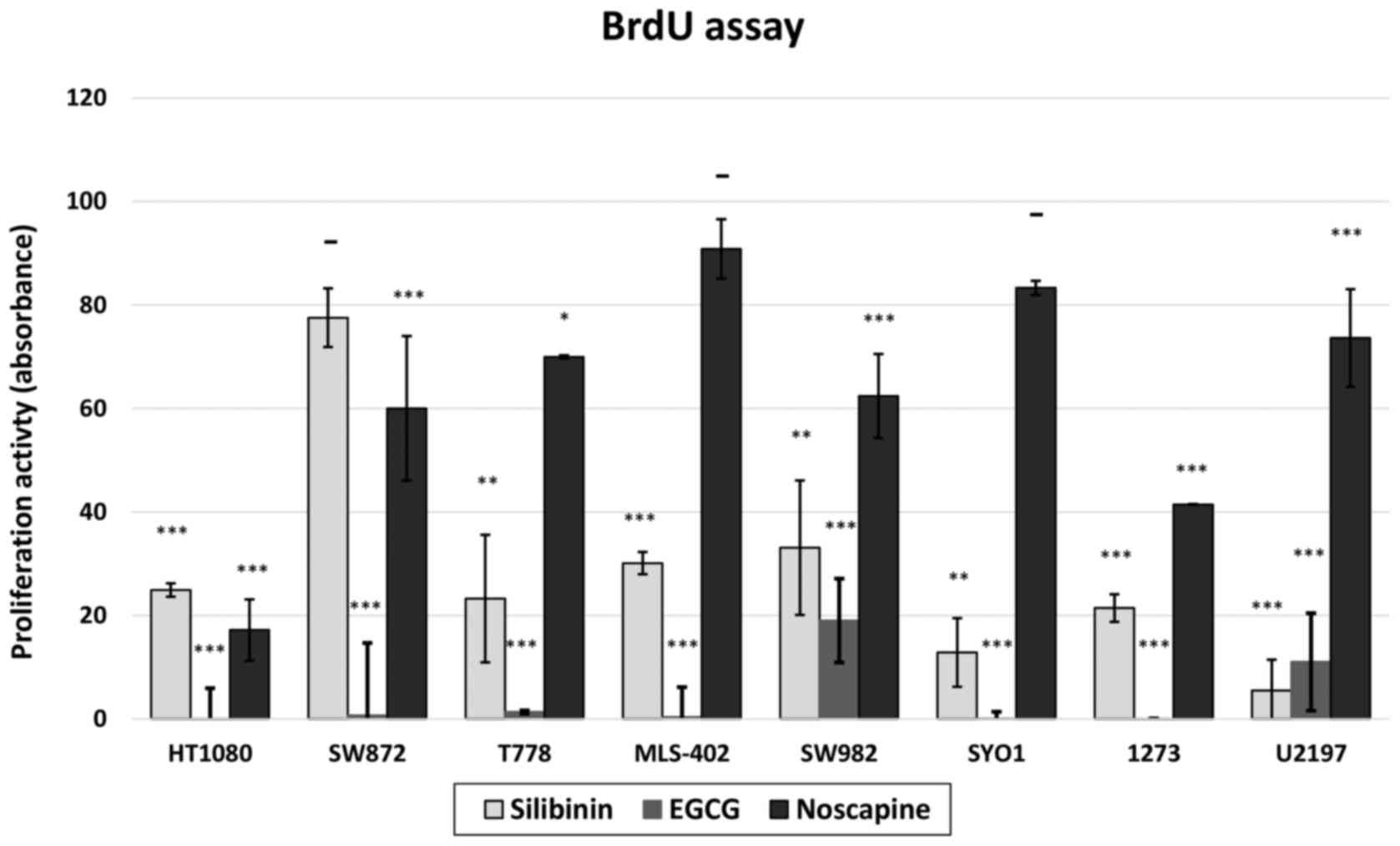
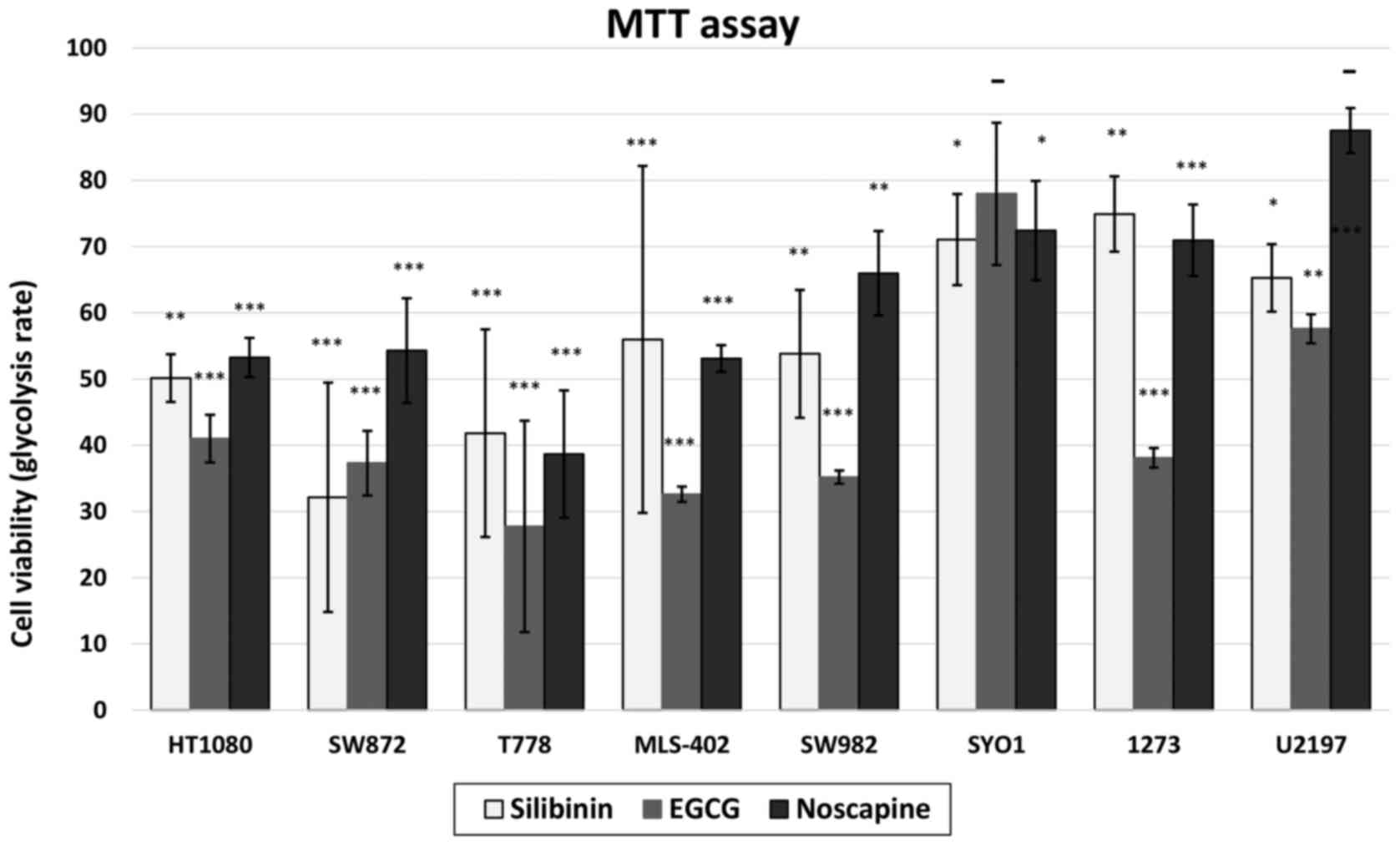
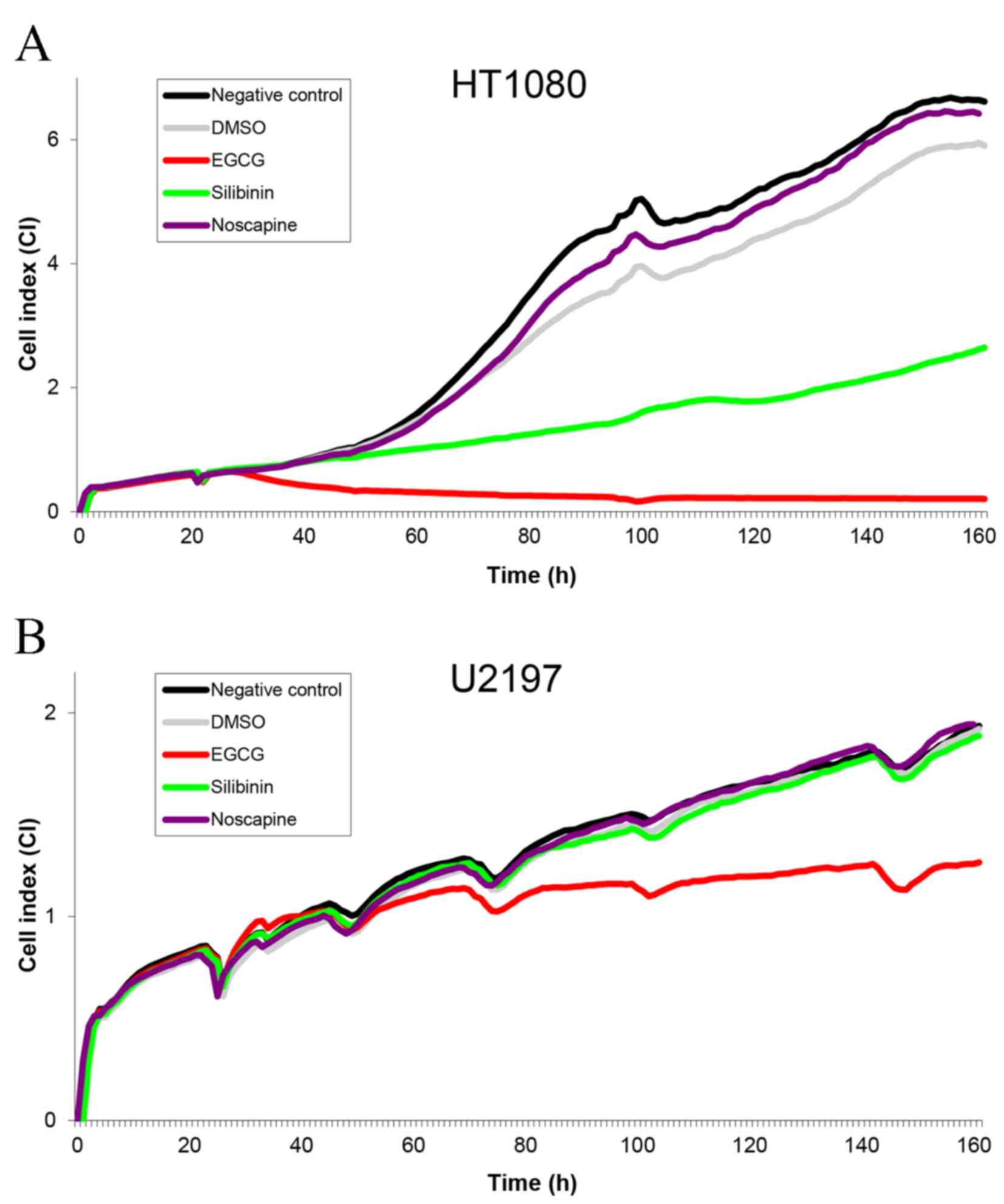
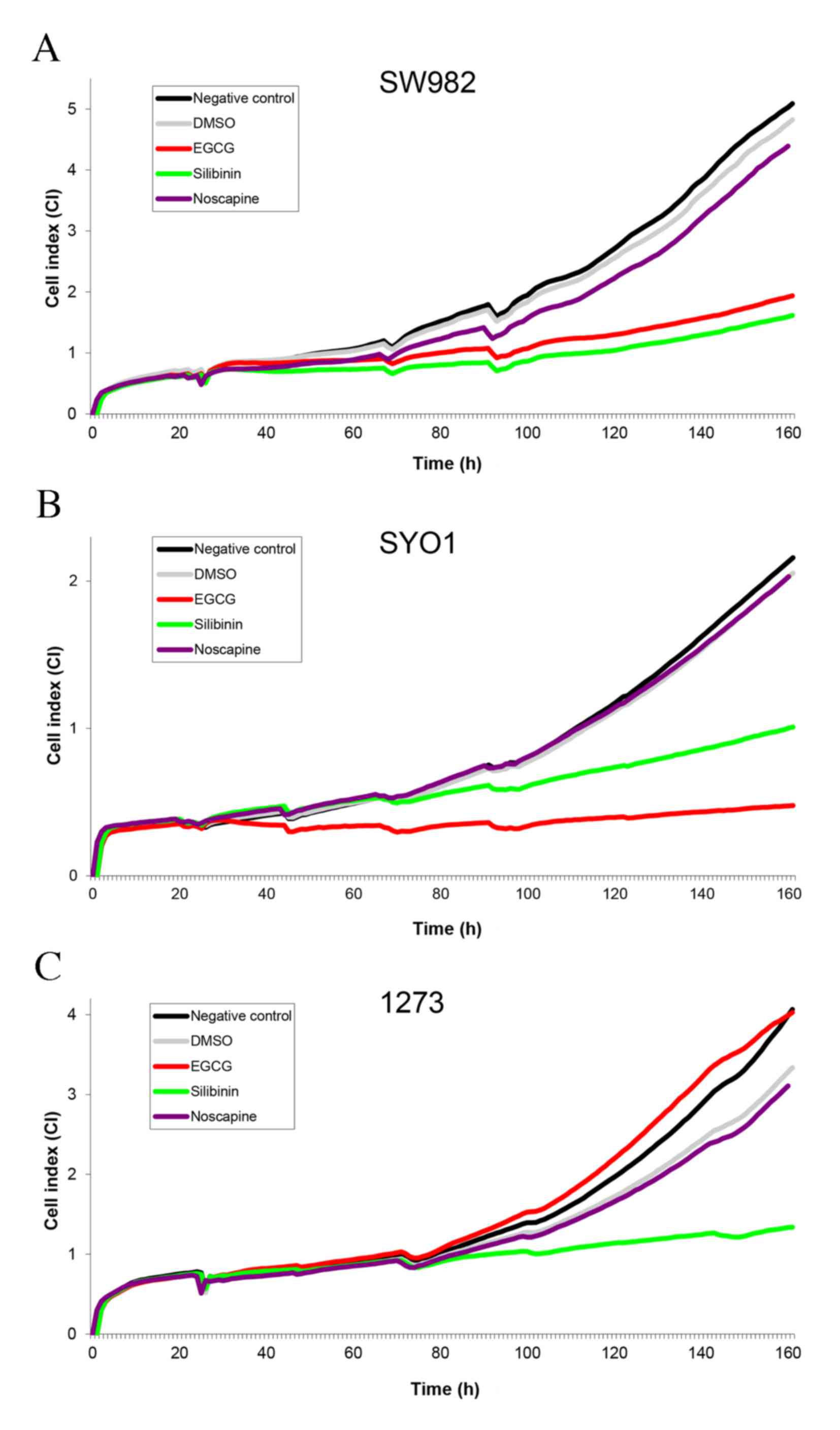
|
1
|
Hoos A, Lewis JJ and Brennan MF:
Weichgewebssarkome-prognostische Faktoren und multimodale Therapie.
Der Chirurg. 71:787–794. 2000. View Article : Google Scholar
|
|
2
|
Patrikidou A, Domont J, Cioffi A and Le
Cesne A: Treating soft tissue sarcomas with adjuvant chemotherapy.
Curr Treat Options Oncol. 12:21–31. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kaushal A and Citrin D: The role of
radiation therapy in the management of sarcomas. Surg Clin North
Am. 88629–646. (viii)2008.PubMed/NCBI
|
|
4
|
O'Brien GC, Cahill RA, Bouchier-Hayes DJ
and Redmond HP: Co-immunotherapy with interleukin-2 and taurolidine
