Introduction
Osteoarthritis (OA) is an age-related degenerative
joint disease that at its late stage is characterized by extensive
cartilage destruction and the loss of chondrocytes. The loss of the
major components of the extracellular matrix (ECM) as a result of
increased synthesis of matrix metalloproteases (MMPs) and
aggrecanases (such as, ADAMTS), as well as reduced
glycosaminoglycan (GAG) synthesis, has been well-documented during
OA progression (1,2). MMPs are zinc-containing,
calcium-dependent proteinases, which collectively degrade all
components of the extracellular matrix and have a critical role in
intrinsic chondrocyte-mediated degenerative changes in the
cartilage matrix in OA (3). MMPs
are often expressed in chondrocytes in response to major
inflammatory cytokines, including interleukin (IL)-1β, which is
produced by the synovium and other joint tissues (4). IL-1β has been thoroughly
investigated, particularly its role as a key mediator of cartilage
destruction in OA due to its ability to induce or upregulate the
expression of proteinases, including MMPs, plasminogen activators
and aggrecanases, and to downregulate the expression of endogenous
proteinase inhibitors (for example, specific tissue inhibitors of
metalloproteinases) (5–7). These products disrupt the metabolic
balance of chondrocytes, resulting in a perturbation of the
chondrocyte phenotype by reducing type II collagen expression
(5,6). This process induces specific
degradation, and consequently the inhibition of the IL-1β pathway
presents a promising means to prevent cartilage degradation during
OA pathogenesis. One of the major endogenous inhibitors of the IL-1
pathway is the IL-1 receptor antagonist (IL-1Ra). IL-1Ra is a
well-known anti-inflammatory cytokine that is a member of the IL-1
family. IL-1Ra binds to the IL-1RI receptor with similar
specificity and affinity but does not activate downstream signals
(8,9). In addition, IL-1Ra has been used as a
gene therapeutic strategy in the treatment of OA, suggesting that
it may also be appropriate for the treatment of early-stage,
cytokine-mediated cartilage degeneration (10,11).
With the aim of enhancing the potential role of
IL-Ra in the treatment of OA, it was proposed that the
encapsulation of this macromolecule in select microspheres capable
of positively interacting with chondrocytes, where the
macromolecule may be internalized by chondrocytes and released as
encapsulated IL-1Ra in a controlled manner.
Chitosan (CS) is a natural copolymer of
D-glucosamine and N-acetylglucosamine, which is derived from chitin
and is structurally similar to GAGs. Chitosan has been reported to
be non-toxic and bioabsorbable (12) and is known to promote wound healing
(13) and contribute to the
maintenance of the chondrogenic phenotype (14), particularly in its morphology.
Furthermore, CS has been widely used in various nanocarriers and is
considered to enhance drug absorption through tight junctions via a
paracellular route by direct interactions of the cationic polymer
molecule with negatively charged cellular membranes. Numerous
studies have successfully applied the use of CS in drug (15,16)
and DNA delivery (17,18), and tissue engineering (19).
The present study aimed to combine the virtues of CS
in the development of IL-Ra-loaded microspheres, as a potential
novel drug-release system for OA treatment. The interaction between
these microspheres and chondrocytes, as well as their potential in
protecting cartilage, was evaluated.
Materials and methods
Materials
CS (molecular weight, 150 kDa; deacetylation, 95%)
and sodium tripolyphosphate (STPP) were obtained from Sigma-Aldrich
(St. Louis, MO, USA). Recombinant rat (rr)IL-1Ra was purchased from
PeproTech (Rocky Hill, NJ, USA). Trypsinase, collagenase II and
Dulbecco’s modified Eagle’s medium (DMEM)/F12 were purchased from
Gibco-BRL (Carlsbad, CA, USA). All of the other chemicals used in
the present study were of the highest available commercial
grade.
Microsphere preparation and
characterization
CS microspheres were prepared according to an
emulsion ionic cross-linking method modified from previously
described methods (20). The
microspheres were spontaneously obtained via inotropic gelation
between the positively charged amino groups of CS and the
negatively charged amino groups of TPP and IL-1Ra proteins. Under
magnetic stirring at room temperature, 3.5 ml of a mixture of an
aqueous solution of TPP (0.06 mg/ml) and IL-1Ra was added to 3.5 ml
of a solution of CS (1%, w/v, pH 5.0). Magnetic stirring was
maintained for 10 min for complete stabilization of the system.
