Introduction
Ulcerative colitis (UC) is an important type of
inflammatory bowel disease (IBD), which is a chronic non-specific
autoimmune disease. Although environmental factors, infections and
immunological and psychological factors can lead to the occurrence
of the disease, the specific pathogenesis remains to be
elucidated.
Intestinal mucosal barrier damage has been
recognized as one of the key factors in the pathogenesis of IBD. A
single layer of epithelial cells covering the intestinal mucosal
surface, form a physical barrier to withstand external stimuli and
infections and maintain homeostasis between the intestinal cavity
and internal tissues (1,2). The function of intestinal mucosal
barrier protection is often referred to as intestinal permeability
in IBD (3). Hyperactivity and
increased permeability of intestinal epithelium is an important
characteristic of IBD (4). A
previous study confirmed that colonic epithelial barrier
dysfunction is considered to be important in the initiation of
intestinal inflammation (5). The
main functional characteristic of intestinal epithelial barrier
dysfunction is intestinal permeability alterations, which not only
easily lead to disease, but may also be an important reason for the
onset of delayed healing (6,7).
Barrier function is dynamic and can be adjusted by a
variety of physiological and pathophysiological stimuli (8). Several lines of evidence support the
theory that myosin light chain kinase (MLCK)-dependent
phosphorylation of MLC has a central role in the pathogenesis of
IBD (9–11). The levels of MLCK are positively
associated with disease severity in UC. Increased MLCK expression
can lead to the destruction of tight junctions (TJs), which is
associated with increased intestinal permeability (12). This phenomenon can be prevented by
MLCK inhibitors (13).
Adrenomedullin (AM) is an endogenous vasoactive
peptide, which is closely associated with cell metabolism, immune
function and endocrine and cardiovascular function, and is
important in a number of tissues and organs (14). Previous studies have suggested that
intestinal epithelial cells may be an important target for the
protective effect of AM. Sakata et al found that AM had a
higher expression in the cecum and colon and may have an important
protective role in the colon (15), however, the specific mechanisms
involved remain to be elucidated. Prior studies have demonstrated
that AM may exert a protective effect in the gut by its
anti-inflammatory properties and by suppressing the generation of
bacteria (16–18). Other studies also suggest that AM
can maintain epithelial barrier function via constraining
inflammatory cytokine production and altering the balance of T
cells (4,19–21).
These data indicate that AM is an important protective peptide for
intestinal epithelial cells and is important in the protection of
the intestinal mucosal barrier. Based on the crucial role of MLCK
in the intestinal mucosal barrier, the present study aimed to
evaluate the effect of AM on intestinal permeability and on the
MLCK-p-MLC pathway in a UC animal model induced by
2,4,6-trinitro-benzene-sulfonic acid (TNBS), and to understand its
relative contribution to disease pathogenesis and further clarify
the mechanism of AM in the disease process of UC.
Materials and methods
Ethics statement
Animal experiments were approved by the ethics
committee of Tongji Medical College Affiliated to Huazhong
University of Science and Technology (Wuhan, China) and conformed
to the provisions of the Declaration of Helsinki (as revised in
Edinburgh 2000). In addition, all animals received humane care and
the procedures were performed in accordance with the Principles of
Laboratory Animal Care.
Animals
A total of 21 male Sprague-Dawley rats weighing
200–250 g (Beijing HFK Bioscience Co., Ltd., Beijing, China) were
used for the experiments. Rats were maintained at the Animal
Experimental Center of Huazhong University of Science and
Technology and kept in specific pathogen free air at a temperature
of 22°C with a relative humidity of 50% and a 12 h light and dark
cycle. Rats were randomly divided into three groups, namely, the
normal, model and AM group, with seven rats in each group.
Introduction of colitis
Male Sprague-Dawley rats were induced with colitis
as previously described (22).
Briefly, rats were fasted for 24 h, then TNBS (Sigma-Aldrich, St.
Louis, MO, USA; 5%, 0.6 ml) was integrated in ethanol (50%, 0.25
ml) and delivered into the lumen of the colon via a ~15 cm diameter
2.0 mm rubber infusion tube inserted into the rectum to a depth of
~8 cm from the anal verge.
Administration of AM
The recombinant AM used in the present study was
purchased from Sino Biological Inc. (Beijing, China). After 48 h of
TNBS administration, AM (1.0 µg of AM diluted in 1.0 ml of
saline) was delivered into the lumen of the colon via a 15 cm long
rubber infusion tube inserted into the rectum to a depth of ~8 cm
from the anal verge. Non AM-treated rats were administered 1.0 ml
of saline without AM. AM and saline were administered once a day
for 7 days.
Histological analysis
Colon tissues were fixed in 4% paraformaldehyde and
histological alterations were observed by hematoxylin and eosin
(H&E) staining in routinely processed paraffin-embedded
sections.