for progressive metastatic melanoma. Ir J Med Sci. 175:10–14. 2006.
View Article : Google Scholar
|
|
5
|
Solomon LR, Cheesbrough JS, Bhargava R,
Mitsides N, Heap M, Green G and Diggle P: Observational study of
need for thrombolytic therapy and incidence of bacteremia using
taurolidine-citrate-heparin, taurolidine-citrate and heparin
catheter locks in patients treated with hemodialysis. Semin Dial.
25:233–238. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Karavasilis V, Seddon BM, Ashley S,
Al-Muderis O, Fisher C and Judson I: Significant clinical benefit
of first-line palliative chemotherapy in advanced soft-tissue
sarcoma: Retrospective analysis and identification of prognostic
factors in 488 patients. Cancer. 112:1585–1591. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Billingsley KG, Lewis JJ, Leung DH, Casper
ES, Woodruff JM and Brennan MF: Multifactorial analysis of the
survival of patients with distant metastasis arising from primary
extremity sarcoma. Cancer. 85:389–395. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pezzi CM, Pollock RE, Evans HL, Lorigan
JG, Pezzi TA, Benjamin RS and Romsdahl MM: Preoperative
chemotherapy for soft-tissue sarcomas of the extremities. Ann Surg.
211:476–481. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Di Paola Donato E and Nielsen OS: EORTC
Soft Tissue and Bone Sarcoma Group: The EORTC soft tissue and bone
sarcoma group. European Organisation for Research and Treatment of
Cancer. Eur J Cancer. 38:(Suppl 4). S138–S141. 2002.PubMed/NCBI
|
|
10
|
Nedea EA and DeLaney TF: Sarcoma and skin
radiation oncology. Hematol Oncol Clin North Am. 20:401–429. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Brodowicz T, Schwameis E, Widder J, Amann
G, Wiltschke C, Dominkus M, Windhager R, Ritschl P, Pötter R, Kotz
R and Zielinski CC: Intensified adjuvant IFADIC chemotherapy for
adult soft tissue sarcoma: A prospective randomized feasibility
trial. Sarcoma. 4:151–160. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Frustaci S, Gherlinzoni F, De Paoli A,
Bonetti M, Azzarelli A, Comandone A, Olmi P, Buonadonna A, Pignatti
G, Barbieri E, et al: Adjuvant chemotherapy for adult soft tissue
sarcomas of the extremities and girdles: Results of the Italian
randomized cooperative trial. J Clin Oncol. 19:1238–1247.
