Introduction
Epilepsy is the most common serious neurological
disorder worldwide (1). It is
characterized by recurring and unprovoked seizures with a
prevalence of 1% and a lifetime incidence of 3% (2). The high drug-resistance rate
(3) in epilepsy creates a great
financial burden for society, hence there has been much research
devoted to the study of the mechanisms of epilepsy (4). Epilepsy is associated with brain
neurons which are activated upon initiation of neuronal damage
cascade reactions (5,6). There have been numerous advances in
the treatment of epilepsy, including the generation of drug
treatments (7), an increasing
number of investigations (8) and
the establishment of novel surgical approaches (9). The specific causative factors and
detailed mechanisms of epilepsy remain unclear.
Micro RNAs (miRNA) are a group of non-coding RNAs
(20–30 nucleotides in length) that regulate the expression of
target genes by binding to the 3′-untranslated regions of target
messenger (m)RNAs (10,11). The majority of mRNAs are regulated
by miRNAs, and each miRNA can target hundreds of genes (12). A previous study indicated that 60%
of all miRNA species are expressed in the brain (13). Thus, miRNAs may be involved in
various biological processes associated with brain funciton,
including learning, memory, neurological diseases (14) and neuroprotection (15).
A study on the metabolic pathways affected by
epileptic processes demonstrated that miRNAs are particularly
important in epigenetic mechanisms (16). A dysfunction of processing,
variable expression levels and alterations in the expression of
individual miRNAs were observed in the temporal lobe of patients
with epilepsy together with hippocampal sclerosis (17). Bot et al (18) measured the miRNA levels in the
dentate gyrus in epileptic rats and suggested that miRNAs can
participate in several molecular events that occur in epileptic
tissue, including the immune response and neuronal plasticity. It
was hypothesized that complex changes in the expression levels of
miRNAs suggest an important role for miRNAs in the molecular
mechanisms of epilepsy. However, data regarding the role of miRNAs
in epilepsy are limited.
In the current study, a bioinformatic-based analysis
was performed on the miRNA expression profiles in rat models of
epilepsy, with the aim to reveal the potential genes or pathways
associated with epilepsy.
Materials and methods
miRNA array data
The expression profile under the accession number
GSE49850 (18) was downloaded from
the public functional genomics data repository, Gene Expression
Omnibus, which was based on the GPL17566 platform (miRCURY LNA
microRNA array; Exiqon, Woburn, MA, USA). A total of 686 miRNAs in
rats were analyzed in GSE49850. Data sets consisted of miRNA
expression levels at 7, 14, 30 and 90 days subsequent to electrical
stimulation of the amygdala, which was used as a model of temporal
lobe epilepsy. The sham-operated time-matched control group (N) and
the stimulated group (C) were included at each time point with 5
replicates of each.
Differentially expressed miRNA
screening
The R statistical software package was used to
analyze the gene expression profiles. The CEL source files were
processed into expression estimates and a background correction was
performed with the normexp method and quartile data normalization
using the Robust Multiarray Average algorithm (19). The probability of genes being
differentially expressed between human degenerative disc disease
samples and normal samples was computed using the limma package
(20). The TimeCourse software
(21,22) was used to analyze the time-course
microarray data and differences in four time points in the N and C
groups. Hotelling’s T2 test was used to identify
differentially expressed genes. P<0.01 was considered to
indicate statistically significant data.
Expression pattern analysis
The mfuzz package (23) in Bioconductor was used for miRNA
cluster analyses in the C and N groups at four time points. Cluster
analysis of the differential expression values in the 40 samples
was performed with a Euclidean distance, using Cluster 3.0 software
(24).
Predication and enrichment of target
genes
The names of differentially expressed miRNAs were
matched to the miRBase (25) to
identify the accession IDs. The target genes associated with miRNAs
were then selected using the miRWalk database (26).
The enrichment of all target genes was conducted
using the Database for Annotation, Visualization and Integrated
Discovery (DAVID) software (27,28).
This program has been widely used to investigate the bio-functions
of genes based on the gene ontology (GO) and the Kyoto
Encyclopaedia of Genes and Genomes (KEGG) databases. In the present
study, DAVID was used to perform the GO and KEGG pathway enrichment
analysis. The biological process, molecular function and cellular
components were included in the enrichment analysis. In each
category, there were at least 5 genes with P<0.01.
Construction of target gene network
Genes and proteins associated with the target genes
were screened in the BIND (29)
database with the BisoGenet (30)
package from the Cytoscape (31)
software to obtain the interaction network. Additionally,
ClusterONE (32) in Cytoscape was
used for the cluster analysis. P<0.05 was considered to indicate
statistically significant data.
Results
Differentially expressed miRNAs in the N
and C groups
The microarray data were processed with background
correction and standardization to obtain the differentially
expressed miRNAs. At a P-value of <0.01, a total of 152
differentially expressed miRNAs (including 144 up- and 8
downregulated) were obtained with Hotelling’s T2 test.
