajamonde A, Fleming T, Eiermann W, Wolter J, Pegram M,
Baselga J and Norton L: Use of chemotherapy plus a monoclonal
antibody against HER2 for metastatic breast cancer that
overexpresses HER2. N Engl J Med. 344:783–792. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cobleigh MA, Vogel CL, Tripathy D, Robert
NJ, Scholl S, Fehrenbacher L, Wolter JM, Paton V, Shak S, Lieberman
G and Slamon DJ: Multinational study of the efficacy and safety of
humanized anti-HER2 monoclonal antibody in women who have
HER2-overexpressing metastatic breast cancer that has progressed
after chemotherapy for metastatic disease. J Clin Oncol.
17:2639–2648. 1999.
|
|
10
|
Romond EH, Perez EA, Bryant J, Suman VJ,
Geyer CE Jr, Davidson NE, Tan-Chiu E, Martino S, Paik S, Kaufman
PA, Swain SM, Pisansky TM, Fehrenbacher L, Kutteh LA, Vogel VG,
Visscher DW, Yothers G, Jenkins RB, Brown AM, Dakhil SR, Mamounas
EP, Lingle WL, Klein PM, Ingle JN and Wolmark N: Trastuzumab plus
adjuvant chemotherapy for operable HER2-positive breast cancer. N
Engl J Med. 353:1673–1684. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Perez EA, Romond EH, Suman VJ, Jeong JH,
Davidson NE, Geyer CE Jr, Martino S, Mamounas EP, Kaufman PA and
Wolmark N: Four-year follow-up of trastuzumab plus adjuvant
chemotherapy for operable human epidermal growth factor receptor
2-positive breast cancer: joint analysis of data from NCCTG N9831
and NSABP B-31. J Clin Oncol. 29:3366–3373. 2011.PubMed/NCBI
|
|
12
|
Smith I, Procter M, Gelber RD, Guillaume
S, Feyereislova A, Dowsett M, Goldhirsch A, Untch M, Mariani G,
Baselga J, Kaufmann M, Cameron D, Bell R, Bergh J, Coleman R,
Wardley A, Harbeck N, Lopez RI, Mallmann P, Gelmon K, Wilcken N,
Wist E, Sánchez Rovira P and Piccart-Gebhart MJ: 2-year follow-up
of trastuzumab after adjuvant chemotherapy in HER2-positive breast
cancer: a randomised controlled trial. Lancet. 369:29–36.
2007.PubMed/NCBI
|
|
13
|
Slamon D, Eiermann W, Robert N, Pienkowski
T, Martin M, Press M, Mackey J, Glaspy J, Chan A, Pawlicki M,
Pinter T, Valero V, Liu MC, Sauter G, von Minckwitz G, Visco F, Bee
V, Buyse M, Bendahmane B, Tabah-Fisch I, Lindsay MA, Riva A and
Crown J: Adjuvant trastuzumab in HER2-positive breast cancer. N
Engl J Med. 365:1273–1283. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bang YJ, Van Cutsem E, Feyereislova A,
Chung HC, Shen L, Sawaki A, Lordick F, Ohtsu A, Omuro Y, Satoh T,
Aprile G, Kulikov E, Hill J, Lehle M, Rüschoff J and Kang YK:
Trastuzumab in combination with chemotherapy versus chemotherapy
alone for treatment of HER2-positive advanced gastric or
gastro-oesophageal junction cancer (ToGA): a phase 3, open-label,
randomised controlled trial. Lancet. 376:687–697. 2010. View Article : Google Scholar
|
|
15
|
Spector NL and Blackwell KL: Understanding
the mechanisms behind trastuzumab therapy for human epidermal
growth factor receptor 2-positive breast cancer. J Clin Oncol.
27:5838–5847. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Albanell J and Baselga J: Unraveling
resistance to trastuzumab (Herceptin): insulin-like growth factor-I
receptor, a new suspect. J Natl Cancer Inst. 93:1830–1832. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Nahta R, Takahashi T, Ueno NT, Hung MC and
Esteva FJ: p27Kip1 down-regulation is associated with
trastuzumab resistance in breast cancer cells. Cancer Res.
64:3981–3986. 2004.
|
|
18
|
Nagata Y, Lan KH, Zhou X, Tan M, Esteva
FJ, Sahin AA, Klos KS, Li P, Monia BP, Nguyen NT, Hortobagyi GN,
Hung MC and Yu D: PTEN activation contributes to tumor inhibition
by trastuzumab, and loss of PTEN predicts trastuzumab resistance in
patients. Cancer Cell. 6:117–127. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Nagy P, Friedländer E, Tanner M, Kapanen
AI, Carraway KL, Isola J and Jovin TM: Decreased accessibility and
lack of activation of ErbB2 in JIMT-1, a Herceptin-resistant,
MUC4-expressing breast cancer cell line. Cancer Res. 65:473–482.
