Introduction
Vascular dementia (VD) occurs as a result of
ischemic injury or oligemia to brain areas, which leads to a
progressive cognition decline, functional ability impairment and
behavioral problems (1). VD is
considered the second most common dementia and prevalence of VD is
increased, accompanied by a corresponding increase in elderly
population. Currently, there is no effective cure for VD, thus
investigators have focused on preventing further brain damage.
The standardized Ginkgo biloba extract, EGb
761, has been reported to have neuroprotective effects against
various neurological disorders, such as Alzheimer's disease,
ischemia and depression (2–4).
Although these results suggest the possibility of using the
Ginkgo biloba extract as an anti-dementia drug, the
properties of each constituent of the extract should be analyzed to
develop an optimally effective anti-dementia drug. EGb 761 contains
two groups of bioactive constituents, the flavonoids (24%) and the
terpenoids (6%), which have been actively investigated for their
neuroprotective and neuromodulatory activities (5). It has been reported that the
effectiveness of the Ginkgo biloba extract EGb 761 in a
middle cerebral artery occlusion (MCAO) model was exclusively due
to the presence of bilobalide (BB), a sesquiterpene lactone that
constitutes 2.9–3.2% of the extract (6,7).
Accumulating evidence suggest that BB has neuroprotective effects
(2,8,9).
Optimum efficacy usually requires extracts at high doses (50–200
mg/kg), while using lower doses of BB may result in an equally high
efficacy.
Chronic cerebral hypoperfusion is mimicked in
animals by bilateral common carotid artery occlusion (2-vessel
occlusion, 2-VO), resulting in the establishment of a suitable
model for investigating the mechanisms of VD (10). The aim of the present study was to
investigate the effects of BB, a specific constituent of the
Ginkgo biloba extract EGb 761, on VD rats. Consequently, we
investigated the effects of BB on the learning and memory
dysfunction in VD rats. BB was hypothesized to improve the learning
and memory dysfunction in VD rats due to its antioxidant and
antiapoptotic properties. Furthermore, we investigated the effects
of BB on oxidative injury, neuronal apoptosis and the expression of
tumor necrosis factor-α (TNF-α) in VD rats.
Materials and methods
Drugs and reagents
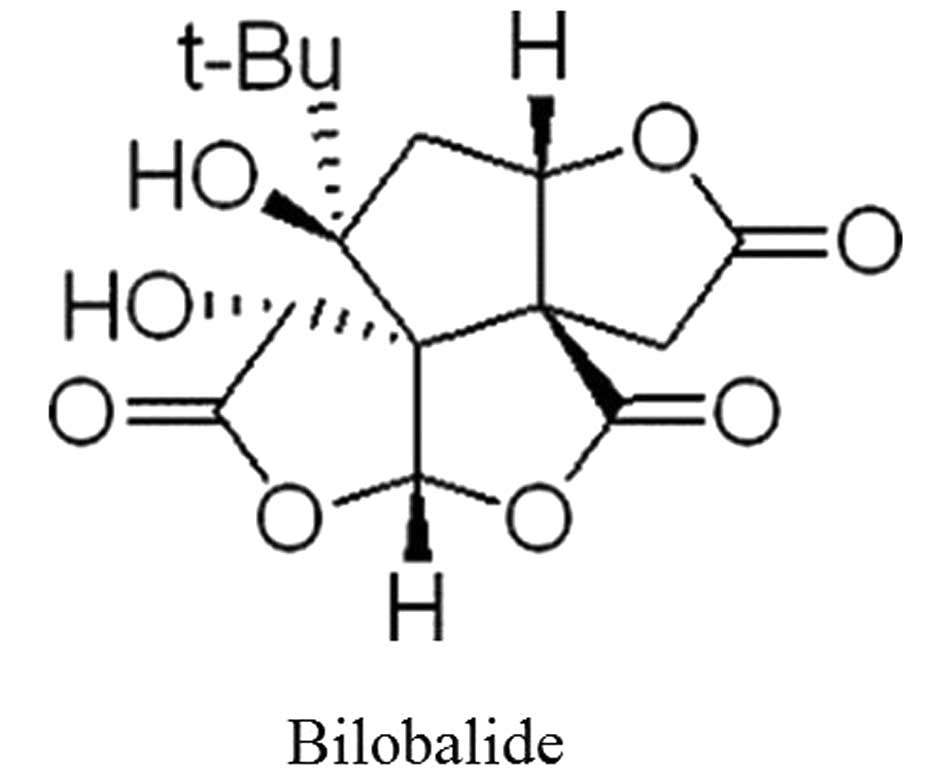
Bilobalide (content of BB, >98%; Fig. 1) was obtained from the Creative
Pharmaceutical Technology Co., Ltd. (Hefei, China).
Streptavidin-biotin complex and diaminobenzidine (DAB) staining
kits were purchased from ZSGB-BIO (Beijing, China). Antibody
against TNF-α was purchased from Beijing Biosynthesis Biotechnology
Co., Ltd. (China). Drugs were dissolved in distilled water, and all
the additional chemicals were of the highest analytical grade
available.
Animals and treatment
The rodent experiments were appropriately performed
to minimize animal suffering and were in accordance with the animal
protocols approved by the local authorities in Anhui, China. Adult
male Sprague Dawley rats (2 to 3-month-old, 180–200 g) were
obtained from the Center of Laboratory Animal of Anhui, China
(Grade II, Certificate no. SCXK 2005-001). The rats were housed
under conditions of controlled lighting (12-h light/dark cycle) and
temperature (25°C). All the animals were allowed ad libitum
access to food and water, and were allowed to acclimatize for ≥3
days prior to experimentation.