The top 10 most significantly differentially expressed miRNAs are
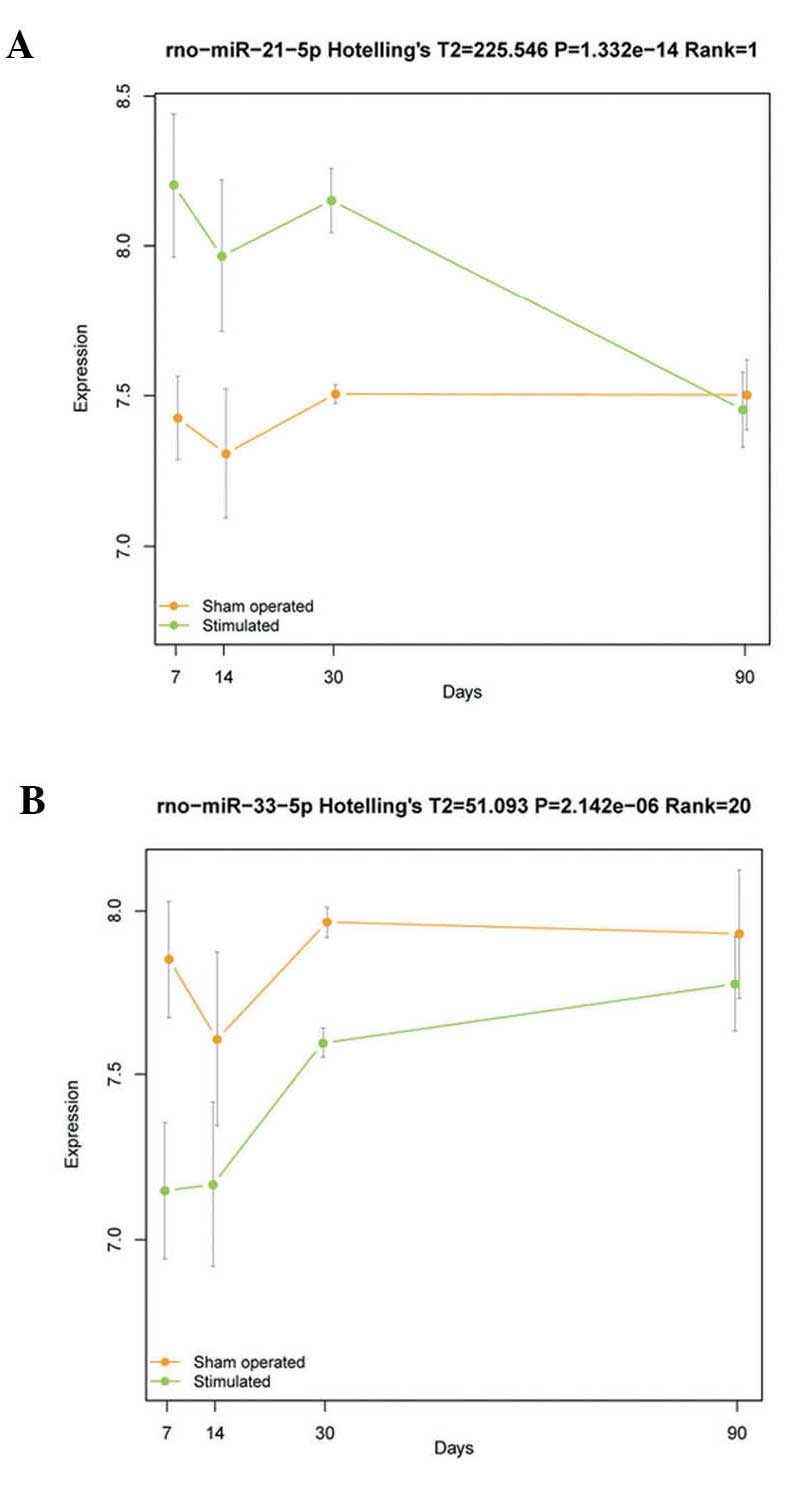
listed in Table I. The
rno-miR-21-5p and rno-miR-33-5p were the most significantly up- and
downregulated differentially expressed miRNAs, respectively
(Fig. 1).
 | Table ITop 10 most significantly
differentially expressed microRNAs. |
Table I
Top 10 most significantly
differentially expressed microRNAs.
