Introduction
Brain metastasis is a common cause of mortality in
patients with cancer. It is estimated that 20–40% of patients with
systemic cancer develop metastases to the central nervous system
(CNS) (1). Brain metastases are
associated with a median survival rate of 4–6 months, despite
treatment with surgery, radiation therapy, and chemotherapy
(2). Targeted toxins (TT) are
extremely potent agents that recognize specific antigens located on
tumor cells, and cause cytotoxicity following cell entry, via
protein synthesis inhibition (3,4).
Previous studies have shown that TT may be appropriate for the
treatment of patients with intracranial metastatic disease or with
radio-resistant tumor histology (4,5).
Unfortunately, due to the large size of TT and the presence of the
partially intact blood-brain barrier (BBB), these compounds poorly
penetrate the CNS, which ultimately influences the therapeutic
efficacy of the delivery system (4,6).
Convection-enhanced delivery (CED) represents a potential approach
for the delivery of TT to the brain, by bypassing the BBB (6,7).
Numerous pre-clinical and early clinical studies have demonstrated
that CED is a useful modality for regional drug delivery in the
treatment of brain tumors (8–10).
In order to develop improved treatment strategies and to improve
the outcome for brain metastasis, there is a current requirement
for the identification of relevant metastatic brain tumor models,
which may be used to test novel therapeutic agents and delivery
systems in pre-clinical studies. When using intracranial xenograft
models, therapeutic efficacy is usually measured by bioluminescence
imaging (BLI), which allows for noninvasive, quantitative, and
real-time monitoring of tumor growth and response to therapy in
small animal models (11–13).
The present study aimed to develop a metastatic
brain tumor xenograft model in athymic mice, using a micro-osmotic
pump system for CED of TT, which could be monitored by BLI. This
model system adapted for BLI may prove useful for pre-clinical
testing of therapeutics for the treatment of patients with
metastatic brain tumors.
Materials and methods
Cell culture
The PC9-BrM3 non-small cell lung cancer human
metastatic brain tumor cell line was obtained from the Memorial
Sloan-Kettering Cancer Center (New York, NY, USA) (14). A luciferase reporter gene (Clontech
Laboratories, Inc., Mountain View, CA, USA) was inserted, according
to a previously described method (11). The cells were cultured in
Dulbecco's modified Eagle's medium supplemented with 10% fetal
bovine serum (Thermo Fisher Scientific, Inc., Logan, UT, USA), and
5% streptomycin (Mediatech, Inc., Manassas, VA, USA). The cells
were grown as monolayers and incubated in a humidified 37°C
atmosphere containing 5% CO2. As determined by Trypan
blue (Thermo Fisher Scientific, Inc.) exclusion, only cells with
>95% viability were used for subsequent experiments.
Animals
Six week old female athymic mice weighing 17–19 g
were purchased from the National Cancer Institute (Hudson, NY,
USA). The mice were housed in an animal facility and were
maintained in a temperature-controlled and light-controlled
environment, with an alternating 12 h light/dark cycle. All
surgeries were performed under sterile conditions and all animal
experimental protocols were approved by the animal care and use
committee of SUNY Upstate Medical University (Syracuse, NY,
USA).