The rats were randomly divided into 7 groups: the
sham-surgery, model, BB (2, 4 and 8 mg/kg), Nimotop (Nimo, 10.5
mg/kg) and Huperzine A (Hupa, 0.047 mg/kg) groups. Nimo and Hupa,
which are standard drugs for dementia treatment, were used as
positive controls. The animals were anesthetized with 10% chloral
hydrate [3 ml/kg, intraperitoneally (i.p.)]. Surgical operation was
conducted as previously described (11). Briefly, through a midline cervical
incision, the bilateral common carotid arteries of each rat were
carefully separated from the cervical sympathetic and vagal nerves.
The bilateral common carotid arteries were then exposed and ligated
with 4-0 silk sutures in the permanent 2-VO group. The rats of the
sham-operated group underwent the same surgical operation without
carotid artery ligations. Mean body temperature (taken rectally)
was maintained during surgery at 37.0±0.5°C. The rats of the BB (2,
4 and 8 mg/kg), Nimo (10.5 mg/kg) and Hupa (0.047 mg/kg) groups
were treated with the respective drugs [intragastric (i.g.)
administration] for 60 days from day 1 following surgery. The rats
of the sham-surgery group were administered equivalent volumes of
vehicle.
Morris water maze test
The Morris water maze (MWM) consists of a black
1.6-m diameter circular pool. The pool was divided into 4 equal
hypothetical quadrants and filled with water (24°C; depth, 30 cm).
In the middle of a certain quadrant, there was a submerged escape
platform (10 cm in diameter), which was placed 2 cm below the
surface of the water. The water maze apparatus, rat handling, and
general testing procedure have been previously described (12).
All the rats were released into water (facing the
wall) at 4 distinct starting quadrant points and were given 4
trials/day (90 sec/trial) from 56 to 59 days following surgery, for
4 consecutive days. Using an on-line image video tracking system,
escape latency (sec) was recorded to determine changes in learning
dysfunction. On day 60, following completion of all the learning
trials for all the rats, the platform was removed from the pool,
and each rat received one 90-sec swim probe trial. The time of
swimming in the quadrant of platform (STP) and the number of rats
crossing the platform (NCP) were recorded to determine changes in
memory dysfunction. Behavioral data were analyzed using analysis of
variance.
Detection of superoxide dismutase (SOD),
nitric oxide synthase (NOS) activity, and malondialdehyde (MDA)
glutathione (GSH) content
Following MWM testing, 8 rats from each group were
anesthetized and sacrificed by decapitation. The brain of each rat
was dissociated and stored at −80°C until assessment. Superoxide
dismutase (SOD) and nitric oxide synthase (NOS) activity, as well
as malondialdehyde (MDA) and glutathione (GSH) content were
determined using the appropriative detection kits (Nianjing
Jiancheng Bioengineering Institute, Nanjing, China).
Histological examination
Following MWM testing, 5 rats from each group were
anesthetized with 10% chloral hydrate (3.0 ml/kg). Following animal
sacrifice, the brain of each rat was removed, fixed in 4%
paraformaldehyde and embedded in paraffin. The brain tissues were
sliced (5-μm) using a section cutter (Leica Biosystems, Wetzlar,
Germany). The sections were then stained with hematoxylin and eosin
(H&E) and examined under a light microscope.
Hoechst 33258 staining
For nuclear staining, paraffin sections were
dehydrated through a gradient of xylene and ethanol, and then
rinsed with phosphate-buffered saline (PBS). The sections were
incubated with 25 mM Hoechst 33258 (ZSGB-BIO) for 15 min at 37°C,
washed with PBS (pH 7.2–7.5), mounted onto slides using antifade
mounting medium, and then examined using fluorescence microscopy
(Ex/Em, 352/461 nm) (Olympus Optical, Tokyo, Japan).
Immunohistochemistry
Paraffin sections were cut (5-μm) and fixed to
slides to ensure adhesion. The sections were incubated with
hydrogen peroxide (3%) to inactivate endogenous peroxidase, and
non-immune goat serum was used as a blocking agent. The sections
were incubated with the primary antibody against TNF-α (1:100)
overnight at 4°C. Immunostaining was visualized by the peroxidase
method with a biotinylated anti-rabbit secondary antibody and DAB
oxidation. The sections were resin-mounted and examined using a
fluorescence microscope (Nikon, Japan). The immunoreactive neurons
which were stained brown were observed, and images were captured
under the microscope. Five sections/group and the three high-power
fields of the hippocampal CA1 region and the brain cortex/section
of the same magnification (x400) were used for quantitative
analysis. The mean optical density of positive neurons in each
section was measured using the Image-Pro Plus 6.0 analysis system
to determine TNF-α expression.
Statistical analysis
Intergroup differences were analyzed using one-way
ANOVA. The Student's t-test was used to determine significant
differences between groups. Results were expressed as the mean ±
SD. P<0.05 was considered to indicate a statistically
significant difference.
Results
Effects of BB on the learning and memory
ability of VD rats
MWM was used to determine the learning and memory
ability of rats. In the memory training experiment, the mean escape
latencies in the rats of all the groups were significantly reduced
with increasing time. The mean escape latencies in the VD rats of
the model group were significantly increased compared with the
model group (P<0.01). The mean escape latencies were differently
decreased following BB (2, 4 and 8 mg/kg), Nimo and Hupa treatment
(P<0.05 or <0.01) (Table I).
