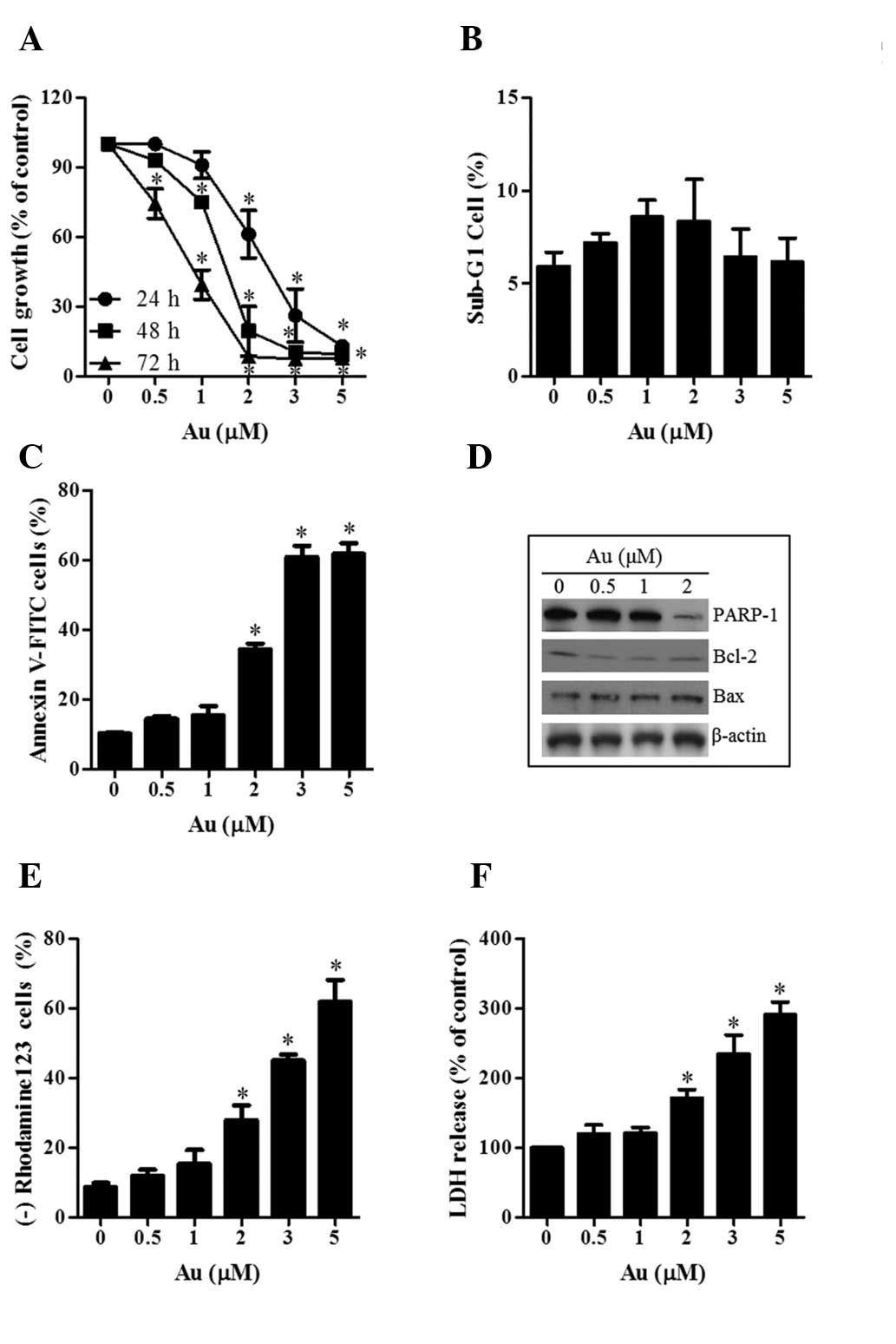
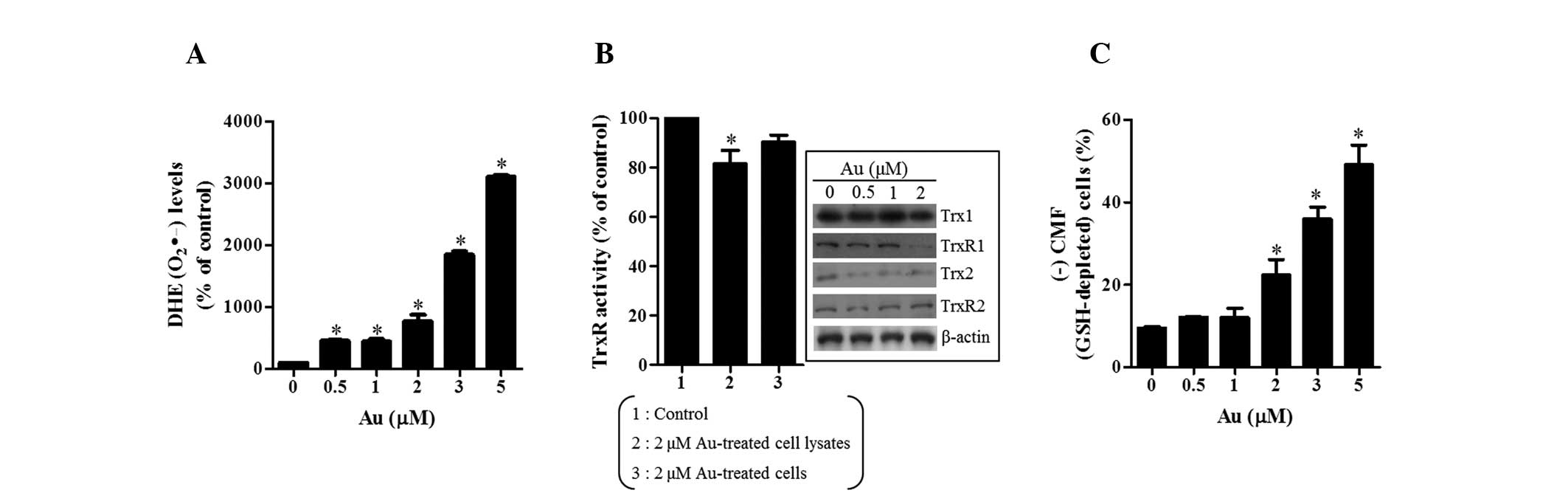
|
1
|
Lu J and Holmgren A: Thioredoxin system in
cell death progression. Antioxid Redox Signal. 17:1738–1747. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Collet JF and Messens J: Structure,
function, and mechanism of thioredoxin proteins. Antioxid Redox
Signal. 13:1205–1216. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Burke-Gaffney A, Callister ME and Nakamura
H: Thioredoxin: friend or foe in human disease? Trends Pharmacol
Sci. 26:398–404. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rundlöf AK and Arnér ES: Regulation of the
mammalian selenoprotein thioredoxin reductase 1 in relation to
cellular phenotype, growth, and signaling events. Antioxida Redox
Signal. 6:41–52. 2004. View Article : Google Scholar
|
|
5
|
Noike T, Miwa S, Soeda J, Kobayashi A and
Miyagawa S: Increased expression of thioredoxin-1, vascular
endothelial growth factor, and redox factor-1 is associated with
poor prognosis in patients with liver metastasis from colorectal
cancer. Hum Pathol. 39:201–208. 2008. View Article : Google Scholar
|
|
6
|
Karlenius TC, Shah F, Di Trapani G, Clarke
FM and Tonissen KF: Cycling hypoxia up-regulates thioredoxin levels
in human MDA-MB-231 breast cancer cells. Biochem Biophys Res
Commun. 419:350–355. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Fernandes AP, Capitanio A, Selenius M,
Brodin O, Rundlöf AK and Björnstedt M: Expression profiles of
thioredoxin family proteins in human lung cancer tissue:
correlation with proliferation and differentiation. Histopathology.
55:313–320. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Poerschke RL and Moos PJ: Thioredoxin
reductase 1 knockdown enhances selenazolidine cytotoxicity in human
lung cancer cells via mitochondrial dysfunction. Biochem Pharmacol.
