Introduction
End-stage liver disease is associated with clear
patient mortality (1), and
orthotopic liver transplantation is the last resort for its
treatment. However, due to factors including the shortage of
donors, immune rejection and a high cost, the clinical use of
orthotopic liver transplantation is restricted (2). Therefore, alternative treatment
strategies are urgently required. The activation of hepatic
stellate cells (HSCs) and subsequent secretion of large amounts of
collagen is considered to be the core mechanism of liver fibrosis
(3,4). Bone marrow-derived mesenchymal stem
cells (BM-MSCs) possess a wide range of clinical applications in
the field of cell therapy and tissue engineering due to their high
plasticity, low immunogenicity, rapid amplification rate in
vitro and stable genetic background (5).
Liver fibrosis is an early stage of liver cirrhosis,
therefore reversing liver fibrosis holds clear clinical potential.
An optimal treatment would prevent the development of liver
fibrosis, in addition to inhibiting the formation of scar tissue in
the liver, in order to stabilize and improve liver function
(6). Previous studies have
suggested that BM-MSCs may serve a role in the treatment of liver
fibrosis (7–9). A previous study identified that the
supernatant of cultured BM-MSCs is able to inhibit the expression
of matrix metalloproteinase-2 (MMP-2) and tissue inhibitor of
matrix metalloproteinase-1 (TIMP-1) in HSCs (10). These results indirectly confirmed
that BM-MSCs are able to influence the proliferation of HSCs via
the exocrine mechanism. However, it remains unclear which cytokines
are secreted by BM-MSCs and which signal transduction pathways are
used in HSCs. The current study aimed to further elucidate whether
BM-MSCs are able to inhibit the proliferation of HSCs, then further
explore alterations in transforming growth factor (TGF)-β1 during
co-culture of BM-MSCs with HSCs.
Materials and methods
Materials
The human HSC cell line (LX2) was donated by
Professor Scott L. Friedman from Icahn School of Medicine at Mount
Sinai (New York City, NY, USA). Low glucose-Dulbecco's modified
Eagle's medium (L-DMEM) and fetal bovine serum (FBS) (GE Healthcate
Life-Sciences, Logan, UT, USA); MTT reagent cartridge
(Sigma-Aldrich, St. Louis, MO, USA); the TGF-β1 ELISA reagent
cartridge (R&D Systems, Inc., Minneapolis, MN, USA); rabbit
anti-human TGF-β1 (1:500 dilution; cat. no. BS1361) and rabbit
anti-human β-actin (1:5,000 dilution; cat. no. AP0733) polyclonal
antibodies (Bioworld Technology, Inc., St. Louis Park, MN, USA),
monoclonal mouse anti-human Smad7 antibody (cat. no. ab55493;
Abcam, Pak Shek Kok, Hong Kong); horseradish peroxidase
(HRP)-conjugated goat anti-rabbit IgG polyclonal antibody (1:5,000;
cat. no. ab6741; Bioworld Technology, Inc.); RNA extraction reagent
RNAiao Plus (Takara Bio, Inc., Otsu, Japan);
PrimeScript® RT-PCR kit (Takara Bio, Inc.); enhanced
chemiluminescence (ECL) light kit (Thermo Fisher Scientific, Inc.,
Waltham, MA, USA); polyvinylidene difluoride (PVDF) membrane (Merck
Millipore, Darmstadt, Germany); semipermeable Transwell insert film
(Corning Incorporated, Corning, NY, USA); Hoechst dye
(Sigma-Aldrich); bicinchoninic acid (BCA) protein concentration
assay kit (Beyotime Institute of Biotechnology, Shanghai, China);
protein lysate (Beyotime Institute of Biotechnology).
Culture of human BM-MSCs and HSCs
Human BM-MSCs and HSCs were cultured in L-DMEM
containing 10% FBS at 37°C in a 5% CO2 incubator. Cells
were inoculated with a density of 1×105 cells/ml in
culture flasks with a base area of 25 cm2. When cells
were 80–90% confluent, they were washed with phosphate-buffered
saline (PBS), digested with 0.25% trypsin (Thermo Fisher
Scientific, Inc.) and subcultured. The growth and morphological
characteristics of BM-MSCs and HSCs (third passage) were examined
using an inverted phase contrast microscope (AE21; Motic China
Group Co., Ltd., Xiamen, China). The growth curve of the cells
cultured for 1–9 days was evaluated by MTT assay.