2001.PubMed/NCBI
|
|
13
|
Judson I, Verweij J, Gelderblom H,
Hartmann JT, Schöffski P, Blay JY, Kerst JM, Sufliarsky J, Whelan
J, Hohenberger P, et al: Doxorubicin alone versus intensified
doxorubicin plus ifosfamide for first-line treatment of advanced or
metastatic soft-tissue sarcoma: A randomised controlled phase 3
trial. Lancet Oncol. 15:415–423. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Swain SM, Whaley FS and Ewer MS:
Congestive heart failure in patients treated with doxorubicin: A
retrospective analysis of three trials. Cancer. 97:2869–2879. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Burningham Z, Hashibe M, Spector L and
Schiffman JD: The epidemiology of sarcoma. Clin Sarcoma Res.
2:142012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jiang L, Tao C, He A and He X:
Overexpression of miR-126 sensitizes osteosarcoma cells to
apoptosis induced by epigallocatechin-3-gallate. World J Surg
Oncol. 12:3832014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lambert JD, Sang S, Hong J and Yang CS:
Anticancer and anti-inflammatory effects of cysteine metabolites of
the green tea polyphenol, (−)-epigallocatechin-3-gallate. J Agric
Food Chem. 58:10016–10019. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kalaiselvi P, Rajashree K, Bharathi Priya
L and Padma VV: Cytoprotective effect of epigallocatechin-3-gallate
against deoxynivalenol-induced toxicity through anti-oxidative and
anti-inflammatory mechanisms in HT-29 cells. Food Chem Toxicol.
56:110–118. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Deng YT and Lin JK: EGCG inhibits the
invasion of highly invasive CL1-5 lung cancer cells through
suppressing MMP-2 expression via JNK signaling and induces G2/M
arrest. J Agric Food Chem. 59:13318–13327. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kobalka AJ, Keck RW and Jankun J:
Synergistic anticancer activity of biologicals from green and black
tea on DU 145 human prostate cancer cells. Cent Eur J Immunol.
40:1–4. 2015.PubMed/NCBI
|
|
21
|
Park IJ, Lee YK, Hwang JT, Kwon DY, Ha J
and Park OJ: Green tea catechin controls apoptosis in colon cancer
cells by attenuation of H2O2-stimulated COX-2 expression via the
AMPK signaling pathway at low-dose H2O2. Ann N Y Acad Sci.
1171:538–544. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Park JS, Khoi PN, Joo YE, Lee YH, Lang SA,
Stoeltzing O and Jung YD: EGCG inhibits recepteur d'origine nantais
expression by suppressing Egr-1 in gastric cancer cells. Int J
Oncol. 42:1120–1126. 2013.PubMed/NCBI
|
|
23
|
Braicu C, Gherman CD, Irimie A and
Berindan-Neagoe I: Epigallocatechin-3-Gallate (EGCG) inhibits cell
proliferation and migratory behaviour of triple negative breast
cancer cells. J Nanosci Nanotechnol. 13:632–637. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zou C, Liu H, Feugang JM, Hao Z, Chow HH
and Garcia F: Green tea compound in chemoprevention of cervical
cancer. Int J Gynecol Cancer. 20:617–624. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Shanafelt TD, Call TG, Zent CS, Leis JF,
LaPlant B, Bowen DA, Roos M, Laumann K, Ghosh AK, Lesnick C, et al:
Phase 2 trial of daily, oral Polyphenon E in patients with
asymptomatic, Rai stage 0 to II chronic lymphocytic leukemia.
