Introduction
Retinoids, derivatives of vitamin A (Vit A), have
essential roles in visual phototransduction. In the initial step of
visual phototransduction, conversion of 11-cis retinal to all-trans
(AT) retinal occurs in photoreceptor cells subjected to light
exposure. Vitamin A deficiency (VAD) reduces this activity and
subsequent downstream reactions in photoreceptor cells. As a
result, patients with VAD exhibit visual abnormalities, including
abnormal dark adaptation and night blindness, which may eventually
progress to a severe visual impairment (1–3).
Consistent with these clinical symptoms, electroretinogram (ERG)
recordings in humans with VAD show decreases in amplitudes or loss
of a- and b-waves, which reflect the function of photoreceptor and
bipolar cells, respectively (1,3). A
previous study reported that a reduced a-wave and a partial
reduction of b-wave are induced by feeding albino rats a Vit A
deficient diet for 8 weeks, with disintegration of the outer
segments of photoreceptor cells (4). In contrast to the established effects
of VAD on the a- and b-waves in ERG and histological alterations in
photoreceptors, the effects of VAD on oscillatory potentials (OPs)
remain to be elucidated. OPs, which are components of the ERG, are
considered to be generated by the inner retinal layer cells,
including amacrine and ganglion cells (5) and the reduction of OPs is known to be
associated with reduced contrast sensitivity in diabetic patients
(6). VAD has also been reported to
reduce contrast sensitivity (7).
Therefore, although, to the best of our knowledge, there are no
studies indicating that VAD directly affects the formation of OPs
or the function of inner retinal layer cells, it was hypothesized
that OPs in ERG recordings may be affected during a state of
VAD.
Another disease with known reduced contrast
sensitivity is Parkinson’s disease (PD). Patients with PD are also
known to have reduced OPs (8,9). In
PD, the level of dopamine in the central nervous system (CNS) is
decreased. Notably, retinoid is implicated in the regulation of
gene expression of dopamine receptors (10–12).
Dopamine receptors are present in amacrine cells and are known to
be involved in visual signal transduction (7,13–15).
This was supported by a previous study demonstrating that dopamine
channel blockers can lead to a decline in OPs (16,17).
Retinoid receptors, including the retinoic acid receptor (RAR) and
the retinoid X receptor (RXR), are present in amacrine cells
(18,19). This suggests that retinoids are
associated with the dopamine pathway in visual transduction in the
inner retinal layer cells. In order to assess this hypothesis, gene
expression of dopamine receptors in the retina and spontaneous
locomotor activity, an indicator of dopamine level in the CNS, were
additionally measured to provide collateral evidence for the
effects of VAD on the dopamine system.
The aim of the present study was to analyze the
effects of VAD on the function of inner retinal layer cells, with a
particular focus on OPs and possible involvement of the dopamine
system. VAD rats were analyzed by ophthalmologic examination,
including ERG recording and histology. The concentration of
AT-retinol in the plasma was measured at specified time points to
monitor the state of VAD. In addition, the present study evaluated
the concentration of retinoids and gene expression of dopamine
receptors in the retina. The reversibility of these parameters was
also evaluated following a 5 week recovery period.
Materials and methods
Animals
In total, 36 male Brown Norway (BN) rats were
purchased at 21 days of age from Charles River Laboratories
International, Inc. (Wilmington, MA, USA). All rats were subject to
examination of the eyes via slit lamp and binocular indirect
ophthalmoscope during the pretreatment period. Rats with no
abnormal findings in either eye were selected and divided into VAD
and control groups. A diet with or without Vit A was fed to the
control and the VAD group, respectively. An AIN93G, Vit A (+) diet
or an AIN93G-Vitamin A (−), Vit A (−) diet, were used. The two feed
diets were obtained from Oriental Bioservice (Kyoto, Japan). Each
animal was housed individually in a stainless steel cage within a
clean air room operating a 12 h light/dark cycle. The room had
controlled illumination (<100 lux), temperature (23–24°C) and
humidity (53–71%). The present study was performed in compliance
with Laboratory Animal Policy at Eisai and was approved by the
Laboratory Animal Care and Use Committee at Eisai Co., Ltd.
(Tsukuba, Japan). All procedures were performed according to the
ARVO Statement for the Use of Animals in Ophthalmic and Vision
Research.
Study design
The first week of feeding the Vit A (−) diet was
defined as week 1. To induce VAD in rats, the Vit A (−) diet was
fed to 18 male BN rats for 10 weeks. Subsequently, a Vit A (+) diet
was fed for 5 weeks (weeks 11–15) in order to examine recovery from
VAD. The Vit A (+) diet was administered to 18 males in a control
group for 15 weeks. In the VAD and control groups, 12 main study
and six recovery rats were euthanized at weeks 10 and 15,
respectively, by exsanguination from the abdominal aorta under
isoflurane anesthesia, for sampling of the eyes. Following
euthanasia, the retina was collected. This stage was performed
under a dim red light. All animals were weighed weekly. Blood was
collected from the jugular vein of each of the 12 rats (euthanized
at week 10) at weeks 6, 8 and 10 to measure plasma concentration of
AT-retinol. Prior to euthanasia, ophthalmologic examination,
including ERG recording was performed on all surviving rats and
measurement of spontaneous locomotor activity was conducted on each
of six rats at weeks 10 and 15. At week 10, six and three right
retinas were used for the measurement of retinoids and
determination of the gene expression levels of dopamine receptors,
respectively, in the VAD and control groups. The retinal retinoids
were also measured using six right retinas in the two groups at
week 15. Six left eyes in the two groups were preserved in
glutaraldehyde-formaldehyde and sectioned then coverslipped for
microscopic examination at week 10 and 15. The paraffin-embedded
sections were stained with hematoxylin and eosin (H&E).
Retinoid determinations in plasma and the
retina
Reagents and standard derivatives
High performance liquid chromatography-grade
solvents and reagents, acetonitrile, methanol, propanol and formic
acid were purchased from Wako Pure Chemicals (Osaka, Japan).
Ultra-pure water was prepared using a Millipore Milli-Q TOC system
(Millipore, Billerica, MA, USA). The standard reagent of retinyl
acetate was obtained from Kanto Chemical Co., Inc. (Tokyo, Japan).
Internal standard solutions (1 mg/ml) were prepared in
acetonitrile. To prepare working sample solutions for liquid
chromatography-mass spectrometry (LC-MS), retinyl acetate stock
solution was diluted to a final concentration of 5.0 μg/ml.