Cell co-culture
The upper and lower double-cell co-culture system
was established as previously described (11,12).
Cells were cultured in a 6-well plastic cell plate with 2 ml
BM-MSCs (10×105 cells/ml) in the upper semi-permeable
membrane (Transwell insert) and with 2 ml HSCs (1×105
cells/ml) in the lower membrane layer. Cells were divided into two
experimental groups: i) Control group, HSCs were cultured alone
(upper, L-DMEM media only); and ii) experimental group, BM-MSCs and
HSCs were co-cultured. The morphological alterations were examined
using an inverted phase contrast microscope at 24 and 48 h
subsequent to incubation.
MTT assay to detect the HSC proliferation
inhibition rate
Following co-culture of the cells for 24, 48 and 72
h, 400 µl MTT solution (5 g/l) was added to each well. Cells
were incubated for 4 h, and the culture medium was discarded. A
total of 2 ml dimethyl sulfoxide (Sigma-Aldrich) was added to each
well and the wells were agitated for 10 min in the dark. Once the
crystals had fully dissolved, cells were transferred to 96-well
plates, each well containing 150 µl and each group
consisting of 10 wells. The absorbance value of each well was
measured at 490 nm in an enzyme-linked immunosorbent monitor
(Thermo Fisher Scientific, Inc.) and recorded as the A value. The
cell growth inhibition rate was calculated as follows: (1 - A value
of experimental group / control group A value) ×100%.
Flow cytometry to detect apoptosis
Following co-culture for 24 or 48 h, the culture
medium was discarded. Cells were washed with PBS and digested with
0.25% trypsin (excluding EDTA). Cells were resuspended in PBS and
centrifuged for 5 min at 800 × g. The supernatant was discarded and
the cells were divided evenly. A total of 5 µl annexin
V-fluorescein isothiocyanate (FITC) (Beckman Coulter, Inc., Brea,
CA, USA) was added into each tube, which were then vortexed prior
to incubation in the dark at room temperature for 15 min. A total
of 5 µl propidium iodide (PI) dye was added to each tube
prior to detection for 5 min. Prior to detection, 200 µl 1X
binding buffer was added into the tubes.
Hoechst staining of apoptotic cell
bodies
Subsequent to co-culture for 24 and 48 h, the
culture medium was removed and 2 ml 4% formaldehyde was added into
each well. Cells were fixed for 15 min at room temperature and were
washed twice with PBS, for 5 min each time. HSCs were stained with
Hoechst dye for 6–9 min in the dark at room temperature. Apoptotic
cell bodies in HSCs were examined using a fluorescence microscope
(BX431-46; Olympus Corporation, Tokyo, Japan).
ELISA for TGF-β1 levels
Following co-culture for 24 and 48 h, the
supernatants from the upper and lower Transwell chambers were
collected and centrifuged at 750 x g for 20 min. TGF-β1
concentration was measured in accordance with the ELISA kit
(R&D Systems, Inc.) instructions.
Total RNA extraction of HSCs and reverse
transcription-quantitative polymerase chain reaction detection
HSCs were collected and counted at 48 h in each
group. The primer sequences used were as follows: β-actin, upstream
sequence TGGCACCCAGCACAATGAA and downstream sequence
CTAAGTCATAGTCCGCCTAGAAGCA; TGF-β1, upstream sequence
TGGAAACCCACAACGAAATC and downstream sequence GTATTTCTGGTACAGCTCCA;
Smad7, upstream sequence TCTGCGAACTAGAGTCTCCC and downstream
sequence ACGCACCAGTGTGACCGATC. The total RNA was extracted using
RNAiso Plus reagent and RNA integrity and concentration were
measured in the spectrophotometer (Tecan Group Ltd., Männedorf,
Switzerland). A total of 500 ng RNA was used to synthesize 10
µl cDNA using the PrimeScript® RT-PCR kit. The
reaction system volume was 10 µl. The reaction conditions
were as follows: 37°C for 15 min then 85°C for 5 sec. Genes were
amplified using a LightCycler amplification system (Bio-Rad
Laboratories, Inc., Hercules, CA, USA). The reaction system volume
was 20 µl. The reaction conditions were as follows: 95°C for
30 sec (20°C/sec) for 1 cycle, then at 95°C for 5 sec (20°C/sec)
and 60°C for 20 sec (20°C/sec) for 40 cycles. TGF-β1, Smad7 mRNA
expression levels were detected. The data were stated as
2−∆∆Ct.