ELISA evaluation of TNF-α and IL-6
Blood samples were acquired by cardiac puncture and
used for the evaluation of ELISA. ELISA for TNF-α (rat TNF-α ELISA
kit, cat. no. ERC102a; NeoBioscience, Shenzhen, China) and IL-6
(rat IL-6 ELISA kit, cat. no. ERC003; NeoBioscience) was performed
according to the manufacturer's instructions. The peroxidase
reaction was terminated by the addition of 100 µl 1 N
H2SO4. The absorbance was examined using a
microplate reader (synergy 2; BioTek Instruments, Inc., Winooski,
VT, USA) at 450 nm.
Western blot analysis
Monoclonal rabbit anti-MLCK antibody (cat. no.
ab76092; Abcam, Cambridge, MA, USA) and polyclonal rabbit
anti-myosin light chain (phospho S20) antibody (cat. no. ab2480;
Abcam) were used to assess the expression of MLCK and p-MLC.
Monoclonal rat anti-ZO-1 antibody (cat. no. MABT11; Millipore,
Billerica, MA, USA) was used to assess ZO-1 expression. Proteins
were extracted from intestinal mucosa. Total protein (50 µg)
from each sample was detected by sodium dodecyl sulfate
polyacrylamide gel electrophoresis, respectively. Subsequently, the
gels were transferred onto polyvinylidene difluoride membranes
(Millipore), which were later blocked in Tris-buffered saline
Tween-20 containing 5% fat-free dry milk for 2 h or overnight at
room temperature. The blots were performed using primary antibodies
against rat MLCK (diluted 1:1,000), rat p-MLCK (diluted 1:1,000)
and rat ZO-1 (diluted 1:1,000), respectively, followed by
incubation with horseradish peroxidase-linked goat anti-rabbit
(diluted 1:10,000; cat. no. LK2001; Tianjin Sungene Biotech Co.,
Ltd., Tianjin, China) or rabbit anti-rat (1:50,000; cat. no.
BA1058; Wuhan Boster Biological Technology, Ltd., Wuhan, China)
secondary antibodies. Finally, the extended signals of
protein-antibody complexes were detected through exposure to X-ray
film. To standardize the protein expression, β-actin was conducted
by immunoblotting techniques as an internal control, respectively.
BandScan 5.0 software (Glyko Inc., Novato, CA, USA) was used to
analyze the gray scale values for different bands.
Measurement of transmission electron
microscopy (TEM)
The integrity of the Tjs of epithelial cells was
assessed by measuring TEM. The rats were sacrificed by
intraperitoneal injection of 0.3 ml/100 g 10% chloral hydrate
(Wuhan Union Hospital Pharmacy, Wuhan, China), and then ~1 cm
square colon tissue was obtained using a sharp blade. The colon
tissue was fixed in 2.5% glutaraldehyde-buffered fixative
(absolutely avoid extrusion or using scissors or forceps to uptake)
and pre-fixed for 5–10 min. The bowel was then cut into an area of
1×1 mm tissue, fixed in 2.5% glutaraldehyde-buffered fixative for
2–4 h, using 0.1 M phosphate buffer (pH 7.4) and washed twice, for
20 min each. The tissue was then postfixed in 1% osmium tetroxide
for 2 h, dehydrated in acetone, and embedded in epoxy resin 618.
Areas of interest were selected from semi-thin sections, which were
initally stained with aqueous uranyl acetate followed by lead
citrate. The sections were then detected under TEM (VEGA3 LMU;
Tescan, Brno, Czekh Republic).
Statistical analysis
Experiments were duplicated at least three times and
the values are presented as the mean ± standard deviation. One-way
analysis of variance was used to analyze the statistical
differences. Statistical analysis was performed using SPSS 13.0
software (SPSS, Inc., Chicago, IL, USA). P<0.05 was considered
to indicate a statistically significant difference.