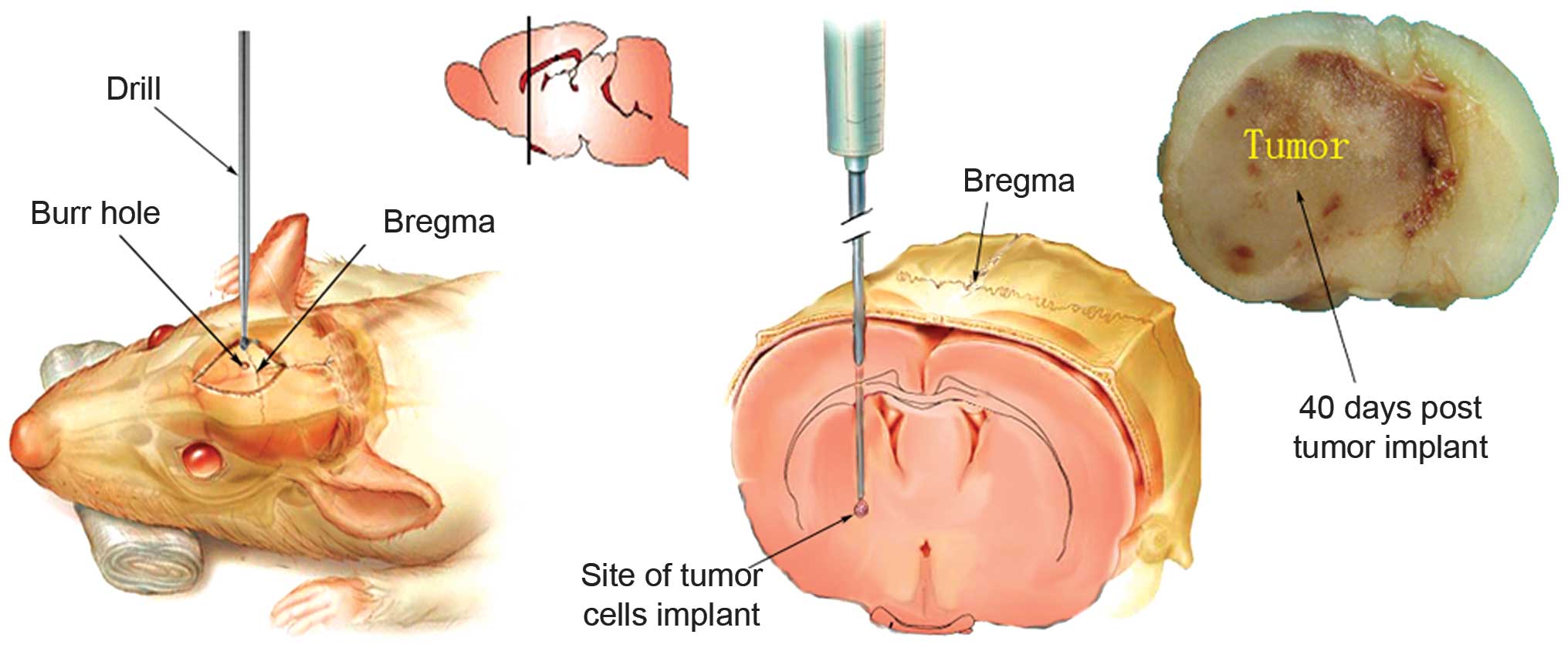
Surgical procedure for implantation of
tumor cells
For intracranial tumor injections, the mice were
anesthetized with an intraperitoneal injection of ketamine solution
(80 mg/kg; Sigma-Aldrich, St. Louis, MO, USA). The anesthetized
mice were then placed into the ear bars of a stereotactic frame
(David Kopf Instruments, Tujunga, CA, USA), as previously described
(9). The scalp was swabbed with
povidone-iodide (PDI, Orangeburg, NY, USA) and a midline incision
was made using a scalpel. A burr hole was drilled through the skull
0.5 mm anterior to the bregma and 2.5 mm lateral to the midline
using a drill (Foredom Electric Co., Bethel, CT, USA). Using the
stereotactic frame a 10 µl Hamilton syringe attached to a 25
gauge needle was used to deliver the tumor cells to a 3.0 mm depth
from the skull surface into the middle of the caudate-putamen
(Fig. 1). Following intracranial
inoculation with 1.4 µl 4×105 metastatic brain
tumor cells over a period of 5 min, the needle was left in place
for a further 5 min prior to being slowly withdrawn. The burr hole
was covered with sterile bone wax (Ethicon, Somerville, NJ, USA),
and the incision was closed with surgical glue.
Growth kinetics of intracranial tumor
xenografts and determination of intracranial tumor volumes
The brains of 16 mice were injected with
4×105 luciferase-modified PC9-BrM3 metastatic brain
tumor cells, and intratumoral luciferase activity was monitored by
BLI. BLI was performed once every 5 days beginning at day five
following tumor cell implantation. Four mice were sacrificed at
days 10, 20, 30, and 40 following implantation and immediately
following BLI. The mice were sacrificed with an overdose of inhaled
CO2, and their brains were removed and placed in 4%
formalin immediately afterwards. The formalin-fixed murine brains
were subsequently deposited in 20% sucrose solution at 4°C,
embedded in paraffin, sectioned at 200 µm intervals, and
then stained with hematoxylin and eosin (HE; Sigma-Aldrich). The
histological analyses of the serial microscopy sections were
performed using a Leica DMR microscope (Leica Microsystems GmbH,
Wetzlar, Germany). The digital images of the tumor-containing
sections were then examined for each mouse in order to determine
the histopathological tumor volumes.