HSCs total protein extraction and western
blot analysis
Following co-culture for 24 and 48 h, cells were
lysed and total protein extracted from HSCs. Protein content was
measured using the BCA protein concentration determination kit. The
sample quantity was 30 µg. Proteins were separated by
SDS-PAGE (10% gel; Bio-Rad Laboratories, Inc.), transferred to a
PVDF membrane, blocked with Tris-buffered saline and Tween 20 and
5% skimmed milk and incubated with anti-human TGF-β1 (1:500) at 4°C
overnight. The membrane was incubated with the HRP-conjugated
secondary antibody, then developed using ECL. Data were analyzed
using Image J 3.0 (Bio-Rad Laboratories, Inc.). The gray ratio of
TGF-β1 proteins and β-actin represented the relative level of the
target protein. Smad7 protein was detected as mentioned above.
Statistical analysis
SPSS software, version 18.0 (SPSS, Inc., Chicago,
IL, USA) was used for statistical analysis. The count data were
analyzed using the Chi-square test, measurement data were subjected
to Student's t-test. Experimental data are expressed as mean ±
standard deviation. P<0.05 was considered to indicate a
statistically significant difference.
Results
Culture of BM-MSCs and HSCs
HSCs were observed to be adherent and oval or
spindle-shaped, and cytoplasmic lipid droplets appeared following
inoculation for 2–3 h, with certain cells beginning to stretch out
during this time. Subsequent to culture of HSCs for 2–3 days, cells
were 80–90% confluent and could be sub-cultured. Lipid droplets in
the cytoplasm gradually reduced or disappeared, and cells displayed
pseudopodia. HSCs exhibiting rapid growth and proliferation when
passaged for 3–4 generations were used for the experiments
(Fig. 1A). Cultured BM-MSCs were
observed to be round or oval and were of varying sizes following
inoculation. Subsequent to the media being replaced, cell adherence
increased and the cells became uniform in shape, tightly packed,
multi-fusiform and began to merge into a sheet. When cells were
digested by trypsin, they became round, began to adhere, gradually
restoring their long spindle morphology over 2–3 h. Subsequent to
culture for 72 h, BM-MSCs were highly confluent, of uniform shape
and fused with typical swirling growth (Fig. 1B). The third passage cells were for
transplantation. The growth curve of HSCs and BM-MSC demonstrated
that HSCs grew faster than BM-MSCs (Fig. 1C).
Inhibitory effect of BM-MSC on HSCs
Hoechst staining revealed the number of apoptotic
bodies in the experimental group was significantly increased
compared with the control group (Fig.
2A–D). The inhibitory effect of BM-MSCs on HSCs was detected by
MTT assay following culture of the cells for 24, 48 and 72 h. The
absorbance values of the control group at 24, 48 and 72 h (at 490
nm) were 0.149±0.012, 0.405±0.007 and 3.146±0.033, respectively.
The absorbance values of the experimental group were 0.149±0.160,
0.358±0.007 and 0.986±0.020, respectively. There was no significant
difference between the control and experimental group at 24 h
(P=1.000), however the absorbance values at 48 and 72 h between the
two groups were significantly different (P<0.001). BM-MSCs
inhibited the proliferation of HSCs, with the inhibitory rates of
BM-MSCs on HSCs at 24, 48 and 72 h observed to be 0, 11.5 and
68.7%, respectively. BM-MSCs did not inhibit the proliferation of
HSCs at 24 h, however significantly inhibited the proliferation of
HSCs in a time-dependent manner at 48 and 72 h (Fig. 2E).
Apoptotic effect of BM-MSCs on HSCs
Following co-culture of BM-MSCs with HSCs for 24 and
48 h, cells were double stained with annexin V-FITC and PI to
detect the rate of apoptosis of HSCs using flow cytometry (Fig. 3A–D). The rate of apoptosis was not
identified to significantly differ between the control and
experimental groups at 24 h (2.08% for control group and 1.00% for
experimental group). However, the rate of apoptosis was
significantly increased in the experimental group at 48 h (7.51%
for control group and 10.28% for experimental group) (Fig. 3E).