LC-MS analysis
An Applied Biosystems AB5000 mass spectrometer
(Applied Biosystems, Foster City, CA, USA) with a Shimadzu
prominence UFLC (Shimadzu, Kyoto, Japan) was used as the
LC-atmospheric pressure chemical ionization (APCI)-MS system. The
Waters Shield RP18 (2.0×150 mm; Waters Corporation, Milford, MA,
USA) with Phenomenex security guard (C18, 2.0×4.0 mm; Phenomenex,
Torrance, CA, USA) was selected as the stationary phase. The
composition of mobile phase A was water/acetonitrile/formic acid
(95:5:0.1) and the mobile phase B was propanol/acetonitrile
(80:20). Linear gradients of mobile phase B were as follows: 0
min-1%, 0.5 min-1%, 1.5 min-25%, 30 min-99 %, 33 min-99%, 33.1
min-1% and 40 min-stop. The flow rate of the mobile phase was 250
μl/min with 450 μl/min of post-column addition of methanol. An
AB5000 mass spectrometer was used in APCI-multiple reaction
monitoring (MRM) mode. Each condition of the MRM channel is listed
in Table I.
 | Table ICondition of multiple reaction
monitoring channels for liquid chromatography/mass spectrometric
analysis. |
Table I
Condition of multiple reaction
monitoring channels for liquid chromatography/mass spectrometric
analysis.
| Retinoid | Q1 (Da) | Q2 (Da) | Dwell time
(msec) | DP (volts) | CE (volts) | CXP (volts) |
|---|
| Retinol (retinol
esters) | 269.2 | 93.2 | 150 | 66 | 27 | 12 |
| Retinal | 285.1 | 161.3 | 150 | 71 | 15 | 18 |
| Retinoic acid | 301.2 | 123.3 | 150 | 66 | 25 | 18 |
The majority of the sample pretreatment processes
were performed under a dim red light. For the plasma sample
pretreatment, 50 μl of plasma samples were transferred into shaded
brown tubes (1.5 ml) and mixed with 190 μl of acetonitrile and 10
μl of 5.0 μg/ml retinyl acetate in acetonitrile. Mixed solutions
were centrifuged at 9,500 × g for 20 min. The supernatant was
transferred to a plastic vial. For the tissue sample pretreatment,
retinas were homogenized in 2.0 ml of water/acetonitrile (25:75).
Retinyl acetate (100 μl of 5.0 μg/ml) in acetonitrile was added to
2.0 ml of tissue homogenate. The sample (250 μl) was transferred to
shaded brown tubes (1.5 ml). Mixed solutions were then centrifuged
at 8,600 × g for 20 min. Supernatants were then transferred to
shaded plastic vials.
The quantity of plasma retinoid was calculated based
on the quantity of retinyl acetate. In the present study, X-cis
retinal included either 11-or 13-cis retinal due to technical
difficulties in measuring the retinal separately.
Ophthalmologic examination
The ophthalmologic examinations with slit lamp or
binocular indirect ophthalmoscope were performed on the eyes of all
animals during the pretreatment period and at weeks 10 and 15.
Prior to observation, the pupils were dilated by topical
application of 0.5% tropicamide and 0.5% phenylephrine
hydrochloride (Mydrin®-P, Santen Pharmaceutical Co.,
Ltd., Osaka, Japan). Images of the fundus were captured using a
Kowa RC-2 fundus camera (Kowa, Tokyo, Japan).
ERG recording
Prior to ERG recording, rats from each group were
adapted to the dark overnight and anesthetized with a combination
of 40 mg/kg ketamine and 0.2 mg/kg xylazine by intramuscular
injection. The pupils were dilated with 0.5% tropicamide and 0.5%
phenylephrine HCl. Rats were placed on an electric heater set at
25°C to prevent a decrease in body temperature until recording.
Dark adaptation was maintained throughout the recording. ERG was
recorded from the right eye of each animal using a contact lens
type electrode (LS-W; Mayo, Inazawa, Japan). The recording
electrode was placed in contact with the cornea, while a reference
electrode was placed in the mouth. ERG responses were amplified and
band-pass filtered between 0.5 and 200 Hz, using a
computer-assisted signal analysis system (MEB2200, Neuropack; Nihon
Kohden, Tokyo, Japan). Light stimuli between 0.0003 and 30
cd·s/m2 were provided by a flash stimulator (SLS-3100;
Nihon Kohden). ERG response amplitudes and implicit time periods
for a- and b-waves were determined based on the International
Society for Clinical Electrophysiology of Vision (ISCEV) standard
(20). OPs were also recorded from
the right eye between 0.0003 and 3.0 cd·s/m2 of light
stimuli using similar methods to the ISCEV standard (20). A band-pass filter was set to 50–500
Hz. The amplitudes and implicit time of OPs were determined as
previously described (21). The
amplitude of OPs was measured between the baseline and peak of each
wavelet and the implicit time was measured between the stimulus
onset and the peak of each wavelet. Each ERG response was recorded
with intervals of >30 sec and the average of three flashes were
calculated at each light stimuli level.
Locomotor activity
Spontaneous locomotor activity was measured using an
automated motion analysis system (SCANET SV-20; Melquest, Toyama,
Japan). Animals (n=6 for control and VAD rats) were assessed in the
SCANET cage for 30 min. Horizontal movements and the frequency of
vertical movements caused by rearing were measured as previously
described (22).
Quantitative polymerase chain reaction
(qPCR) analysis
Gene expression was investigated using reverse
transcription qPCR analysis (RT-qPCR). Total RNA was extracted from
whole unilateral retina using an RNeasy mini kit (Qiagen, Hilden,
Germany) at week 10 and converted to cDNA using
SuperScript® III (Invitrogen Life Technologies,
Carlsbad, CA, USA). Gene expression levels were quantified using an
ABI7900HT Sequence Detection System (Invitrogen Life Technologies).
The quantity of mRNA of dopamine receptor 1 (D1) and 2 (D2) was
determined using specific probe sets (Assay ID: dopamine receptor
D1A: Rn03062203_s1, dopamine receptor D2: Rn01418275_m1) of TaqMan
Gene Expression Assays (Invitrogen Life Technologies). PCR was
performed on 10 ng of the obtained cDNA. PCR conditions were as
follows: 50°C for 2 min, 95°C for 10 min, 40 cycles of 95°C for 15
sec and 60°C for 1 min.
Statistical analysis
Student’s t-test was used to compare the amplitude
and implicit time of ERG components, the relative amplitude of
b-waves and OPs to a-waves, the concentration of retinal retinoids
and gene expression. Wilcoxon analysis was applied to locomotor
activity data. The body weights and concentration of plasma
retinoid were analyzed with repeated measures of analysis of
variance. P<0.05 was considered to indicate a statistically
significant difference.