Results
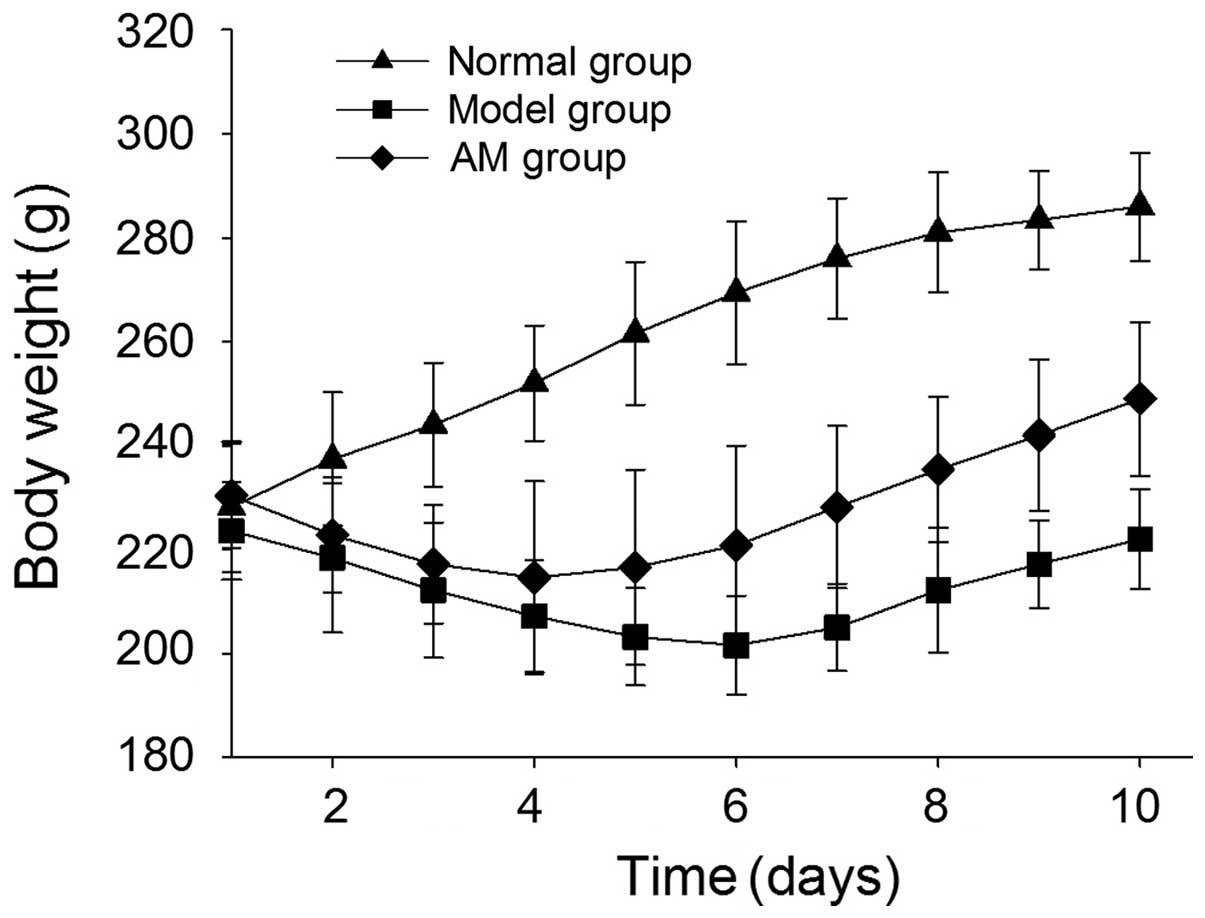
Symptoms of rats in different groups
The activity, diet, stool traits, and body weight of
each rat were observed. Rats administered TNBS exhibited
bradykinesia, reduced food intake, sustained weight loss, and their
stools contained blood and mucus, as compared with the normal rats.
Conversely, the effects of TNBS were markedly ameliorated in the
AM-treated rats, and the reduced body weight of the rats was also
improved, as compared with the untreated group (Fig. 1).
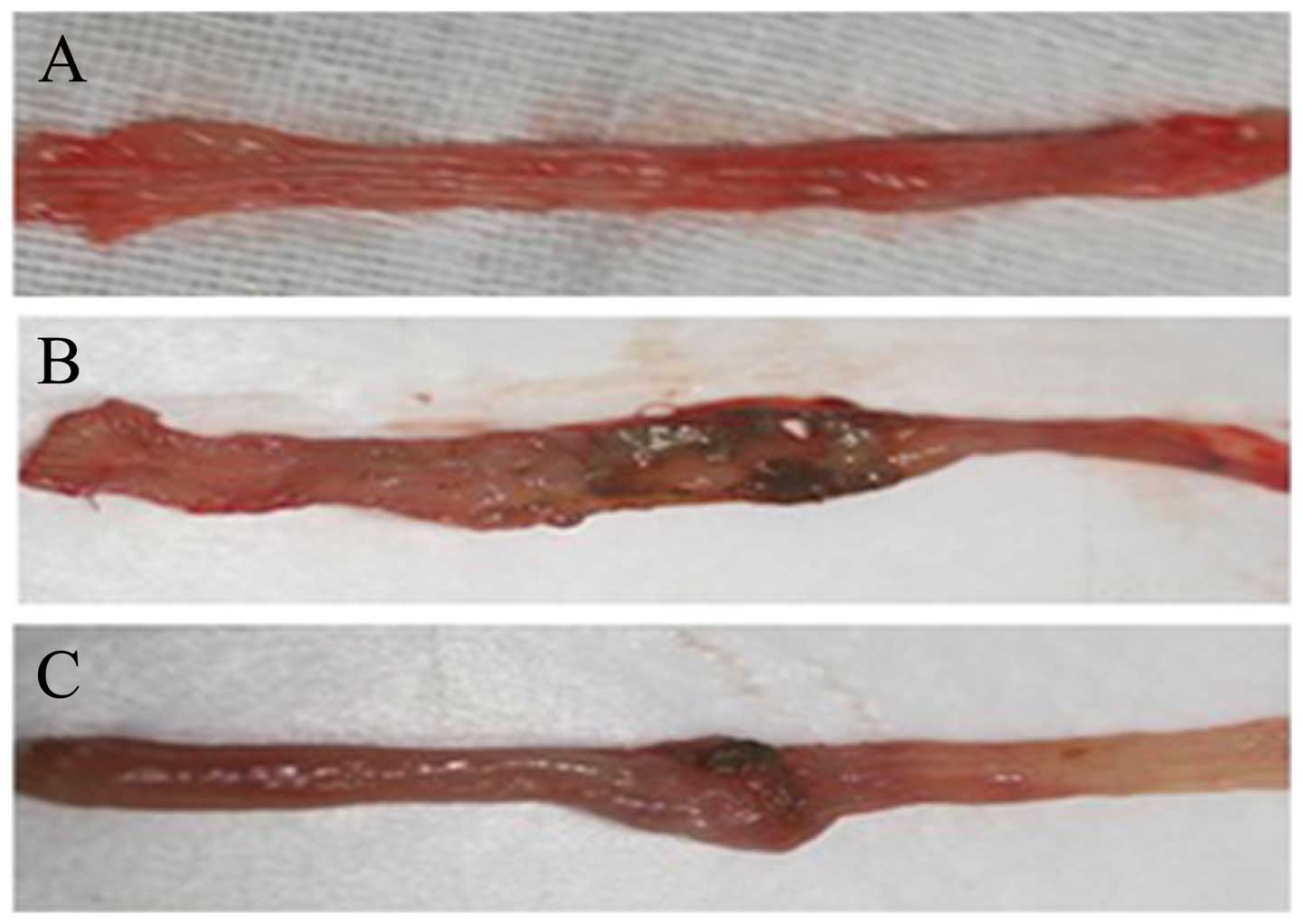
Histological variations
Histological examination demonstrated a normal
morphology in normal rats (Fig.
