Introduction
Persistent inflammatory microenvironment induced by
bacteria, including Mycoplasma, is conducive to tumor development
in host tissues (1). Mycoplasma
promotes multistage malignant transformation of host cells
following a long-term infection (2,3).
Being different from other bacteria, Mycoplasma lacks a rigid cell
wall, which facilitates fusion with the host cell membrane
(4). Following fusion, Mycoplasma
may lead to various alterations in the affected cells, which
involves the Mycoplasma membrane components inserting into the host
cell membrane. Among those mycoplasmal components, the
lipid-associated membrane proteins have been reported to activate
the Jak/Stat3 pathway through mediating host small GTPases of the
Rho family, including Rac1 (5,6).
Rac1 is a significant member of Rho-family small
GTPases, which are pleiotropic in controlling actin cytoskeleton
reorganization (7) and Stat3 is an
important transcription factor that may be activated by the
GTP-bound form of active Rac1 (8–10).
In terms of subcellular localization, active Rac1 contributes to
Stat3 nuclear translocation by forming a complex with a Rac1/Cdc42
GTPase-activating protein (11,12),
whereas cytoplasmic Stat3 regulates Rac1 activity to modulate
directional cell migration (13).
The functional and physical interactions between Rac1 and Stat3
potentiate cell proliferation and migration in certain cell types
(8,13–15).
There are a number of bacterial virulence factors
that possess a WxxxE motif, which may subvert cellular functions
via interacting with host small GTPases of the Rho family. These
identified factors include: The Shigella effectors, IpgB1
and IpgB2 (16), the E.
coli effector, Map (17), the
Salmonella effector, SifA (18)
and Pantoea stewartii subsp. stewartii and Pseudomonas
syringae effectors, WtsE and AvrE1, respectively (19).
Through computational analysis of Mycoplasma
genomes, the chromosome partition protein Smc (Smc) conserved among
Mycoplasma species was observed to possess an invariant WxxxE motif
at its N terminus, which is associated with Rho family small
GTPases, as well as a probable coiled coil domain downstream of the
WxxxE motif, which may interact with Stat3 as predicted by an
online program designed for predicting the parallel coiled coil
interaction (20). Notably, Stat3
is a binding partner of Rac1 (8);
therefore, the present study aimed to determine whether the
N-terminal domain of Smc interacts with Rac1 and/or Stat3.
The N-terminal sequence (amino acids 9–248) of Smc
(UniProt: Q98PK8) was cloned from Mycoplasma pulmonis, a
common Mycoplasma contaminant in cell cultures. The cloned
240-amino acid N-terminus of Smc possesses a characteristic
GTP-binding motif of GxxxxGKS/T that is observed in all small
GTPases. With regard to such a similarity with small GTPases, this
small GTPase-like protein fragment of 240 amino acids from Smc is
referred to as SGLP.
In the current study, the interaction of SGLP with
Rac1 and Stat3 was investigated and the mechanism by which SGLP may
be involved in affecting tumor cell migration and proliferation
through interaction with Rac1 and Stat3 was demonstrated.
Materials and methods
Antibodies
The primary antibodies used were: p-Stat3 (9145)
from Cell Signaling Technology, Inc. (Beverly, MA, USA);
glutathione S-transferase (GST; sc-33614) and Rac1 (sc-217) from
Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA); FLAG (T510-2)
and green fluorescent protein (GFP; T508-2) from Signalway Antibody
(Baltimore, MD, USA); Myc-Tag (AM1007a) from Abgent (San Diego, CA,
USA); anti-bromodeoxyuridine (BrdU; 560810) from BD Biosciences
(San Jose, CA, USA) and Stat3 (51076-2-AP) from Proteintech Group
(Chicago, IL, USA). Other antibodies used, including
FITC-conjugated goat anti-mouse, Alexa Fluor 488-conjugated goat
anti-rabbit, Alexa Fluor 594-conjugated goat anti-mouse antibodies
and HRP-conjugated goat antibodies against rabbit and mouse, were
purchased from Jiayuan Biotech Co. Ltd. (Wuhan, China).
Overlapping polymerase chain reaction
(PCR) and plasmid construction
The point mutation for tryptophan and the mutation
of WVLGE to AVLGA were generated by overlapping PCR using primers
as follows:i) point mutation for tryptophan of SGLP, N-terminus
5′-ATGGATCCAAATCATTTGCAGAGCCAATTC-3′ and
5′-GTTCACCCAAGACCCATTTAATGGCATC-3′ and C-terminus
5′-GCCATTAAATGGGTCTTGGGTGAA CAAT-3′ and 5′-CACCGAATTCCTCAATTTCAAATT
CTTTTAGTTTG-3′ and ii) point mutation for WxxxE to AxxxA,
N-terminus 5′-GATATCAAATCATTTGCAGAGCCAAT-3′ and
5′-TGTGCACCCAAGACCGCTTTAATG-3′ and C-terminus
5′-AAAGCGGTCTTGGGTGCACAATC-3′ and 5′-GCGAATTCCTACTCAATTTCAAATTC-3′.
SGLP sequences were cloned into pGEX4T1, pEGFP-C1 and
pCDF1-MCS2-EF1-Puro vectors (System Biosciences, Mountain View, CA,
USA). In addition, the FLAG-tag DYKDDDDK and Kozak sequences were
inserted into the pCDF1-MCS2-EF1-Puro vector. DN-Rac1 (T17N) and
CA-Rac1 (Q61L) pRK5 plasmids were purchased from Addgene Inc.
(Cambridge, MA, USA).