Cancer. 119:363–370. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
de la Torre R, de Sola S, Hernandez G,
Farré M, Pujol J, Rodriguez J, Espadaler JM, Langohr K, Cuenca-Royo
A, Principe A, et al: Safety and efficacy of cognitive training
plus epigallocatechin-3-gallate in young adults with Down's
syndrome (TESDAD): A double-blind, randomised, placebo-controlled,
phase 2 trial. Lancet Neurol. 15:801–810. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhao H, Xie P, Li X, Zhu W, Sun X, Sun X,
Chen X, Xing L and Yu J: A prospective phase II trial of EGCG in
treatment of acute radiation-induced esophagitis for stage III lung
cancer. Radiother Oncol. 114:351–356. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Trudel D, Labbé DP, Araya-Farias M, Doyen
A, Bazinet L, Duchesne T, Plante M, Grégoire J, Renaud MC,
Bachvarov D, et al: A two-stage, single-arm, phase II study of
EGCG-enriched green tea drink as a maintenance therapy in women
with advanced stage ovarian cancer. Gynecol Oncol. 131:357–361.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Dostal AM, Samavat H, Bedell S, Torkelson
C, Wang R, Swenson K, Le C, Wu AH, Ursin G, Yuan JM and Kurzer MS:
The safety of green tea extract supplementation in postmenopausal
women at risk for breast cancer: Results of the Minnesota Green Tea
Trial. Food Chem Toxicol. 83:26–35. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Garcia FA, Cornelison T, Nuño T, Greenspan
DL, Byron JW, Hsu CH, Alberts DS and Chow HH: Results of a phase II
randomized, double-blind, placebo-controlled trial of Polyphenon E
in women with persistent high-risk HPV infection and low-grade
cervical intraepithelial neoplasia. Gynecol Oncol. 132:377–382.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Singh BN, Shankar S and Srivastava RK:
Green tea catechin, epigallocatechin-3-gallate (EGCG): Mechanisms,
perspectives and clinical applications. Biochem Pharmacol.
82:1807–1821. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Shanafelt TD, Call TG, Zent CS, LaPlant B,
Bowen DA, Roos M, Secreto CR, Ghosh AK, Kabat BF, Lee MJ, et al:
Phase I trial of daily oral Polyphenon E in patients with
asymptomatic Rai stage 0 to II chronic lymphocytic leukemia. J Clin
Oncol. 27:3808–3814. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Mengs U, Pohl RT and Mitchell T:
Legalon® SIL: The antidote of choice in patients with
acute hepatotoxicity from amatoxin poisoning. Curr Pharm
Biotechnol. 13:1964–1970. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Flaig TW, Glodé M, Gustafson D, van
Bokhoven A, Tao Y, Wilson S, Su LJ, Li Y, Harrison G, Agarwal R, et
al: A study of high-dose oral silybin-phytosome followed by
prostatectomy in patients with localized prostate cancer. Prostate.
70:848–855. 2010.PubMed/NCBI
|
|
35
|
Flaig TW, Gustafson DL, Su LJ, Zirrolli
JA, Crighton F, Harrison GS, Pierson AS, Agarwal R and Glodé LM: A
phase I and pharmacokinetic study of silybin-phytosome in prostate
cancer patients. Invest New Drugs. 25:139–146. 2007.PubMed/NCBI
|
|
36
|
Kaur M, Velmurugan B, Tyagi A, Deep G,
Katiyar S, Agarwal C and Agarwal R: Silibinin suppresses growth and
induces apoptotic death of human colorectal carcinoma LoVo cells in
culture and tumor xenograft. Mol Cancer Ther. 8:2366–2374. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Singh RP and Agarwal R: Prostate cancer
chemoprevention by silibinin: Bench to bedside. Mol Carcinog.
45:436–442. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Leon IE, Porro V, Di Virgilio AL, Naso LG,
Williams PA, Bollati-Fogolín M and Etcheverry SB: Antiproliferative
and apoptosis-inducing activity of an oxidovanadium (IV) complex
with the flavonoid silibinin against osteosarcoma cells. J Biol
Inorg Chem. 19:59–74. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Vue B, Zhang S, Zhang X, Parisis K, Zhang
Q, Zheng S, Wang G and Chen QH: Silibinin derivatives as
anti-prostate cancer agents: Synthesis and cell-based evaluations.
Eur J Med Chem. 109:36–46. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Duan WJ, Li QS, Xia MY, Tashiro S, Onodera
S and Ikejima T: Silibinin activated p53 and induced autophagic
death in human fibrosarcoma HT1080 cells via reactive oxygen
species-p38 and c-Jun N-terminal kinase pathways. Biol Pharm Bull.
34:47–53. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ghaly PE, El-Magd RM Abou, Churchill CD,
Tuszynski JA and West FG: A new antiproliferative noscapine
analogue: Chemical synthesis and biological evaluation. Oncotarget.