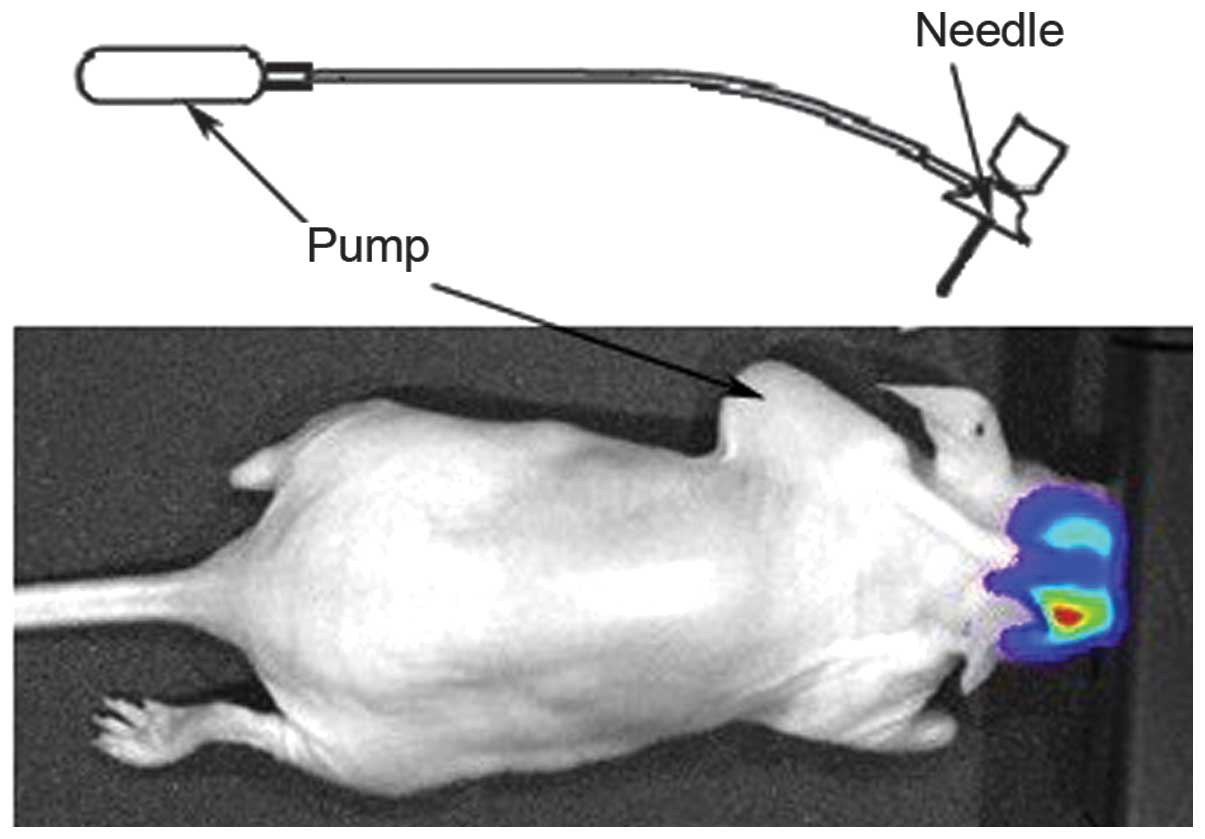
Micro-osmotic pump implantation and
treatment of metastatic brain tumors with TT in mice
The osmotic mini-pump system (ALZET model 1007D;
Durect Corporation, Cupertino, CA, USA) consisted of four
components: A micro-osmotic pump, a flow moderator, a catheter
tube, and a brain infusion cannula (Fig. 2). In order to evaluate the
therapeutic response of the metastatic brain tumor xeno-grafts to
TT, 12 mice that received the caudate-putamen implantation of
luciferase-modified brain metastatic tumor cells were randomly
assigned to either TT (DTATEGF) treatment or control groups. The
mice were assigned to the groups following the initial BLI image
examination, which was performed 4 days following tumor cell
implantation. The pump systems were assembled for intracranial
infusion according to the manufacturer's instructions, and both a
25 gauge 3 mm brain infusion needle and a 15 mm catheter tube were
attached to the pump. The drug-loaded pumps were implanted
subcutaneously into the mouse back. In the treatment group, each
pump reservoir was filled with 1 µg TT in 100 µl
solution. DTATEGF TT is a novel recombinant bispecific TT
consisting of a truncated diphtheria toxin (DT), an aminoterminal
(AT) fragment of the urokinase-type plasminogen activator, and a
fragment of human epidermal growth factor (EGF), and was developed
in our laboratory (5). In the
control group, the pumps were filled with 100 µl BIC3KDEL
(University of Minnesota, Minneapolis, MN, USA), a T-cell targeting
control toxin. The pump flow rate was set to 0.5 µl/h, and
the solutions were delivered continuously for 7 days. On day eight
following pump implantation, the mice were anesthetized again and
all components of the pump system were removed. The mice were
monitored each day for the development of symptoms associated with
tumor growth and pump implantation. Following tumor cell
implantation, the mice were monitored by BLI at days 4, 14, 30, 60,
and 90. The mean BLI values for the treatment and control groups
were then calculated and plotted according to the corresponding day
of imaging. The mice were sacrificed by CO2 inhalation
when they demonstrated physical symptoms indicative of significant
compromise to neurological function, or following >20% body
weight loss.