Results
Body weight
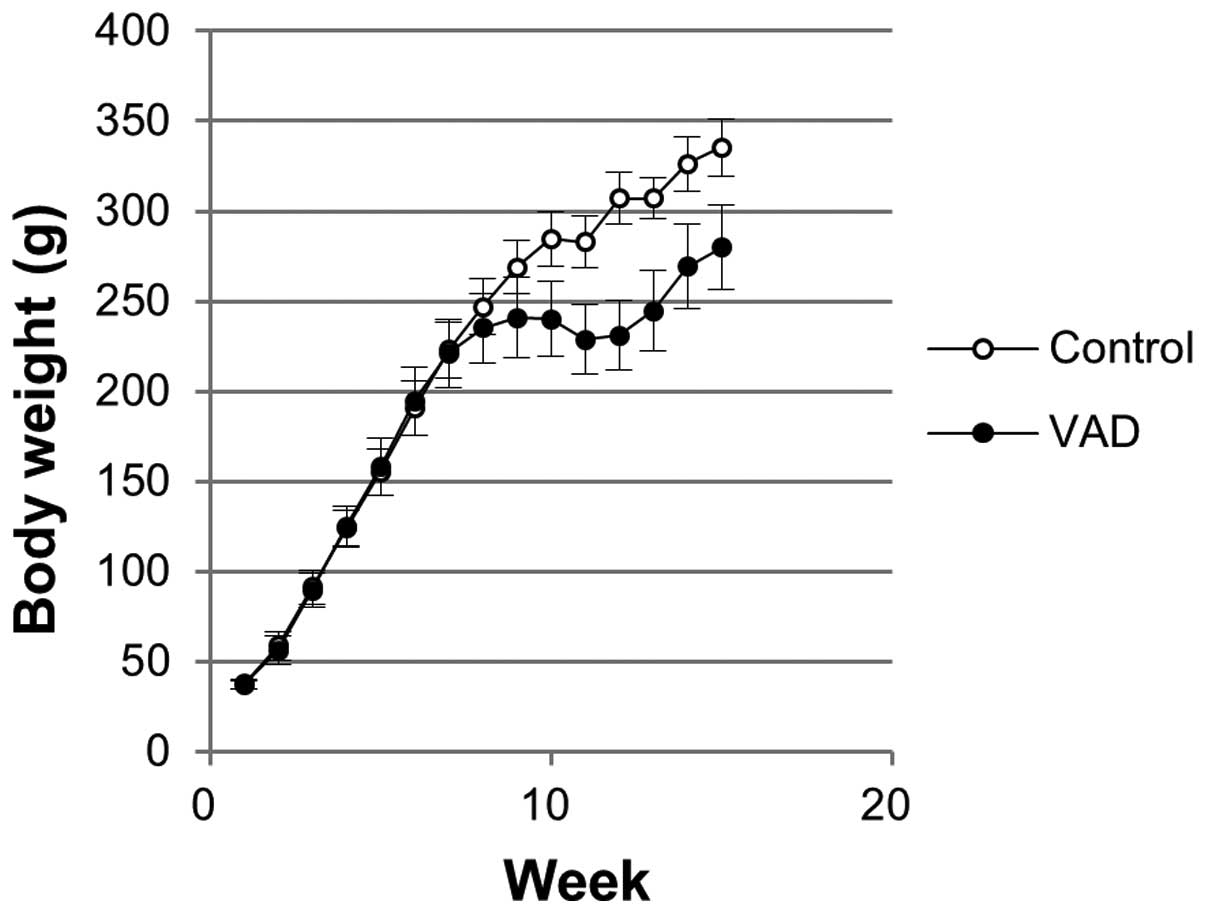
Body weights within the study period are shown in
Fig. 1. A significantly lower body
weight was observed from week 8 in VAD rats. During the recovery
period, the body weight of VAD rats increased with similar growth
rate compared with the control group. However, the difference in
body weight between the control and VAD rats at week 10 (44.5 g)
was maintained at week 15 (55.3 g).
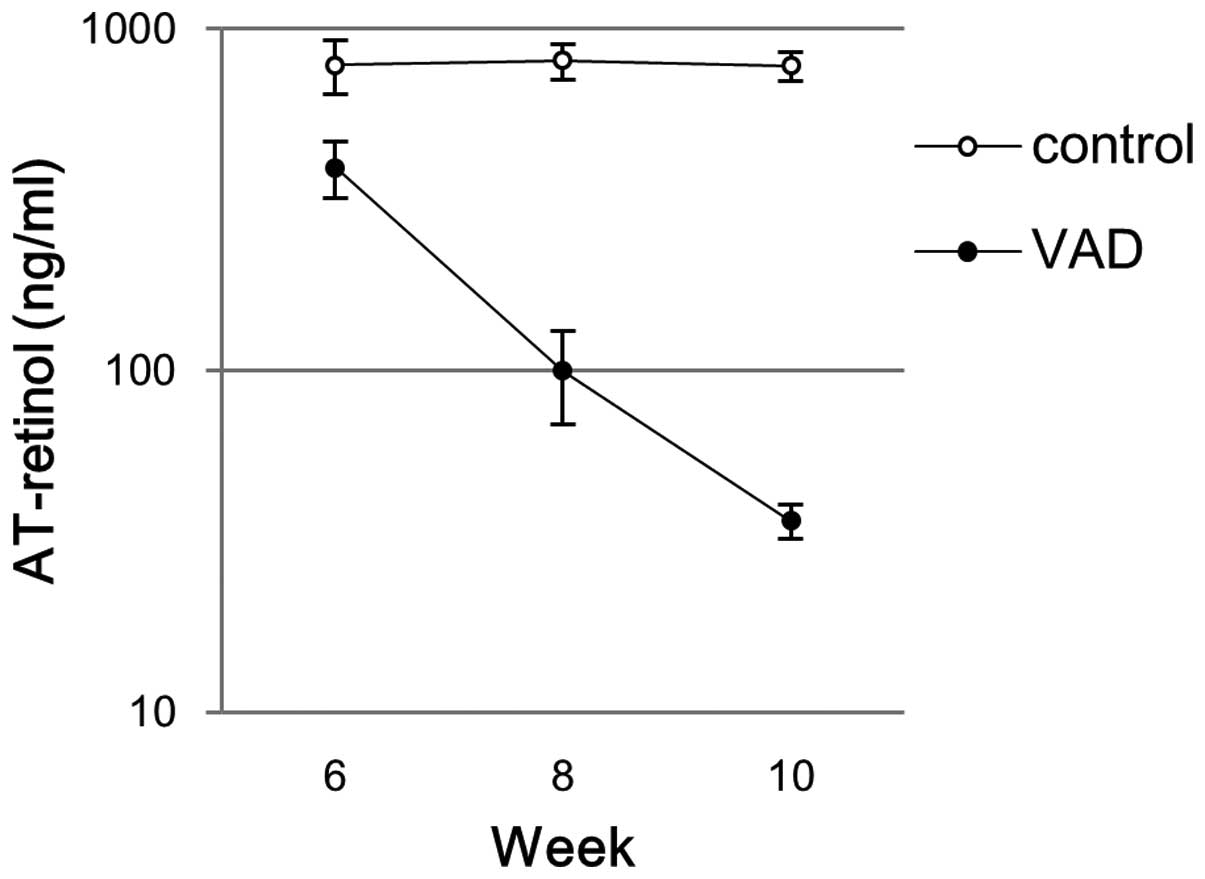
Concentration of plasma AT-retinol
The concentration of plasma AT-retinol is shown in
Fig. 2. The plasma AT-retinol
levels in the control group were stable throughout the experimental
period between 6 and 10 weeks with an average concentration of ~800
ng/ml (minimal-maximal concentrations: 576–1012 ng/ml). At week 6,
in the VAD group, the average concentration of plasma AT-retinol
decreased to 393 ng/ml (min-max: 261–516 ng/ml). The average plasma
concentrations of AT-retinol in VAD rats decreased further to 100
ng/ml (min-max: 57–161 ng/ml) and 36 ng/ml (min-max: 30–44 ng/ml)
at week 8 and 10, respectively.