| microRNA | Hotelling’s
T2 | P-value |
|---|
| rno-miR-21-5p | 225.54606 | 1.332268 ×
10−14 |
| rno-miR-218a-5p | 215.12361 | 2.697842 ×
10−14 |
| rno-miR-370-5p | 176.86949 | 4.736211 ×
10−13 |
| rno-miR-132-5p | 133.86876 | 2.341205 ×
10−11 |
| rno-miR-212-3p | 101.48246 | 8.938348 ×
10−10 |
| rno-miR-212-5p | 96.00433 | 1.796583 ×
10−9 |
| rno-miR-298-5p | 89.53393 | 4.253404 ×
10−9 |
| rno-miR-352 | 85.14339 | 7.825937 ×
10−9 |
| rno-miR-363-5p | 80.30582 | 1.571388 ×
10−8 |
| rno-miR-124-3p | 74.28944 | 3.892660 ×
10−8 |
Cluster analysis
The expression patterns of 152 differentially
expressed miRNA in 40 samples exhibited a significant distribution
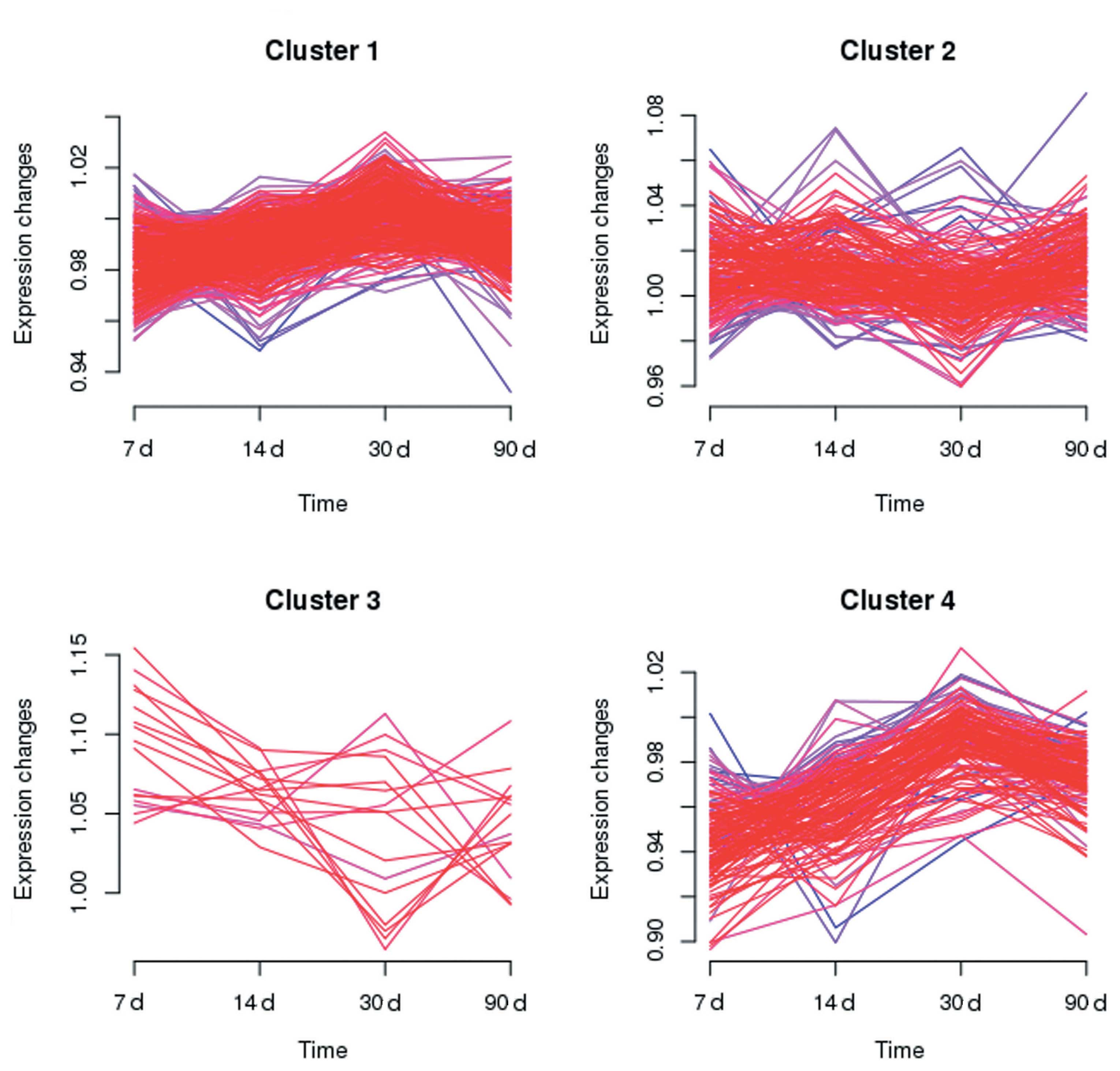
character (Fig 2). Multiple
clustering between the N and C groups was carried out with mfuzz at
four time points (Fig. 3).
Clusters 1 and 4 represent slow and rapid increases in the
expression, respectively; whilst clusters 2 and 3 represent slow
and rapid reductions in the expression, respectively.