2A). However, apparent effusion and adhesion, a shorter bowel
length and a thinner intestinal wall was observed in the model
group, and a section of the colon revealed superficial ulcers,
bleeding and erosion in the mucosa (Fig. 2B). By contrast, following AM
administration pathological alterations were significantly less
severe than in the model group and the superficial ulcers of
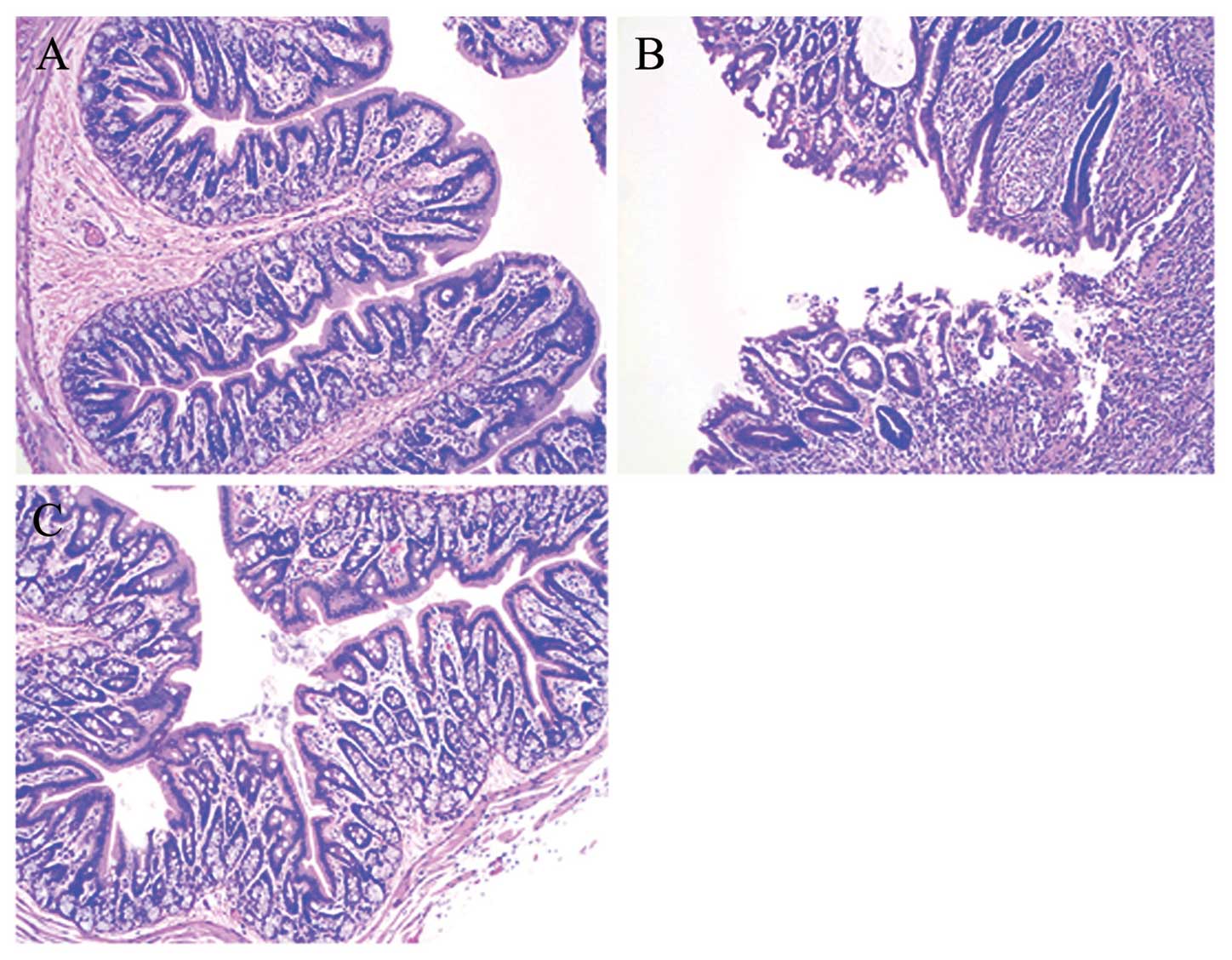
intestinal mucosa were significantly reduced (Fig. 2C). H&E staining demonstrated
mucosal atrophy, partial microvilli loss, edema of the lamina
propria and marked inflammatory cell infiltration in the
TNBS-induced group (Fig. 3A)
compared with the normal group, which exhibited a clear intestinal
wall structure and regularly arranged microvilli (Fig. 3B). Edema and inflammatory cell
infiltration were reduced following AM administration (Fig. 3C).