2005.PubMed/NCBI
|
|
20
|
Dhillon J, Astanehe A, Lee C, Fotovati A,
Hu K and Dunn SE: The expression of activated Y-box binding
protein-1 serine 102 mediates trastuzumab resistance in breast
cancer cells by increasing CD44+ cells. Oncogene.
29:6294–6300. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhang S, Huang WC, Li P, Guo H, Poh SB,
Brady SW, Xiong Y, Tseng LM, Li SH, Ding Z, Sahin AA, Esteva FJ,
Hortobagyi GN and Yu D: Combating trastuzumab resistance by
targeting SRC, a common node downstream of multiple resistance
pathways. Nat Med. 17:461–469. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Fernandes H, Cohen S and Bishayee S:
Glycosylation-induced conformational modification positively
regulates receptor-receptor association: a study with an aberrant
epidermal growth factor receptor (EGFRvIII/DeltaEGFR) expressed in
cancer cells. J Biol Chem. 276:5375–5383. 2001. View Article : Google Scholar
|
|
23
|
Meric F, Lee WP, Sahin A, Zhang H, Kung HJ
and Hung MC: Expression profile of tyrosine kinases in breast
cancer. Clin Cancer Res. 8:361–367. 2002.PubMed/NCBI
|
|
24
|
Unger MA, Rishi M, Clemmer VB, Hartman JL,
Keiper EA, Greshock JD, Chodosh LA, Liebman MN and Weber BL:
Characterization of adjacent breast tumors using oligonucleotide
microarrays. Breast Cancer Res. 3:336–341. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Raponi M, Belly RT, Karp JE, Lancet JE,
Atkins D and Wang Y: Microarray analysis reveals genetic pathways
modulated by tipifarnib in acute myeloid leukemia. BMC Cancer.
4:562004. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sliwkowski MX, Lofgren JA, Lewis GD,
Hotaling TE, Fendly BM and Fox JA: Nonclinical studies addressing
the mechanism of action of trastuzumab (Herceptin). Semin Oncol.
26(Suppl): 60–70. 1999.PubMed/NCBI
|
|
27
|
Schroeder JA, Thompson MC, Gardner MM and
Gendler SJ: Transgenic MUC1 interacts with epidermal growth factor
receptor and correlates with mitogen-activated protein kinase
activation in the mouse mammary gland. J Biol Chem.
276:13057–13064. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Nielsen DL, Andersson M and Kamby C:
HER2-targeted therapy in breast cancer. Monoclonal antibodies and
tyrosine kinase inhibitors. Cancer Treat Rev. 35:121–136. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
du Manoir JM, Francia G, Man S, Mossoba M,
Medin JA, Viloria-Petit A, Hicklin DJ, Emmenegger U and Kerbel RS:
Strategies for delaying or treating in vivo acquired resistance to
trastuzumab in human breast cancer xenografts. Clin Cancer Res.
12:904–916. 2006.
|
|
30
|
Fessler SP, Wotkowicz MT, Mahanta SK and
Bamdad C: MUC1* is a determinant of trastuzumab
(Herceptin) resistance in breast cancer cells. Breast Cancer Res
Treat. 118:113–124. 2009.
|
|
31
|
Valabrega G, Capellero S, Cavalloni G,
Zaccarello G, Petrelli A, Migliardi G, Milani A, Peraldo-Neia C,
Gammaitoni L, Sapino A, Pecchioni C, Moggio A, Giordano S, Aglietta
M and Montemurro F: HER2-positive breast cancer cells resistant to
trastuzumab and lapatinib lose reliance upon HER2 and are sensitive
to the multitargeted kinase inhibitor sorafenib. Breast Cancer Res
Treat. 130:29–40. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ramanathan RK, Hwang JJ, Zamboni WC,
Sinicrope FA, Safran H, Wong MK, Earle M, Brufsky A, Evans T,
Troetschel M, Walko C, Day R, Chen HX and Finkelstein S: Low
overexpression of HER-2/neu in advanced colorectal cancer limits
the usefulness of trastuzumab (Herceptin) and irinotecan as
therapy. A phase II trial Cancer Invest. 22:858–865. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Pietras RJ, Fendly BM, Chazin VR, Pegram
MD, Howell SB and Slamon DJ: Antibody to HER-2/neu receptor blocks
DNA repair after cisplatin in human breast and ovarian cells.
Oncogene. 9:1829–1838. 1994.PubMed/NCBI
|
|
34
|
Ritter CA, Bianco R, Dugger T, Forbes J,
Qu S, Rinehart C, King W and Arteaga CL: Mechanisms of resistance
development against trastuzumab (Herceptin) in an in vivo breast
cancer model. Int J Clin Pharmacol Ther. 42:642–643. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
O’Brien NA, Browne BC, Chow L, Wang Y,
Ginther C, Arboleda J, Duffy MJ, Crown J, O’Donovan N and Slamon
DJ: Activated phosphoinositide 3-kinase/AKT signaling confers
resistance to trastuzumab but not lapatinib. Mol Cancer Ther.