Cell culture, transfection and stable
cell line establishment
HEK 293T and HeLa cells were cultured in Dulbecco’s
modified Eagle’s medium (DMEM; Hyclone, Logan, UT, USA) and
supplemented with 10% fetal bovine serum (FBS; Hyclone) at 37°C in
a humidified 5% CO2 atmosphere. Lentiviral packaging was
conducted in HEK 293T cells using pPACK™ Packaging mix (System
Biosciences) according to the manufacturer’s instructions. The
lentiviral titers were determined by an UltraRapid Titering kit
(System Biosciences). HeLa cells were transduced with SGLP
lentiviral particles with 6 μg/ml polybrene (Santa Cruz
Biotechnology, Inc.) for 12 h. Transduced cells were selected with
2 μg/ml puromycin (Sigma-Aldrich, St. Louis, MO, USA) for two weeks
and the stable cell lines were maintained in growth medium with 1
μg/ml puromycin. Lipofectamine™ 2000 (Invitrogen Life Technologies,
Carlsbad, CA, USA) was used for transfection of plasmids and small
interfering RNAs according to the manufacturer’s instructions.
GST pull-down assay, immunoprecipitation
and western blot analysis
GST-tagged protein expression was induced by adding
0.1 mM isopropyl-β-d-thiogalactopyranoside to BL21 E. coli
cultures at 30°C for 6 h. GST-tagged proteins were purified by
affinity chromatography with glutathione Sepharose™ 4B beads
(Amersham Biosciences, Uppsala, Sweden) according to the
manufacturer’s instructions. HeLa cells (~1×107) were
treated with 100 ng/ml IL-6 for 24 h prior to harvesting. Equal
quantities of precleared lysates (~2 mg) were incubated with
sepharose beads and 10 μg GST or GST-SGLP for 2 h at 4°C. The beads
were washed three times with washing buffer (pH 7.2, 10 mM
NaPO4, 10 mM NaN3, 150 mM NaCl and 0.1%
Tween-20). The bait and pray proteins were then eluted from beads
with elution buffer (pH 8.0, 50 mM Tris-Cl and 30 mM GSH) and
subjected to western blot analysis. The active Rac1 pull-down assay
was conducted according to a modified method from Teng et
al(13). The GST-Pak1-PBD,
which contains the peptide ISLPSDFEHTIHVGF (CRIB domain), was
purchased from Millipore (Billerica, MA, USA) and active Rac1, as
well as total Rac1, were probed separately. For
immunoprecipitation, the GFP- or GFP-SGLP-transfected HeLa cell
lysates (1 mg) were precleared with 25 μl protein G sepharose beads
(Amersham Biosciences) and incubated with 2 μg primary antibodies
at 4°C for 2 h, followed by incubation with 25 μl protein G
sepharose beads at 4°C for 2 h. Western blot analyses were
performed as previously described (21).
Confocal microscopy, fluorescence
resonance energy transfer (FRET) and photobleaching analyses
Immunofluorescence images were captured using an
Olympus FluoView™ FV1000 confocal microscope. HeLa cells expressing
SGLP and vector control were labeled with 250 nM Mitotracker at
37°C for 15 min. The cells were then fixed, permeabilized and
stained with 50 μg/ml fluorescein isothiocyanate (FITC)-phalloidin
at room temperature for 2 h followed by DAPI counterstaining. The
ImageJ plugins for colocalization analysis are described and may be
downloaded at http://fiji.sc/wiki/index.php/Colocalization_Analysis.
The colocalization of FLAG-SGLP with Stat3 was assessed with the
ImageJ plugin as described by Fay et al(22) and the Pearson’s correlation
coefficients were also calculated using ImageJ. For FRET analysis,
images were recorded in three channels for donor, acceptor and
transfer, respectively. The donor and acceptor bleed-through was
determined and the pseudocolor image of FRET was captured using the
ImageJ plugin ‘FRET and Colocalization Analyzer’, as described by
Hachet-Hass et al(23).
Photobleaching of DsRed-Rac1 was achieved by continuous excitation
at 561 nm with full-lamp intensity. Images of GFP-SGLP and
DsRed-Rac1 were recorded at intervals of 30 sec.
Transwell migration assay
Transwell assays were conducted using Transwell
migration chambers with an 8-μm pore size (Corning Inc., Acton, MA,
USA) according to the manufacturer’s instructions. HeLa cells
(~1×105) suspended in 500 μl of serum-free DMEM were
seeded in each chamber. The migration chamber was placed into a
24-well plate with 500 μl DMEM containing 10% FBS and incubated at
37°C with 5% CO2. Following 8 h, cells on the upper
surface were removed with a cotton swab. The migrated cells on the
lower surface were fixed and stained with 1% crystal violet. Five
microscopic fields (magnification, ×200) were counted per filter in
three independent experiments.
BrdU incorporation by flow cytometry
HeLa cells were incubated with 50 μM BrdU at 37°C
for 30 min followed by fixation in 80% ethanol at 4°C overnight.
The cells were sequentially incubated with 2 M HCl (room
temperature, 30 min) and 0.1 M
Na2B4O7 buffer (pH 8.5) for DNA
denaturation. Following extensive washing, the cells were
immunostained at room temperature with an anti-BrdU antibody (1:50)
for 2 h and a secondary FITC-conjugated antibody (1:200) for 1 h.
The cells were then resuspended in phosphate-buffered saline
containing 50 μg/ml propidium iodide and 125 μg/ml RNAase, prior to
flow cytometry.
Statistical analysis
Analysis of data from three independent experiments
were conducted using GraphPad Prism 5.0 software (GraphPad
Software, Inc., La Jolla, CA, USA) and expressed as the mean ± SD.