TNF-α and IL-6
ELISA was used to detect the expression of TNF- and
IL-6. The results demonstrated that the expression of TNF-α and
IL-6 in the serum of model rats was significantly higher than in
the normal group (P<0.05), whereas a lower expression was
observed following AM treatment compared with the model group
(P<0.05; Fig. 4).
MLCK and p-MLC protein
MLC has a crucial role in barrier function, an
increase in the phosphorylation of MLC can mediate epithelial
barrier dysfunction. The present study therefore assessed the
effect of AM on MLC phosphorylation using western blot analysis. As
shown in Fig. 5, MLCK and p-MLC
expression significantly increased in the model group compared with
normal rats (*P<0.05). However, following AM administration,
MLCK and p-MLC expression significantly decreased compared with the
untreated colitis rats (P<0.05; Fig. 5).
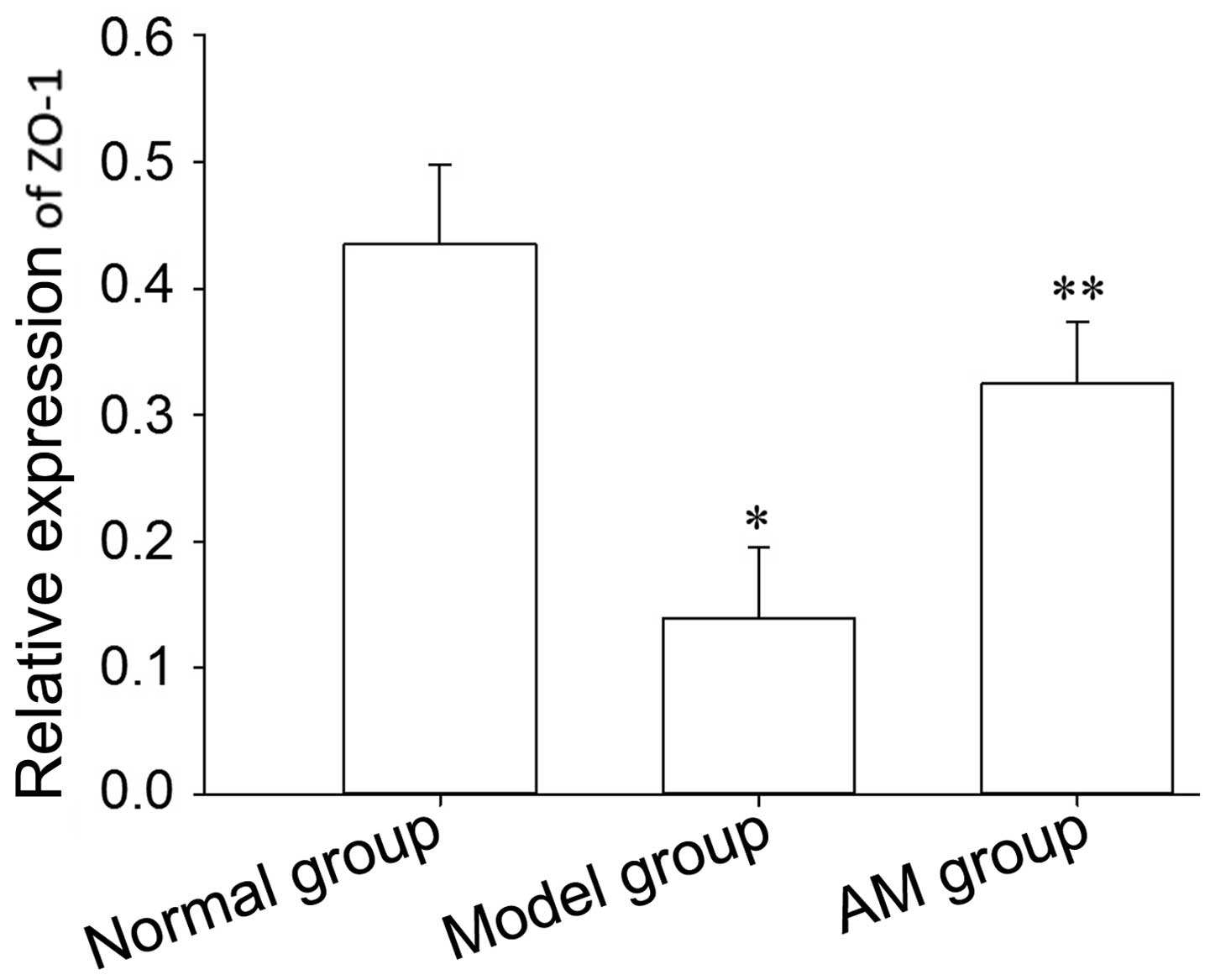
ZO-1 expression
The TJ is also involved in intestinal mucosal
barrier damage. In order to observe alterations in TJ proteins, the
present study used western blot analysis to detect the expression
level of ZO-1. Western blot analysis demonstrated a decreased
expression of ZO-1 in model rats, however, the expression of ZO-1
was decreased in the AM-treated group (Fig. 6).