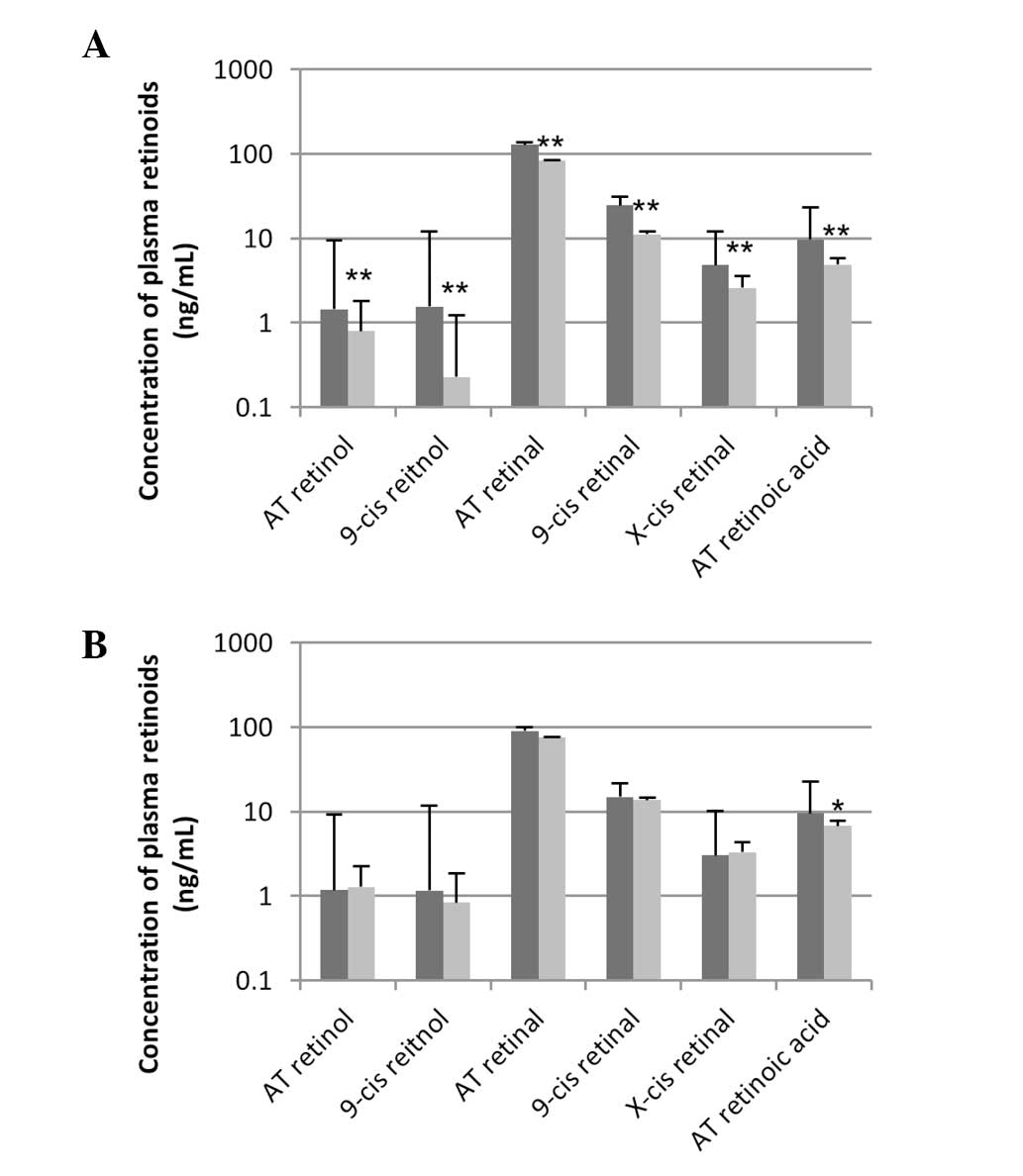
Retinal retinoids
The levels of all measurable retinal retinoids were
significantly lower in VAD rats compared with the control rats at
week 10 (Fig. 3A). AT-retinol,
AT-retinal and AT-retinoic acid in the VAD group decreased
(1.4-->0.8 ng/ml, 129.0-->83.8 ng/ml and 9.7-->4.9 ng/ml,
respectively). 9-cis retinol and retinal decreased by 85 and 54%,
respectively. X-cis retinal containing 11- and 13-cis retinal
decreased by 46%. These alterations almost returned to control
levels by week 15 (Fig. 3B).
ERG recording
Typical ERG wave forms, which were recorded at
0.0003–30 cd·s/m2 at week 10, are shown in Fig. 4. The reduction of OPs in the VAD
rats was clearly evident from these traces. The association between
light intensity and amplitude or implicit time of a- and b-waves is
shown in Fig. 5A and B,
respectively. The amplitude of the b-wave was significantly
decreased to ~50% of the control value at a light intensity
>0.003 cd·s/m2 at week 10. The amplitude of the
a-wave was also significantly decreased at light intensities
>0.3 cd·s/m2. There were significant reductions in
the implicit time of b-waves in the VAD rats compared with the
control at light intensities >3.0 cd·s/m2. Partial
prolongations of the implicit time of the a-wave were observed at
light intensities <0.03 cd·s/m2 with statistical
significance. However, no significant differences were identified
in the implicit time of a-wave between VAD and control rats at
light intensity >3.0 cd·s/m2. No differences were
observed in the amplitude and implicit time between VAD and control
rats at week 15 (Fig. 5C and
D).
OPs of VAD rats were smaller than those of control
rats at week 10 (Fig. 4C and D). A
summary of amplitudes and implicit time periods of OPs recorded at
0.03–3.0 cd·s/m2 is shown in Tables II and III. OPs recorded at <0.01
cd·s/m2 were not listed in these tables as they could
not be detected in the majority of control animals. The mean
amplitudes of OP1, 2 and 3 and summed OPs (∑OPs=OP1 + OP2 + OP3 +
OP4) in VAD rats were significantly decreased to 35–71% of control
rats at week 10. In several VAD rats, loss of all OP wavelets was
observed at <0.3 cd·s/m2. At 3.0 and 30
cd·s/m2, loss of OP peaks was noted. The mean implicit
time of OP1 and 2 were slightly prolonged at multi-intensity at
week 10. These changes returned to control levels at week 15.