Next, the microspheres were transferred to Eppendorf tubes and
isolated by centrifugation in a glycerol bed (16,000 × g, 30 min,
25°C). The supernatants were collected and the microspheres were
then resuspended in ultrapure water by shaking on a vortex mixer
(Beijing Donglinchangsheng Biotechnology Co., Ltd., Beijing,
China). Next, the microspheres were centrifuged from the fixed
volumes of microsphere suspension (16,000 × g, 30 min, 25°C)
without a glycerol bed. The supernatants were discarded and the
sediments were freeze-dried. CS microspheres were prepared, and the
sizes and shapes of the microspheres were examined using a scanning
electron microscope (JSM-6510; Jeol, Ltd., Tokyo, Japan).
In vitro release profiles
Approximately 30 mg microspheres were placed in 1.5
ml microcentrifuge tubes containing 1 ml phosphate-buffered saline
(PBS), and incubated at 37°C in a shaking bath at 108 × g at the
indicated time periods for up to seven days. Periodically, the
microsphere suspension was centrifuged, at 10,000 × g for 10 min,
and the supernatant was collected for IL-1Ra analysis. The tubes
obtained were resuspended in fresh PBS. Samples were then assayed
for the IL-1Ra concentration using enzyme-linked immunosorbent
assay kits (PeproTech) according to the manufacturer’s
instructions.
Chondrocyte isolation and culture
conditions
Eight-day-old Sprague Dawley rats were obtained from
the Experimental Animal Center of Wuhan University (Wuhan, China)
and were housed under standard conditions (temperature, 21±1°C;
humidity, 55–60%) with food and water available ad libitum.
The care and use of the animals followed the recommendations and
guidelines of the National Institutes of Health and were approved
by the Wuhan University Animal Care and Use Committee (Wuhan,
China).
Rat cartilage was isolated from the knee joints and
placed into PBS. Briefly, the cartilage tissues were cut into small
pieces (<1 mm3) and digested in 0.2% trypsin and 0.2%
type II collagenase for 30 min and 2 h, respectively. Following
washing twice with DMEM, the released cells were cultured in
DMEM/F12 medium supplemented with 10% fetal bovine serum (FBS) and
antibiotics (1% v/v penicillin/streptomycin) at 37°C with 5%
CO2. The cell viability was determined using a cell
viability analyzer (viability, >90%; Beckman Coulter, Inc.,
Pasadena, CA, USA). The primary cells were maintained in a
monolayer culture throughout the study. After the cells reached
70–80% confluence, the medium was changed to DMEM/F12 without 10%
FBS and antibiotics for 6 h. IL-1β (10 ng/ml) was then added to the
culture medium without rinsing for an additional 48 h. The cultured
chondrocytes were divided into four groups: (i) Blank group, with
no treatment; (ii) controls, treated with IL-1β only; (iii) treated
with IL-1β and CS microspheres; and (iv) treated with IL-1β and
CS-IL-1Ra microspheres. All of the cells were cultured in DMEM/F12,
containing 10% FBS without antibiotics, and incubated for 4 h after
the treatment. Four samples were taken from each group and each
experiment was repeated five times. Four samples were taken from
each group and each experiment was repeated five times.
Cell proliferation and GAG synthesis
Following 72 h of co-culture, the microsphere
solution was discarded and fresh DMEM media containing 0.5 mg/ml
3-(4, 5-dimethylthiazol-2-yl)-2, 5-dippphenyltetrazolium bromide
was added to the chondrocytes and incubated at 37°C with 5%
CO2 for 4 h. The resulting precipitate was dissolved in
dimethylsulfoxide and the absorbance was measured at 570 nm using a
microplate reader (Shimadzu, Kyoto, Japan).
The dimethylmethylene blue (DMMB) spectrophotometric
assay was used to determine the GAG contents in the cell
supernatants. The cell supernatants were digested in 0.5 mg/ml
papain for 2 h at 65°C. Following digestion, the supernatants were
centrifuged at 1,500 × g for 8 min. Next, the DMMB solution (Sigma,
St. Louis, MO, USA) was added to the digested cell supernatants,
and the absorbance was measured at 525 nm using a UV-1601
spectrophotometer (Shimadzu). A standard curve was derived from the
mixed-isomer shark chondroitin sulfate and the GAG content was
calculated.