In addition, NCP and STP in the rats of the model group were also
significantly reduced compared with the sham-surgery group
(P<0.05 or <0.01). NCP (P<0.05) and STP (P<0.05 or
<0.01) in the rats of the BB (2, 4 and 8 mg/kg), Nimo and Hupa
groups were differentially decreased compared with the model group
(Table II).
 | Table IEffect of BB on mean escape latencies
in VD rats. |
Table I
Effect of BB on mean escape latencies
in VD rats.
| | Mean escape latencies
(sec) |
|---|
| |
|
|---|
| Group | Dose (mg/kg) | Day 1 | Day 2 | Day 3 | Day 4 |
|---|
| Sham | - | 58.99±37.20 | 31.19±28.77 | 19.82±15.03 | 12.15±9.62 |
| Model | - | 80.11±22.96a | 64.27±34.37a | 56.48±36.67a | 55.61±35.40a |
| Nimo | 10.5 | 68.03±34.96 | 50.25±31.28 | 43.82±31.15 | 40.22±21.35b |
| Hupa | 0.047 | 56.33±32.08c | 45.96±31.59b | 30.36±27.81c | 26.24±26.75c |
| BB | 2 | 72.35±25.05 | 49.87±34.66 | 33.58±30.82c | 22.12±18.37c |
| 4 | 77.47±24.35 | 55.95±31.70 | 29.90±24.65c | 28.75±22.05c |
| 8 | 68.90±29.56 | 52.99±33.45 | 32.65±30.36c | 27.23±25.14c |
 | Table IIEffect of BB on STP and NCP in VD
rats. |
Table II
Effect of BB on STP and NCP in VD
rats.
| Group | Dose (mg/kg) | STP (sec) | NCP |
|---|
| Sham | - | 32.90±5.89 | 6.38±2.62 |
| Model | - | 25.56±5.34a | 1.50±1.60b |
| Nimo | 10.5 | 31.28±6.62 | 4.63±2.97c |
| Hupa | 0.047 | 35.19±7.84c | 5.00±2.33d |
| BB | 2 | 28.44±5.77 | 4.75±3.20c |
| 4 | 33.83±9.00c | 4.63±3.54c |
| 8 | 33.39±8.48c | 5.63±2.56d |
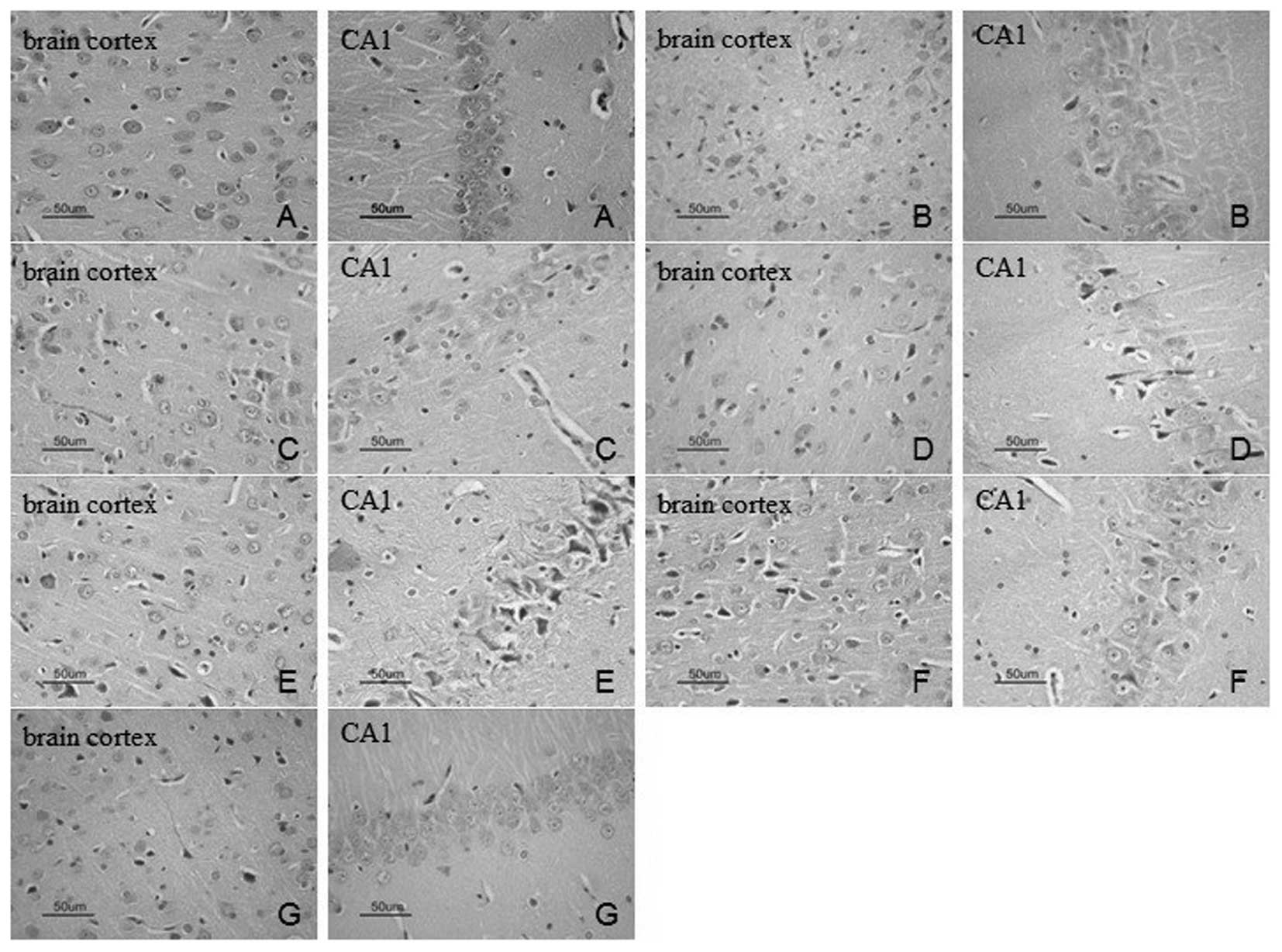
Effects of BB on neuronal morphology in
the brain cortex and hippocampal CA1 region
H&E and Hoechst 33258 staining were used to
investigate the neuronal morphology and apoptosis of the brain
cortex and the hippocampal CA1 region of the rats. H&E staining
indicated that there were no significant neuronal abnormalities in
the brain cortex and the hippocampal CA1 region of the rats in the
sham-surgery group (Fig. 2A).