TEM of TJs. Microvilli damage and TJ
destruction are major contributors to epithelial dysfunction. In
our experiments, TEM demonstrated the integrity of epithelial TJs
and neat arrangement of microvilli in the normal group (Fig. 7A), however, a widened or fractured
TJ, sparse microvilli and expansion of endoplasmic reticulum was
observed in the model group (Fig.
7B). These were improved significantly following AM treatment
(Fig. 7C).
Discussion
IBD, including UC and Crohn's disease affects a
large number of individuals. However, the specific mechanism
involved remains to be elucidated. One important factor is
intestinal mucosal barrier damage, which is often referred to as
increased intestinal permeability (1,2).
There are different degrees of intestinal mucosal barrier
dysfunction in a variety of IBDs and bacterial dysentery. The
integrity of the intestinal epithelial barrier has an important
function to prevent electrolyte loss and toxin penetration
(23). Inflammatory cytokines can
alter the structure of intestinal epithelial TJs, resulting in
impaired epithelial barrier function (24,25).
During the pathogenesis of IBD, TNF-α has been widely recognized as
one of the key proinflammatory cytokines, which can increase
paracellular permeability, damage the mucosal barrier and mediate
intestinal inflammation, while TNF-α antagonist therapy can
effectively alleviate intestinal inflammation and repair the
intestinal mucosa tissues (26).
TNF-α may be directly involved in intestinal barrier function
damage. Previous studies have demonstrated that the dysfunction of
intestinal epithelial cells induced by TNF-α requires the
involvement of MLCK and p-MLC, while inhibiting the synthesis of
MLCK can significantly reduce the permeability of the epithelial
barrier (9,27,28),
disclosing a key role of the MLCK-p-MLC pathway in intestinal
epithelial barrier dysfunction induced by TNF-α. In rat colitis
induced by TNBS, an increased level of TNF-α was detected in the
serum, at the same time, the protein expression levels of MLCK and
p-MLC were also upregulated, however, this was prevented by the
administration of AM, indicating its ability to inhibit activation
of the MLCK-p-MLC pathway by TNF-α.
In addition to TNF-α, whether other cytokines are
involved in the destruction of the gut barrier, remains to be
elucidated. In the present study, serum IL-6 expression correlated
with the increase of intestinal epithelial permeability in UC rats
induced by TNBS. This result is also in agreement with a previous
study demonstrating that IL-6 has a detrimental role in DSS-induced
colitis (29). Notably, IL-6 may
also be involved in the destruction of intestinal barrier function,
demonstrating that AM may antagonize activation of the MLCK-p-MLC
pathway by inhibiting expression of pro-inflammatory cytokines,
including TNF-α and IL-6.
The intestinal mucosal barrier is important in
maintaining intestinal function (30,31).
Previous studies have demonstrated that TNF-α can induce intestinal
barrier dysfunction and the mechanism may be associated with the
upregulation of MLCK expression (32). Turner et al (1,33)
demonstrated that TNF-α-induced barrier dysfunction requires myosin
II activation by MLCK-dependent MLC phosphorylation. Therefore,
TNF-α can induce MLC phosphorylation and p-MLC has a crucial role
in TNF-α-induced barrier disruption. MLCK is a protein that
catalyzes the phosphorylation of MLC, and the expression of MLCK
was consistent with p-MLC. Our data revealed that MLCK and p-MLC
expression significantly increased compared with normal rats.
Notably, ZO-1 protein expression clearly decreased and intestinal
epithelial cell permeability increased at the same time. The
MLCK-p-MLC pathway is not only essential for mucosal damage, but
also causes epithelial barrier damage possibly through controlling
the permeability of TJs.
AM is a type of endogenous vasoactive peptide, which
is upregulated during inflammation in numerous areas (14,34).
In the treatment of IBD, AM has been confirmed to have a beneficial
effect on the intestine by its anti-inflammatory and antibacterial
properties and by suppressing inflammatory cytokines (17–19).
In an intestinal ischemia-reperfusion injury rat model, AM was
demonstrated to have a protective effect on intestinal mucosal
structure and function (35).