Reverse transcription-polymerase chain
reaction (RT-PCR) for IL-1β, MMP-1, MMP-3 and MMP-13
Total RNA was extracted from chondrocytes using
TRIzol® (Invitrogen Life Technologies, Carlsbad, CA,
USA) reagent according to the manufacturer’s instructions. Total
RNA was quantified using a spectrophotometer (DU730; Beckman
Coulter, Inc, Brea, CA, USA) at 260 nm and the purity was assessed
by determining the ratio of A260/A280. All of the samples had
ratios >1.75. Reverse transcription was performed at 42°C for 60
min in a total volume of 25 μl under standard conditions with 1 μg
total RNA and 0.5 μg random hexamer primers (Takara Biotechnology
Co., Ltd., Dalian, China). The cDNA was amplified using PCR with
the following primer sets: Forward: 5′-GAGCCTGTCATCTTCGAAACG-3′ and
reverse: 5′-GCACGGGTGCGTCACA-3′ for IL-1β; forward:
5′-GCTAACCTTTGATGCTATAACTACGA-3′ and reverse:
5′-TTTGTGCGCATGTAGAATCT-3′ for MMP-1; forward:
5′-CAAAACATATTTCTTTGTAGAGGACAA-3′ and reverse:
5′-TTCAGCTATTTGCTTGGGAAA-3′ for MMP-3; and forward: 5′-TGGTCCA
GGAGATGAAGACC-3′ and reverse: 5′-TGGCATCAAGGGATAAGGAA-3′ for
MMP-13. As a control, GAPDH was amplified in parallel using the
following primers: Forward: 5′-ACCACAGTCCATGCCATCAC-3′ and reverse:
5′-TCCACCACCCTGTTGCTGTA-3′. The PCR amplification was performed
under standard conditions using 25 μl reaction mixture consisting
of 0.5 μl of 10 mm dNTP, 2.5 μl of 10× buffer (containing
Mg2+), 1 μl upstream primer (50 μg/ml), 1 μl downstream
primer (50 μg/ml), 4 μl cDNA and 1 unit Taq enzyme. The
reaction conditions consisted of 30 cycles of denaturation at 94°C
for 60 sec, annealing at 53°C for 60 sec, extension at 72°C for 60
sec and a final extension at 72°C for 5 min. The PCR amplification
products were analyzed on a 1.5% agarose gel stained with ethidium
bromide and measured semiquantitatively using the Bio-Rad
Multi-Analyst system (Bio-Rad Laboratories, Hercules, CA, USA).
Western blotting analyses for IL-1β
MMP-1, MMP-3 and MMP-13
The proteins were extracted from harvested
chondrocytes. The protein concentrations were determined using the
Bicinchoninic Acid Protein Assay kit (Beyotime Institute of
Biotechnology, Haimen, China). Following adjusting to equal
quantities of proteins (50 μl protein/lane), the proteins were
separated using sodium dodecyl sulfate-polyacrylamide gel
electrophoresis (SDS-PAGE) under reducing conditions and then
transferred onto polyvinylidene difluoride membranes (Bio-Rad
Laboratories). The membranes were blocked in PBS, pH 7.4,
containing 5% non-fat dry milk, and initially incubated with rabbit
polyclonal anti-IL-1β (EMD Millipore, Billerica, MA, USA), rabbit
polyclonal MMP-1 (EMD Millipore), rabbit monoclonal MMP-3 (Abcam,
Cambridge, MA, USA) and rabbit polyclonal MMP-13 (Abcam). Next, the
membranes were incubated with horseradish peroxidase-conjugated
secondary antibodies (goat anti-rabbit IgG; Santa Cruz
Biotechnology Inc., Dallas, TX, USA), followed with visualization
using an enhanced chemiluminescence kit (PerkinElmer, Waltham, MA,
USA).
Statistical analysis
SPSS 19.0 (SPSS, Inc., Armonk, NY, USA) software was
applied for data analysis. The data are presented as the mean ±
standard deviation of five independent experiments. Each
experimental condition was performed in triplicate wells, where the
replicates from each culture were averaged and combined as one
value for analysis. Significant differences among the mean values
of multiple groups were evaluated using one-way analysis of
variance and Student-Newman-Keuls q-test. P<0.05 was considered
to indicate a statistically significant difference.