In vivo BLI of intracranial tumors
In vivo BLI was performed using the IVIS 50
Imaging system (Caliper Life Sciences, Alameda, CA, USA), which
captured the luminescence signal emitted from the engrafted tumor.
The data were analyzed using Living Image 2.5 software (Caliper
Life Sciences). Prior to imaging, the mice were anesthetized with
inhalation of isoflurane gas, and injected intraperitoneally with
150 mg/kg D-luciferin aqueous solution (PerkinElmer, Inc., Waltham,
MA, USA). The images were subsequently captured 10 min following
injection. Signal intensity was quantified within a region of
interest over the head, as defined by Living Image software. All
images represent 2 min exposure time, and the total numbers of
photons/sec/cm2/steradian were recorded.
Statistical analysis
The data were statistically analyzed with a
Student's t-test using commercially available software (GraphPad
Prism; GraphPad Software, Inc. La Jolla, CA, USA). The statistical
differences between each comparison were calculated using a
log-rank test. The correlation between bioluminescence and
corresponding tumor volume was calculated using a linear regression
analysis. P<0.05 was considered to indicate a statistically
significant difference.
Results
General outcomes
Injection of luciferase-modified human metastatic
brain tumor cells into athymic mice resulted in 100% tumor growth.
During all experimental procedures, no morbidity or mortality was
observed in response to the pump system or the surgical procedure.
The mice implanted with pumps showed no evidence of clinical side
effects over the study period. No mice experienced irritation at
the wound site or attempted to remove the pump system. No
neurological deficits were observed immediately following cell