Expression of TGF-β1 and SMAD7 mRNA
The TGF-β1 and Smad7 mRNA expression levels in the
control and experimental groups were as follows: Control group,
1.00±0.00 and 1.00±0.00, respectively; experimental group,
0.401±0.301 and 9.697±2.591, respectively. Following co-culture for
48 h, TGF-β1 mRNA in the experimental group was significantly lower
than in the control group, and Smad7 mRNA was significantly higher
than that in the control group (P<0.05; Fig. 4).
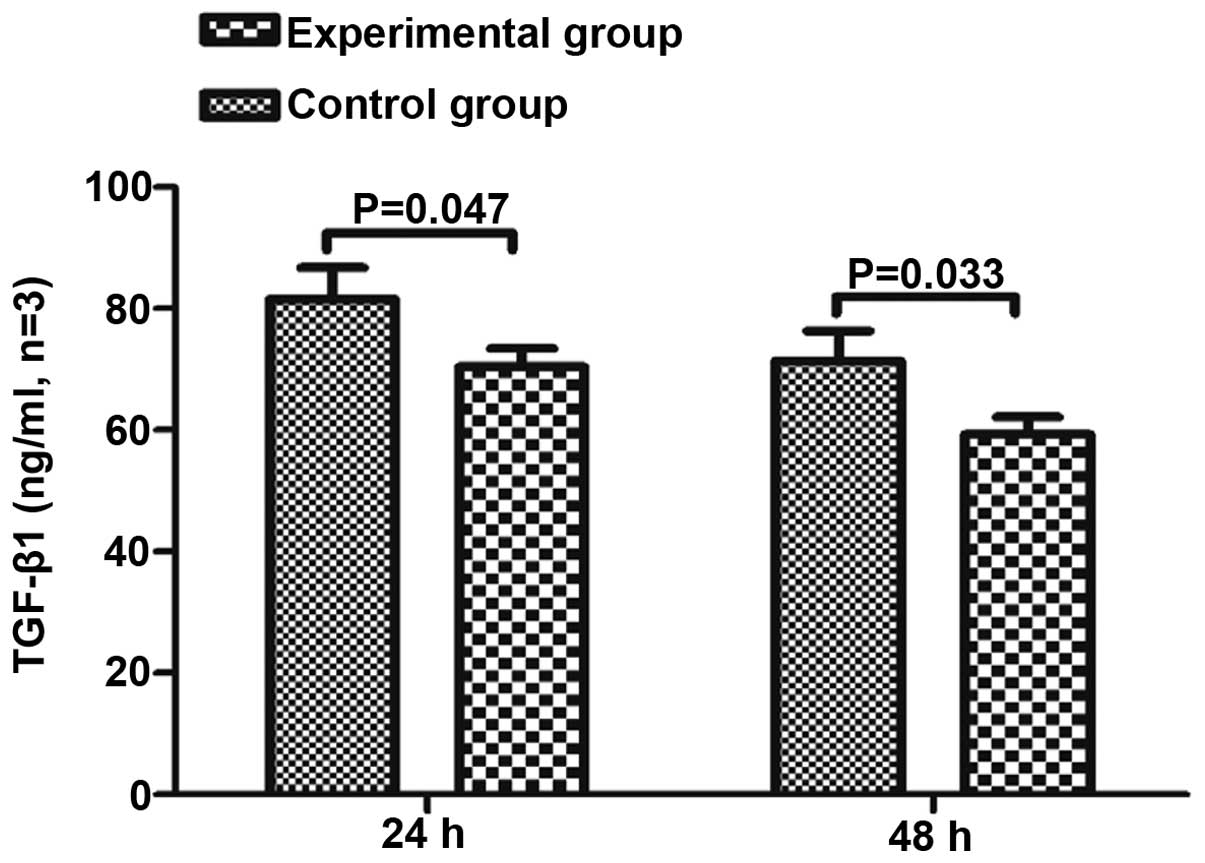
Concentration of TGF-β1 in the
supernatant
To clarify whether BM-MSCs impact the secretion of
TGF-β1 from HSCs, the TGF-β1 concentration in the supernatant was
measured in the BM-MSCs and HSCs co-culture group, BM-MSCs group
and the HSCs group at 24 and 48 h. The concentration of TGF-β1 in
the BM-MSCs single culture group was below the ELISA kits minimum
detection value, thus could not be measured. The concentration of
TGF-β1 in the co-culture group was significantly lower than in the
HSC single culture group at 24 and 48 h (P<0.05; Fig. 5).