Results
Characterization of microspheres
The CS-IL-1Ra microspheres were fabricated using the
emulsion ionic cross-linking method. The ability of CS to form a
gel following contact with polyanions (TPP and IL-1Ra) by promoting
the inter- and intramolecular linkage enables the formation of the
microspheres. The morphology of the microspheres is demonstrated in
Fig. 1. The SEM demonstrated that
all of the resulting microspheres were distributed uniformly
(Fig. 1A). The microspheres were
spherical and the surfaces were nearly smooth. The surfaces of the
microspheres appeared to be porous (Fig. 1B), and their diameters ranged from
7 to 18 μM. Compared with the CS-IL-1Ra group (Fig. 1C), the mean size of the
microspheres in the CS group was smaller (Fig. 1D).
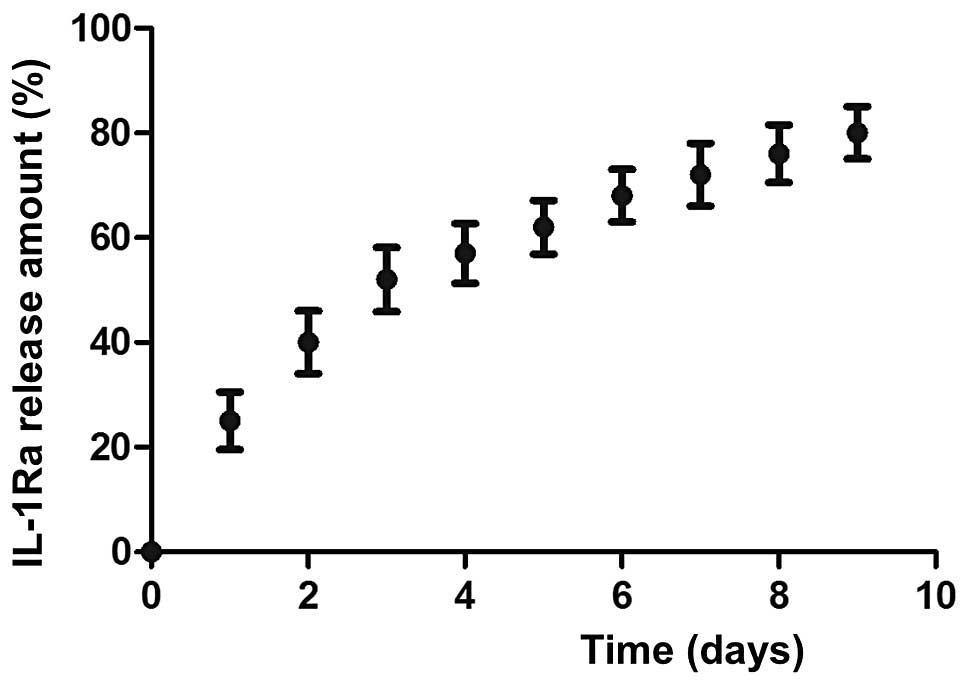
In vitro release profiles
The release kinetics of IL-1Ra from the CS
microspheres are shown in Fig. 2.
In the CS-IL-1Ra microspheres, the IL-1Ra protein was released
slowly. The release kinetics were monitored over seven days, with a
final release of ~80% of IL-1Ra within seven days of incubation.
The IL-1Ra release kinetics were characterized by an initial burst
release, which was reduced to a linear release.
Assay of cell proliferation
The monolayer cultured chondrocyte proliferation was
significantly increased in the CS-IL-1Ra group and the CS group
compared with the control group. Furthermore, chondrocyte
proliferation was significantly higher in the CS-IL-1Ra group
compared with the CS group. The results are demonstrated in
Table I.
 | Table IEffects of microphere treatment on
IL-1β-stimulated chondrocytes. |
Table I
Effects of microphere treatment on
IL-1β-stimulated chondrocytes.
| Group | Blank | 10 ng/ml IL-1β | 10 ng/ml IL-1β
+CS | 10 ng/ml IL-1β
+CS-IL-1Ra |
|---|
| GAG content
(μg/ml) | 12.53±1.24 | 4.09±1.67a | 7.38±0.51b | 10.43±0.82b |
| Cell
proliferation | 0.92±0.02 | 0.39±0.11a | 0.62±0.07b | 0.81±0.04b,c |
GAG contents in cell supernatants
Compared with the control group, the GAG contents
were higher in the CS-IL-1Ra (10.43±0.82 μg/ml) and CS (7.38±0.51
μg/ml) groups, while the GAG content in the CS-IL-1Ra group was
marginally increased in the CS group, as shown in Table I.