Target gene analysis
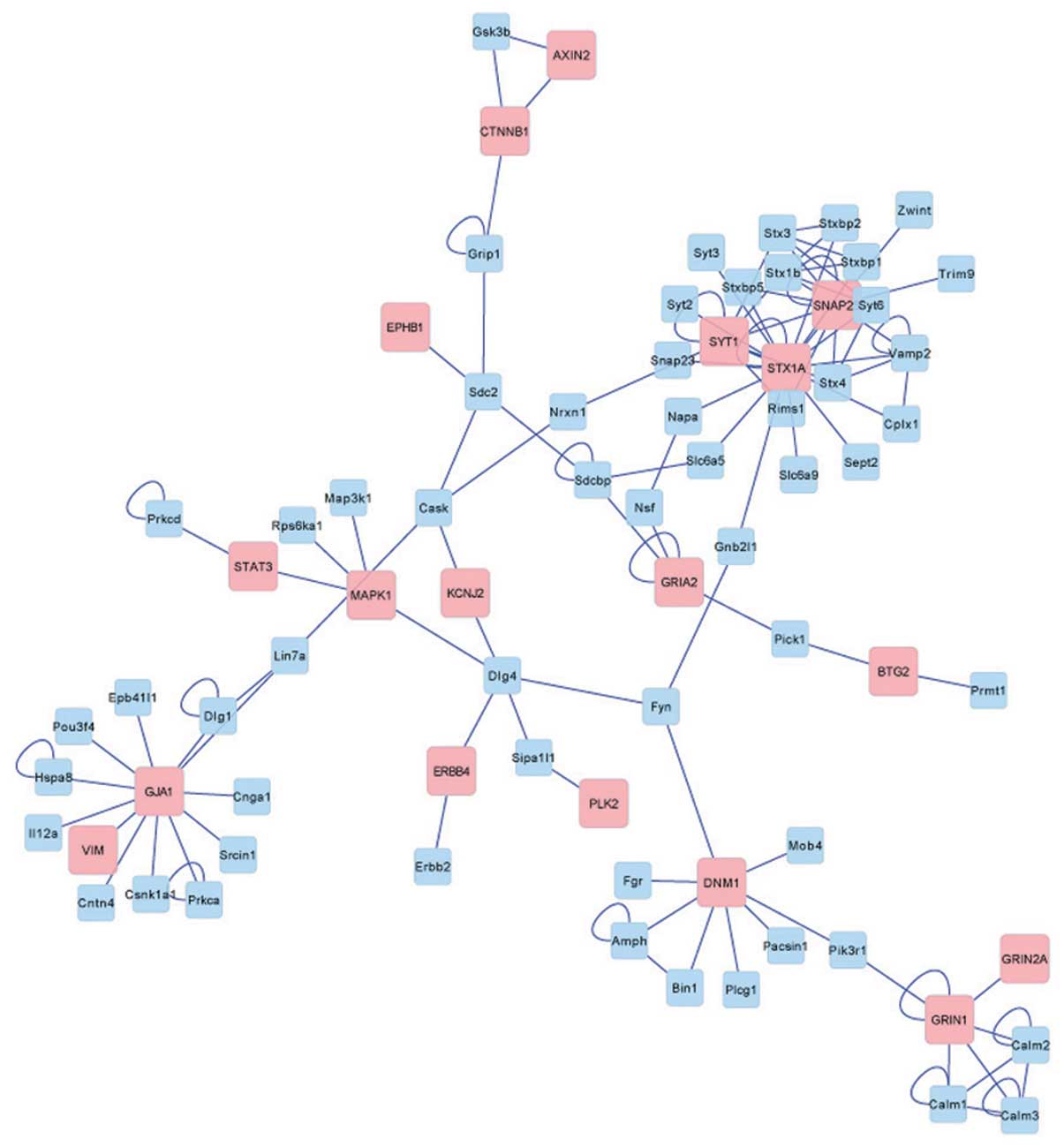
Following removal of the duplicate genes, 526 target
genes associated with miRNA were identified. The interaction
network (Fig. 4) was constructed
when these target genes were matched to the BIND database. The
result of the network construction revealed that a total of 18
target genes, including GRIN1, GRIN2A, STX3, STX1A, STX1B, MAPK1
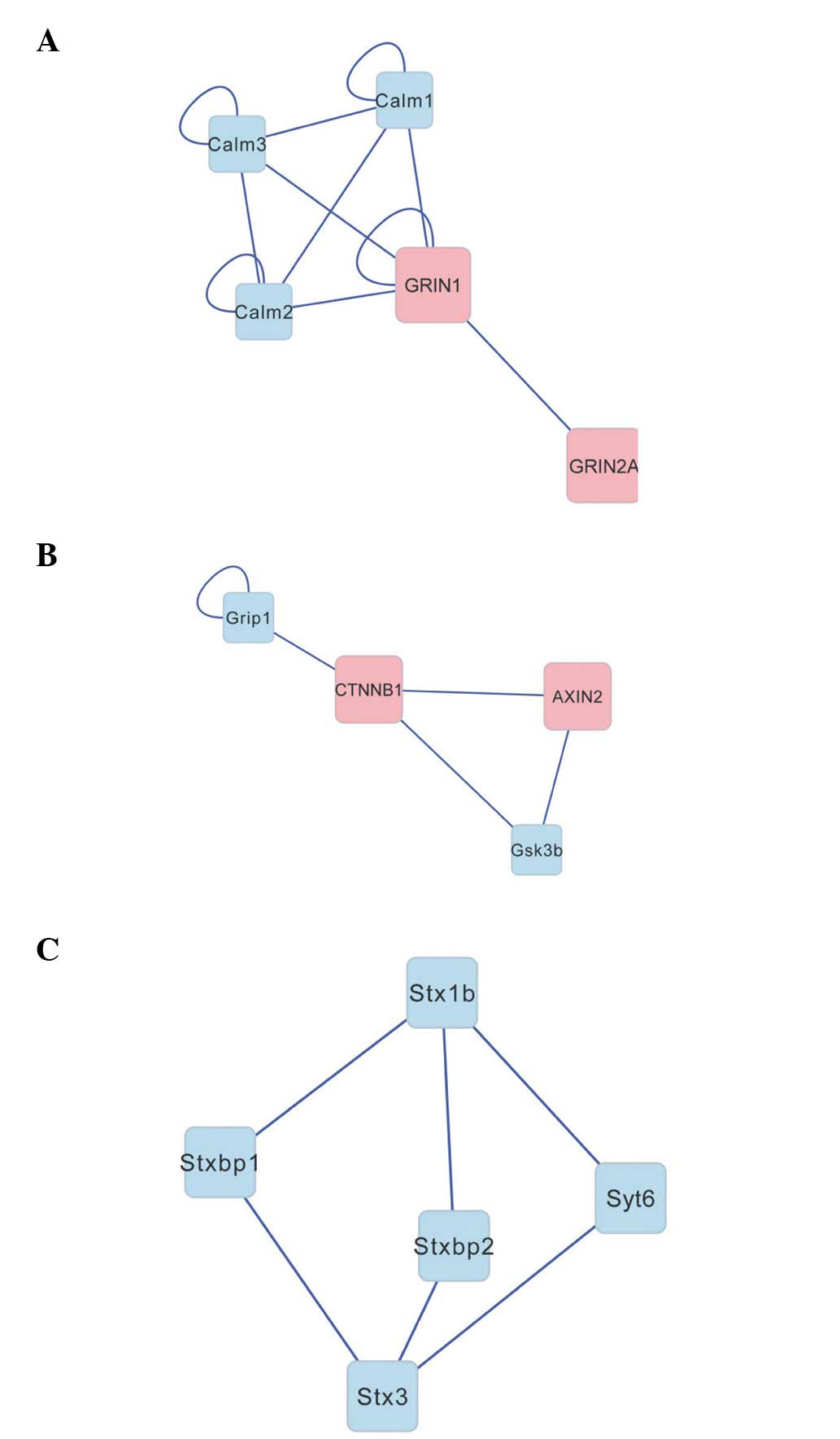
and Calm 1–3, were included in the network. The cluster analysis by
ClusterONE software revealed three significant categories (Fig. 5), including: GRIN and Calm
(density=0.7, quality=0.875, P=0.007); CTNNB1 and AXIN2
(density=0.667, quality=0.8, P=0.019); and the STX family
(density=0.6, quality=0.429, P=0.049).
GO terms and KEGG pathway enrichment analysis
indicated that there were 30 KEGG pathways and 630 GO terms in 113
types of cluster categories, including external stimuli response,
gene transcription regulation and steroid response. The 3 most
significantly enriched terms in each category are described in
Table II.
 | Table IITop 3 most significant Gene Onology
terms and KEGG pathway enrichment. |
Table II
Top 3 most significant Gene Onology
terms and KEGG pathway enrichment.
| Category | Term | Count | Fold
enrichment | FDR |
|---|
| Biological
process | GO:0010033~response
to organic substance | 135 | 4.238731 | 1.45E-47 |
| GO:0009719~response