81:211–221. 2011. View Article : Google Scholar
|
|
9
|
Fu JN, Li J, Tan Q, et al: Thioredoxin
reductase inhibitor ethaselen increases the drug sensitivity of the
colon cancer cell line LoVo towards cisplatin via regulation of G1
phase and reversal of G2/M phase arrest. Invest New Drugs.
29:627–636. 2011. View Article : Google Scholar
|
|
10
|
Madeira JM, Gibson DL, Kean WF and
Klegeris A: The biological activity of auranofin: implications for
novel treatment of diseases. Inflammopharmacology. 20:297–306.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Suarez-Almazor ME, Spooner CH, Belseck E
and Shea B: Auranofin versus placebo in rheumatoid arthritis.
Cochrane Database Syst Rev. CD0020482000.PubMed/NCBI
|
|
12
|
Han S, Kim K, Kim H, et al: Auranofin
inhibits overproduction of pro-inflammatory cytokines,
cyclooxygenase expression and PGE2 production in macrophages. Arch
Pharm Res. 31:67–74. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Han S, Kim K, Song Y, et al: Auranofin, an
immunosuppressive drug, inhibits MHC class I and MHC class II
pathways of antigen presentation in dendritic cells. Arch Pharm
Res. 31:370–376. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liu C, Liu Z, Li M, et al: Enhancement of
auranofin-induced apoptosis in MCF-7 human breast cells by
selenocystine, a synergistic inhibitor of thioredoxin reductase.
PloS One. 8:e539452013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nakaya A, Sagawa M, Muto A, Uchida H,
Ikeda Y and Kizaki M: The gold compound auranofin induces apoptosis
of human multiple myeloma cells through both down-regulation of
STAT3 and inhibition of NF-kappaB activity. Leuk Res. 35:243–249.
2011. View Article : Google Scholar
|
|
16
|
Rigobello MP, Scutari G, Boscolo R and
Bindoli A: Induction of mitochondrial permeability transition by
auranofin, a gold(I)-phosphine derivative. Br J Pharmacol.
