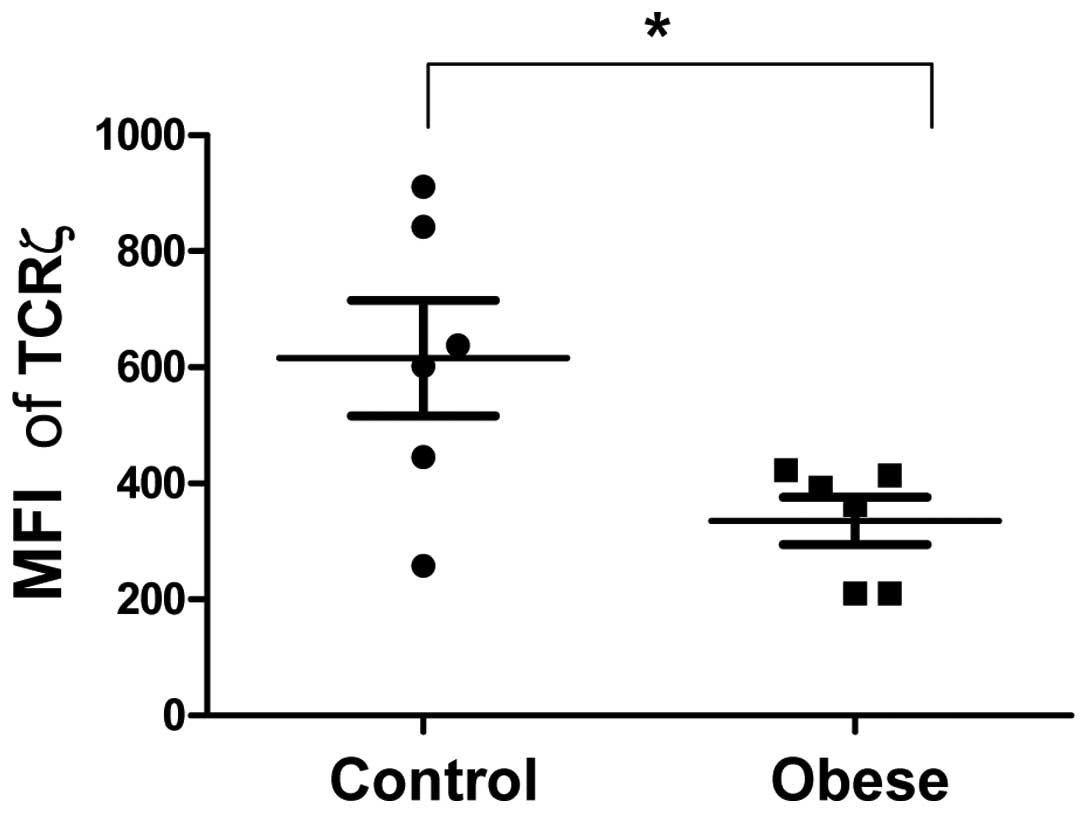
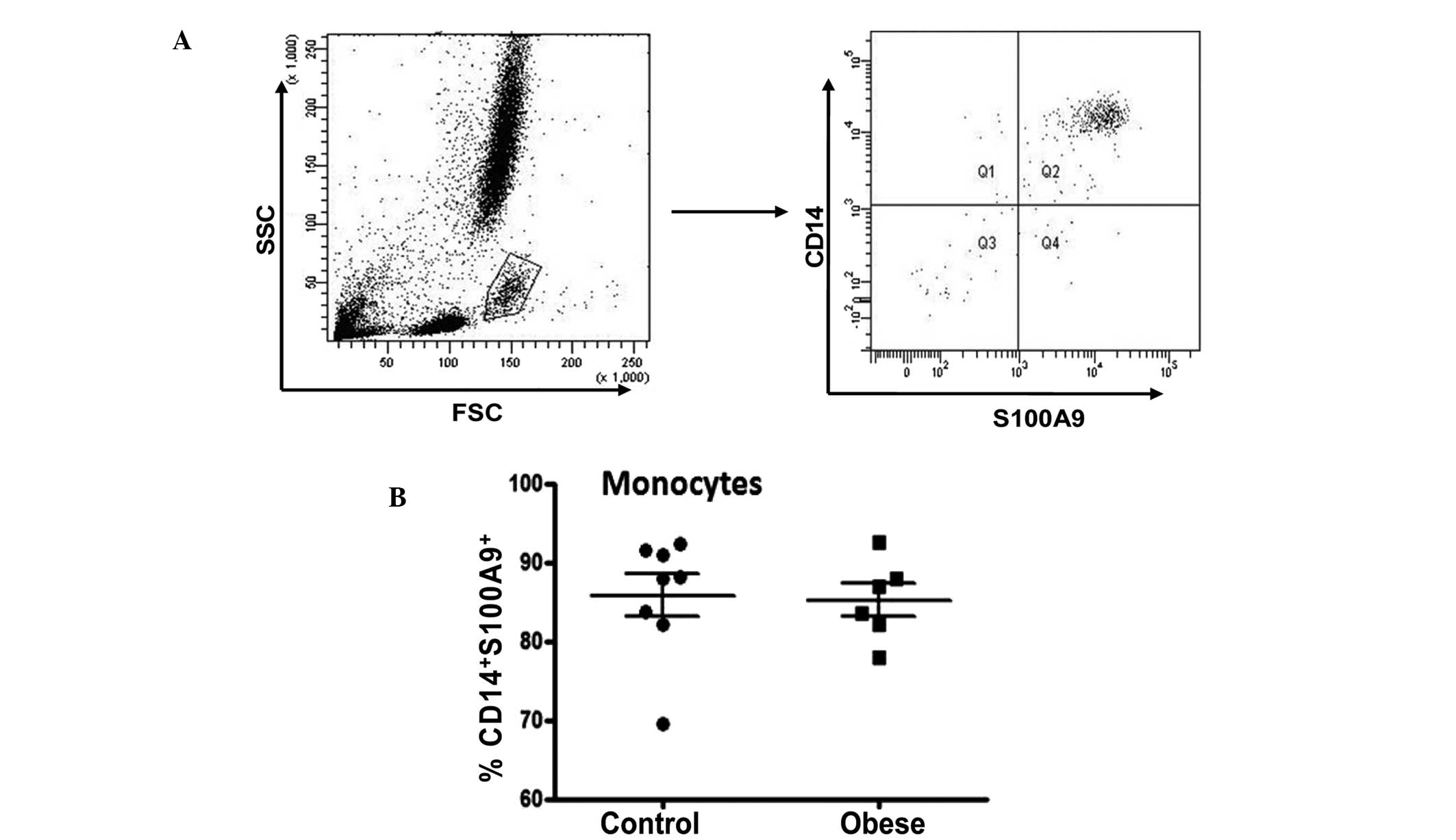
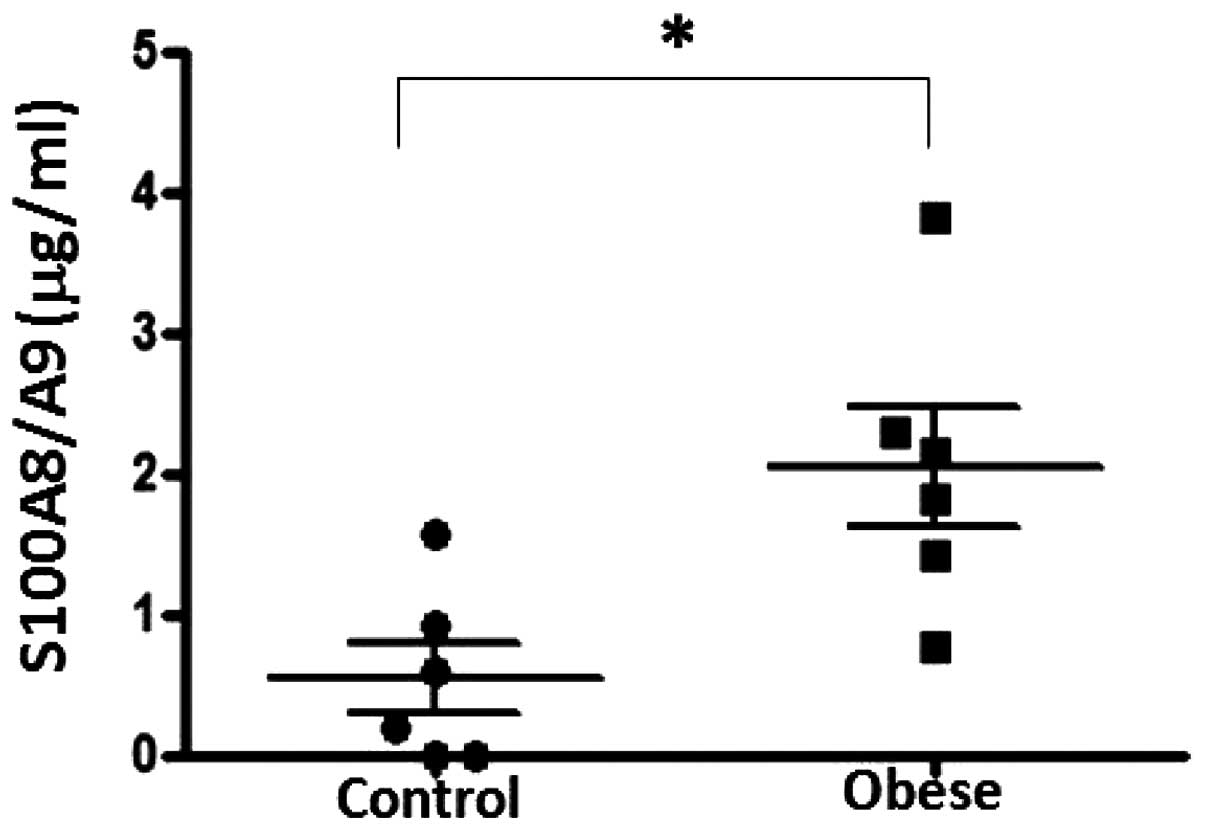
|
1
|
Renehan AG, Tyson M, Egger M, Heller RF
and Zwahlen M: Body-mass index and incidence of cancer: a
systematic review and meta-analysis of prospective observational
studies. Lancet. 371:569–578. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rader DJ: Effect of insulin resistance,
dyslipidemia, and intra-abdominal adiposity on the development of
cardiovascular disease and diabetes mellitus. Am J Med. 120(Suppl
1): S12–S18. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
de Leal VO and Mafra D: Adipokines in
obesity. Clin Chim Acta. 419:87–94. 2013. View Article : Google Scholar
|
|
4
|
van der Weerd K, Dik WA, Schrijver B, et
al: Morbidly obese human subjects have increased peripheral blood
CD4+ T cells with skewing toward a Treg- and
Th2-dominated phenotype. Diabetes. 61:401–408. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
de Heredia FP, Gómez-Martinez S and Marcos
A: Obesity, inflammation and the immune system. Proc Nutr Soc.
71:332–338. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wagner NM, Brandhorst G, Czepluch F, et