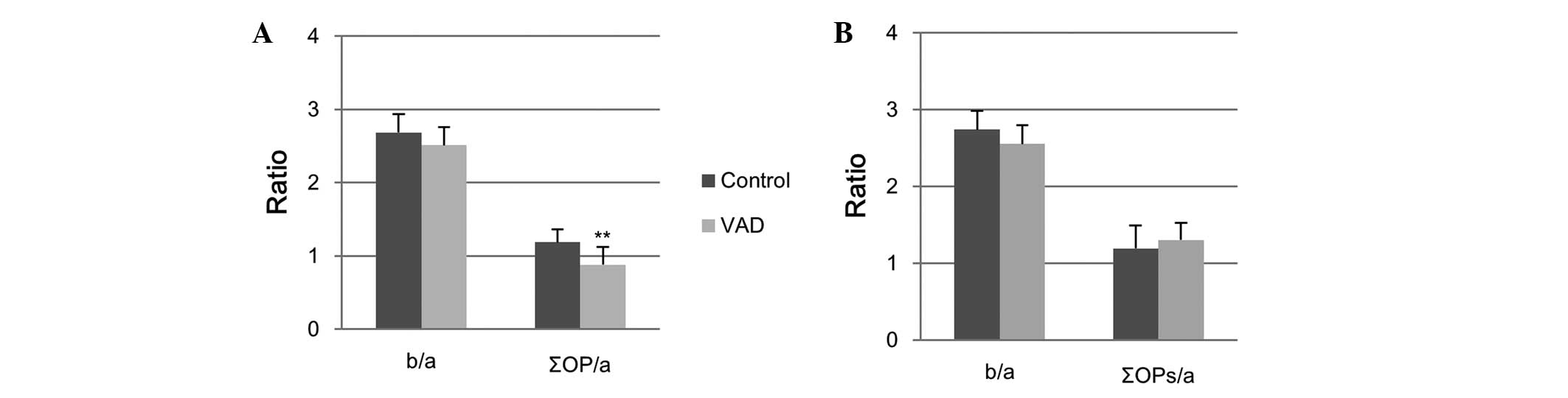
Relative amplitudes of b-wave (b/a) and ∑OPs (∑OPs/a) to a-wave at
3.0 cd·s/m2 are shown in Fig. 6. While the b/a of VAD rats was
comparable to the control value, ∑OPs/a was significantly decreased
at week 10 (Fig. 6A) and returned
to the level of the control following the recovery period (Fig. 6B). The data suggest that VAD
significantly affected OPs more so than a- and b-waves.
 | Table IISummary of the amplitude and implicit
time of each OP wavelet and summed OPs at Week 10. |
Table II
Summary of the amplitude and implicit
time of each OP wavelet and summed OPs at Week 10.
| Light
intensity | Amplitude (μV) | Implicit time
(msec) |
|---|
|
|
|---|
| OP1 | OP2 | OP3 | OP4 | ∑OP | OP1 | OP2 | OP3 | OP4 |
|---|
| 0.03
cd·s/m2 |
| Control |
| AV | 34.6 | 83.8 | 45.8 | 6.8 | 170.9 | 30.1 | 35.6 | 42.0 | 52.4 |
| SD | 5.2 | 12.4 | 18.7 | 10.9 | 34.9 | 1.2 | 1.0 | 1.2 | 3.6 |
| VAD |
| AV | 15.3b | 37.8b | 21.6b | 2.6 | 77.2b | 34.8b | 40.6b | 47.2b | 55.7 |
| SD | 12.9 | 29.7 | 16.7 | 5.3 | 59.4 | 2.6 | 2.7 | 3.2 | 5.1 |
| 0.3
cd·s/m2 |
| Control |
| AV | 75.8 | 134.8 | 44.8 | 3.4 | 258.8 | 26.1 | 31.6 | 39.1 | 50.0 |
| SD | 17.8 | 30.6 | 12.7 | 3.4 | 58.8 | 2.3 | 2.2 | 2.2 | 3.3 |
| VAD |
| AV | 32.6b |
68.6** | 31.8a | 5.5 |
130.9b | 28.9b | 34.6b | 41.0 | 50.0 |
| SD | 19.6 | 26.4 | 21.3 | 8.9 | 64.0 | 2.8 | 2.6 | 3.4 | 5.3 |
| 3.0
cd·s/m2 |
| Control |
| AV | 120.1 | 161.6 | 50.4 | 3.2 | 335.4 | 24.3 | 30.3 | 38.3 | 48.1 |
| SD | 27.2 | 38.3 | 13.9 | 2.6 | 78.8 | 2.2 | 2.4 | 2.5 | 1.7 |
| VAD |
| AV | 45.4b | 77.3b | 33.1b | 5.1 |
161.0b | 26.9a | 32.6a | 39.3 | 47.1 |
| SD | 25.3 | 34.9 | 19.6 | 6.7 | 73.9 | 3.3 | 2.9 | 4.1 | 4.2 |
| 30.0
cd·s/m2 |
| Control |
| AV | 129.5 | 181.5 | 54.5 | 5.8 | 371.4 | 22.4 | 28.7 | 37.1 | 48.0 |
| SD | 28.3 | 45.8 | 15.1 | 3.4 | 87.6 | 2.7 | 2.6 | 2.6 | 2.8 |
| VAD |
| AV | 45.4b | 84.3b | 36.1b | 4.6 |
170.4b | 24.6a | 30.5a | 37.5 | 45.7 |
| SD | 23.2 | 35.9 | 20.9 | 5.6 | 73.8 | 2.5 | 2.4 | 3.0 | 3.9 |
 | Table IIISummary of the amplitude and implicit
time of each OP wavelet and summed OPs at week 15. |
Table III
Summary of the amplitude and implicit
time of each OP wavelet and summed OPs at week 15.
| Light
intensity | Amplitude (μV) | Implicit time
(msec) |
|---|
|
|
|---|
| OP1 | OP2 | OP3 | OP4 | ∑OP | OP1 | OP2 | OP3 | OP4 |
|---|
| 0.03
cd·s/m2 |
| Control |
| AV | 36.2 | 67.2 | 38.2 | 3.3 | 144.8 | 29.9 | 35.2 | 41.5 | 51.1 |
| SD | 20.0 | 40.8 | 21.7 | 5.3 | 80.2 | 0.9 | 1.2 | 1.1 | 2.1 |
| VAD |