Student’s t-test was used for comparisons among groups. P<0.05
was considered to indicate a statistically significant
difference.
Results
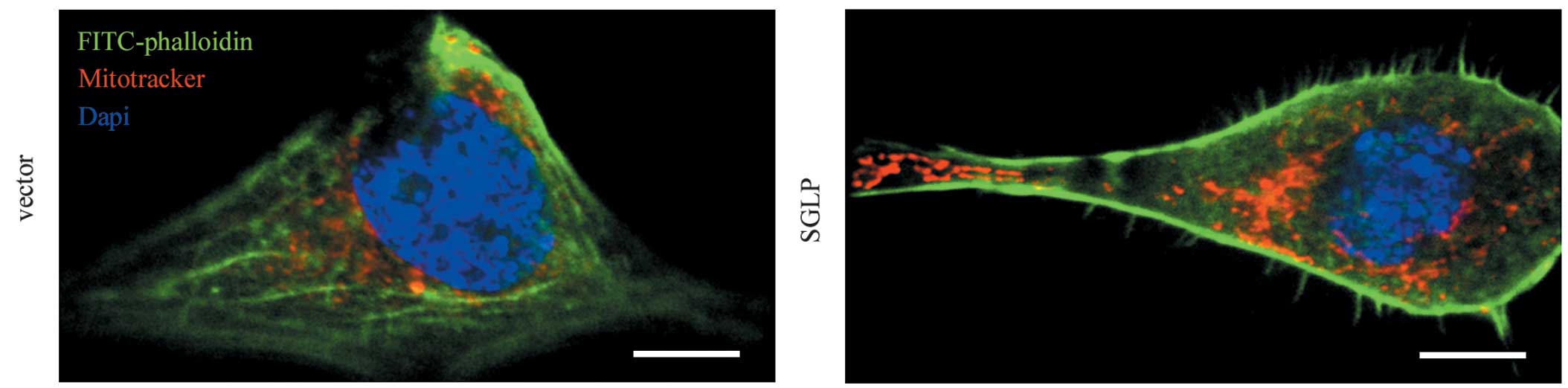
SGLP induces actin filament
reorganization in HeLa cells
As SGLP possesses a WxxxE motif, which is required
for a number of bacterial virulence factors in subverting host
cellular actin cytoskeleton, the actin filament patterns in HeLa
cells expressing SGLP or vector control by FITC-phalloidin staining
were examined. The actin cytoskeleton was observed to be markedly
altered following transduction of HeLa cells with lentiviral
particles expressing SGLP, which was characterized by reduced
stress fibers and increased lamellipodia and filopodia (Fig. 1). As the Rho family members of
small GTPases are key regulators of stress fibers, lamellipodia and
filopodia (24), this result
indicated that SGLP is associated with functions of host Rho small
GTPases.
SGLP interacts with host Rac1 and
Stat3
The results presented in Fig. 1 revealed that SGLP, which resembles
a number of bacterial virulence factors that contain a WxxxE motif,
was associated with Rho-family small GTPases. One of the members of
Rho small GTPases is Rac1, which is a binding partner of Stat3
(8). In addition, using
computational analysis, SGLP was observed to possess a coiled-coil
domain that may interact with the corresponding coiled-coil domain
of Stat3 (20). Therefore, it was
hypothesized that there are probable functional and/or physical
interactions of SGLP with Rac1 and Stat3. Thus, a possible
interaction between SGLP and Rac1 and Stat3 was investigated.
A GST pull-down assay revealed that the purified
GST-SGLP protein binds to host endogenous Rac1 and Stat3 (Fig. 2A). In addition, in HeLa cells,
endogenous Rac1 and Stat3 were co-immunoprecipitated with GFP-SGLP
using a GFP antibody (Fig. 2B).
Immunofluorescence analyses revealed that FLAG-tagged SGLP
colocalized with Stat3 in the nucleus and cytoplasm (Fig. 2C). The Pearson’s correlation
coefficient of colocalization between SGLP and Stat3 was ~0.75 as
measured by ImageJ, whereas that of the control was <0.2 in 9
cells from three independent experiments. GFP-SGLP also colocalized
with DsRed-Rac1 in HeLa cells coexpressing these two proteins
(Fig. 2D). Although the DsRed-Rac1
distribution was homogeneous throughout the cell, which makes it
difficult to interpret for colocalization directly, the FRET
between GFP-SGLP and DsRed-Rac1 revealed a significant
colocalization of SGLP and Rac1 compared with the control (Fig. 2D). Furthermore, consecutive
confocal images showed that the GFP-SGLP (donor) intensity
increased following photobleaching of the DsRed-Rac1 (acceptor)
(Fig. 2E). A linear correlation
was observed between the intensity of donor recovery vs. acceptor
photobleaching, indicating that GFP-SGLP interacts with DsRed-Rac1
(Fig. 2F).