However, the rats in the model group exhibited acidophilia
degeneration, nuclear condensation and disorder of the array of
neurons in these regions (Fig.
2B). Neuronal degeneration was alleviated in the rats of the BB
(4 and 8 mg/kg), Nimo and Hupa groups compared with the model group
(Fig. 2).
We examined the neuronal apoptosis of the brain
cortex and the hippocampal CA1 region of rats using Hoechst 33258
staining. The average number of apoptotic cells in these regions,
which appear smaller than normal and in which the chromatin appears
to be condensed, were counted (n=5). The percentage of nuclear
condensation in the brain cortex and the hippocampal CA1 region in
the rats of the model group was significantly increased compared
with sham-surgery group (P<0.05 or <0.01). The percentage of
nuclear condensation in the brain cortex and the hippocampal CA1
region in the rats of the BB (4 and 8 mg/kg), Hupa and Nimo groups
was differentially decreased compared with the model group
(P<0.05 or <0.01; Fig.
3).
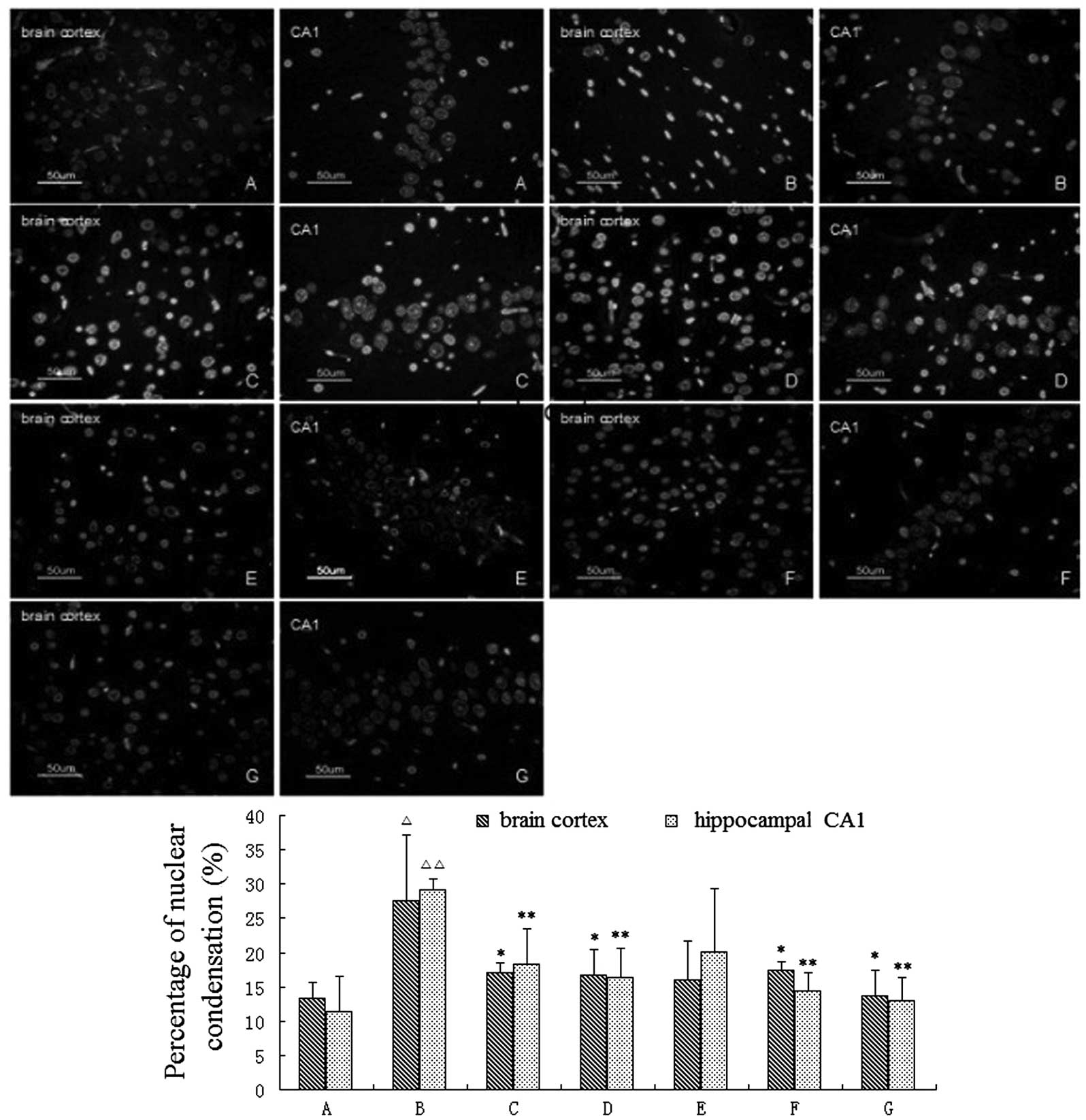 | Figure 3Effects of bilobalide (BB) on neuronal
apoptosis in the brain cortex and hippocampal CA1 region (Hoechst
33258 staining; magnification, ×400). Neuronal apoptosis in the
brain cortex and hippocampal CA1 region was significantly increased
in the rats of the (B) model group compared to the (A) sham-surgery
group. However, the number of apoptotic cells in the brain cortex
and the hippocampal CA1 region in the rats of the BB (4 and 8
mg/kg), Nimo and Hupa groups were significantly decreased. (A)
Sham-surgery, (B) model, (C) Nimo, (D) Hupa, (E) BB 2 mg/kg, (F) BB
4 mg/kg and (G) BB 8 mg/kg groups (n=5 rats/group).