| AV | 30.3 | 67.7 | 41.7 | 4.0 | 143.7 | 28.9 | 34.8 | 41.3 | 51.0 |
| SD | 17.5 | 37.4 | 25.7 | 5.5 | 80.0 | 0.7 | 0.5 | 0.7 | 2.1 |
| 0.3
cd·s/m2 |
| Control |
| AV | 76.8 | 139.7 | 45.3 | 10.3 | 272.2 | 25.0 | 30.5 | 38.3 | 47.6 |
| SD | 16.7 | 32.0 | 14.5 | 6.9 | 59.5 | 0.8 | 0.7 | 1.8 | 1.8 |
| VAD |
| AV | 73.8 | 139.0 | 40.5 | 6.0 | 259.3 | 25.0 | 30.3 | 38.4 | 49.4 |
| SD | 16.9 | 27.6 | 11.1 | 3.6 | 55.1 | 0.5 | 0.4 | 0.8 | 0.7 |
| 3.0
cd·s/m2 |
| Control |
| AV | 126.8 | 174.8 | 54.3 | 8.5 | 364.5 | 23.4 | 29.1 | 37.5 | 46.6 |
| SD | 19.8 | 40.3 | 13.8 | 4.8 | 72.5 | 0.6 | 0.6 | 1.6 | 1.7 |
| VAD |
| AV | 117.8 | 165.2 | 45.3 | 2.5 | 330.8 | 23.0 | 28.9 | 37.5 | 47.9 |
| SD | 24.7 | 26.9 | 14.6 | 3.6 | 66.4 | 0.4 | 0.7 | 0.7 | 1.3 |
| 30.0
cd·s/m2 |
| Control |
| AV | 137.2 | 194.5 | 54.7 | 6.3 | 392.7 | 21.5 | 27.5 | 36.1 | 46.5 |
| SD | 24.2 | 43.8 | 15.0 | 5.5 | 79.8 | 0.5 | 0.6 | 0.8 | 1.4 |
| VAD |
| AV | 124.7 | 181.5 | 45.8 | 3.2 | 355.2 | 21.1 | 27.2 | 36.0 | 46.6 |
| SD | 26.4 | 34.4 | 12.8 | 1.5 | 70.5 | 0.3 | 0.5 | 0.5 | 1.0 |
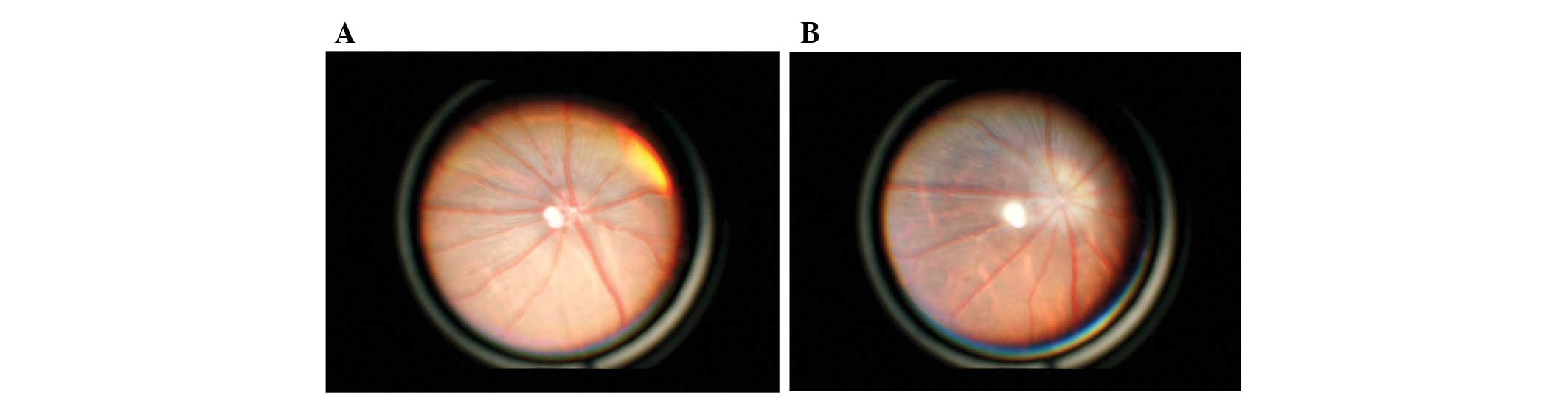
Ophthalmologic examination
Binocular indirect ophthalmoscope observation of the
fundus revealed peri-papillary opacification and was identified in
all VAD rats at week 10 (Table IV
and Fig. 7). At week 15, the
region of opacification became smaller, however, did not disappear.
There were no other VAD-associated changes in anterior and
posterior chambers and the optic media during the experimental
period.
 | Table IVSummary of peri-papillary
opacification identified at weeks 10 and 15. |
Table IV
Summary of peri-papillary
opacification identified at weeks 10 and 15.
| Control group | Vitamin VAD
group |
|---|
| Week 10 |
| Right eye | 0/18 | 18/18 |
| Left eye | 0/18 | 18/18 |
| Week 15 |
| Right eye | 0/6 | 6/6 |
| Left eye | 0/6 | 6/6 |
Histopathological examination
Six left eyes of control and VAD rats were
histopathologically observed at weeks 10 and 15 using H&E
staining. There were no notable findings in the retina, including
photoreceptor cells, inner retinal layer cells and papilla.
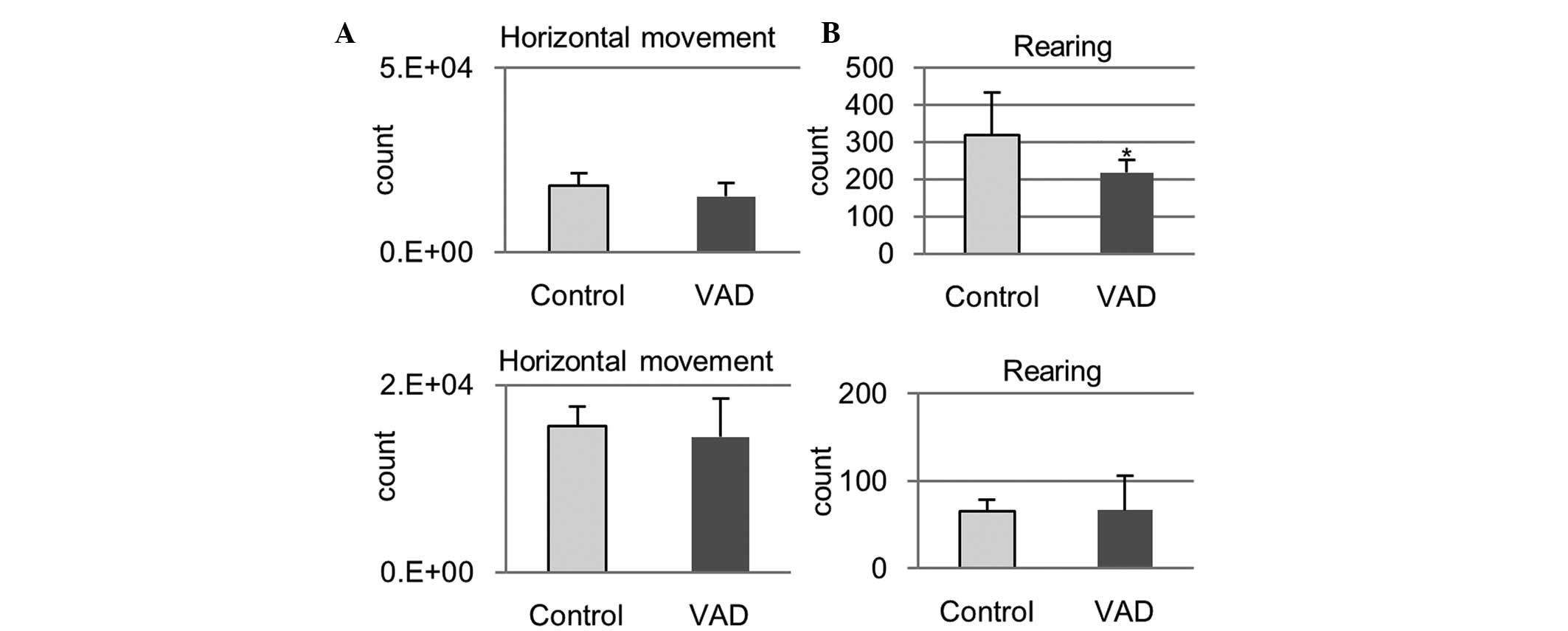
Locomotor activity
In VAD rats, horizontal movements tended to decrease
and the frequency of rearing was significantly lower at week 10
compared with the control rats (Fig.
8, horizontal movement at week 10, P=0.31; rearing at week 10,
P=0.04). At week 15, no apparent differences between VAD and
control rats were observed under these parameters (Fig. 8, horizontal movement at week 15,
P=0.438; rearing at week 15, P=0.485).
