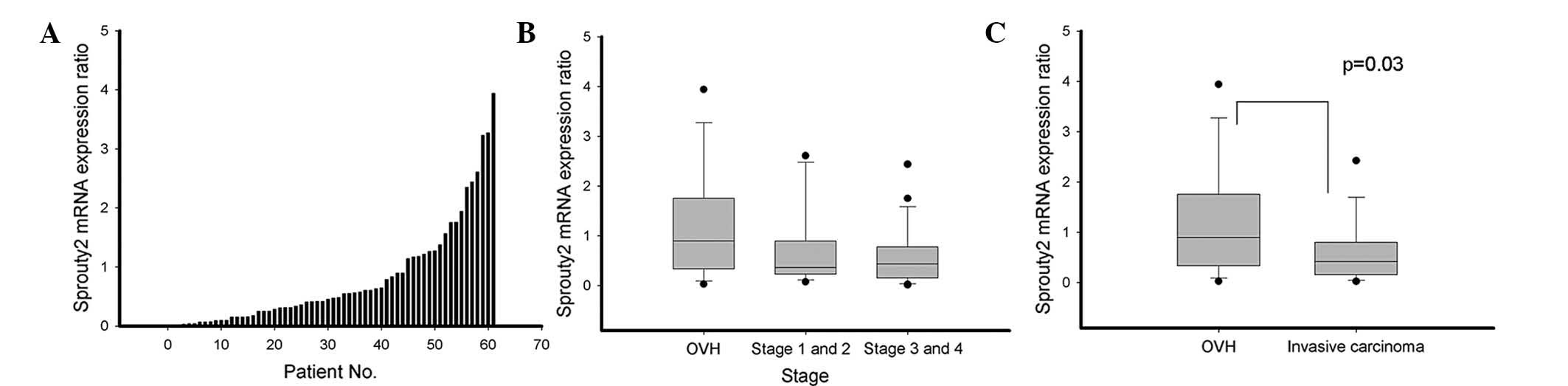
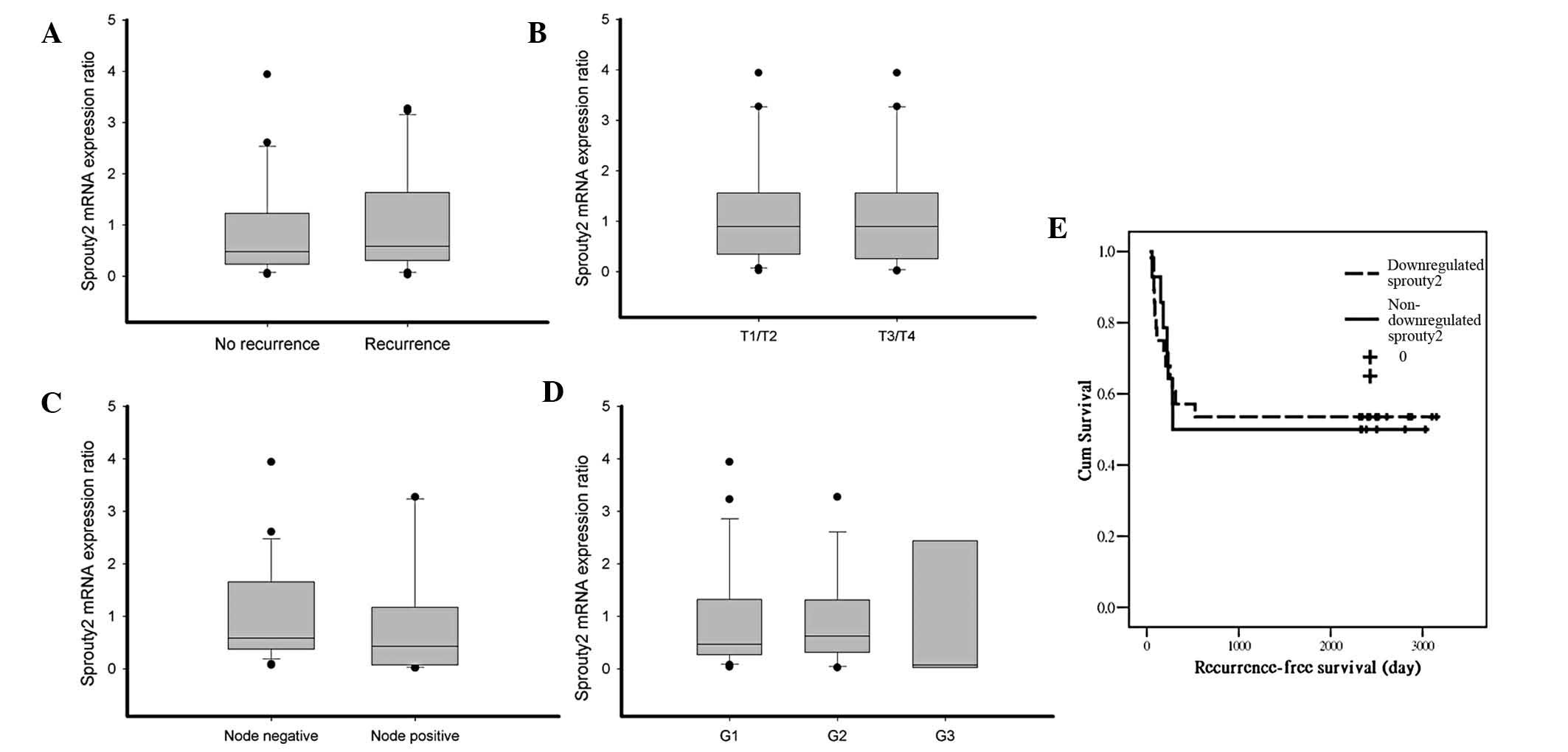
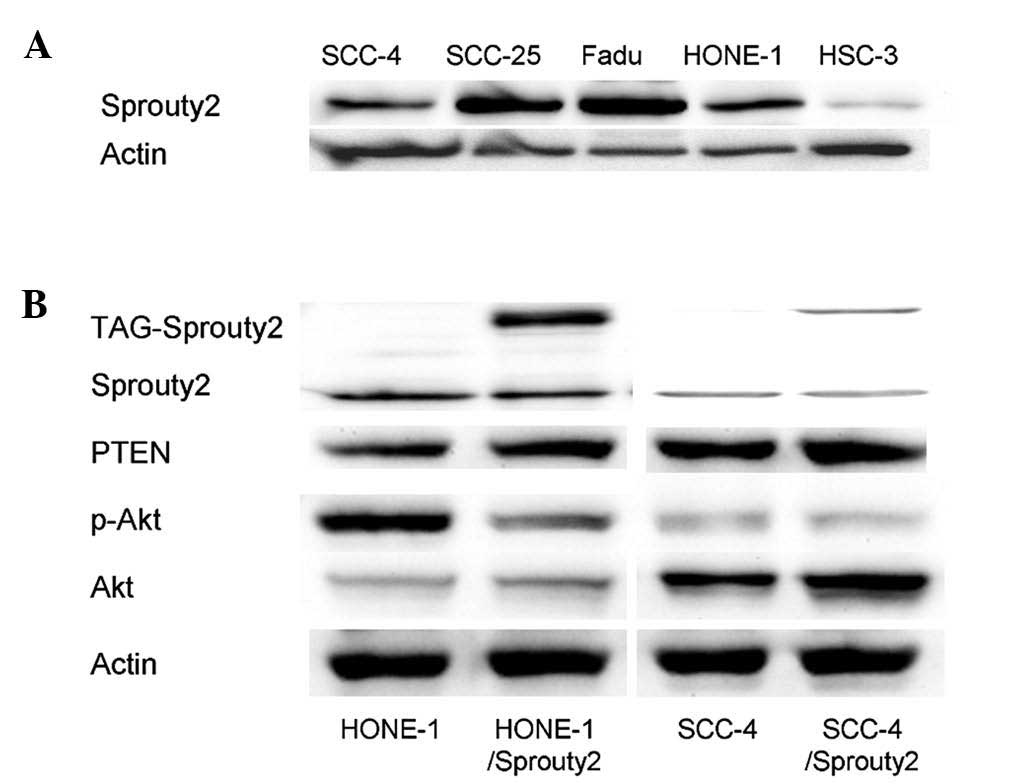
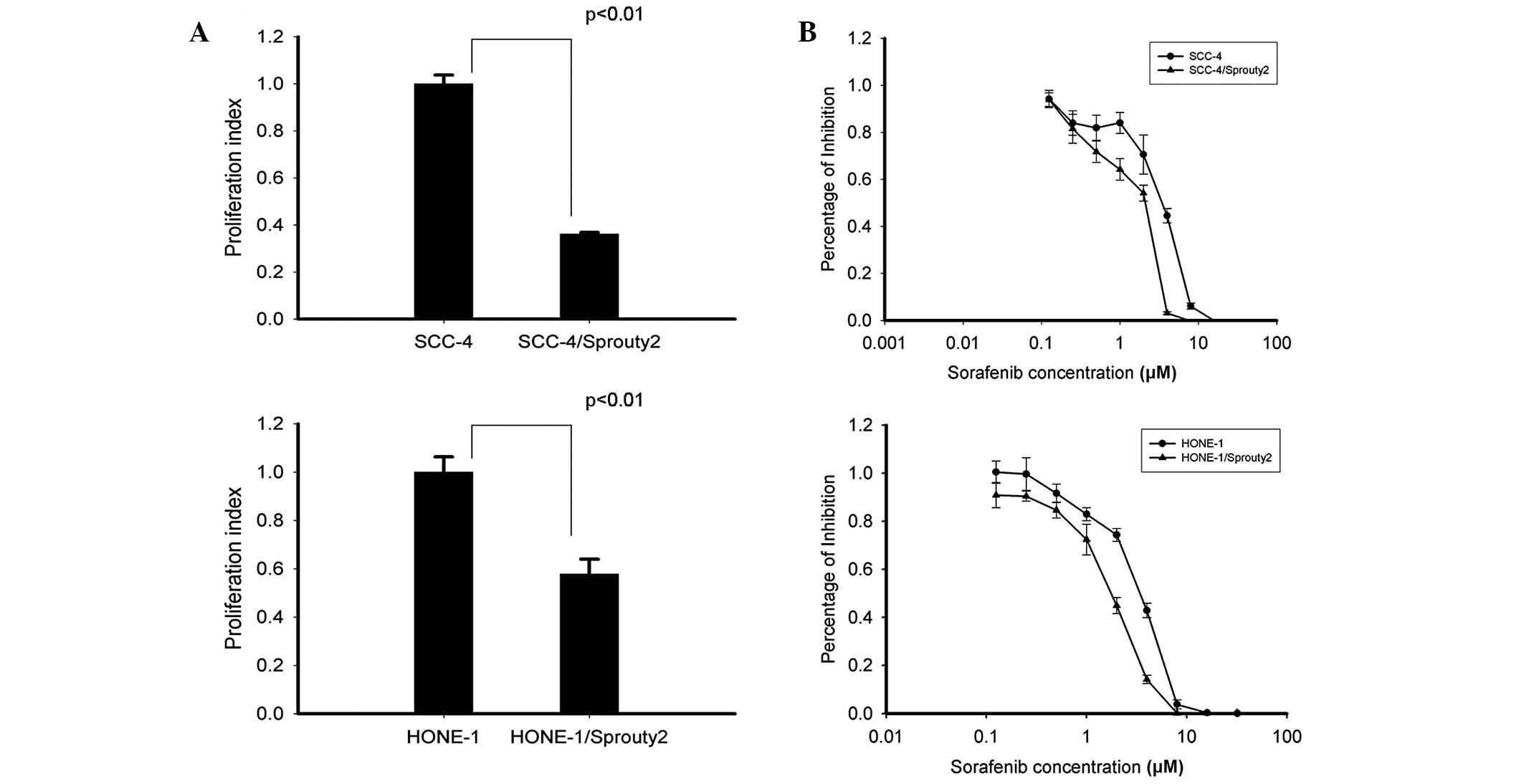
|
1
|
Jemal A, Siegel R, Ward E, et al: Cancer
statistics, 2008. CA Cancer J Clin. 58:71–96. 2008. View Article : Google Scholar
|
|
2
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar
|
|
3
|
Ko YC, Huang YL, Lee CH, Chen MJ, Lin LM
and Tsai CC: Betel quid chewing, cigarette smoking and alcohol
consumption related to oral cancer in Taiwan. J Oral Pathol Med.
24:450–453. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Stransky N, Egloff AM, Tward AD, et al:
The mutational landscape of head and neck squamous cell carcinoma.
Science. 333:1157–1160. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ang KK, Berkey BA, Tu X, et al: Impact of
epidermal growth factor receptor expression on survival and pattern
of relapse in patients with advanced head and neck carcinoma.
Cancer Res. 62:7350–7356. 2002.PubMed/NCBI
|
|
6
|
Rubin Grandis J, Melhem MF, Gooding WE, et
al: Levels of TGF-alpha and EGFR protein in head and neck squamous
cell carcinoma and patient survival. J Natl Cancer Inst.
90:824–832. 1998.
|
|
7
|
Kalyankrishna S and Grandis JR: Epidermal
growth factor receptor biology in head and neck cancer. J Clin
Oncol. 24:2666–2672. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sharafinski ME, Ferris RL, Ferrone S and
Grandis JR: Epidermal growth factor receptor targeted therapy of
squamous cell carcinoma of the head and neck. Head Neck.