9:1489–1502. 2010.PubMed/NCBI
|
|
36
|
Scheuer W, Freiss T, Burtscher H,
Bossenmaier B, Endl J and Hasmann M: Strongly enhanced antitumor
activity of trastuzumab and pertuzumab combination treatment on
HER2-positive human xenograft tumor models. Cancer Res.
69:9330–9336. 2009. View Article : Google Scholar
|
|
37
|
Vogel CL, Cobleigh MA, Tripathy D, Gutheil
JC, Harris LN, Fehrenbacher L, Slamon DJ, Murphy M, Novotny WF,
Burchmore M, Shak S, Stewart SJ and Press M: Efficacy and safety of
trastuzumab as a single agent in first-line treatment of
HER2-overexpressing metastatic breast cancer. J Clin Oncol.
20:719–726. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Reim F, Dombrowski Y, Ritter C, Buttmann
M, Häusler S, Ossadnik M, Krockenberger M, Beier D, Beier CP, Dietl
J, Becker JC, Hönig A and Wischhusen J: Immunoselection of breast
and ovarian cancer cells with trastuzumab and natural killer cells:
selective escape of
CD44high/CD24low/HER2low breast
cancer stem cells. Cancer Res. 69:8058–8066. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Packer LM, Williams SJ, Callaghan S,
Gotley DC and McGuckin MA: Expression of the cell surface mucin
gene family in adenocarcinomas. Int J Oncol. 25:1119–1126.
2004.PubMed/NCBI
|
|
40
|
Scibetta AG, Albanese I, Morris J, Cooper
L, Downward J, Rowe P and Taylor-Papadimitriou J: Regulation of
MUC1 expression in human mammary cell lines by the c-ErbB2 and ras
signaling pathways. DNA Cell Biol. 20:265–274. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Hollingsworth MA and Swanson BJ: Mucins in
cancer: protection and control of the cell surface. Nat Rev Cancer.
4:45–60. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Li Y, Kuwahara H, Ren J, Wen G and Kufe D:
The c-Src tyrosine kinase regulates signaling of the human DF3/MUC1
carcinoma-associated antigen with GSK3 beta and beta-catenin. J
Biol Chem. 276:6061–6064. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Baselga J, Gelmon KA, Verma S, Wardley A,
Conte P, Miles D, Bianchi G, Cortes J, McNally VA, Ross GA,
Fumoleau P and Gianni L: Phase II trial of pertuzumab and
trastuzumab in patients with human epidermal growth factor receptor
2-positive metastatic breast cancer that progressed during prior
trastuzumab therapy. J Clin Oncol. 28:1138–1144. 2010. View Article : Google Scholar
|
|
44
|
Krop IE, LoRusso O, Miller KD, Modi S,
Yardley D, Rodriguez G, Lu M, Burrington B, Agresta S and Rugo H: A
phase 2 study of the HER2 antibody-drug conjugate trastuzumab-DM1
(TDM-1) in patients (PTS) with HER2-positive metastatic breast
cancer (MBC) previously treated with trastuzumab, lapatinib, and
chemotherapy [abstract 2770]. Ann Oncol. 21(Suppl 8):
viii972010.
|
|
45
|
Burris HA III, Rugo HS, Vukelja SJ, Vogel
CL, Borson RA, Limentani S, Tan-Chiu E, Krop IE, Michaelson RA,
Girish S, Amler L, Zheng M, Chu YW, Klencke B and O’Shaughnessy JA:
Phase II study of the antibody drug conjugate trastuzumab-DM1 for
the treatment of human epidermal growth factor receptor 2
(HER2)-positive breast cancer after prior HER2-directed therapy. J
Clin Oncol. 29:398–405. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Konecny GE, Pegram MD, Venkatesan N, Finn
R, Yang G, Rahmeh M, Untch M, Rusnak DW, Spehar G, Mullin RJ, Keith
BR, Gilmer TM, Berger M, Podratz KC and Slamon DJ: Activity of the
dual kinase inhibitor lapatinib (GW572016) against
HER-2-overexpressing and trastuzumab-treated breast cancer cells.
Cancer Res. 66:1630–1639. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Geyer CE, Forster J, Lindquist D, Chan S,
Romieu CG, Pienkowski T, Jagiello-Gruszfeld A, Crown J, Chan A,
Kaufman B, Skarlos D, Campone M, Davidson N, Berger M, Oliva C,
Rubin SD, Stein S and Cameron D: Lapatinib plus capecitabine for
HER2-positive advanced breast cancer. N Engl J Med. 355:2733–2743.
2006. View Article : Google Scholar : PubMed/NCBI
|




