Expression of TGF-β1 and Smad7
protein
The expression of TGF-β1 and Smad7 protein was
measured in the control group and experimental group at 24 and 48
h, and levels were observed to be significantly different between
the groups (Fig. 6). Expression of
TGF-β1 protein in the experimental group was lower than in the
control group at 24 and 48 h (24 h control group, 0.902±0.012; 24 h
experimental group, 0.854±0.018; 48 h control group, 0.715±0.053;
48 h experimental group, 0.532±0.066), and Smad7 protein levels
were higher than that of the control group (24 h control group,
0.661±0.043; 24 h experimental group, 1.134±0.059; 48 h control
group, 1.322±0.095; 48 h experimental group, 1.834±0.161).
Discussion
Liver fibrosis is an early stage in the development
of liver cirrhosis. In-depth study of the mechanisms controlling
the development of liver fibrosis may aid in reducing the
occurrence of liver fibrosis (13), thereby preventing the progression
of liver cirrhosis and improving quality of life for patients.
Previous studies have confirmed that HSCs serve a key role in the
development of liver fibrosis (14,15).
Following the activation of HSCs by a variety of stimulating
factors, they are able to proliferate, secrete excessive TIMP-1 and
inhibit MMP-1 activity, leading to an imbalance between collagen
deposition and degradation (16).
TGF-β1 is a key factor for the promotion of HSCs activation and
expression of the extracellular matrix (17).
In the present study, a co-culture system was
established in which BM-MSCs cannot pass through the Transwell
insert while cytokines are able to pass freely. BM-MSCs were seeded
in the upper layer and HSCs in the lower layer, and the
proliferative and apoptotic effects of BM-MSCs on HSCs were
observed. The results of the MTT assay suggested that BM-MSCs did
not significantly inhibit the proliferation of HSCs at 24 h.
However, BM-MSCs significantly inhibited the proliferation of HSCs
when they were co-cultured for 48 and 72 h. Additionally, BM-MSCs
did not promote apoptosis of HSCs at 24 h, however induced
significant apoptosis at 48 h. The number of apoptotic cell bodies
in the experimental group was significantly increased compared with
the control group. Therefore, it was concluded that BM-MSCs inhibit
proliferation and induce apoptosis in HSCs. The non-contact
co-culture method excluded the contact inhibition effect of BM-MSC
on HSCs. These data suggest that BM-MSCs may secrete cytokines that
pass through the Transwell insert mesh and inhibit proliferation
and promote apoptosis of HSCs. The present study demonstrated that
HSCs synthesise and secrete a reduced quantity of TGF-β1 and
greater Smad7 following co-culture with BM-MSCs. These data further
confirm that BM-MSCs inhibit the proliferation and induce apoptosis
of HSCs through the secretion of certain cytokines involved in the
TGFβ/Smad signaling pathway. Further experiments are required to
identify the cytokines secreted by BM-MSCs.
In conclusion, the current study indicated that
BM-MSC may serve a role in inhibiting the proliferation of HSCs and
promoting apoptosis of HSCs through paracrine signaling, suggesting
that infusion of the BM-MSC culture supernatant may be used to
clinically treat liver fibrosis patients. However, in the present
study TGF-β1 and Smad7 mRNA and protein expression levels were
observed to be altered following co-culture for 24 h. However, from
the results at 24 h, the alterations of TGF-β1 and Smad7 did not
appear to affect cell proliferation and apoptosis during this time.
This suggests that the effect of alterations in fibrogenic factors
and fibrosis inhibitors on the mRNA and protein levels in HSCs
co-cultured for 24 h may not be large enough to inhibit
proliferation and promote apoptosis of HSCs. Following 48 h of
co-culture, alterations at the gene and protein levels were
observed, in addition to inhibition of HSC proliferation, and an
increase in the number of apoptotic cells. In the early stages of
the experiment, three observation time points were used, 24, 48 and
72 h, however from the MTT assay it was established that co-culture
of BM-MSCs and HSCs for 72 h significantly inhibited the
proliferation of HSCs. Additionally, the density of HSCs in the
control group culture was observed to impact upon levels of
apoptosis, with the number of adherent cells unable to meet the
requirements of the follow-up experiments. Therefore, two
observation time points, 24 and 48 h, were used in the follow-up
experiment.
There are several limitations of the current study.
The experiments used cultured BM-MSCs and HSCs cell lines in
vitro, which cannot accurately reflect the effect of BM-MSCs
in vivo. In addition, the results did not account for the
influence of the inoculation on cell concentration. Therefore,
future studies should address these limitations.