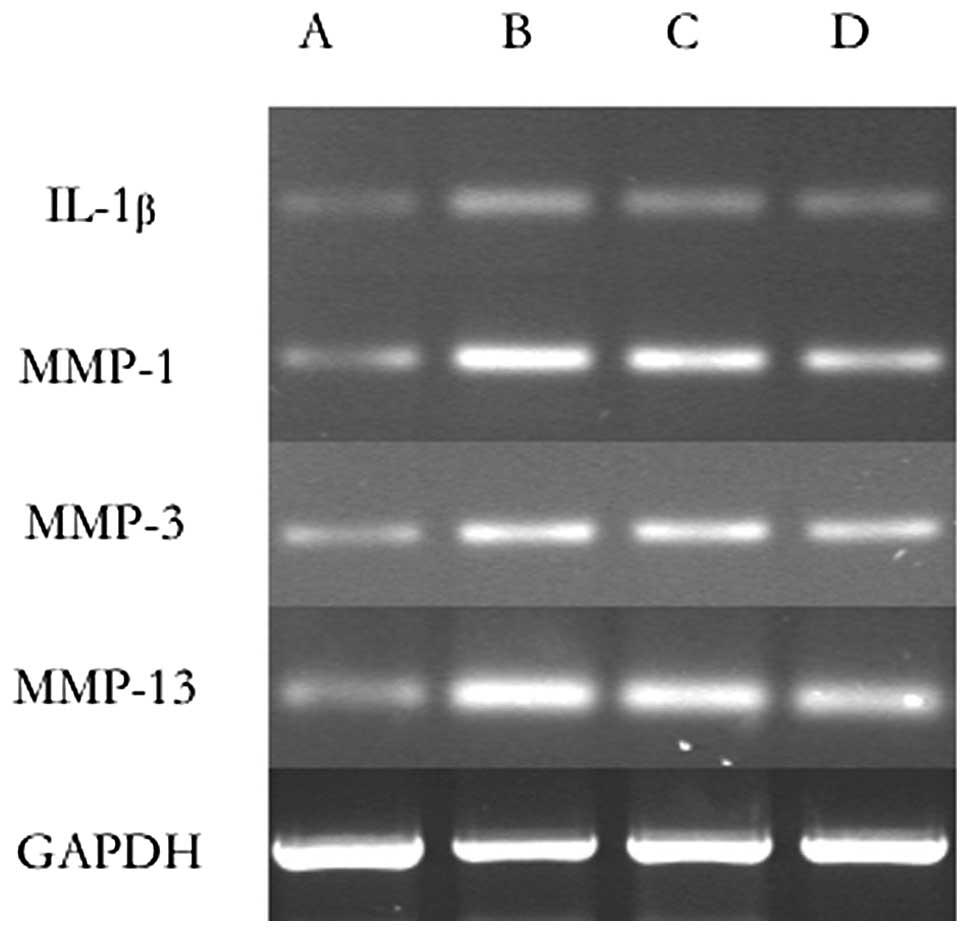
RT-PCR for IL-1β, MMP-1, MMP-3 and
MMP-13
A significant decrease in the expression of IL-1β,
MMP-1, MMP-3 and MMP-13 in all of the experimental groups compared
with the control group was observed as demonstrated in Table II and Fig. 3.
 | Table IImRNA expression of MMP-1, -3, -13 and
IL-1β in chondrocytes. |
Table II
mRNA expression of MMP-1, -3, -13 and
IL-1β in chondrocytes.