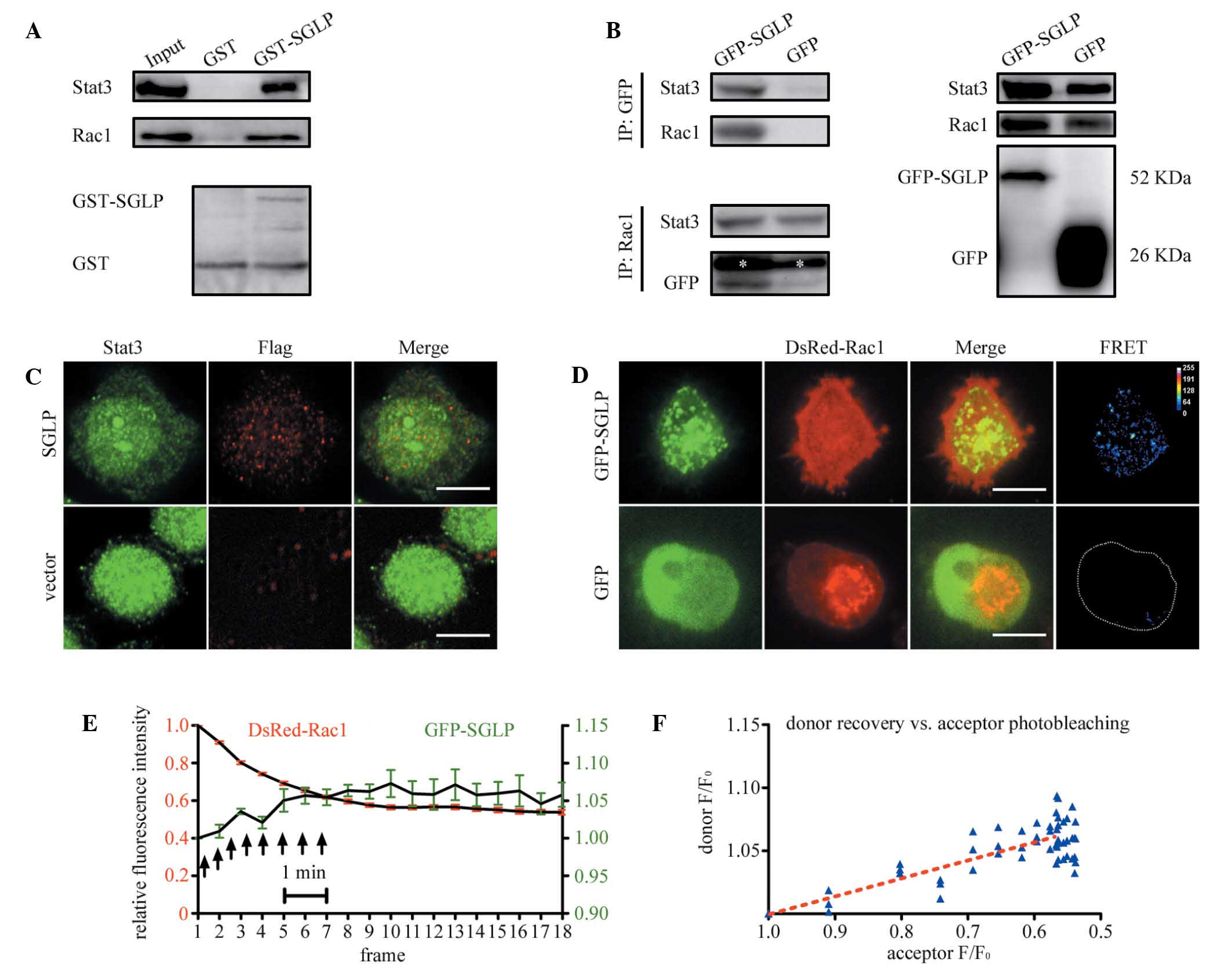 | Figure 2SGLP interacts with Rac1 and Stat3.
(A) GST-tagged SGLP (~10 μg) was used to pull-down endogenous Rac1
and Stat3 from IL-6 stimulated HeLa cell lysates (2 mg). The bait
protein, GST-SGLP and control GST were also immunoblotted with a
GST antibody. (B) HeLa cell lysates expressing GFP-SGLP were
immunoprecipitated with a GFP antibody and then probed with
anti-Stat3 and anti-Rac1 antibodies. The cell lysates were also
immunoprecipitated with a Rac1 antibody and then probed with
anti-Stat3 and anti-GFP antibodies. The white asterisk indicates
the location of the heavy chain. The right panel shows the cell
lysates of input probed with indicated antibodies. (C) HeLa cells
expressing FLAG-SGLP were fixed, permeabilized and immunostained
with anti-Stat3 (rabbit) and anti-FLAG (mouse) antibodies in
conjugation with Alexa Fluor 488 and 594 secondary antibodies,
respectively. The confocal images were analyzed for colocalization
using an ImageJ plugin. The boxed region is enlarged. Scale bar, 10
μm. (D) HeLa cells coexpressing GFP-SGLP and DsRed-Rac1 were fixed
in 4% paraformaldehyde and observed by confocal microscopy. As
DsRed-Rac1 was homogenously expressed, the FRET between GFP-SGLP
and DsRed-Rac1 was measured, and the FRET indices of GFP-SGLP and
GFP control were plotted as pseudocolor images. Scale bar, 10 μm.
(E) HeLa cells, as in (D), were observed by confocal microscopy
following photobleaching of DsRed-Rac1. Images were recorded for
DsRed-Rac1 (red) and GFP-SGLP (green) every 30 sec during and
following photobleaching, and the relative fluorescence intensities
were plotted. The black arrows indicate the duration of full-lamp
excitation. The negative GFP control did not exhibit such a
recovery following photobleaching of DsRed-Rac1 (data not shown).