7:40518–40530. 2016.PubMed/NCBI
|
|
42
|
Yang ZR, Liu M, Peng XL, Lei XF, Zhang JX
and Dong WG: Noscapine induces mitochondria-mediated apoptosis in
human colon cancer cells in vivo and in vitro. Biochem Biophys Res
Commun. 421:627–633. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Li S, He J, Li S, Cao G, Tang S, Tong Q
and Joshi HC: Noscapine induced apoptosis via downregulation of
survivin in human neuroblastoma cells having wild type or null p53.
PloS One. 7:e400762012. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Quisbert-Valenzuela EO and Calaf GM:
Apoptotic effect of noscapine in breast cancer cell lines. Int J
Oncol. 48:2666–2674. 2016.PubMed/NCBI
|
|
45
|
Lopus M and Naik PK: Taking aim at a
dynamic target: Noscapinoids as microtubule-targeted cancer
therapeutics. Pharmacol Rep. 67:56–62. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Mukkavilli R, Gundala SR, Yang C, Jadhav
GR, Vangala S, Reid MD and Aneja R: Noscapine recirculates
enterohepatically and induces self-clearance. Eur J Pharm Sci.
77:90–99. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Stratford EW, Castro R, Daffinrud J, Skårn
M, Lauvrak S, Munthe E and Myklebost O: Characterization of
liposarcoma cell lines for preclinical and biological studies.
Sarcoma. 2012:1486142012.PubMed/NCBI
|
|
48
|
Aman P, Ron D, Mandahl N, Fioretos T, Heim
S, Arheden K, Willén H, Rydholm A and Mitelman F: Rearrangement of
the transcription factor gene CHOP in myxoid liposarcomas with
t(12;16)(q13;p11). Genes, chromosomes cancer. 5:278–285. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Kawai A, Naito N, Yoshida A, Morimoto Y,
Ouchida M, Shimizu K and Beppu Y: Establishment and
characterization of a biphasic synovial sarcoma cell line, SYO-1.
Cancer Lett. 204:105–113. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Xie Y, Skytting B, Nilsson G, Gasbarri A,
Haslam K, Bartolazzi A, Brodin B, Mandahl N and Larsson O: SYT-SSX
is critical for cyclin D1 expression in synovial sarcoma cells: A
gain of function of the t(X;18)(p11.2;q11.2) translocation. Cancer
Res. 62:3861–3867. 2002.PubMed/NCBI
|
|
51
|
Becerikli M, Jacobsen F, Rittig A, Köhne
W, Nambiar S, Mirmohammadsadegh A, Stricker I, Tannapfel A,
Wieczorek S, Epplen JT, et al: Growth rate of late passage sarcoma
cells is independent of epigenetic events but dependent on the
amount of chromosomal aberrations. Exp Cell Res. 319:1724–1731.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Kang HG, Jenabi JM, Liu XF, Reynolds CP,
Triche TJ and Sorensen PH: Inhibition of the insulin-like growth
factor I receptor by epigallocatechin gallate blocks proliferation
and induces the death of Ewing tumor cells. Mol Cancer Ther.
9:1396–1407. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Chougule MB, Patel AR, Jackson T and Singh
M: Antitumor activity of Noscapine in combination with Doxorubicin
in triple negative breast cancer. PloS One. 6:e177332011.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Koval OA, Sakaeva GR, Fomin AS, Nushtaeva
AA, Semenov DV, Kuligina EV, Gulyaeva LF, Gerasimov AV and Richter
VA: Sensitivity of endometrial cancer cells from primary human
tumor samples to new potential anticancer peptide lactaptin. J
Cancer Res Ther. 11:345–351. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Italiano A, Toulmonde M, Cioffi A, Penel
N, Isambert N, Bompas E, Duffaud F, Patrikidou A, Lortal B, Le
Cesne A, et al: Advanced well-differentiated/dedifferentiated
liposarcomas: Role of chemotherapy and survival. Ann Oncol.
23:1601–1607. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Italiano A, Garbay D, Cioffi A, Maki RG
and Bui B: Advanced pleomorphic liposarcomas: Clinical outcome and
impact of chemotherapy. Ann Oncol. 23:2205–2206. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Jones RL, Fisher C, Al-Muderis O and
Judson IR: Differential sensitivity of liposarcoma subtypes to
chemotherapy. Eur J Cancer. 41:2853–2860. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Patel SR, Burgess MA, Plager C,
Papadopoulos NE, Linke KA and Benjamin RS: Myxoid liposarcoma.