inoculation, pump implantation, or drug delivery. The pump system
never migrated from its implanted position, and all mice tolerated
the device well. In the latter stages of the experiments, mild
contralateral forelimb weakness was observed and gradually worsened
over time. The body weights of the mice began to significantly
decrease and the animals deteriorated rapidly in their last week of
life prior to being sacrificed, as a consequence of progressive
tumor growth.
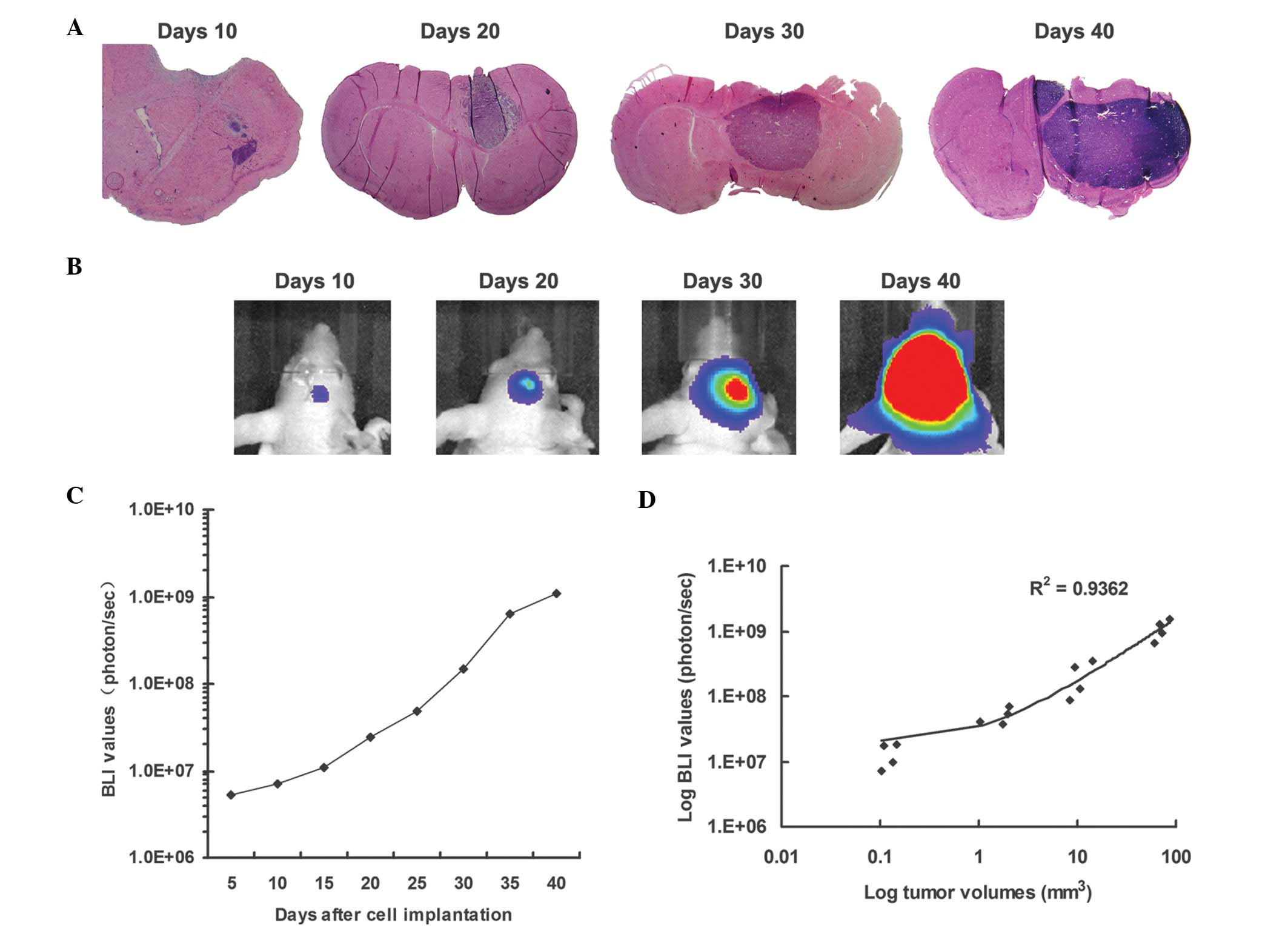
Real-time imaging of xenograft brain
metastases growth as measured by BLI
In a time course study involving tumor monitoring by
BLI, the mice were injected with luciferase-modified metastatic
brain tumor cells, and sacrificed at selected time points following
implantation. Bioluminescence was readily detectable on day five
following tumor cell implantation, and the normalized luminescence
plot showed progressively increasing luminescence until the mice
developed neurological symptoms. Small tumors were detected on day
10, and tumor size increased up to day 40. The implanted tumor
cells exhibited logarithmic growth 10 days following tumor cell
implantation. On day 40, the presence of solid tumors was evident
in the right caudate-putamen, with extension over the brain surface
(Fig. 3A and B).
Correlation between bioluminescence and
corresponding tumor volume
The HE-stained sections of each tumor-bearing brain
were serially registered and used to estimate tumor volume. The
tumor volumes were compared with the corresponding luminescence
readings obtained prior to mouse sacrifice (Fig. 3C). Linear regression analyses
revealed a marked correlation (R2=0.94) between the
strength of luminescence and intracerebral tumor volume (Fig. 3C).