△P<0.05, △△P<0.01 vs. sham-surgery
group; *P<0.05, **P<0.01 vs. model
group. |
Effects of BB on brain tissue SOD, NOS
activities and MDA, GSH contents
SOD activity and GSH content were significantly
decreased (P<0.01), while the MDA content and NOS activity were
clearly increased (P<0.01) in the rats of the model group
compared with the sham-surgery group. BB 2, 4 and 8 mg/kg), Nimo
and Hupa treatment differentially increased SOD activity and GSH
content (P<0.05 or <0.01), but decreased MDA content and NOS
activity in VD rats compared with the rats in the model group
(P<0.05 or <0.01) (Tables
III and IV).
 | Table IIIEffects of BB on brain tissue SOD
activity and MDA content in VD rats. |
Table III
Effects of BB on brain tissue SOD
activity and MDA content in VD rats.
| Group | Dose (mg/kg) | SOD (KU/g
protein) | MDA (μmol/g
protein) |
|---|
| Sham | - | 125.54±8.06 | 3.31±0.78 |
| Model | - |
104.78±10.63a | 4.91±0.62a |
| Nimo | 10.5 | 119.53±9.80b | 3.76±0.94b |
| Hupa | 0.047 |
117.67±10.85b | 3.87±0.90b |
| BB | 2 | 115.80±9.23b | 4.08±0.71b |
| 4 | 116.88±8.96b | 3.83±0.84b |
| 8 | 118.46±6.93c | 3.65±0.59c |
 | Table IVEffects of BB on brain tissue NOS
activity and GSH content in VD rats. |
Table IV
Effects of BB on brain tissue NOS
activity and GSH content in VD rats.
| Group | Dose (mg/kg) | NOS (KU/g
protein) | GSH (μmol/g
protein) |
|---|
| Sham | - | 0.44±0.09 | 29.06±6.50 |
| Model | - | 0.75±0.15a | 16.30±4.29a |
| Nimo | 10.5 | 0.54±0.17b | 23.11±5.57b |
| Hupa | 0.047 | 0.53±0.16b | 23.45±5.28c |
| BB | 2 | 0.61±0.15 | 22.16±5.96b |
| 4 | 0.54±0.19b | 22.32±3.83b |
| 8 | 0.51±0.17c | 24.57±3.60c |
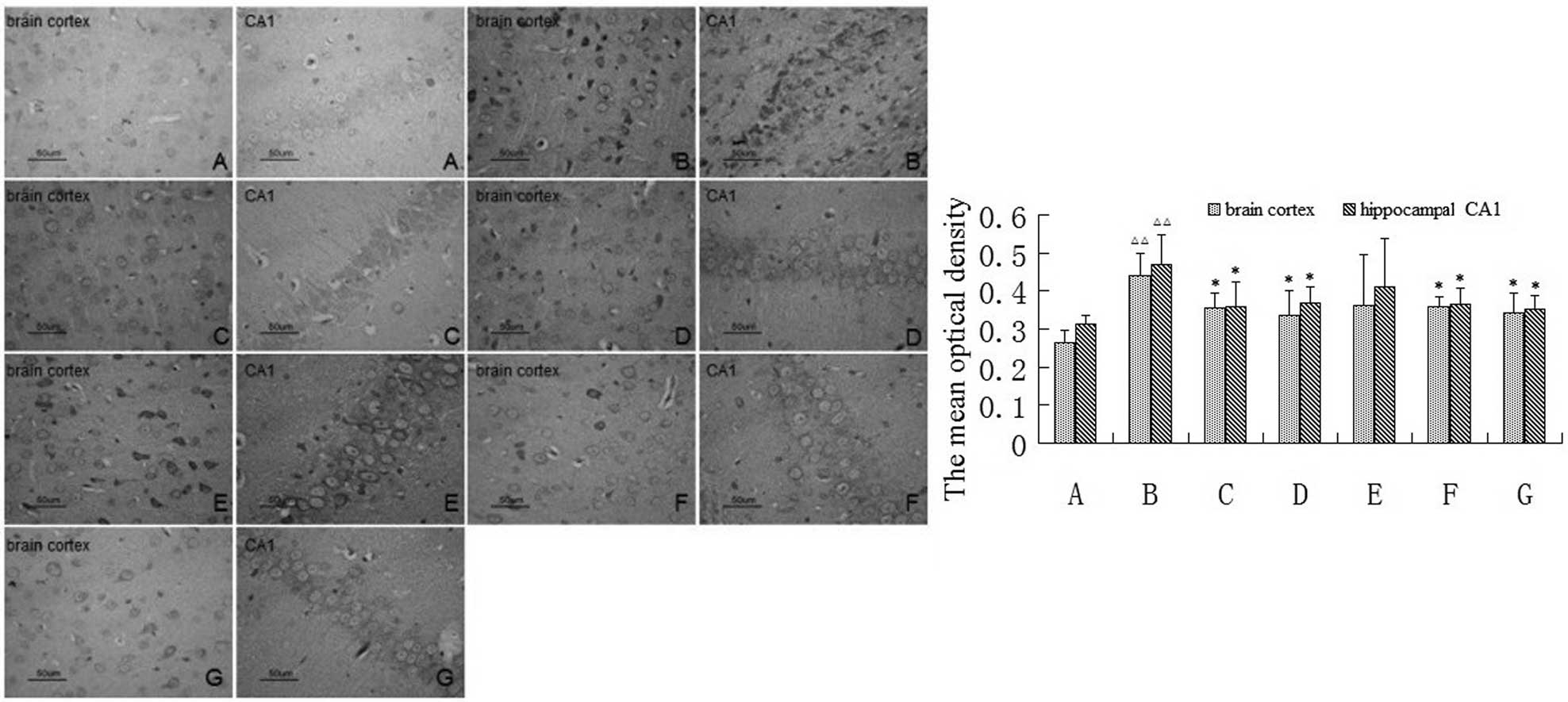
Effect of BB on the expression of TNF-α
in the brain cortex and the hippocampal CA1 region
The effect of BB on TNF-α immunoreactivity in the
brain cortex and the hippocampal CA1 region in VD rats was
investigated. Under normal conditions, immunoreactive cells are
abundant and appear brown in color following immunohistochemistry.
Many TNF-α immunoreactive neuronal cells were observed in the brain
cortex and the hippocampal CA1 region in the rats of the model
group compared with the sham-surgery group. However, the number of
TNF-α immunoreactive cells in the BB (4 and 8 mg/kg), Nimo and
Hupa-treated rats was significantly decreased compared with the
model group (Fig. 4).
The mean optical density of the brain cortex and the
hippocampal CA1 region in the rats of the model group was
significantly increased compared with the sham-surgery group
(P<0.01). BB (4 and 8 mg/kg), Nimo and Hupa treatment obviously
decreased the expression of TNF-α in these regions of VD rats