| Group | Blank | 10 ng/ml IL-1β | 10 ng/ml IL-1β
+CS | 10 ng/ml IL-1β
+CS-IL-1Ra |
|---|
| MMP-1 | 0.34±0.02 | 1.66±0.09a | 0.96±0.07b | 0.58±0.05b |
| MMP-3 | 0.47±0.11 | 1.45±0.18a | 0.82±0.08b | 0.63±0.11b |
| MMP-13 | 0.41±0.08 | 0.83±0.12a | 0.75±0.09b | 0.68±0.08b |
| IL-1β | 0.26±0.04 | 1.40±0.15a | 0.73±0.07b | 0.55±0.06b |
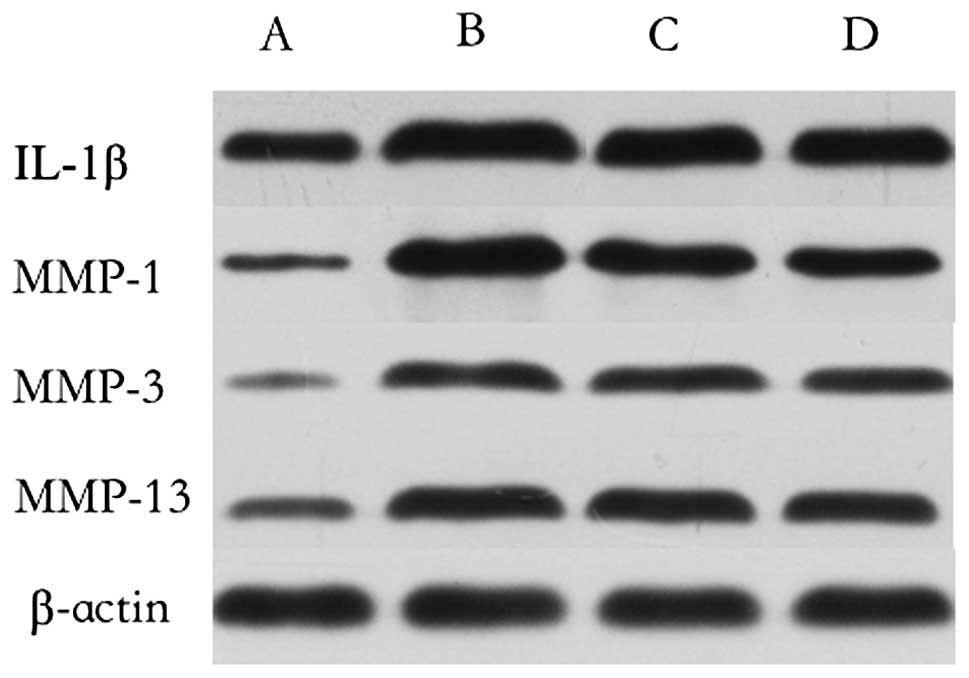
Western blotting analyses for IL-1β,
MMP-1, MMP-3 and MMP-13
The expression of IL-1β, MMP-1, MMP-3 and MMP-13 was
decreased in the CS-IL-1Ra and CS-IL-1Ra groups compared with the
control group. Compared with the CS group, the CS-IL-1Ra group
exhibited a greater decrease in the expression of IL-1β, MMP-1,
MMP-3 and MMP-13 (Fig. 4).
Discussion
The aim of the present study was to clarify the
feasibility of using novel microspheres containing IL-1Ra as a drug
release system to prevent the degeneration of articular cartilage.
The results demonstrated that proliferation and ECM production,
including GAGs, in chondrocytes cultured with microspheres evolved
normally compared with the control group. The IL-1Ra
controlled-release method was long-acting and efficacious. The
expression of inflammatory cytokine production was shown to be
decreased. On the basis of previous studies and the results of the
present study, it was concluded that the controlled-release of
IL-1Ra from CS microspheres demonstrated potential as a novel
method of OA treatment.
GAG is a type of polysaccharide, which together with
collagen type II forms the main component of the cartilage matrix.
The secretion of GAGs in in vitro cultured chondrocytes is
regarded as an indication that the chondrocyte phenotype is
maintained (21). The present
study demonstrated that microspheres were able to inhibit
IL-1β-stimulated production of MMP-1, MMP-3 and MMP-13 in
chondrocytes. Therefore, treatment with microspheres may prevent
the IL-1β-induced breakdown of GAGs and maintain the multi-capacity
of chondrocytes in cartilage by blocking MMPs, resulting in the
delay of OA progression. MMPs destroy arch-like cartilage
structures, which results in disruption of the elastic tissue via
the degradation of collagen II, elastic fibers, proteoglycans and
damage to the molecular sieve function (3). Such an effect renders articular
cartilage more sensitive to catabolic enzymes. As revealed in
Table II, the expression of MMPs
was decreased, while GAG production was significantly increased in
the CS-IL-1Ra and CS groups, but the CS-IL-1Ra group showed a
greater decrease. These data indicated that IL-1Ra was key in
decreasing the production of MMPs in microspheres. Furthermore,
IL-1Ra is a naturally occurring inflammatory inhibitor protein,
which is produced by corneal epithelial cells, monocytes,
neutrophils, macrophages and fibroblasts. It inhibits the activity
of IL-1α and IL-1β by competitively inhibiting binding to type I
and type II receptors, and subsequently blocking the IL-1β-mediated
downstream inflammatory signaling pathway (22). In addition, IL-1Ra may be
chondroprotective as it inhibits IL-1β. It has been reported that
IL-1β may promote the destruction of the cartilage matrix via an
increase in inflammatory cytokine production by chondrocytes,
including IL-8, TNF-α and IL-1β (23); a positive feedback mechanism.