In summary, the present study identified that
BM-MSCs are able to inhibit the proliferation of HSCs and promote
their apoptosis, and the mechanism may be associated with
inhibition of the TGF-β1/Smad pathway in HSCs.
References
|
1
|
Mutimer DJ and Lok A: Management of HBV-
and HCV-induced end stage liver disease. Gut. 61(Suppl 1): i59–i67.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Olson JC, Wendon JA, Kramer DJ, Arroyo V,
Jalan R, Garcia-Tsao G and Kamath PS: Intensive care of the patient
with cirrhosis. Hepatology. 54:1864–1872. 2011. View Article : Google Scholar
|
|
3
|
Friedman SL: Hepatic stellate cells:
Protean, multifunctional, and enigmatic cells of the liver. Physiol
Rev. 88:125–172. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kisseleva T and Brenner DA: Role of
hepatic stellate cells in fibrogenesis and the reversal of
fibrosis. J Gastroenterol Hepatol. 22(Suppl 1): S73–S78. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sancho-Bru P, Najimi M, Caruso M, Pauwelyn
K, Cantz T, Forbes S, Roskams T, Ott M, Gehling U, Sokal E, et al:
Stem and progenitor cells for liver repopulation: Can we
standardise the process from bench to bedside? Gut. 58:594–603.
2009. View Article : Google Scholar
|
|
6
|
Mormone E, George J and Nieto N: Molecular
pathogenesis of hepatic fibrosis and current therapeutic
approaches. Chem Biol Interact. 193:225–231. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Rabani V, Shahsavani M, Gharavi M, Piryaei
A, Azhdari Z and Baharvand H: Mesenchymal stem cell infusion
therapy in a carbon tetrachloride-induced liver fibrosis model
affects matrix metalloproteinase expression. Cell Biol Int.
34:601–605. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mohamadnejad M, Alimoghaddam K, Bagheri M,
Ashrafi M, Abdollahzadeh L, Akhlaghpoor S, Bashtar M, Ghavamzadeh A
and Malekzadeh R: Randomized placebo-controlled trial of
mesenchymal stem cell transplantation in decompensated cirrhosis.
Liver Int. 33:1490–1496. 2013.PubMed/NCBI
|
|
9
|
Nunes de Carvalho S, Helal-Neto E, de
Andrade DC, Costa Cortez EA, Thole AA, Barja-Fidalgo C and de
Carvalho L: Bone marrow mononuclear cell transplantation increases
metalloproteinase-9 and 13 and decreases tissue inhibitors of
metalloproteinase-1 and 2 expression in the liver of cholestatic
rats. Cells Tissues Organs. 198:139–148. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang S, Zhang LT and Chen H: Effect of
human bone marrow mesenchymal stem cells culture supernatant on
related enzyme expression in hepatic stellate cells. J Clin Rehab
Tissue Eng Res. 16:8374–8379. 2012.
|
|
11
|
Parekkadan B, van Poll D, Megeed Z,
Kobayashi N, Tilles AW, Berthiaume F and Yarmush ML:
Immunomodulation of activated hepatic stellate cells by mesenchymal
stem cells. Biochem Biophys Res Commun. 363:247–252. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Shi L, Li G, Wang J, Sun B, Yang L, Wang
G, Wang D, Mu L, Chen H, Jin L, et al: Bone marrow stromal cells
control the growth of hepatic stellate cells in vitro. Dig Dis Sci.
53:2969–2974. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Friedman SL: Hepatic fibrosis - overview.
Toxicology. 254:120–129. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Friedman SL: Mechanisms of disease:
Mechanisms of hepatic fibrosis and therapeutic implications. Nat
Clin Pract Gastroenterol Hepatol. 1:98–105. 2004. View Article : Google Scholar
|
|
15
|
Friedman SL: Seminars in medicine of the
Beth Israel Hospital, Boston. The cellular basis of hepatic
fibrosis. Mechanisms and treatment strategies. N Engl J Med.
328:1828–1835. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Friedman SL: Mechanisms of hepatic
fibrogenesis. Gastroenterology. 134:1655–1669. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Li JH, Huang XR, Zhu HJ, Johnson R and Lan
HY: Role of TGF-beta signaling in extracellular matrix production
under high glucose conditions. Kidney Int. 63:2010–2019. 2003.
View Article : Google Scholar : PubMed/NCBI
|