(P<0.05) (Fig. 4).
Discussion
The 2-VO rat is a suitable model for investigating
the pathophysiology of learning and memory impairment in human
dementia, as well as for assessing the therapeutic potential and/or
exploring possible mechanisms of putative anti-dementia drugs
(13). BB, a sesquiterpene
trilactone constituent of Ginkgo biloba leaf extracts, has
been suggested to exert protective and trophic effects on neurons
(8,9,14).
MWM has been proven to be an effective method for the ethological
evaluation of rats, and has been used to evaluate the spatial
learning and memory abilities of rats (15). In the present study, the spatial
learning and memory abilities of rats were decreased in the model
group, while they were found to be improved following BB (4 and 8
mg/kg) treatment. These results suggest that daily administration
of BB rescues cognitive deficits in VD rats.
The mechanism underlying the permanent occlusion of
both common carotid arteries responsible for inducing learning and
memory deficits is intricate. Neuronal apoptosis has been found to
be related to deficits in spatial learning and memory in rats
induced by chronic cerebral hypoperfusion (16). Hippocampus plays an important role
in learning and memory processing, and is highly sensitive to
ischemic insults (11,17). Cerebral ischemia inducing neuronal
degeneration has also been observed in other structures, such as
striatum, brain cortex and thalamus (18,19).
In the present study, histological changes and neuronal apoptosis
in the brain cortex and the hippocampal CA1 region of rats were
investigated. Histological examination plays an important role in
the evaluation of neuronal damage and drug action (20). H&E and Hoechst 33258 staining
showed that the number of apoptotic cells was significantly
increased in the rats of the model group, and that BB (4 and 8
mg/kg) treatment alleviated the neuronal impairment and apoptosis.
These results suggest that BB has an antiapoptotic activity.
Oxidative stress is involved in neuronal apoptosis
(21). It is well known that free
radicals participate in the regulation of neuron degeneration and
apoptosis, and are potentially involved in the pathogenesis of
neurodegenerative diseases such as VD (22,23).
MDA is an important biomarker of lipid peroxidation that is induced
by reactive oxygen species. Specific quantification of MDA is able
to determine the extent of lipid damage as a result of oxidative
stress in a variety of lipid systems, such as plasma, organs and
cell membranes (24). Free
radicals cause oxidative damage to important biological molecules.