to endogenous stimulus | 93 | 4.729099 | 2.14E-34 |
| GO:0009725~response
to hormone stimulus | 85 | 4.856225 | 7.51E-32 |
| Cellular
component |
GO:0031974~membrane-enclosed lumen | 96 | 2.246048 | 1.55E-11 |
|
GO:0043233~organelle lumen | 93 | 2.236448 | 6.30E-11 |
|
GO:0044421~extracellular region part | 64 | 2.711679 | 4.82E-10 |
| Molecular
function | GO:0046983~protein
dimerization activity | 69 | 3.918162 | 1.51E-19 |
|
GO:0003700~transcription factor
activity | 73 | 3.398632 | 2.97E-17 |
| GO:0019899~enzyme
binding | 57 | 3.473577 | 5.11E-13 |
| KEGG pathway | rno05210:Colorectal
cancer | 23 | 6.0353 | 8.78E-09 |
| rno05220:Chronic
myeloid leukemia | 20 | 5.667934 | 9.28E-07 |
| rno05215:Prostate
cancer | 20 | 4.723278 | 2.46E-05 |
Discussion
Epilepsy is the most common serious neurological
disorder worldwide (1), and the
pathogenesis of the disease remains unclear. A bioinformatics
analysis based on the expression profiles of miRNAs in epilepsy was
performed to reveal the potential genes or pathways associated with
epilepsy. The results of the present study demonstrated a total of
152 differentially expressed miRNAs, and 526 target genes of the
differentially expressed miRNAs. Functional analysis indicated that
these genes were largely involved in stimulus responses. The
interaction network clarified that the GRIN and STX gene family,
which are associated with synaptic signal transmission, had a
significant interaction.