Bar represents ± SD of nine cells from three independent
experiments. (F) A plot of the linear correlation of donor recovery
vs. acceptor photobleaching from (E). F0 represents the
original fluorescence intensity. SGLP, small GTPase-like protein
fragment; GST, glutathione S-transferase; IL, interleukin; GFP,
green fluorescent protein. |
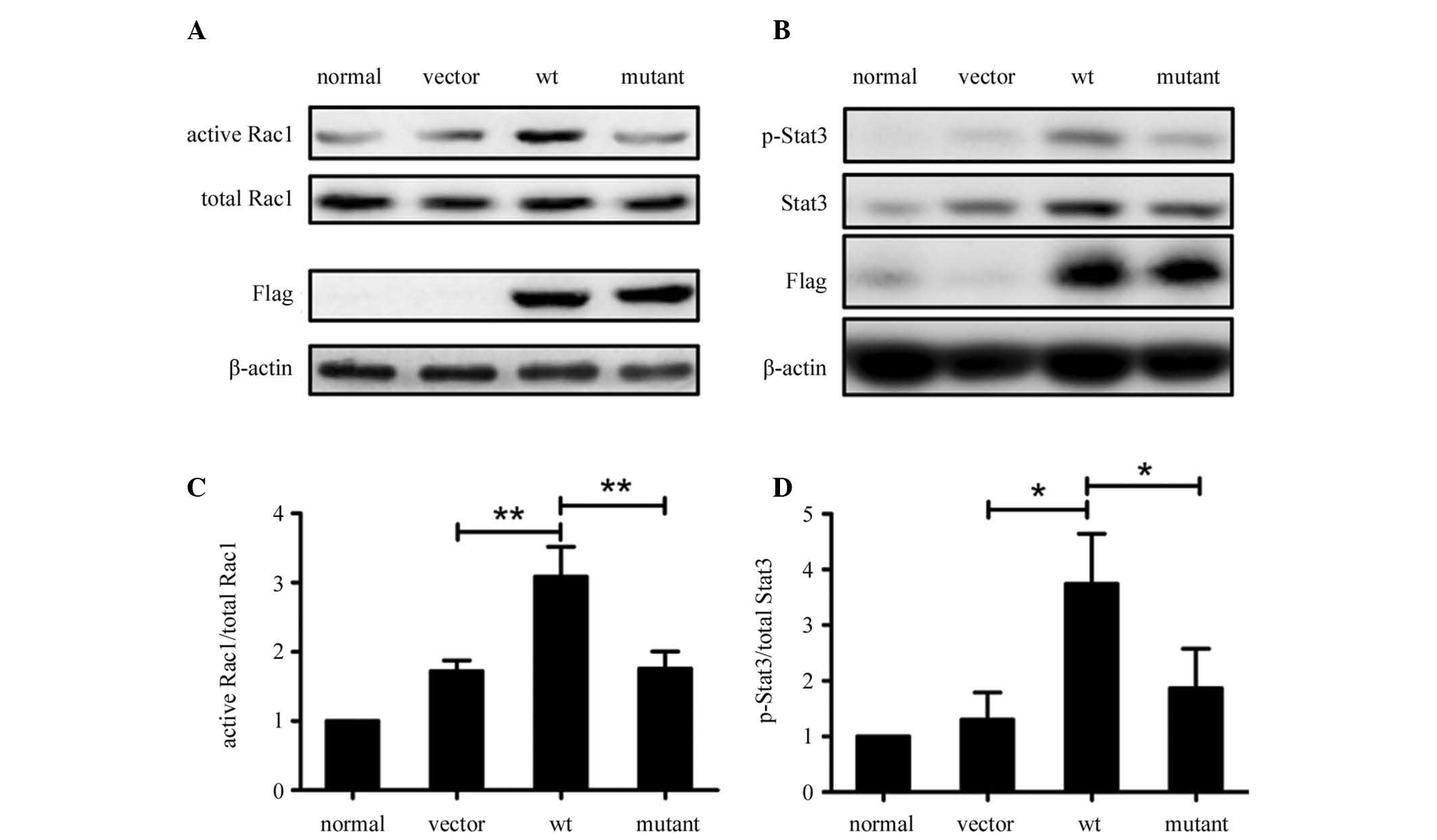
WxxxE motif is required for SGLP-induced
activation of Rac1 and phosphorylation of Stat3
The results in Fig.
2 revealed that SGLP interacts with Rac1 and Stat3, therefore
the study focused on the effect of SGLP on the function of Rac1 and
Stat3. As the WxxxE motif is required for a number of bacterial
virulence factors to activate cellular Rho-family small GTPases,
including Rac1 (16), it was
hypothesized that SGLP may also activate Rac1 and that the WxxxE
motif is crucial for this effect. Therefore the study investigated
whether SGLP activates Rac1, as well as whether the WxxxE motif is
responsible for the activation of Rac1. The wild-type (wt) WVLGE
sequence within wt SGLP was mutated to AVLGA to generate an SGLP
mutant. HeLa cell lines stably expressing wt SGLP, SGLP mutant or
vector, as well as parental HeLa cells were studied. Active Rac1
levels were determined by a GST-Pak1 pull-down assay followed by
western blot analysis. In the presence of the WxxxE motif, wt SGLP
induced greater Rac1 activation in HeLa cells as compared with the
SGLP mutant and the vector control, as well as the parental cells
(Fig. 3A and C). In addition,
western blot analysis revealed that wt SGLP increased the
phosphorylation of Stat3 at tyrosine-705 residue (p-Stat3) in HeLa
cells and that the SGLP mutant did not exhibit such a prominent
effect on Stat3 phosphorylation (Fig.
3B and D).