Consequently, organisms exhibit increased SOD, catalase and
glutathione peroxidase activities in response to oxidative stress
(25). Increased SOD activity
could be regarded as an indication that the antioxidant mechanism
of the brain is activated in response to oxidative stress (26). Free radicals directly damage
antioxidant enzymes and reduce their activities (27). In the present study, GSH content
was found to be significantly decreased in VD rats. Therefore, the
increment in MDA content and SOD activity found in the rats of the
model group suggests an obviously increased oxidative process,
while the decrease in the GSH content indicates the presence of
deficiencies in the endogenous antioxidant ability. Nitric oxide
(NO) might exacerbate the damage by enhancing the post-ischemic
release of excitatory neurotransmitters (28). Changes in the level of NO produced
by NOS occur in acute cerebral ischemia and Alzheimer's disease
(AD) (29). In the present study,
BB (2, 4 and 8 mg/kg) significantly increased SOD activity and GSH
content, and decreased MDA content and NOS activity in VD rats.
Previous studies have demonstrated that agents with antioxidant and
radical scavenger properties have been effective in the treatment
of VD in animals or in clinical trials (30–32).
In the present study, learning and memory dysfunction is suggested
to have been alleviated by BB due to its antioxidant properties,
since oxidative stress in all cases plays an important role in
2-VO-induced neurodegenerative processes (33).
TNF-α, a pro-inflammatory cytokine that is produced
by macrophages, adipocytes and astrocytes, appears to be an
unspecific but potent factor in the development of several
psychiatric diseases, including depression and dementia. Elevated
TNF-α levels have been found in the cerebrospinal fluid of patients
with AD and VD, suggesting an important role of TNF-α production by
the central nervous system in dementia (34). TNF-α has been shown to induce
neuronal death in several neurodegenerative disorders including AD
and VD (35). The results of the
present study have shown that BB (4 and 8 mg/kg) significantly
decreased the expression of TNF-α in the brain cortex and the
hippocampal CA1 region of VD rats. This suggests that the
inhibitory effect of BB on neuronal apoptosis is associated with
the regulation of TNF-α expression by BB.
In conclusion, the present study has demonstrated
that BB has a potential antioxidant activity, inhibits neuronal
apoptosis and reduces the expression of TNF-α in the brain cortex
and the hippocampal CA1 region of VD rats. These effects might be
related to its antioxidant and antiapoptotic activities. However,
additional studies are needed for the elucidation of the detailed
mechanism between the two activities of BB.
Acknowledgements
This study was supported by the Doctor Foundation of
Anhui Medical University (XJ201011) and the Teaching Quality
Project of Anhui Higher Education (20101941). The authors thank
Dake Huang, Shan Huang and Li Gui (Synthetic Laboratory of Basic
Medicine College, Anhui Medical University, Hefei, China) for their
excellent technical assistance.
References
|
1
|
Román GC: Vascular dementia:
distinguishing characteristics, treatment, and prevention. J Am
Geriatr Soc. 51:S296–S304. 2003.PubMed/NCBI
|
|
2
|
Ahlemeyer B and Krieglstein J:
Neuroprotective effects of Ginkgo biloba extract. Cell Mol
Life Sci. 60:1779–1792. 2003. View Article : Google Scholar
|
|
3
|
Chandrasekaran K, Mehrabian Z, Spinnewyn
B, Chinopoulos C, Drieu K and Fiskum G: Neuroprotective effects of
bilobalide, a component of Ginkgo biloba extract (EGb 761)
in global brain ischemia and in excitotoxicity-induced neuronal
death. Pharmacopsychiatry. 36(Suppl 1): S89–S94. 2003.PubMed/NCBI
|
|
4
|
Tanaka Y, Marumo T, Omura T and Yoshida S:
Serum S100B indicates successful combination treatment with
recombinant tissue plasminogen activator and MK-801 in a rat model
of embolic stroke. Brain Res. 1154:194–199. 2007. View Article : Google Scholar
|
|
5
|
Smith JV and Luo Y: Studies on molecular
mechanisms of Ginkgo biloba extract. Appl Microbiol
Biotechnol. 64:465–472. 2004. View Article : Google Scholar
|
|
6
|
Klein J, Chatterjee SS and Löffelholz K:
Phospholipid breakdown and choline release under hypoxic
conditions: inhibition by bilobalide, a constituent of Ginkgo
biloba. Brain Res. 755:347–350. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Weichel O, Hilgert M, Chatterjee SS, Lehr
M and Klein J: Bilobalide, a constituent of Ginkgo biloba,
inhibits NMDA-induced phospholipase A2 activation and phospholipid
breakdown in rat hippocampus. Naunyn Schmiedebergs Arch Pharmacol.
360:609–615. 1999.PubMed/NCBI
|
|
8
|
Rossi R, Basilico F, Rossoni G, Riva A,
Morazzoni P and Mauri PL: Liquid chromatography/atmospheric
pressure chemical ionization ion trap mass spectrometry of
bilobalide in plasma and brain of rats after oral administration of
its phospholipidic complex. J Pharm Biomed Anal. 50:224–227. 2009.
View Article : Google Scholar
|
|
9
|
Defeudis FV: Bilobalide and
neuroprotection. Pharmacol Res. 46:565–568. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sarti C, Pantoni L, Bartolini L and
Inzitari D: Persistent impairment of gait performances and working
memory after bilateral common carotid artery occlusion in the adult
Wistar rat. Behav Brain Res. 136:13–20. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ni J, Ohta H, Matsumoto K and Watanabe H:
Progressive cognitive impairment following chronic cerebral
hypoperfusion induced by permanent occlusion of bilateral carotid
arteries in rats. Brain Res. 653:231–236. 1994. View Article : Google Scholar
|
|
12
|
Janus C, Pearson J, McLaurin J, et al: A
beta peptide immunization reduces behavioural impairment and
plaques in a model of Alzheimer's disease. Nature. 408:979–982.