However, the effect of AM on MLCK and p-MLC of UC rats has not been
reported. In our experimental colitis model induced by TNBS, the
expression of TNF-α, IL-6, MLCK and p-MLC significantly increased
compared with nor malrats. AM can decrease the level of TNF-α and
IL-6, downregulate the expression of MLCK, inhibit the
phosphorylation of MLC and improve the epithelial TJ so as to
reduce intestinal mucosal permeability, indicating that AM possibly
improves the permeability of the intestinal mucosa by inhibiting
the expression of pro-inflammatory cytokines and reducing MLCK and
p-MLC levels. Due to the complexity of the signaling pathway, only
the MLCK-P-MLC pathway, which is closely associated with colonic
mucosa permeability, was examined in the present study. Whether AM
can mediate intestinal barrier dysfunction via other molecules,
will be further investigated in subsequent experiments.
In conclusion, the current study demonstrated that
AM treatment can improve intestinal mucosa permeability by
downregulating the expression of proinflammatory cytokines and
inhibiting activation of the MLCK-p-MLC pathway so as to ameliorate
intestinal barrier function. The suppressive effects of AM on p-MLC
may represent a mechanism responsible for the beneficial effects of
AM. These findings further explain the pathogenesis of UC and may
provide a new therapeutic strategy for the treatment of UC.
Acknowledgments
This study was supported by the Health and Family
Planning Commission of Hubei Province (grant no. 2013Z-Y06).
References
|
1
|
Turner JR: Intestinal mucosal barrier
function in health and disease. Nat Rev Immunol. 9:799–809. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Marchiando AM, Graham WV and Turner JR:
Epithelial barriers in homeostasis and disease. Annu Rev Pathol.
5:119–144. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Munkholm P, Langholz E, Hollander D,
Thornberg K, Orholm M, Katz KD and Binder V: Intestinal
permeability in patients with Crohn's disease and ulcerative
colitis and their first degree relatives. Gut. 35:68–72. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ashizuka S, Inagaki-Ohara K, Kuwasako K,
Kato J, Inatsu H and Kitamura K: Adrenomedullin treatment reduces
intestinal inflammation and maintains epithelial barrier function
in mice administered dextran sulphate sodium. Microbiol Immunol.
53:573–581. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gardiner KR, Anderson NH, Rowlands BJ and
Barbul A: Colitis and colonic mucosal barrier dysfunction. Gut.
37:530–535. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Clayburgh DR, Shen L and Turner JR: A
porous defense: The leaky epithelial barrier in intestinal disease.
Lab Invest. 84:282–291. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gibson PR: Increased gut permeability in
Crohn's disease: Is TNF the link? Gut. 53:1724–1725. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nusrat A, Turner JR and Madara JL:
Molecular physiology and pathophysiology of tight junctions. IV
Regulation of tight junctions by extracellular stimuli: Nutrients,
cytokines, and immune cells. Am J Physiol Gastrointest Liver
Physiol. 279:G851–G857. 2000.PubMed/NCBI
|
|
9
|
Ye D, Ma I and Ma TY: Molecular mechanism
of tumor necrosis factor-alpha modulation of intestinal epithelial
tight junction barrier. Am J Physiol Gastrointest Liver Physiol.
290:G496–G504. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gu L, Li N, Gong J, Li Q, Zhu W and Li J:
Berberine ameliorates intestinal epithelial tight-junction damage
and down-regulates myosin light chain kinase pathways in a mouse
model of endotoxinemia. J Infect Dis. 203:1602–1612. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Costantini TW, Loomis WH, Putnam JG, et
al: Pentoxifylline modulates intestinal tight junction signaling
after burn injury: Effects on myosin light chain kinase. J Trauma.
66:17–24; discussion 24–25. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Shen L: Tight junctions on the move:
Molecular mechanisms for epithelial barrier regulation. Ann NY Acad
Sci. 1258:9–18. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu X, Xu J, Mei Q, Han L and Huang J:
Myosin light chain kinase inhibitor inhibits dextran sulfate
sodium-induced colitis in mice. Dig Dis Sci. 58:107–114. 2013.
View Article : Google Scholar
|
|
14
|
Elsasser TH and Kahl S: Adrenomedullin has
multiple roles in disease stress: Development and remission of the
inflammatory response. Microsc Res Tech. 57:120–129. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sakata J, Asada Y, Shimokubo T, Kitani M,
Inatsu H, Kitamura K, Kangawa K, Matsuo H, Sumiyoshi A and Eto T:
Adrenomedullin in the gastrointestinal tract. Distribution and gene
expression in rat and augmented gastric adrenomedullin after
fasting. J Gastroenterol. 33:828–834. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ashizuka S, Ishikawa N, Kato J, Yamaga J,
Inatsu H, Eto T and Kitamura K: Effect of adrenomedullin
administration on acetic acid-induced colitis in rats. Peptides.