Experience with chemotherapy. Cancer. 74:1265–1269. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Vlenterie M, Litière S, Rizzo E, Marréaud
S, Judson I, Gelderblom H, Le Cesne A, Wardelmann E, Messiou C,
Gronchi A and van der Graaf WT: Outcome of chemotherapy in advanced
synovial sarcoma patients: Review of 15 clinical trials from the
European Organisation for Research and Treatment of Cancer Soft
Tissue and Bone Sarcoma Group; setting a new landmark for studies
in this entity. Eur J Cancer. 58:62–72. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Saeed NM, El-Naga RN, El-Bakly WM,
Abdel-Rahman HM, Salah ElDin RA and El-Demerdash E:
Epigallocatechin-3-gallate pretreatment attenuates
doxorubicin-induced cardiotoxicity in rats: A mechanistic study.
Biochem Pharmacol. 95:145–155. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Khan MA, Singh M, Khan MS, Ahmad W, Najmi
AK and Ahmad S: Alternative approach for mitigation of
doxorubicin-induced cardiotoxicity using herbal agents. Curr Clin
Pharmacol. 9:288–297. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Zheng J, Lee HC, Bin Sattar MM, Huang Y
and Bian JS: Cardioprotective effects of epigallocatechin-3-gallate
against doxorubicin-induced cardiomyocyte injury. Eur J Pharmacol.
652:82–88. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Li W, Nie S, Xie M, Chen Y, Li C and Zhang
H: A major green tea component, (−)-epigallocatechin-3-gallate,
ameliorates doxorubicin-mediated cardiotoxicity in cardiomyocytes
of neonatal rats. J Agric Food Chem. 58:8977–8982. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Cheng T, Liu J, Ren J, Huang F, Ou H, Ding
Y, Zhang Y, Ma R, An Y, Liu J and Shi L: Green tea catechin-based
complex micelles combined with doxorubicin to overcome
cardiotoxicity and multidrug resistance. Theranostics. 6:1277–1292.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Stearns ME, Amatangelo MD, Varma D, Sell C
and Goodyear SM: Combination therapy with
epigallocatechin-3-gallate and doxorubicin in human prostate tumor
modeling studies: Inhibition of metastatic tumor growth in severe
combined immunodeficiency mice. Am J Pathol. 177:3169–3179. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Chen L, Ye HL, Zhang G, Yao WM, Chen XZ,
Zhang FC and Liang G: Autophagy inhibition contributes to the
synergistic interaction between EGCG and doxorubicin to kill the
hepatoma Hep3B cells. PloS One. 9:e857712014. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Liang G, Tang A, Lin X, Li L, Zhang S,
Huang Z, Tang H and Li QQ: Green tea catechins augment the
antitumor activity of doxorubicin in an in vivo mouse model for
chemoresistant liver cancer. Int J Oncol. 37:111–123.
2010.PubMed/NCBI
|
|
68
|
Singh RP, Mallikarjuna GU, Sharma G,
Dhanalakshmi S, Tyagi AK, Chan DC, Agarwal C and Agarwal R: Oral
silibinin inhibits lung tumor growth in athymic nude mice and forms
a novel chemocombination with doxorubicin targeting nuclear factor
kappaB-mediated inducible chemoresistance. Clin Cancer Res.
10:8641–8647. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Tyagi AK, Agarwal C, Chan DC and Agarwal
R: Synergistic anti-cancer effects of silibinin with conventional
cytotoxic agents doxorubicin, cisplatin and carboplatin against
human breast carcinoma MCF-7 and MDA-MB468 cells. Oncol Rep.
11:493–499. 2004.PubMed/NCBI
|
|
70
|
Tyagi AK, Singh RP, Agarwal C, Chan DC and
Agarwal R: Silibinin strongly synergizes human prostate carcinoma
DU145 cells to doxorubicin-induced growth Inhibition, G2-M arrest,
and apoptosis. Clin Cancer Res. 8:3512–3519. 2002.PubMed/NCBI
|
|
71
|
Rašković A, Stilinović N, Kolarović J,
Vasović V, Vukmirović S and Mikov M: The protective effects of
silymarin against doxorubicin-induced cardiotoxicity and
hepatotoxicity in rats. Molecules. 16:8601–8613. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Chlopcíková S, Psotová J, Miketová P and
Simánek V: Chemoprotective effect of plant phenolics against
anthracycline-induced toxicity on rat cardiomyocytes. Part I.
Silymarin and its flavonolignans. Phytother Res. 18:107–110. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Psotová J, Chlopcíková S, Grambal F,
Simánek V and Ulrichová J: Influence of silymarin and its
flavonolignans on doxorubicin-iron induced lipid peroxidation in
rat heart microsomes and mitochondria in comparison with quercetin.
Phytother Res. 16:(Suppl 1). S63–S67. 2002. View Article : Google Scholar : PubMed/NCBI
|