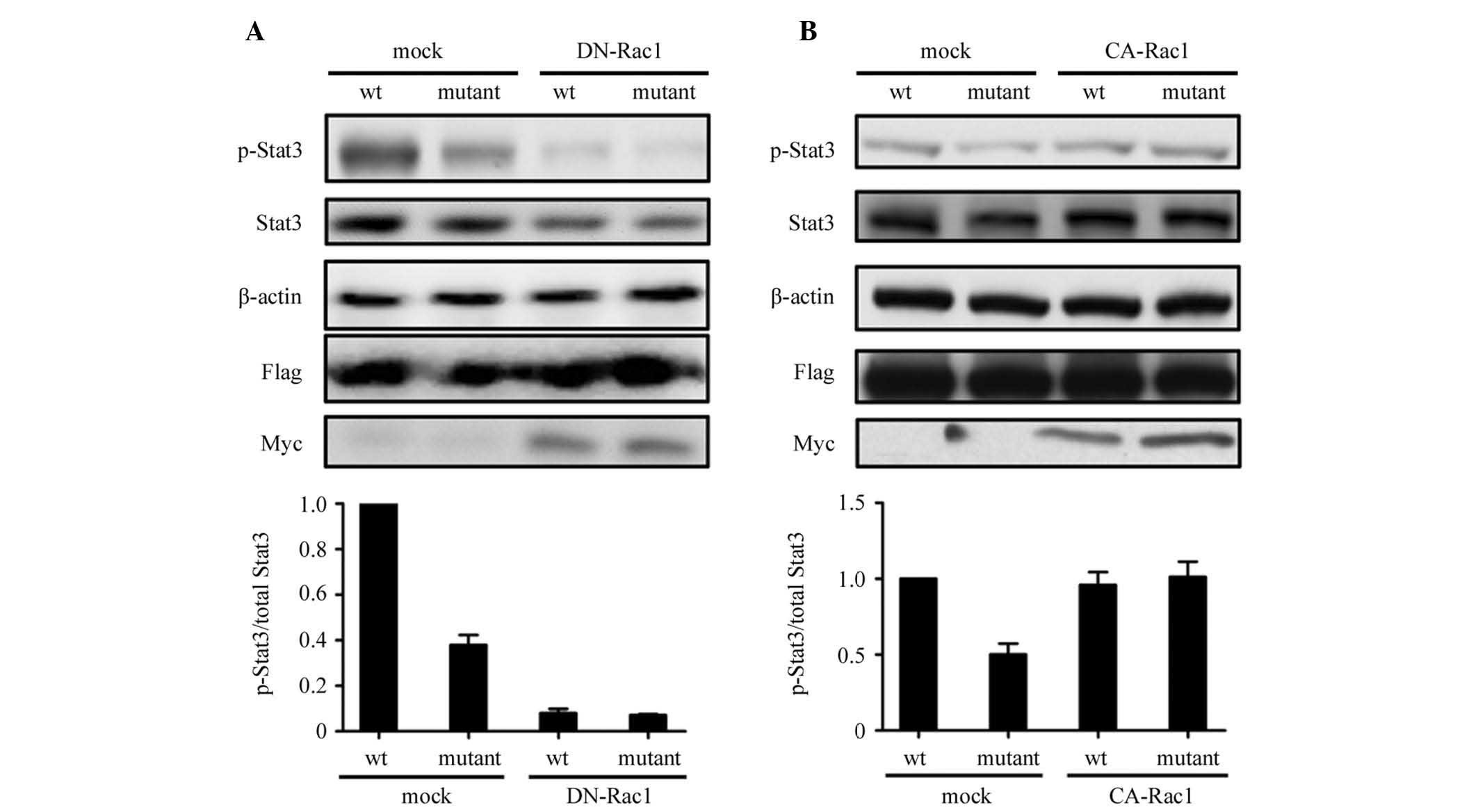
SGLP-induced Stat3 activation is
dependent upon Rac1 activity
Fig. 3 shows that
wt SGLP increased cellular p-Stat3 levels, which reflects its role
in the activation of Stat3. Thus, the mechanism by which SGLP
activates Stat3 was investigated. As Rac1 mediates Stat3 activity
through direct binding to Stat3 and an indirect activation loop of
autocrine IL-6 (8,25), it was examined whether the
SGLP-induced activation of Stat3 was dependent on Rac1 activity.
The wt and mutant SGLP-transduced HeLa cells were transfected with
dominant negative Rac1 (DN-Rac1) or constitutive active Rac1
(CA-Rac1) plasmids separately. Overexpression of DN-Rac1 was
observed to eliminate the increase in p-Stat3 levels in wt
SGLP-transduced HeLa cells (Fig.
4A), whereas overexpression of CA-Rac1 rescued the decrease in
p-Stat3 levels in mutant SGLP-transduced HeLa cells (Fig. 4B). The results suggested that SGLP
activates Stat3 depending on the active form of Rac1.