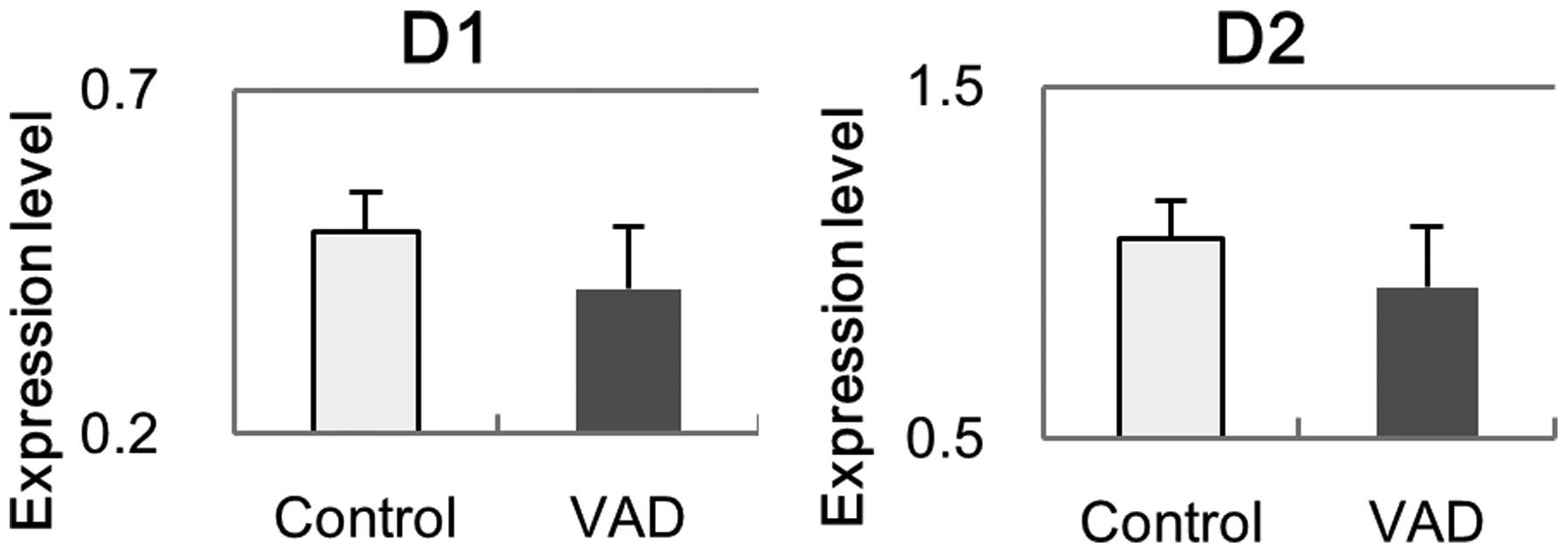
Gene expression of DR
The mRNA of D1 and D2 in the retina was investigated
with RT-qPCR at week 10 (Fig. 9).
The expression levels of D1 and D2 in VAD rats were partially
decreased compared with the control rats, however, no statistical
significance was identified (D1, P=0.13; D2, P=0.16).
Discussion
It has previously been demonstrated that VAD causes
dysfunction of photoreceptor cells (23). VAD induces a decrease in 11-cis
retinal, which is necessary for the synthesis of rhodopsin. This
leads to insufficient phototransduction from light to electrical
signals and a decline in a-waves in an ERG. The present results
were consistent with these changes. In VAD animals, occurrence of
papilledema is also reported as a result of increased cerebrospinal
fluid pressure (24). In the
present study, peri-papillary opacification, one of the properties
of papilledema (25), was noted in
all VAD rats. These results and the significant reduction of plasma
AT-retinol confirm that VAD was induced in rats administered a Vit
A (−) diet for 10 weeks in the present study. By contrast, no
histopathological alterations were observed in the retina of VAD
rats at week 10, nor was there body weight loss, xerophthalmia,
xerosis or keratomalacia indicative of an advanced VAD state
(4). These results indicate that
only functional impairments of the retina were induced by feeding a
Vit A (−) diet to BN rats for 10 weeks and the VAD rats in the
present study were considered to be at an early stage of VAD
disease.
In outer retinal layer cells, the electrical
stimulus is transmitted to and activates bipolar cells through
glutamate released from photoreceptor cells. The activation of
bipolar cells is reflected in the b-wave in an ERG (5). Accordingly, it is expected that the
magnitude of the b-wave depends on that of the a-wave. No
difference in the b/a ratio between control and VAD rats was
identified in the present study suggesting that the there was no
interference in the signal transmission from photoreceptor cells to
bipolar cells by VAD. The signal from bipolar cells subsequently
activates the inner retinal layer cells, including amacrine cells.
The reduction of a-waves may lead to the decreased input to bipolar
cells and subsequently reduced signal transduction from bipolar
cells to amacrine cells, which generate OPs. Therefore, there may
be a corresponding decrease in OPs following the reduction of the
a-wave. In the present study, there was decreased amplitude of the
a-wave with strong light intensities in VAD rats with decreases in
OPs at week 10. However, analysis demonstrated a significant
reduction of ∑OPs/a in VAD rats at week 10, despite having no
changes in b/a, indicating a greater magnitude of reduction in OPs
than the reduction secondary to a-wave reduction alone. OPs are
considered to be generated by the activation of amacrine cells
(3), which release several
neurotransmitters, including dopamine, γ-aminobutyric acid (GABA)
and glycine in order to stimulate other amacrine or ganglion cells
(16,26); inhibitors of these
neurotransmitters are known to decrease OPs (16). The significant reduction of ∑OPs/a
in VAD rats possibly indicates that VAD affects not only the
functions of photoreceptor cells but also signal transduction of
inner retinal layer cells via these neurotransmitters.
In the CNS, 9-cis retinoic acid, a retinoid, is
known to regulate gene expression of dopamine receptors via nuclear
receptors, RAR and RXR (10–12,27).
The greater decrease in 9-cis retinoic acid was suggested by the
notable reduction of endogenous precursors, 9-cis retinol and
retinal (28). The retinoid
receptor mutant mouse shows decreases in locomotor activity, which
is an indication of dopamine levels in the CNS (29). The significant reduction in rearing
count in the present study indicates that a similar decrease in
dopamine receptors by retinoids is present in VAD rats. A previous
study have reported that dopamine acts as an important chemical
messenger within amacrine cells or between amacrine cells and
ganglion cells in the retina activating the dopamine pathway via D1
and D2 dopamine receptors (8).
Based on the decreases in 9-cis retinol and retinal in the retina
and rearing count, it was hypothesized that VAD induced
downregulation of dopamine receptors in the inner retinal layer
cells with a decrease in OPs. Although no statistically significant
difference was identified in the mRNA levels of D1 and D2 in the
retina between VAD and control rats, there was a reduction in these
levels. This may indicate that additional factors contribute to the
decline of OPs in VAD rats. A previous study indicated that
retinoic acid is required for activation of glutamic acid
decarboxylase that converts glutamate to GABA (30). The decline of OPs in VAD rats may
be affected by a variety of factors associated with abnormalities
in the levels of neurotransmitters and decreased photoreceptor
output.