32:1412–1421. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lalla RV, Boisoneau DS, Spiro JD and
Kreutzer DL: Expression of vascular endothelial growth factor
receptors on tumor cells in head and neck squamous cell carcinoma.
Arch Otolaryngol Head Neck Surg. 129:882–888. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Neuchrist C, Erovic BM, Handisurya A, et
al: Vascular endothelial growth factor C and vascular endothelial
growth factor receptor 3 expression in squamous cell carcinomas of
the head and neck. Head Neck. 25:464–474. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hacohen N, Kramer S, Sutherland D, Hiromi
Y and Krasnow MA: Sprouty encodes a novel antagonist of FGF
signaling that patterns apical branching of the Drosophila
airways. Cell. 92:253–263. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tefft JD, Lee M, Smith S, et al: Conserved
function of mSpry-2, a murine homolog of Drosophila sprouty,
which negatively modulates respiratory organogenesis. Curr Biol.
9:219–222. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Minowada G, Jarvis LA, Chi CL, et al:
Vertebrate Sprouty genes are induced by FGF signaling and can cause
chondrodysplasia when overexpressed. Development. 126:4465–4475.
1999.PubMed/NCBI
|
|
14
|
Impagnatiello MA, Weitzer S, Gannon G,
Compagni A, Cotten M and Christofori G: Mammalian sprouty-1 and -2
are membrane-anchored phosphoprotein inhibitors of growth factor
signaling in endothelial cells. J Cell Biol. 152:1087–1098. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gross I, Bassit B, Benezra M and Licht JD:
Mammalian sprouty proteins inhibit cell growth and differentiation
by preventing ras activation. J Biol Chem. 276:46460–46468. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sutterlüty H, Mayer CE, Setinek U, et al:
Down-regulation of Sprouty2 in non-small cell lung cancer
contributes to tumor malignancy via extracellular signal-regulated
kinase pathway-dependent and -independent mechanisms. Mol Cancer
Res. 5:509–520. 2007.PubMed/NCBI
|
|
17
|
Fong CW, Chua MS, McKie AB, et al: Sprouty
2, an inhibitor of mitogen-activated protein kinase signaling, is
down-regulated in hepatocellular carcinoma. Cancer Res.
66:2048–2058. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Feng YH, Wu CL, Tsao CJ, et al:
Deregulated expression of sprouty2 and microRNA-21 in human colon
cancer: Correlation with the clinical stage of the disease. Cancer
Biol Ther. 11:111–121. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bancroft CC, Chen Z, Dong G, et al:
Coexpression of proangiogenic factors IL-8 and VEGF by human head
and neck squamous cell carcinoma involves coactivation by MEK-MAPK
and IKK-NF-kappaB signal pathways. Clin Cancer Res. 7:435–442.
2001.PubMed/NCBI
|
|
20
|
Mineta H, Miura K, Ogino T, et al:
Prognostic value of vascular endothelial growth factor (VEGF) in
head and neck squamous cell carcinomas. Br J Cancer. 83:775–781.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Williamson SK, Moon J, Huang CH,
Guaglianone PP, LeBlanc M, Wolf GT, et al: Phase II evaluation of
sorafenib in advanced and metastatic squamous cell carcinoma of the
head and neck: Southwest Oncology Group Study S0420. J Clin Oncol.
2010.28(20): 3330–5
|
|
22
|
Fritzsche S, Kenzelmann M, Hoffmann MJ, et
al: Concomitant down-regulation of SPRY1 and SPRY2 in prostate
carcinoma. Endocr Relat Cancer. 13:839–849. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Song K, Gao Q, Zhou J, et al: Prognostic
significance and clinical relevance of Sprouty 2 protein expression
in human hepatocellular carcinoma. Hepatobiliary Pancreat Dis Int.
11:177–184. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Edwin F, Singh R, Endersby R, Baker SJ and
Patel TB: The tumor suppressor PTEN is necessary for human Sprouty
2-mediated inhibition of cell proliferation. J Biol Chem.
281:4816–4822. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Edwin F, Anderson K, Ying C and Patel TB:
Intermolecular interactions of Sprouty proteins and their
implications in development and disease. Mol Pharmacol. 76:679–691.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lo TL, Yusoff P, Fong CW, et al: The
ras/mitogen-activated protein kinase pathway inhibitor and likely
tumor suppressor proteins, sprouty 1 and sprouty 2 are deregulated
in breast cancer. Cancer Res. 64:6127–6136. 2004. View Article : Google Scholar
|
|
27
|
Velasco A, Pallares J, Santacana M, et al:
Promoter hypermethylation and expression of sprouty 2 in
endometrial carcinoma. Hum Pathol. 42:185–193. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kwak HJ, Kim YJ, Chun KR, et al:
Downregulation of Spry2 by miR-21 triggers malignancy in human
gliomas. Oncogene. 30:2433–2442. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lee SA, Ho C, Roy R, et al: Integration of
genomic analysis and in vivo transfection to identify sprouty 2 as
a candidate tumor suppressor in liver cancer. Hepatology.