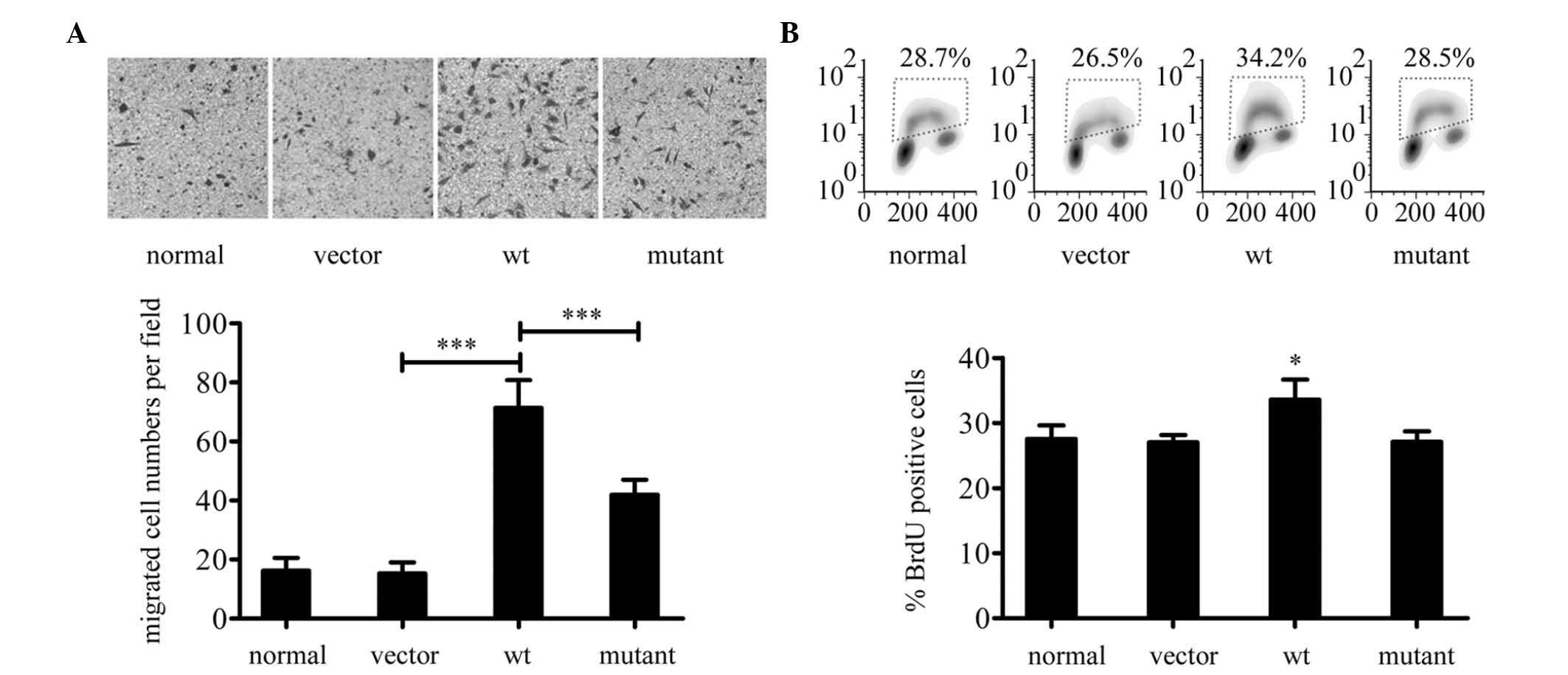
SGLP promotes migration and proliferation
of HeLa cells
The aforementioned observations suggest that SGLP
activates Rac1 and Stat3. Since Rac1 and Stat3 are involved in cell
migration and proliferation (14),
the possibility of whether SGLP may affect migration and/or
proliferation of HeLa cells was determined. In a Transwell assay,
wt SGLP was observed to increase the transwell migration of HeLa
cells relative to the control and SGLP (mutant) (Fig. 5A), which is consistent with the
levels of active Rac1 demonstrated in Fig. 3A. These observations suggest that
the effect of wt SGLP on HeLa cell migration is associated with the
wt SGLP-induced Rac1 activation. BrdU incorporation rates in HeLa
cells were then investigated by flow cytometry. Wt SGLP was
observed to promote BrdU incorporation (Fig. 5B), which suggests that SGLP may
exert a pro-proliferative effect in HeLa cells.
Discussion
In the current study, SGLP, an N terminal protein
fragment of Smc from Mycoplasma pulmonis, was identified as
a mycoplasmal virulence factor. Homologues of SGLP may be observed
in a minimum of 25 Mycoplasma species, which exhibit high levels of
homology around the sequences encompassing the WVLGE motif. This
conservation in evolution reflects that SGLP and its homologues may
be considered as a group of common virulence factors of Mycoplasma
species.
The study observed that SGLP-transduced HeLa cells
exhibited reduced stress fibers and increased filopodia and
lamellipodia. These phenotypic changes in actin filament
distribution may have resulted from alterations in Rho-family small
GTPase activity (26,27). The host Rho-family small GTPases
are hypothesized to be involved in SGLP-induced cellular
alterations. An interaction between SGLP and members of Rho-family
small GTPases, including Rac1 was hypothesized. In addition, Stat3
directly binds to Rac1 and modulates its function (8,13).
The possibility of SGLP interacting with Rac1 and/or Stat3 was
investigated. SGLP was observed to form a protein complex with Rac1
and Stat3, which suggests that SGLP may interact directly with Rac1
and Stat3. SGLP was also confirmed to colocalize with Rac1 and
Stat3 in the nucleus and cytoplasm, which reflects that SGLP may
interact with Rac1 and Stat3 at various locales within host
cells.
As SGLP is a binding partner of Rac1, the effect of
SGLP on Rac1 activity was determined. SGLP was observed to increase
the active Rac1 level. Since the WxxxE motif is crucial for a
number of bacterial proteins to alter host Rho-family small GTPase
activity (16–18), SGLP was hypothesized to cause a
similar effect on Rac1 depending on the WxxxE motif. The SGLP
mutant with a mutated AxxxA sequence did not induce Rac1
activation, which is consistent with the hypothesis. As active Rac1
promotes cell migration (28), it
was assumed that SGLP-induced Rac1 activation may also contribute
to tumor cell migration. SGLP was observed to increase cell
migration of tumor cells in vitro, while the SGLP mutant did
not exert this effect. These results confirmed the hypothesis that
the WxxxE motif is required for SGLP to increase Rac1 activity and
that SGLP-induced Rac1 activation may be responsible for the
observed increase in cell migration. However, this explanation does
not exclude other small GTPases that may also contribute to
SGLP-associated increase in tumor cell migration.