As important biological indicators, the expression
of miRNAs has been proved to be strongly associated with various
diseases, including brain tumors (33), breast cancer (34), lung tumor (35) and Alzheimer’s disease (36). In studies of epilepsy, the
association between the expression of miRNAs and the processes of
the disease have been demonstrated in humans (17) and animal models (18). In previous pilocarpine-induced
status epilepticus studies in rat models, authors have theorized
that the majority of miRNAs were downregulated following
pilocarpine application (37),
while other studies indicated that the majority of miRNAs were
upregulated (38). In the present
study, the majority of the 152 differentially expressed miRNAs were
upregulated, including miR-21. miRNA-21 is able to mediate tumor
growth (39). A number of previous
studies have indicated that the downregulation of miRNA-21 is
strongly associated with the processes of tumors or cancer
(40–42). However, miRNA-21 was significantly
upregulated in the present study as a differentially expressed
miRNA. This result indicates that the upregulation of miRNA-21 may
be involved in the initiation of the cell signaling pathways
associated with epilepsy, which is supported by a study by Marquez
et al (43). Furthermore,
the expression of target genes associated with these differentially
expressed miRNAs were mostly downregulated. Bot et al
(18) indicated that the target
genes may be involved in several biological functions, including
regulation of transcription, wound response, apoptosis, cell
proliferation and immune response. The enrichment analysis of
target genes in the present study demonstrated that their function
is to respond to stimuli such as macromolecular substances and
hormones. With the downregulation of target genes, the external
stimuli response for cells is weakened. Thus, the signal response
pathways in these cells may be damaged. This may be one reason for
the neuronal dysregulation observed in the epileptic brain
(44).
In the present study, the cluster analysis of the
target genes in the network construction revealed three significant
categories including the GRIN, Calm and STX families.
N-methyl-D-aspartate (NMDA) receptors are vital for healthy brain
development (45). The NMDA-type
glutamate receptors GRIN1 and GRIN2A, are two members of the GRIN
family. Mutations in GRIN2A can induce idiopathic focal epilepsy
(46), while GRIN1 has been
associated with schizophrenia susceptibility (47). Calm1–3 are three members of the
calmodulin family (48) that are
associated with calcium signaling receptor activation. The
sialyltransferase family (STX) (49) consists of various members such as
STX3, STX1A and STX1B. These influencial factors, in particular
STX1A, are involved in the regulation of synaptic vesicles, which
are used to store neurotransmitters. As a target gene for
differentially expressed miRNA, STX1A is a key regulator of ion
channels of the nervous system, although it was not included in the
cluster categories in the present study.
The results of the interaction network for these
target genes indicated that signal transduction of normal nerve
cells suffers significant interference in the process of epilepsy,
thus the nerve cells may not function properly, leading to the
occurrence of disease. Understanding the mechanisms of the
differential expression of miRNAs in epilepsy is crucial for
understanding the pathophysiology of epilepsy and may constitute
interesting candidate targets for therapy. However, to understand
the mechanism of the different miRNAs on the function of epilepsy
requires in-depth knowledge of the targets of each miRNA, and a
larger scale of miRNA-target gene screening. In the current study,
the targets of the miRNAs were unknown and require further
investigation.
In conclusion, the present study, based on miRNA
expression profiles, demonstrated that the development of epilepsy
is closely associated with the neuronal calcium signaling pathways
involving the GRIN and STX families. The results suggested an
important function for miRNAs in molecular mechanisms of epilepsy,
and provided a potential direction for future research into the
disease, and its treatments.
Acknowledgements
This study was supported by the National Natural
Science Foundation of China (grant no. 81301116).
References
|
1
|
Brodie MJ, Schachter SC and Kwan P: Fast
Facts: Epilepsy. 5th edition. Health Press Limited; Oxford, UK:
2012
|
|
2
|
Banerjee PN and Hauser WA: Incidence and
prevalence. Epilepsy: A Comprehensive Textbook. Engel J and Pedley
TA: Lippincott Williams & Wilkins; Philadelphia, PA: pp. 45–56.
2008
|
|
3
|
Sisodiya S: Etiology and management of
refractory epilepsies. Nat Clin Pract Neurol. 3:320–330. 2007.