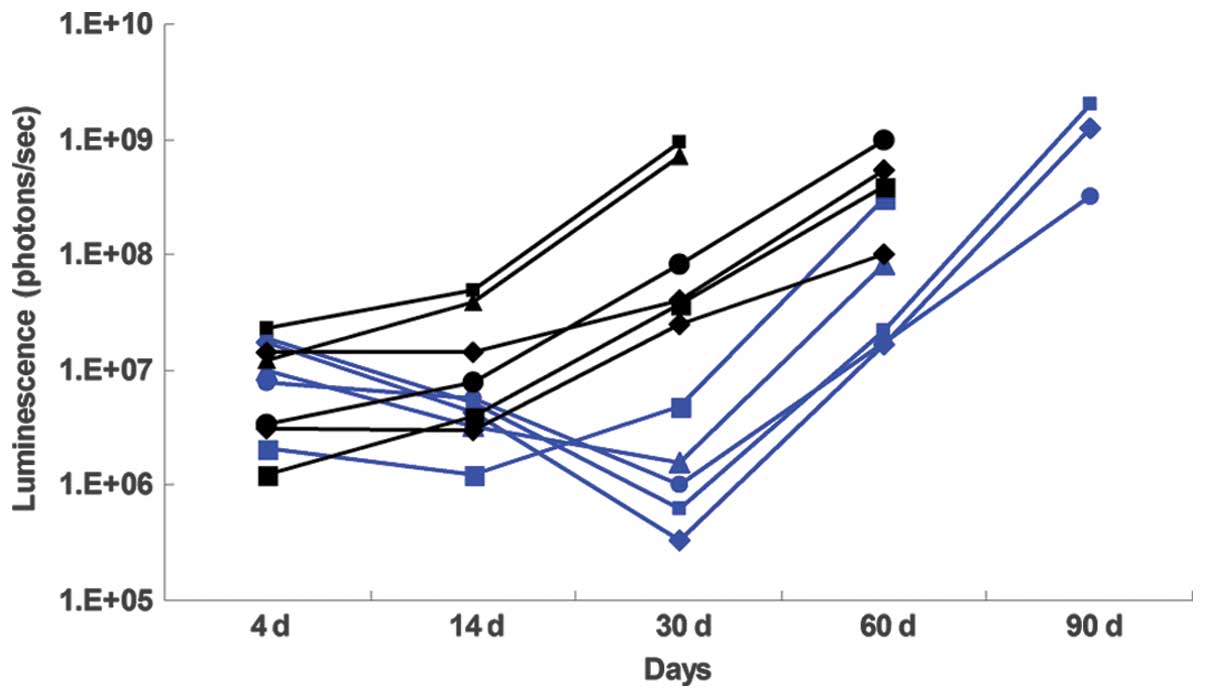
TT intracranial metastatic tumor
efficacy
BLI monitoring was used to evaluate the therapeutic
response of intracranial metastatic brain tumor xenografts. The
control group normalized luminescence plot showed a progressive
increase in luminescence until the mice developed neurological
symptoms such as moribund state, which was used an indicator for
sacrifice. Luminescence in the TT (DTATEGF)-treated mice was
substantially reduced at day 14 when the treatment was completed,
and remained low until day 30 following tumor cell implantation, as
compared with the control group (Fig.
4). Survival analyses revealed that the median survival
duration of the TT-treated mice was significantly extended, as
compared with the control group (87 days, vs. 63 days,
P=0.006).
Discussion
The systemic delivery of TT to the CNS is restricted
due to the inability of numerous compounds to cross the BBB, and
the systemic toxicity of TT. Currently, a useful modality for
regional drug delivery for brain tumor treatment is CED (6,8,10).
CED is a strategy pioneered by Bobo et al (15) whereby drugs are delivered directly
into the tumor or adjacent brain parenchyma via implanted
catheters. CED bypasses the BBB and unlike systemic therapy,
decreases the potential for systemic toxicity. Previous studies
have described the distribution and concentration of
chemotherapeutic agents delivered via CED in vivo (16–18).
Previous studies have demonstrated the efficacy of
CED-delivered TT (10,12,13).
Numerous studies have been performed using CED via a syringe or
shunt catheter, where high flow rates are used to infuse large
volumes of drug over a short period of time. The results of these
studies demonstrated that the infusion may create a deforming force
on the underlying tissue, which may in turn cause tearing of the
tissue or reflux of the infusate along the catheter track, away
from the target tissue (19–22).
In the murine model generated in the present study,
CED was performed via a micro-osmotic pump system, to provide a
continuous positive-pressure microinfusion, a method that has
numerous advantages. Firstly, a drug may be continuously delivered
at a constant rate for days without the need for anesthesia or the
frequent handling of small animals. In addition, the small,
flexible pump system may be fixed to the skull and therefore will
not interfere with the normal activities of the mice. The system
does not require an external syringe or shunt catheter, thus
decreasing the chance for infection. Furthermore, the pump is fully
biocompatible and the infusion volume is well tolerated. A DT-based
TT delivery by CED was previously shown to exhibit immunological
privilege (9). Therefore, the
model used in the present study is a simple and effective tool for
performing CED treatment experiments in small animals with brain
tumors.
Quantitative measurements of therapy-induced changes
in tumor growth have been difficult to obtain in animal models, as
they require the sacrifice of large numbers of animals at multiple
time points. Numerous non-invasive approaches to imaging tumor
growth in small animals have become available, including magnetic
resonance imaging (MRI), computerized tomography (CT), positron
emission tomography (PET), single photon emission computed
tomography (SPECT), and ultrasonography (23–25).