26:2610–2615. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kataoka Y, Miyazaki S, Yasuda S, Nagaya N,
Noguchi T, Yamada N, Morii I, Kawamura A, Doi K, Miyatake K, et al:
The first clinical pilot study of intravenous adrenomedullin
administration in patients with acute myocardial infarction. J
Cardiovasc Pharmacol. 56:413–419. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Gonzalez-Rey E, Fernandez-Martin A, Chorny
A and Delgado M: Therapeutic effect of urocortin and adrenomedullin
in a murine model of Crohn's disease. Gut. 55:824–832. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Talero E, Alvarez de Sotomayor M,
Sánchez-Fidalgo S and Motilva V: Vascular contribution of
adrenomedullin to microcirculatory improvement in experimental
colitis. Eur J Pharmacol. 670:601–607. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hayashi Y, Narumi K, Tsuji S, Tsubokawa T,
Nakaya MA, Wakayama T, Zuka M, Ohshima T, Yamagishi M and Okada T:
Impact of adrenomedullin on dextran sulfate sodium-induced
inflammatory colitis in mice: Insights from in vitro and in vivo
experimental studies. Int J Colorectal Dis. 26:1453–1462. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Carrizo GJ, Wu R, Cui X, Dwivedi AJ, Simms
HH and Wang P: Adrenomedullin and adrenomedullin-binding protein-1
down-regulate inflammatory cytokines and attenuate tissue injury
after gut ischemia-reperfusion. Surgery. 141:245–253. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zuo D, Liu X, Shou Z, Fan H, Tang Q, Duan
X, Cao D, Zou Z and Zhang L: Study on the interactions between
transplanted bone marrow-derived mesenchymal stem cells and
regulatory T cells for the treatment of experimental colitis. Int J
Mol Med. 32:1337–1344. 2013.PubMed/NCBI
|
|
23
|
Li X, Wang Q, Xu H, Tao L, Lu J, Cai L and
Wang C: Somatostatin regulates tight junction proteins expression
in colitis mice. Int J Clin Exp Pathol. 7:2153–2162.
2014.PubMed/NCBI
|
|
24
|
Greig E and Sandle GI: Diarrhea in
ulcerative colitis. The role of altered colonic sodium transport.
Ann NY Acad Sci. 915:327–332. 2000. View Article : Google Scholar
|
|
25
|
Schmitz H, Barmeyer C, Fromm M, Runkel N,
Foss HD, Bentzel CJ, Riecken EO and Schulzke JD: Altered tight
junction structure contributes to the impaired epithelial barrier
function in ulcerative colitis. Gastroenterology. 116:301–309.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Fischer A, Gluth M, Pape UF, Wiedenmann B,
Theuring F and Baumgart DC: Adalimumab prevents barrier dysfunction
and antagonizes distinct effects of TNF-α on tight junction
proteins and signaling pathways in intestinal epithelial cells. Am
J Physiol Gastrointest Liver Physiol. 304:G970–G979. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ma TY, Boivin MA, Ye D, Pedram A and Said
HM: Mechanism of TNF-{alpha} modulation of Caco-2 intestinal
epithelial tight junction barrier: Role of myosin light-chain
kinase protein expression. Am J Physiol Gastrointest Liver Physiol.
288:G422–G430. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Marchiando AM, Shen L, Graham WV, Weber
CR, Schwarz BT, Austin JR 2nd, Raleigh DR, Guan Y, Watson AJ,
Montrose MH, et al: Caveolin-1-dependent occludin endocytosis is
required for TNF-induced tight junction regulation in vivo. J Cell
Biol. 189:111–126. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sommer J, Engelowski E, Baran P, Garbers
C, Floss DM and Scheller J: Interleukin-6, but not the
interleukin-6 receptor plays a role in recovery from dextran sodium
sulfate-induced colitis. Int J Mol Med. 34:651–660. 2014.PubMed/NCBI
|
|
30
|
Irvine EJ and Marshall JK: Increased
intestinal permeability precedes the onset of Crohn's disease in a
subject with familial risk. Gastroenterology. 119:1740–1744. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wyatt J, Vogelsang H, Hübl W, Waldhöer T
and Lochs H: Intestinal permeability and the prediction of relapse
in Crohn's disease. Lancet. 341:1437–1439. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang F, Graham WV, Wang Y, Witkowski ED,
Schwarz BT and Turner JR: Interferon-gamma and tumor necrosis
factor-alpha synergize to induce intestinal epithelial barrier
dysfunction by up-regulating myosin light chain kinase expression.
Am J Pathol. 166:409–419. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Taylor CT, Dzus AL and Colgan SP:
Autocrine regulation of epithelial permeability by hypoxia: Role
for polarized release of tumor necrosis factor alpha.
Gastroenterology. 114:657–668. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ueda S, Nishio K, Minamino N, Kubo A, Akai
Y, Kangawa K, Matsuo H, Fujimura Y, Yoshioka A, Masui K, et al:
Increased plasma levels of adrenomedullin in patients with systemic
inflammatory response syndrome. Am J Respir Crit Care Med.
160:132–136. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Higuchi S, Wu R, Zhou M, Marini CP,
Ravikumar TS and Wang P: Gut hyperpermeability after ischemia and
reperfusion: Attenuation with adrenomedullin and its binding
protein treatment. Int J Clin Exp Pathol. 1:409–418.
2008.PubMed/NCBI
|