47:1200–1210. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lee SA, Ladu S, Evert M, et al:
Synergistic role of Sprouty2 inactivation and c-Met up-regulation
in mouse and human hepatocarcinogenesis. Hepatology. 52:506–517.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Frank MJ, Dawson DW, Bensinger SJ, et al:
Expression of sprouty2 inhibits B-cell proliferation and is
epigenetically silenced in mouse and human B-cell lymphomas. Blood.
113:2478–2487. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ishida M, Ichihara M, Mii S, et al:
Sprouty2 regulates growth and differentiation of human
neuroblastoma cells through RET tyrosine kinase. Cancer Sci.
98:815–821. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ma Y, Yu S, Zhao W, Lu Z and Chen J:
miR-27a regulates the growth, colony formation and migration of
pancreatic cancer cells by targeting Sprouty2. Cancer Lett.
298:150–158. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
McKie AB, Douglas DA, Olijslagers S, et
al: Epigenetic inactivation of the human sprouty2 (hSPRY2)
homologue in prostate cancer. Oncogene. 24:2166–2174. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Holgren C, Dougherty U, Edwin F, et al:
Sprouty-2 controls c-Met expression and metastatic potential of
colon cancer cells: sprouty/c-Met upregulation in human colonic
adenocarcinomas. Oncogene. 29:5241–5253. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Barbáchano A, Ordóñez-Morán P, García JM,
et al: SPROUTY-2 and E-cadherin regulate reciprocally and dictate
colon cancer cell tumourigenicity. Oncogene. 29:4800–4813.
2010.PubMed/NCBI
|
|
37
|
Bloethner S, Chen B, Hemminki K, et al:
Effect of common B-RAF and N-RAS mutations on global gene
expression in melanoma cell lines. Carcinogenesis. 26:1224–1232.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Sasaki A, Taketomi T, Kato R, et al:
Mammalian Sprouty4 suppresses Ras-independent ERK activation by
binding to Raf1. Nat Cell Biol. 5:427–432. 2003. View Article : Google Scholar
|
|
39
|
Mason JM, Morrison DJ, Basson MA and Licht
JD: Sprouty proteins: multifaceted negative-feedback regulators of
receptor tyrosine kinase signaling. Trends Cell Biol. 16:45–54.
2006. View Article : Google Scholar
|
|
40
|
Wong ES, Fong CW, Lim J, et al: Sprouty2
attenuates epidermal growth factor receptor ubiquitylation and
endocytosis, and consequently enhances Ras/ERK signalling. EMBO J.
21:4796–4808. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wilhelm SM, Carter C, Tang L, et al: BAY
43–9006 exhibits broad spectrum oral antitumor activity and targets
the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in
tumor progression and angiogenesis. Cancer Res. 64:7099–7109.
2004.
|
|
42
|
Chang YS, Adnane J, Trail PA, et al:
Sorafenib (BAY 43–9006) inhibits tumor growth and vascularization
and induces tumor apoptosis and hypoxia in RCC xenograft models.
Cancer Chemother Pharmacol. 59:561–574. 2007.
|
|
43
|
Ezzoukhry Z, Louandre C, Trécherel E, et
al: EGFR activation is a potential determinant of primary
resistance of hepatocellular carcinoma cells to sorafenib. Int J
Cancer. 131:2961–2969. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Shibayama Y, Nakano K, Maeda H, et al:
Multidrug resistance protein 2 implicates anticancer
drug-resistance to sorafenib. Biol Pharm Bull. 34:433–435. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Chen KF, Chen HL, Tai WT, et al:
Activation of phosphatidylinositol 3-kinase/Akt signaling pathway
mediates acquired resistance to sorafenib in hepatocellular
carcinoma cells. J Pharmacol Exp Ther. 337:155–161. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Faratian D, Sims AH, Mullen P, et al:
Sprouty 2 is an independent prognostic factor in breast cancer and
may be useful in stratifying patients for trastuzumab therapy. PLoS
One. 6:e237722011. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Feng YH, Tsao CJ, Wu CL, et al: Sprouty2
protein enhances the response to gefitinib through epidermal growth
factor receptor in colon cancer cells. Cancer Sci. 101:2033–2038.
2010. View Article : Google Scholar : PubMed/NCBI
|