Simultaneously, the expression of these inflammatory cytokines was
decreased by the addition of IL-1Ra (24). It was also reported that human
osteoarthritis-affected chondrocytes transfected with IL-1Ra in
vivo demonstrated resistance to IL-1β-mediated damage, thus
indicating that IL-1β released by osteoarthritis-affected
chondrocytes is an important in the pathophysiology of
osteoarthritis (23). In the
present study, the CS-IL-1Ra microspheres effectively suppressed
the expression of IL-1β. The suppression of inflammatory cytokine
activity within the joint may also be an important mechanism
underlying the clinical activity of microspheres in OA treatment.
Therefore, the results of the present study demonstrate that IL-1Ra
with CS may be a potential treatment strategy for patients with
OA.
A considerable limitation of this study is that the
results were derived from an in vitro experimental model.
Whether CS microspheres loaded with IL-1Ra attenuate the
degeneration of articular cartilage may only be determined by
performing in vivo experiments. Although CS have been
observed to evoke a minimal foreign body response in a number of
studies (25,26), the biocompatibility or
immunological reaction of these materials to joint tissue remains
unclear and the process of degradation or absorption of these
materials in the articular environment remains unknown. This is the
next challenge in validating our hypothesis.
Despite the limitation mentioned above, the data
derived from this study suggest promise in the utilization of CS
microspheres as IL-1Ra carriers for the treatment of OA. CS
microspheres, as a novel drug-release system, may be applied to
cartilage tissue engineering. On the basis of these results,
bioactive substances, such as cell growth factors, basic fibroblast
growth factor and insulin-like growth factor-I, may also be loaded
into microspheres to enhance chondrogenesis in OA treatment.
Acknowledgements
The present study was supported by the National
Natural Science Foundation (grant no. 81071494), the Natural
Science Foundation of Hubei Province (grant no. 2011CHB021) and
Young Scientists Foundation of Health Department of Hubei Province
(grant no. QJX2012-12) in China. All of the authors listed in this
manuscript actively participated in designing the experiments,
analyzing and interpreting the data and helping to enhance the
intellectual content of this manuscript.
References
|
1
|
Hardingham T: Extracellular matrix and
pathogenic mechanisms in osteoarthritis. Curr Rheumatol Rep.
10:30–36. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lorenzo P, Bayliss MT and Heinegård D:
Altered patterns and synthesis of extracellular matrix
macromolecules in early osteoarthritis. Matrix Biol. 23:381–391.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Suzuki M, Hashizume M, Yoshida H, Shiina M
and Mihara M: IL-6 and IL-1 synergistically enhanced the production
of MMPs from synovial cells by up-regulating IL-6 production and
IL-1 receptor I expression. Cytokine. 51:178–183. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Martel-Pelletier J, Boileau C, Pelletier
JP and Roughley PJ: Cartilage in normal and osteoarthritis
conditions. Best Pract Res Clin Rheumatol. 22:351–384. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Billinghurst RC, Wu W, Ionescu M, et al:
Comparison of the degradation of type II collagen and proteoglycan
in nasal and articular cartilages induced by interleukin-1 and the
selective inhibition of type II collagen cleavage by collagenase.
Arthritis Rheum. 43:664–672. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yasuda T and Poole AR: A fibronectin
fragment induces type II collagen degradation by collagenase
through an interleukin-1-mediated pathway. Arthritis Rheum.
46:138–148. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wheaton AJ, Borthakur A, Shapiro EM, et
al: Proteoglycan loss in human knee cartilage: quantitation with
sodium MR imaging - feasibility study. Radiology. 231:900–905.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gabay C, Lamacchia C and Palmer G: IL-1
pathways in inflammation and human diseases. Nat Rev Rheumatol.