136:1162–1168. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Rigobello MP, Folda A, Baldoin MC, Scutari
G and Bindoli A: Effect of auranofin on the mitochondrial
generation of hydrogen peroxide. Role of thioredoxin reductase.
Free Rad Res. 39:687–695. 2005. View Article : Google Scholar
|
|
18
|
Haverkos H, Rohrer M and Pickworth W: The
cause of invasive cervical cancer could be multifactorial. Biomed
Pharmacother. 54:54–59. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
De Marco F, Bucaj E, Foppoli C, et al:
Oxidative stress in HPV-driven viral carcinogenesis: redox
proteomics analysis of HPV-16 dysplastic and neoplastic tissues.
PloS One. 7:e343662012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kim NH, Park HJ, Oh MK and Kim IS:
Antiproliferative effect of gold(I) compound auranofin through
inhibition of STAT3 and telomerase activity in MDA-MB 231 human
breast cancer cells. BMB Rep. 46:59–64. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Marzano C, Gandin V, Folda A, Scutari G,
Bindoli A and Rigobello MP: Inhibition of thioredoxin reductase by
auranofin induces apoptosis in cisplatin-resistant human ovarian
cancer cells. Free Rad Biol Med. 42:872–881. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Rigobello MP, Gandin V, Folda A, et al:
Treatment of human cancer cells with selenite or tellurite in
combination with auranofin enhances cell death due to redox shift.
Free Rad Biol Med. 47:710–721. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Han YH, Moon HJ, You BR, Kim SZ, Kim SH
and Park WH: Effects of carbonyl cyanide p-(trifluoromethoxy)
phenylhydrazone on the growth inhibition in human pulmonary
adenocarcinoma Calu-6 cells. Toxicology. 265:101–107. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Han YH, Moon HJ, You BR and Park WH: The
effect of MG132, a proteasome inhibitor on HeLa cells in relation
to cell growth, reactive oxygen species and GSH. Oncol Rep.
22:215–221. 2009.PubMed/NCBI
|
|
25
|
You BR and Park WH: Zebularine inhibits
the growth of HeLa cervical cancer cells via cell cycle arrest and
caspase-dependent apoptosis. Mol Biol Rep. 39:9723–9731. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Han YH, Kim SH, Kim SZ and Park WH:
Carbonyl cyanide p-(trifluoromethoxy) phenylhydrazone (FCCP) as an
O2 (*−) generator induces apoptosis via the
depletion of intracellular GSH contents in Calu-6 cells. Lung
Cancer. 63:201–209. 2009. View Article : Google Scholar
|
|
27
|
Han YH and Park WH: Propyl gallate
inhibits the growth of HeLa cells via regulating intracellular GSH
level. Food Chem Toxicol. 47:2531–2538. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
You BR and Park WH: Suberoyl bishydroxamic
acid-induced apoptosis in HeLa cells via ROS-independent,
GSH-dependent manner. Mol Biol Rep. 40:3807–3816. 2013. View Article : Google Scholar
|
|
29
|
Martinou JC and Youle RJ: Mitochondria in
apoptosis: Bcl-2 family members and mitochondrial dynamics. Dev
Cell. 21:92–101. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Niquet J, Allen SG, Baldwin RA and
Wasterlain CG: Evidence of caspase-3 activation in hyposmotic
stress-induced necrosis. Neurosci Letters. 356:225–227. 2004.
View Article : Google Scholar
|
|
31
|
Honda A, Abe S, Hiroki E, et al:
Activation of caspase 3, 9, 12, and Bax in masseter muscle of mdx
mice during necrosis. J Muscle Res Cell Motil. 28:243–247. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Park SJ and Kim IS: The role of p38 MAPK
activation in auranofin-induced apoptosis of human promyelocytic
leukaemia HL-60 cells. Br J Pharmacol. 146:506–513. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hatem E, Berthonaud V, Dardalhon M, et al:
Glutathione is essential to preserve nuclear function and cell
survival under oxidative stress. Free Radic Biol Med. 67:103–114.
2014. View Article : Google Scholar
|
|
34
|
Ortega AL, Mena S and Estrela JM:
Glutathione in cancer cell death. Cancers. 3:1285–1310. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Franco R and Cidlowski JA: Glutathione
efflux and cell death. Antioxid Redox Signal. 7:1694–1713. 2012.
View Article : Google Scholar
|
|
36
|
Sobhakumari A, Love-Homan L, Fletcher EV,
et al: Susceptibility of human head and neck cancer cells to
combined inhibition of glutathione and thioredoxin metabolism. PloS
One. 7:e481752012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Scarbrough PM, Mapuskar KA, Mattson DM,
Gius D, Watson WH and Spitz DR: Simultaneous inhibition of
glutathione- and thioredoxin-dependent metabolism is necessary to
potentiate 17AAG-induced cancer cell killing via oxidative stress.
Free Radic Biol Med. 52:436–443. 2012. View Article : Google Scholar
|