2000. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sarti C, Pantoni L, Bartolini L and
Inzitari D: Cognitive impairment and chronic cerebral
hypoperfusion: what can be learned from experimental models. J
Neurol Sci. 203–204:263–266. 2002.PubMed/NCBI
|
|
14
|
Bruno C, Cuppini R, Sartini S, Cecchini T,
Ambrogini P and Bombardelli E: Regeneration of motor nerves in
bilobalide-treated rats. Planta Med. 59:302–307. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
D'Hooge R and De Deyn PP: Applications of
the Morris water maze in the study of learning and memory. Brain
Res Brain Res Rev. 36:60–90. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bennett SA, Tenniswood M, Chen JH,
Davidson CM, Keyes MT, Fortin T and Pappas BA: Chronic cerebral
hypoperfusion elicits neuronal apoptosis and behavioral impairment.
Neuroreport. 9:161–166. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Pappas BA, de la Torre JC, Davidson CM,
Keyes MT and Fortin T: Chronic reduction of cerebral blood flow in
the adult rat: late-emerging CA1 cell loss and memory dysfunction.
Brain Res. 708:50–58. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Freund TF, Buzsáki G, Leon A, Baimbridge
KG and Somogyi P: Relationship of neuronal vulnerability and
calcium binding protein immunoreactivity in ischemia. Exp Brain
Res. 83:55–66. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Pereira LO, Nabinger PM, Strapasson AC,
Nardin P, Gonçalves CA, Siqueira IR and Netto CA: Long-term effects
of environmental stimulation following hypoxia-ischemia on the
oxidative state and BDNF levels in rat hippocampus and frontal
cortex. Brain Res. 1247:188–195. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hunter AJ, Mackay KB and Rogers DC: To
what extent have functional studies of ischaemia in animals been
useful in the assessment of potential neuroprotective agents?
Trends Pharmacol Sci. 19:59–66. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Coyle JT and Puttfarcken P: Oxidative
stress, glutamate, and neurodegenerative disorders. Science.
262:689–695. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Markesbery WR: Oxidative stress hypothesis
in Alzheimer's disease. Free Radic Biol Med. 23:134–147. 1997.
View Article : Google Scholar
|
|
23
|
Chong ZZ, Li F and Maiese K: Oxidative
stress in the brain: novel cellular targets that govern survival
during neurodegenerative disease. Prog Neurobiol. 75:207–246. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lazzarino G, Tavazzi B, Di Pierro D,
Vagnozzi R, Penco M and Giardina B: The relevance of
malondialdehyde as a biochemical index of lipid peroxidation of
postischemic tissues in the rat and human beings. Biol Trace Elem
Res. 47:165–170. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Halliwell B: Reactive oxygen species in
living systems: source, biochemistry, and role in human disease. Am
J Med. 91:S14–S22. 1991.PubMed/NCBI
|
|
26
|
Bannister JV, Bannister WH and Rotilio G:
Aspects of the structure, function, and applications of superoxide
dismutase. CRC Crit Rev Biochem. 22:111–180. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Escobar JA, Rubio MA and Lissi EA: Sod and
catalase inactivation by singlet oxygen and peroxyl radicals. Free
Radic Biol Med. 20:285–290. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Montague PR, Gancayco CD, Winn MJ,
Marchase RB and Friedlander MJ: Role of NO production in NMDA
receptor-mediated neurotransmitter release in cerebral cortex.
Science. 263:973–977. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Iadecola C: Bright and dark sides of
nitric oxide in ischemic brain injury. Trends Neurosci. 20:132–139.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tabet N, Mantle D, Walker Z and Orrell M:
Vitamins, trace elements, and antioxidant status in dementia
disorders. Int Psychogeriatr. 13:265–275. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
DeFeudis FV and Drieu K: Ginkgo
biloba extract (EGb 761) and CNS functions: basic studies and
clinical applications. Curr Drug Targets. 1:25–58. 2000. View Article : Google Scholar
|
|
32
|
Xiong Z, Liu C, Wang F, Li C, Wang W, Wang
J and Chen J: Protective effects of breviscapine on ischemic
vascular dementia in rats. Biol Pharm Bull. 29:1880–1885. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Farkas E, Luiten PG and Bari F: Permanent,
bilateral common carotid artery occlusion in the rat: a model for
chronic cerebral hypoperfusion-related neurodegenerative diseases.
Brain Res Rev. 54:162–180. 2007. View Article : Google Scholar
|
|
34
|
Tarkowski E, Blennow K, Wallin A and
Tarkowski A: Intracerebral production of tumor necrosis
factor-alpha, a local neuroprotective agent, in Alzheimer disease
and vascular dementia. J Clin Immunol. 19:223–230. 1999. View Article : Google Scholar
|
|
35
|
Zuliani G, Ranzini M, Guerra G, Rossi L,
Munari MR, Zurlo A, Volpato S, Atti AR, Blè A and Fellin R: Plasma
cytokines profile in older subjects with late onset Alzheimer's
disease or vascular dementia. J Psychiatr Res. 41:686–693. 2007.
View Article : Google Scholar : PubMed/NCBI
|