The present study demonstrated for the first time,
to the best of our knowledge, that VAD affects the generation of
OPs. The decreased OPs possibly reflect reduced contrast
sensitivity in patients with VAD and may also be used for
prediction of, or for early detection of other conditions with
reduced contrast sensitivity. The model used in the present study
may also provide an animal model for research associated with
abnormalities in OPs.
Acknowledgements
The authors would like to thank Dr A. Fukamizu of
Tsukuba University for invaluable advice.
References
|
1
|
Dowling JE and Wald G: Vitamin A
deficiency and night blindness. Proc Natl Acad Sci USA. 44:648–661.
1958. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
McLaren DS: Vitamin A deficiency
disorders. J Indian Med Assoc. 97:320–323. 1999.
|
|
3
|
Lam Byron L: Nutritional, Toxic, and
Pharmacological Effects. Electrophysiology of Vision: Clinical
Testing and Applications. 1. 1st Edition. Taylor & Francis
Group; Florida: pp. 407–435. 2005, View Article : Google Scholar
|
|
4
|
Dowling JE and Wald G: Nutritional night
blindness. Ann NY Acad Sci. 74:256–265. 1959. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lam Byron L: Full-Field Electroretinogram.
Electrophysiology of Vision. Clinical Testing and Applications. 1.
1st Edition. Taylor & Francis Group; Florida: pp. 1–47. 2005,
View Article : Google Scholar
|
|
6
|
Kawasaki K, Yonemura K, Yokogawa Y, Saito
N and Kawakita S: Correlation between ERG oscillatory potential and
psychophysical contrast sensitivity in diabetes. Doc Ophthalmol.
64:209–215. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Leguire LE, Pappa KS, Kachmer ML, Rogers
GL and Bremer DL: Loss of contrast sensitivity in cystic fibrosis.
Am J Ophthalmol. 111:427–429. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Witkovsky P: Dopamine and retinal
function. Doc Ophthalmol. 108:17–40. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Jaffe MJ, Bruno G, Campbell G, Lavine RA,
Karson CN and Weinberger DR: Ganzfeld electroretinographic findings
in parkinsonism: untreated patients and the effect of levodopa
intravenous infusion. J Neurol Neurosurg Psychiatry. 50:847–852.
1987. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Samad TA, Krezel W, Chambon P and Borrelli
E: Regulation of dopaminergic pathways by retinoids: activation of
the D2 receptor promoter by members of the retinoic acid
receptor-retinoid × receptor family. Proc Natl Acad Sci USA.
94:14349–14354. 1997. View Article : Google Scholar
|
|
11
|
Wang HF and Liu FC: Regulation of multiple
dopamine signal transduction molecules by retinoids in the
developing striatum. Neuroscience. 134:97–105. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bremner JD and McCaffery P: The
neurobiology of retinoic acid in affective disorders. Prog
Neuropsychopharmacol Biol Psychiatry. 32:315–331. 2008. View Article : Google Scholar
|
|
13
|
Veruki ML and Wässle H:
Immunohistochemical localization of dopamine D1 receptors in rat
retina. Eur J Neurosci. 8:2286–2297. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Veruki ML: Dopaminergic neurons in the rat
retina express dopamine D2/3 receptors. Eur J Neurosci.
9:1096–1100. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nguyen-Legros J, Versaux-Botteri C and
Vernier P: Dopamine receptor localization in the mammalian retina.
Mol Neurobiol. 19:181–204. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wachtmeister L: Oscillatory potentials in
the retina: what do they reveal. Prog Retin Eye Res. 17:485–521.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gelatt Kirk N: Ophthalmic Examination and
Diagnostics. Veterinary Ophthalmology. 2. 4th edition. Oxfordshire
Blackwell Publishing; Oxford, UK: pp. 484–535. 2007
|
|
18
|
Fischer AJ, Wallman J, Mertz JR and Stell
WK: Localization of retinoid binding proteins, retinoid receptors,
and retinaldehyde dehydrogenase in the chick eye. J Neurocytol.
28:597–609. 1999. View Article : Google Scholar
|
|
19
|
Mori M, Ghyselinck NB, Chambon P and Mark
M: Systematic immunolocalization of retinoid receptors in
developing and adult mouse eyes. Invest Ophthalmol Vis Sci.
42:1312–1318. 2001.PubMed/NCBI
|
|
20
|
Marmor MF, Fulton AB, Holder GE, Miyake Y,
Brigell M and Bach M; International Society for Clinical
Electrophysiology of Vision. ISCEV standard for full-field clinical
electroretinography (2008 update). Doc Ophthalmol. 118:69–77. 2009.
View Article : Google Scholar
|
|
21
|
Yamashita H, Yamada-Nakayama C, Sugihara
K, et al: Functional and morphological effects of β-estradiol in
eyes with N-methyl-D-Aspartate-induced retinal neurotoxicity in
rats. Exp Eye Res. 93:75–81. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Matsumoto A, Okada Y, Nakamichi M, et al:
Disease progression of human SOD1 (G93A) transgenic ALS model rats.
J Neurosci Res. 83:119–133. 2006. View Article : Google Scholar
|
|
23
|
Katz ML, Chen DM, Stientjes HJ and Stark
WS: Photoreceptor recovery in retinoid-deprived rats after vitamin
A replenishment. Exp Eye Res. 56:671–682. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gelatt Kirk N: Ocular Manifestations of
Systemic Diseases. Veterinary Ophthalmology. 2. 4th edition.
Oxfordshire Blackwell Publishing; Oxford, UK: pp. 1617–1643.
2007
|
|
25
|
Scott CJ, Kardon RH, Lee AG, Frisén L and
Wall M: Diagnosis and grading of papilledema in patients with
raised intracranial pressure using optical coherence tomography vs
clinical expert assessment using a clinical staging scale. Arch
Ophthalmol. 128:705–711. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wachtmeister L: Further studies of the
chemical sensitivity of the oscillatory potentials of the
electroretinogram (ERG) I. GABA- and glycine antagonists. Acta
Ophthalmol (Copenh). 58:712–725. 1980. View Article : Google Scholar
|
|
27
|
Bondioni S, Angioni AR, Corbetta S, et al:
Effect of 9-cis retinoic acid on dopamine D2 receptor expression in
pituitary adenoma cells. Exp Biol Med (Maywood). 233:439–446. 2008.
View Article : Google Scholar
|
|
28
|
Romert A, Tuvendal P, Simon A, Dencker L
and Eriksson U: The identification of a 9-cis retinol dehydrogenase
in the mouse embryo reveals a pathway for synthesis of 9-cis
retinoic acid. Proc Natl Acad Sci USA. 95:4404–4409. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Krezel W, Ghyselinck N, Samad TA, et al:
Impaired locomotion and dopamine signaling in retinoid receptor
mutant mice. Science. 279:863–867. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bain G, Ramkumar TP, Cheng JM and Gottlieb
DI: Expression of the genes coding for glutamic acid decarboxylase
in pluripotent cell lines. Brain Res Mol Brain Res. 17:23–30. 1993.
View Article : Google Scholar : PubMed/NCBI
|