Each technique has its own advantages and disadvantages. MRI, CT,
and ultrasonography produce anatomical images of structures in the
body, whereas PET and SPECT image physiological processes and thus
produce functional images. These imaging modalities are expensive
and require technical expertise. Therefore, the use of two
relatively novel alternative non-invasive approaches, fluorescence
imaging and BLI, is expanding rapidly (11,25).
BLI detects the emission of photons based on energy-dependent
reactions catalyzed by cells or organisms that have been
genetically modified to express luciferase. BLI is a
cost-effective, efficient, and accurate means for assessing tumor
growth and response to therapy (11). The applications for BLI have been
numerous, and include monitoring tumor cell growth, visualizing
tumor regression in response to chemotherapy, determining cell
migration, and monitoring the locations as well as the timing of
promoter-dependent luciferase expression (11,12,13,25).
In the present study, the bioluminescence signal was readily
detectable on day 5 following tumor cell implantation, and brain
tumor growth could be monitored in real time via BLI. Since this
approach involved the direct physical measurement of intracranial
tumor for comparison with the BLI results, the extent of the
correlation between tumor volume and the strength of the observed
luminescence signal was high. With regards to treatment response,
the anti-tumor effect of TT was readily detected by BLI, and the
effects of the treatment detected by BLI indicated a corresponding
increase in animal survival. These results suggested that BLI is a
rapid, easy to perform, sensitive, and suitable method to establish
early tumor response to treatment, and may be used as an accurate
model to assess the effect of therapy on survival. Although this
monitoring method has only been validated in experimental models,
the results of the present study highlight the potential value of
this technique in the rapid evaluation of treatment response in
pre-clinical oncology trials. This model may be used in further
studies that measure therapeutic index and pharmacokinetics, in
order to determine whether other means of drug delivery are
superior.
Although the efficacy of a bispecific TT was not the
focus of the present study, the results demonstrated that TT could
be delivered by CED via a micro-osmotic pump system to the
intracranial metastatic brain tumor and delay tumor growth, as
indicated by BLI monitoring, and could also significantly extend
the survival of the animal subjects. Based on these results, TT is
a reasonable treatment alternative that may be used when radiation
therapy fails due to systemic toxicity or tumor resistance, and
further study regarding this promising agent is warranted.
In conclusion, the present study demonstrated the
development of a novel metastatic brain tumor xenograft model for
CED of TT via a micro-osmotic pump system that was measured by
real-time BLI, which in turn facilitates the pre-clinical testing
of novel therapies for the treatment of cancer. The results
obtained from the present study indicated that this xenograft model
has numerous unique features that may prove useful in the
investigation of the pathobiology of intracranial tumor growth, and
for monitoring systemic and intracranial responses to anti-tumor
agents. Notably, the results demonstrated that TT delivered by CED
via a micro-osmotic pump system is an effective drug against
metastatic tumor growth, and that BLI accurately defines metastatic
tumor growth and is able to demonstrate tumor response to TT. This
metastatic brain tumor xenograft model combined with CED and BLI
technology may serve as a valuable tool for preclinical screening
for drugs effective in targeting metastatic brain tumors.
Acknowledgments
The present study was supported by the Scientific
Research Foundation for Selected Overseas Chinese Scholars, State
Education Ministry (2013); and the Technology Foundation for
Selected Overseas Chinese Scholar, Ministry of Human Resources and
Social Security of the People's Republic of China (2014).
References
|
1
|
Arnold SM and Patchell RA: Diagnosis and
management of brain metastases. Hematol Oncol Clin North Am.
15:1085–1107. 2001. View Article : Google Scholar
|
|
2
|
Gaspar L, Scott C, Rotman M, et al:
Recursive partitioning analysis (RPA) of prognostic factors in
three Radiation Therapy Oncology Group (RTOG) brain metastases
trials. Int J Radiat Oncol Biol Phys. 37:745–751. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hall WA: Immunotoxin therapy. Neurosurg
Clin N Am. 7:537–546. 1996.PubMed/NCBI
|
|
4
|
Rustamzadeh E, Low WC, Vallera DA and Hall
WA: Immunotoxin therapy for CNS tumor. J Neurooncol. 64:101–116.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Huang J, Li YM, Massague J, Sicheneder A,
Vallera DA and Hall WA: Intracerebral infusion of the bispecific
targeted toxin DTATEGF in a mouse xenograft model of a human
metastatic non-small cell lung cancer. J Neurooncol. 109:229–238.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hall WA: Convection-enhanced delivery:
Neurosurgical issues. Curr Drug Targets. 10:126–130. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Vogelbaum MA: Convection enhanced delivery
for treating brain tumors and selected neurological disorders:
Symposium review. J Neurooncol. 83:97–109. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Vogelbaum MA, Sampson JH, Kunwar S, et al:
Convection-enhanced delivery of cintredekin besudotox
(interleukin-13-PE38QQR) followed by radiation therapy with and
without temozolomide in newly diagnosed malignant gliomas: Phase 1
study of final safety results. Neurosurgery. 61:1031–1038. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Oh S, Ohlfest JR, Todhunter DA, et al:
Intracranial elimination of human glioblastoma brain tumors in nude
rats using the bispecific ligand-directed toxin, DTEGF13 and
convection enhanced delivery. J Neurooncol. 95:331–342. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hall WA, Rustamzadeh E and Asher AL:
Convection-enhanced delivery in clinical trials. Neurosurg Focus.