al: Circulating regulatory T cells are reduced in obesity and may
identify subjects at increased metabolic and cardiovascular risk.
Obesity (Silver Spring). 21:461–468. 2013. View Article : Google Scholar
|
|
7
|
Lindau D, Gielen P, Kroesen M, Wesseling P
and Adema GJ: The immunosuppressive tumour network: myeloid-derived
suppressor cells, regulatory T cells and natural killer T cells.
Immunology. 138:105–115. 2013. View Article : Google Scholar :
|
|
8
|
Umansky V and Sevko A: Overcoming
immunosuppression in the melanoma microenvironment induced by
chronic inflammation. Cancer Immunol Immunother. 61:275–282. 2012.
View Article : Google Scholar
|
|
9
|
Irving BA, Chan AC and Weiss A: Functional
characterization of a signal transducing motif present in the T
cell antigen receptor zeta chain. J Exp Med. 177:1093–1103. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Irving BA and Weiss A: The cytoplasmic
domain of the T cell receptor zeta chain is sufficient to couple to
receptor-associated signal transduction pathways. Cell. 64:891–901.
1991. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Srivastava MK, Andersson Å, Zhu L, et al:
Myeloid suppressor cells and immune modulation in lung cancer.
Immunotherapy. 4:291–304. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhao F, Hoechst B, Duffy A, et al: S100A9
a new marker for monocytic human myeloid-derived suppressor cells.
Immunology. 136:176–183. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Schouppe E, Van Overmeire E, Laoui D, et
al: Modulation of CD8(+) T-cell activation events by monocytic and
granulocytic myeloid-derived suppressor cells. Immunobiology.
218:1385–1391. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kono K, Ressing ME, Brandt RM, et al:
Decreased expression of signal-transducing zeta chain in peripheral
T cells and natural killer cells in patients with cervical cancer.
Clin Cancer Res. 2:1825–1828. 1996.PubMed/NCBI
|
|
15
|
Fatty Liver and Alcoholic Liver Disease
Study Group of Chinese Liver Disease Association. Diagnostic
criteria of nonalcoholic fatty liver disease. Zhoonghua Gan Zang
Bing Za Zhi. 11:712003.(In Chinese).
|
|
16
|
Kusmartsev S, Su Z, Heiser A, et al:
Reversal of myeloid cell-mediated immunosuppression in patients
with metastatic renal cell carcinoma. Clin Cancer Res.