View Article : Google Scholar
|
|
4
|
Engelborghs S, D’Hooge R and De Deyn PP:
Pathophysiology of epilepsy. Acta Neurol Belg. 100:201–213.
2000.
|
|
5
|
Pitkänen A and Lukasiuk K: Molecular and
cellular basis of epileptogenesis in symptomatic epilepsy. Epilepsy
Behav. 14(Suppl 1): 16–25. 2009.PubMed/NCBI
|
|
6
|
Pitkänen A and Lukasiuk K: Mechanisms of
epileptogenesis and potential treatment targets. Lancet Neurol.
10:173–186. 2011.PubMed/NCBI
|
|
7
|
Löscher W, Klitgaard H, Twyman RE and
Schmidt D: New avenues for anti-epileptic drug discovery and
development. Nat Rev Drug Discov. 12:757–776. 2013.PubMed/NCBI
|
|
8
|
Sørensen AT and Kokaia M: Novel approaches
to epilepsy treatment. Epilepsia. 54:1–10. 2013.
|
|
9
|
Binder JR: Use of fMRI language
lateralization for quantitative prediction of naming and verbal
memory outcome in left temporal lobe epilepsy surgery. fMRI: Basics
and Clinical Aplications. Ulmer S and Jansen O: Springer; New York,
NY: pp. 77–93. 2010, View Article : Google Scholar
|
|
10
|
Carthew RW and Sontheimer EJ: Origins and
mechanisms of miRNAs and siRNAs. Cell. 136:642–655. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Croce CM and Calin GA: miRNAs, cancer, and
stem cell division. Cell. 122:6–7. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ebert MS and Sharp PA: Roles for microRNAs
in conferring robustness to biological processes. Cell.
149:515–524. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sempere LF, Freemantle S, Pitha-Rowe I,
Moss E, Dmitrovsky E and Ambros V: Expression profiling of
mammalian microRNAs uncovers a subset of brain-expressed microRNAs
with possible roles in murine and human neuronal differentiation.
Genome Biol. 5:R132004. View Article : Google Scholar
|
|
14
|
Wang W, Kwon EJ and Tsai LH: MicroRNAs in
learning, memory, and neurological diseases. Learn Mem. 19:359–368.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Saugstad JA: MicroRNAs as effectors of
brain function with roles in ischemia and injury, neuroprotection,
and neurodegeneration. J Cereb Blood Flow Metab. 30:1564–1576.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jimenez-Mateos EM and Henshall DC:
Epilepsy and microRNA. Neuroscience. 238:218–229. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mckiernan RC, Jimenez-Mateos EM, Bray I,
et al: Reduced mature microRNA levels in association with dicer
loss in human temporal lobe epilepsy with hippocampal sclerosis.
PLoS One. 7:e359212012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bot AM, Dębski KJ and Lukasiuk K:
Alterations in miRNA levels in the dentate gyrus in epileptic rats.
PloS One. 8:e760512013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Irizarry RA, Hobbs B, Collin F, et al:
Exploration, normalization, and summaries of high density
oligonucleotide array probe level data. Biostatistics. 4:249–264.
2003. View Article : Google Scholar
|
|
20
|
Diboun I, Wernisch L, Orengo CA and
Koltzenburg M: Microarray analysis after RNA amplification can
detect pronounced differences in gene expression using limma. BMC
Genomics. 7:2522006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gillespie CS, Lei G, Boys RJ, Greenall A
and Wilkinson DJ: Analysing time course microarray data using
Bioconductor: a case study using yeast2 Affymetrix arrays. BMC Res
Notes. 3:812010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tai Y: Timecourse: Statistical Analysis
for Developmental Microarray Time Course Data. R package version 1.
2007
|
|
23
|
Kumar L and E Futschik M: Mfuzz: a
software package for soft clustering of microarray data.
Bioinformation. 2:5–7. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
de Hoon MJ, Imoto S, Nolan J and Miyano S:
Open source clustering software. Bioinformatics. 20:1453–1454.
2004.PubMed/NCBI
|
|
25
|
Kozomara A and Griffiths-Jones S: miRBase:
integrating microRNA annotation and deep-sequencing data. Nucleic
Acids Res. 39(Database issue): D152–D157. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Dweep H, Sticht C, Pandey P and Gretz N:
miRWalk - database: prediction of possible miRNA binding sites by
‘walking’ the genes of three genomes. J Biomed Inform. 44:839–847.
2011.
|
|
27
|
Huang da W, Sherman BT and Lempicki RA:
Systematic and integrative analysis of large gene lists using DAVID
bioinformatics resources. Nat Protoc. 4:44–57. 2009.PubMed/NCBI
|
|
28
|
Huang da W, Sherman BT and Lempicki RA:
Bioinformatics enrichment tools: paths toward the comprehensive
functional analysis of large gene lists. Nucleic Acids Res.