6:232–241. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gouze E, Pawliuk R, Gouze JN, et al:
Lentiviral-mediated gene delivery to synovium: potent
intra-articular expression with amplification by inflammation. Mol
Ther. 7:460–466. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Morisset S, Frisbie DD, Robbins PD, Nixon
AJ and McIlwraith CW: IL-1ra/IGF-1 gene therapy modulates repair of
microfractured chondral defects. Clin Orthop Relat Res.
462:221–228. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chen B, Qin J, Wang H, Magdalou J and Chen
L: Effects of adenovirus-mediated bFGF, IL-1Ra and IGF-1 gene
transfer on human osteoarthritic chondrocytes and osteoarthritis in
rabbits. Exp Mol Med. 42:684–695. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sobol M, Bartkowiak A, de Haan B and de
Vos P: Cytotoxicity study of novel water-soluble chitosan
derivatives applied as membrane material of alginate microcapsules.
J Biomed Mater Res A. 101:1907–1914. 2013. View Article : Google Scholar
|
|
13
|
Lih E, Lee JS, Park KM and Park KD:
Rapidly curable chitosan-PEG hydrogels as tissue adhesives for
hemostasis and wound healing. Acta Biomater. 8:3261–3269. 2012.
View Article : Google Scholar
|
|
14
|
Chang SH, Hsiao YW and Lin HY:
Low-frequency electromagnetic field exposure accelerates
chondrocytic phenotype expression on chitosan substrate.
Orthopedics. 34:202011.
|
|
15
|
Haupt JL, Frisbie DD, McIlwraith CW, et
al: Dual transduction of insulin-like growth factor-I and
interleukin-1 receptor antagonist protein controls cartilage
degradation in an osteoarthritic culture model. J Orthop Res.
23:118–126. 2005. View Article : Google Scholar
|
|
16
|
Lee JE, Kim KE, Kwon IC, et al: Effects of
the controlled-released TGF-beta1 from chitosan microspheres on
chondrocytes cultured in a collagen/chitosan/glycosaminoglycan
scaffold. Biomaterials. 25:4163–4173. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wu H, Wang S, Fang H, et al:
Chitosan-polycaprolactone copolymer microspheres for transforming
growth factor-β1 delivery. Colloids Surf B Biointerfaces. 82:602–8.
2011.PubMed/NCBI
|
|
18
|
Zhao J, Fan X, Zhang Q, et al:
Chitosan-plasmid DNA nanoparticles encoding small hairpin RNA
targeting MMP-3 and -13 to inhibit the expression of
dedifferentiation related genes in expanded chondrocytes. J Biomed
Mater Res A. 102:373–380. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Suh JK and Matthew HW: Application of
chitosan-based polysaccharide biomaterials in cartilage tissue
engineering: a review. Biomaterials. 21:2589–2598. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zeng W, Huang J, Hu X, et al: Ionically
cross-linked chitosan microspheres for controlled release of
bioactive nerve growth factor. Int J Pharm. 421:283–290. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Simonaro CM: Cartilage and chondrocyte
pathology in the mucopolysaccharidoses: The role of
glycosaminoglycan-mediated inflammation. J Pediatr Rehabil Med.
3:85–88. 2010.PubMed/NCBI
|
|
22
|
Jacques C, Gosset M, Berebaum F and Gabay
C: The role of IL-1 and IL-1Ra in joint inflammation and cartilage
degradation. Vitam Horm. 74:371–403. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Baragi VM, Renkiewicz RR, Jordan H, et al:
Transplantation of transduced chondrocytes protects articular
cartilage from interleukin 1-induced extracellular matrix
degradation. J Clin Invest. 96:2454–2460. 1995. View Article : Google Scholar
|
|
24
|
Aida Y, Maeno M, Suzuki N, et al: The
effect of IL-1beta on the expression of inflammatory cytokines and
their receptors in human chondrocytes. Life Sci. 79:764–771. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Pattani A, Patravale VB, Panicker L and
Potdar PD: Immunological effects and membrane interactions of
chitosan nanoparticles. Mol Pharm. 6:345–352. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Song S, Zhou F, Nordquist RE, et al:
Glycated chitosan as a new non-toxic immunological stimulant.
Immunopharmacol Immunotoxicol. 31:202–208. 2009. View Article : Google Scholar : PubMed/NCBI
|