14:e22003. View Article : Google Scholar
|
|
11
|
Rehemtulla A, Stegman LD, Cardozo SJ, et
al: Rapid and quantitative assessment of cancer treatment response
using in vivo bioluminescence imaging. Neoplasia. 2:491–495. 2000.
View Article : Google Scholar
|
|
12
|
Paroo Z, Bollinger RA, Braasch DA, et al:
Validating bioluminescence imaging as a high-throughput,
quantitative modality for assessing tumor burden. Mol Imaging.
3:117–124. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Szentirmai O, Baker CH, Lin N, et al:
Noninvasive bioluminescence imaging of luciferase expressing
intracranial U87 xenografts: Correlation with magnetic resonance
imaging determined tumor volume and longitudinal use in assessing
tumor growth and antiangiogenic treatment effect. Neurosurgery.
58:365–372. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Nguyen DX, Chiang AC, Zhang XH, et al:
WNT/TCF signaling through LEF1 and HOXB9 mediates lung
adenocarcinoma metastasis. Cell. 138:51–62. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bobo RH, Laske DW, Akbasak A, Morrison PF,
Dedrick RL and Oldfield EH: Convection-enhanced delivery of
macromolecules in the brain. Proc Natl Acad Sci USA. 91:2076–2080.
1994. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sampson JH, Brady ML, Petry NA, et al:
Intracerebral infusate distribution by convection-enhanced delivery
in humans with malignant gliomas: Descriptive effects of target
anatomy and catheter positioning. Neurosurgery. 60:ONS89–ONS99.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mamot C, Nguyen JB, Pourdehnad M, et al:
Extensive distribution of liposomes in rodent brains and brain
tumors following convection-enhanced delivery. J Neurooncol.
68:1–9. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Saini M, Roser F, Samii M and Bellinzona
M: A model for intratumoural chemotherapy in the rat brain. Acta
Neurochir (Wien). 146:731–734. 2004. View Article : Google Scholar
|
|
19
|
Olson JJ, Zhang Z, Dillehay D and Stubbs
J: Assessment of a balloon-tipped catheter modified for
intracerebral convection-enhanced delivery. J Neurooncol.
89:159–168. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Morrison PF, Chen MY, Chadwick RS, Lonser
RR and Oldfield EH: Focal delivery during direct infusion to brain:
Role of flow rate, catheter diameter, and tissue mechanics. Am J
Physiol. 277:R1218–R1229. 1999.PubMed/NCBI
|
|
21
|
Griffitt W, Glick RP, Lichtor T, Haughton
DE and Cohen EP: Development of a new mouse brain tumor model using
implantable micro-cannulas. J Neurooncol. 41:117–120. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Morreale VM, Herman BH, Der-Minassian V,
et al: A brain-tumor model utilizing stereotactic implantation of a
permanent cannula. J Neurosurg. 78:959–965. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ray P, Wu AM and Gambhir SS: Optical
bioluminescence and positron emission tomography imaging of a novel
fusion reporter gene in tumor xenografts of living mice. Cancer
Res. 63:1160–1165. 2003.PubMed/NCBI
|
|
24
|
Dickinson PJ, LeCouteur RA, Higgins RJ, et
al: Canine model of convection-enhanced delivery of liposomes
containing CPT-11 monitored with real-time magnetic resonance
imaging: Laboratory investigation. J Neurosurg. 108:989–998. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Shah K, Bureau E, Kim DE, et al: Glioma
therapy and real-time imaging of neural precursor cell migration
and tumor regression. Ann Neurol. 57:34–41. 2005. View Article : Google Scholar
|