37:1–13. 2009.PubMed/NCBI
|
|
29
|
Bader GD, Donaldson I, Wolting C,
Ouellette BF, Pawson T and Hogue CW: BIND - the Biomolecular
Interaction Network Database. Nucleic Acids Res. 29:242–245. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Martin A, Ochagavia ME, Rabasa LC, Miranda
J, Fernandez-de-Cossio J and Bringas R: BisoGenet: a new tool for
gene network building, visualization and analysis. BMC
bioinformatics. 11:912010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Shannon P, Markiel A, Ozier O, et al:
Cytoscape: a software environment for integrated models of
biomolecular interaction networks. Genome Res. 13:2498–2504. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Nepusz T, Yu H and Paccanaro A: Detecting
overlapping protein complexes in protein-protein interaction
networks. Nat Methods. 9:471–472. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
James CD: Aberrant miRNA expression in
brain tumors: a subject attracting an increasing amount of
attention. Neuro Oncol. 15:4052013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kong W, He L, Richards E, et al:
Upregulation of miRNA-155 promotes tumour angiogenesis by targeting
VHL and is associated with poor prognosis and triple-negative
breast cancer. Oncogene. 33:679–689. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Peng Y, Dai Y, Hitchcock C, et al: Insulin
growth factor signaling is regulated by microRNA-486, an
underexpressed microRNA in lung cancer. Proc Natl Acad Sci USA.
110:15043–15048. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Bekris LM, Lutz F, Montine TJ, et al:
MicroRNA in Alzheimer’s disease: an exploratory study in brain,
cerebrospinal fluid and plasma. Biomarkers. 18:455–466. 2013.
|
|
37
|
Hu K, Xie YY, Zhang C, et al: MicroRNA
expression profile of the hippocampus in a rat model of temporal
lobe epilepsy and miR-34a-targeted neuroprotection against
hippocampal neurone cell apoptosis post-status epilepticus. BMC
Neurosci. 13:1152012. View Article : Google Scholar
|
|
38
|
Song YJ, Tian XB, Zhang S, et al: Temporal
lobe epilepsy induces differential expression of hippocampal miRNAs
including let-7e and miR-23a/b. Brain Res. 1387:134–140. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Si ML, Zhu S, Wu H, Lu Z, Wu F and Mo YY:
miR-21-mediated tumor growth. Oncogene. 26:2799–2803. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Asangani IA, Rasheed SA, Nikolova DA, et
al: MicroRNA-21 (miR-21) post-transcriptionally downregulates tumor
suppressor Pdcd4 and stimulates invasion, intravasation and
metastasis in colorectal cancer. Oncogene. 27:2128–2136. 2008.
View Article : Google Scholar
|
|
41
|
Yao Q, Xu H, Zhang QQ, Zhou H and Qu LH:
MicroRNA-21 promotes cell proliferation and down-regulates the
expression of programmed cell death 4 (PDCD4) in HeLa cervical
carcinoma cells. Biochem Biophys Res Commun. 388:539–542. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Su H, Yang JR, Xu T, et al: MicroRNA-101,
down-regulated in hepatocellular carcinoma, promotes apoptosis and
suppresses tumorigenicity. Cancer Res. 69:1135–1142. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Marquez RT, Wendlandt E, Galle CS, Keck K
and Mccaffrey AP: MicroRNA-21 is upregulated during the
proliferative phase of liver regeneration, targets Pellino-1 and
inhibits NF-kappaB signaling. Am J Physiol Gastrointest Liver
Physiol. 298:G535–G541. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Li MM, Li XM, Zheng XP, Yu JT and Tan L:
MicroRNAs dysregulation in epilepsy. Brain Res. Oct 3–2013.(Epub
ahead of print).
|
|
45
|
Barkus C, McHugh SB, Sprengel R, Seeburg
PH, Rawlins JN and Bannerman DM: Hippocampal NMDA receptors and
anxiety: at the interface between cognition and emotion. Eur J
Pharmacol. 626:49–56. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Lemke JR, Lal D, Reinthaler EM, et al:
Mutations in GRIN2A cause idiopathic focal epilepsy with rolandic
spikes. Nat Genet. 45:1067–1072. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
47
|
Saadat M: N-methyl-D-aspartate receptor
NR1 subunit gene (GRIN1) G1001C polymorphism and susceptibility to
schizophrenia: a meta-analysis. EXCLI J. 9:11–16. 2010.
|
|
48
|
Wylie DC and Vanaman TC: Structure and
evolution of the calmodulin family of calcium regulatory proteins.
Calmodulin. Cohen P and Klee CB: Elsevier; New York, NY: pp. 1–15.
1988
|
|
49
|
Harduin-Lepers A, Vallejo-Ruiz V,
Krzewinski-Recchi MA, Samyn-Petit B, Julien S and Delannoy P: The
human sialyltransferase family. Biochimie. 83:727–737. 2001.
View Article : Google Scholar : PubMed/NCBI
|