14:8270–8278. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ozao-Choy J, Ma G, Kao J, et al: The novel
role of tyrosine kinase inhibitor in the reversal of immune
suppression and modulation of tumor microenvironment for
immune-based cancer therapies. Cancer Res. 69:2514–2522. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wang L, Chang EW, Wong SC, et al:
Increased myeloid-derived suppressor cells in gastric cancer
correlate with cancer stage and plasma S100A8/A9 proinflammatory
proteins. J Immunol. 190:794–804. 2013. View Article : Google Scholar
|
|
19
|
Filipazzi P, Valenti R, Huber V, et al:
Identification of a new subset of myeloid suppressor cells in
peripheral blood of melanoma patients with modulation by a
granulocyte-macrophage colony-stimulation factor-based antitumor
vaccine. J Clin Oncol. 25:2546–2553. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gebhardt C, Németh J, Angel P and Hess J:
S100A8 and S100A9 in inflammation and cancer. Biochem Pharmacol.
72:1622–1631. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Foell D, Wittkowski H, Ren Z, et al:
Phagocyte-specific S100 proteins are released from affected mucosa
and promote immune responses during inflammatory bowel disease. J
Pathol. 216:183–192. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Auffray C, Sieweke MH and Geissmann F:
Blood monocytes: development, heterogeneity, and relationship with
dendritic cells. Annu Rev Immunol. 27:669–692. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hansmann L, Groeger S, von Wulffen W, Bein
G and Hackstein H: Human monocytes represent a competitive source
of interferon-alpha in peripheral blood. Clin Immunol. 127:252–264.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lin Y, Gustafson MP, Bulur PA, et al:
Immunosuppressive CD14+HLA-DR(low)/− monocytes in B-cell
non-Hodgkin lymphoma. Blood. 117:872–881. 2011. View Article : Google Scholar :
|
|
25
|
Najjar YG and Finke JH: Clinical
perspectives on targeting of myeloid derived suppressor cells in
the treatment of cancer. Front Oncol. 3:492013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Monneret G, Lepape A, Voirin N, et al:
Persisting low monocyte human leukocyte antigen-DR expression
predicts mortality in septic shock. Intensive Care Med.
32:1175–1183. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mentula P, Kylänpää-Bäck ML, Kemppainen E,
et al: Decreased HLA (human leucocyte antigen)-DR expression on
peripheral blood monocytes predicts the development of organ
failure in patients with acute pancreatitis. Clin Sci (Lond).
105:409–417. 2003. View Article : Google Scholar
|
|
28
|
Antoniades CG, Berry PA, Davies ET, et al:
Reduced monocyte HLA-DR expression: a novel biomarker of disease
severity and outcome in acetaminophen-induced acute liver failure.
Hepatology. 44:34–43. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zimmermann HW, Trautwein C and Tacke F:
Functional role of monocytes and macrophages for the inflammatory
response in acute liver injury. Front Physiol. 3:562012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Trayhurn P and Wood IS: Adipokines:
inflammation and the pleiotropic role of white adipose tissue. Br J
Nutr. 92:347–355. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Trayhurn P, Bing C and Wood IS: Adipose
tissue and adipokines - energy regulation from the human
perspective. J Nutr. 136(Suppl 7): 1935S–1939S. 2006.
|
|
32
|
Rahimi RS and Landaverde C: Nonalcoholic
fatty liver disease and the metabolic syndrome: clinical
implications and treatment. Nutr Clin Pract. 28:40–51. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Höchst B, Schildberg FA, Sauerborn P, et
al: Activated human hepatic stellate cells induce myeloid derived
suppressor cells from peripheral blood monocytes in a
CD44-dependent fashion. J Hepatol. 59:528–535. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Baniyash M: TCR zeta-chain downregulation:
curtailing an excessive inflammatory immune response. Nat Rev
Immunol. 4:675–687. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Whiteside TL: Down-regulation of
zeta-chain expression in T cells: a biomarker of prognosis in
cancer? Cancer Immunol Immunother. 53:865–878. 2004.PubMed/NCBI
|
|
36
|
Sinha P, Okoro C, Foell D, et al:
Proinflammatory S100 proteins regulate the accumulation of
myeloid-derived suppressor cells. J Immunol. 181:4666–4675. 2008.
View Article : Google Scholar : PubMed/NCBI
|