Stat3 activation is involved in a number of
signaling pathways and it is pivotal in receptor tyrosine
kinase-mediated pro-proliferative signal transduction (29). SGLP was observed to induce Stat3
activation. SGLP was also identified to exhibit a pro-proliferative
effect on tumor cells in terms of BrdU incorporation rates, which
is consistent with the observed SGLP-induced activation of Stat3.
In view of the dependency of SGLP-induced Stat3 activation on Rac1
activity, it was hypothesized that SGLP may induce Rac1 activation,
which, in turn, contributes to Stat3 activation. Although the role
of exact domains of SGLP in forming the protein complex with Rac1
and Stat3 is somewhat unclear, the objective of the current study
was to reveal and highlight a molecular mechanism by which
Mycoplasma may affect host cellular responses.
In conclusion, the current findings suggest that,
through interaction with Rac1 and Stat3, SGLP may potentiate tumor
cell migration and proliferation in vitro. However, its
involvement in promoting tumor development in vivo remains
uncertain. The identification of an individual mycoplasmal
virulence factor that interacts with and activates Rac1 and
subsequently triggers Stat3 activation is novel. Further studies of
the role of SGLP in Mycoplasma-associated pathogenesis are likely
to contribute to knowledge of Mycoplasma-induced malignant
transformation of mammalian cells.
References
|
1
|
Sinkovics JG: Molecular biology of
oncogenic inflammatory processes. I Non-oncogenic and oncogenic
pathogens, intrinsic inflammatory reactions without pathogens, and
microRNA/DNA interactions (Review). Int J Oncol. 40:305–349.
2012.
|
|
2
|
Zhang S, Tsai S and Lo SC: Alteration of
gene expression profiles during mycoplasma-induced malignant
cell transformation. BMC Cancer. 6:1162006. View Article : Google Scholar
|
|
3
|
Tsai S, Wear DJ, Shih JW and Lo SC:
Mycoplasmas and oncogenesis: persistent infection and multistage
malignant transformation. Proc Natl Acad Sci USA. 92:10197–10201.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rottem S: Interaction of mycoplasmas with
host cells. Physiol Rev. 83:417–432. 2003.PubMed/NCBI
|
|
5
|
Choi SY, Lim JW, Shimizu T, Kuwano K, Kim
JM and Kim H: Reactive oxygen species mediate Jak2/Stat3 activation
and IL-8 expression in pulmonary epithelial cells stimulated with
lipid-associated membrane proteins from Mycoplasma
pneumoniae. Inflamm Res. 61:493–501. 2012. View Article : Google Scholar
|
|
6
|
Rawadi G, Zugaza JL, Lemercier B, et al:
Involvement of small GTPases in Mycoplasma fermentans
membrane lipoproteins-mediated activation of macrophages. J Biol
Chem. 274:30794–30798. 1999.PubMed/NCBI
|
|
7
|
Takai Y, Sasaki T and Matozaki T: Small
GTP-binding proteins. Physiol Rev. 81:153–208. 2001.PubMed/NCBI
|
|
8
|
Simon AR, Vikis HG, Stewart S, Fanburg BL,
Cochran BH and Guan KL: Regulation of STAT3 by direct binding to
the Rac1 GTPase. Science. 290:144–147. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gupta SC, Hevia D, Patchva S, Park B, Koh
W and Aggarwal BB: Upsides and downsides of reactive oxygen species
for cancer: the roles of reactive oxygen species in tumorigenesis,
prevention, and therapy. Antioxid Redox Signal. 16:1295–1322. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Millonig G, Ganzleben I, Peccerella T, et
al: Sustained submicromolar H2O2 levels
induce hepcidin via signal transducer and activator of
transcription 3 (STAT3). J Biol Chem. 287:37472–37482. 2012.
|
|
11
|
Tonozuka Y, Minoshima Y, Bao YC, et al: A
GTPase-activating protein binds STAT3 and is required for
IL-6-induced STAT3 activation and for differentiation of a leukemic
cell line. Blood. 104:3550–3557. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kawashima T, Bao YC, Nomura Y, et al: Rac1
and a GTPase-activating protein, MgcRacGAP, are required for
nuclear translocation of STAT transcription factors. J Cell Biol.
175:937–946. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Teng TS, Lin B, Manser E, Ng DC and Cao X:
Stat3 promotes directional cell migration by regulating Rac1
activity via its activator betaPIX. J Cell Sci. 122:4150–4159.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Debidda M, Wang L, Zang H, Poli V and
Zheng Y: A role of STAT3 in Rho GTPase-regulated cell migration and
proliferation. J Biol Chem. 280:17275–17285. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Azare J, Leslie K, Al-Ahmadie H, et al:
Constitutively activated Stat3 induces tumorigenesis and enhances
cell motility of prostate epithelial cells through integrin beta 6.
Mol Cell Biol. 27:4444–4453. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Alto NM, Shao F, Lazar CS, et al:
Identification of a bacterial type III effector family with G
protein mimicry functions. Cell. 124:133–145. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bulgin R, Raymond B, Garnett JA, et al:
Bacterial guanine nucleotide exchange factors SopE-like and WxxxE
effectors. Infect Immun. 78:1417–1425. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Jackson LK, Nawabi P, Hentea C, Roark EA
and Haldar K: The Salmonella virulence protein SifA is a G
protein antagonist. Proc Natl Acad Sci USA. 105:14141–14146.
2008.
|
|
19
|
Ham JH, Majerczak DR, Nomura K, et al:
Multiple activities of the plant pathogen type III effector
proteins WtsE and AvrE require WxxxE motifs. Mol Plant Microbe
Interact. 22:703–712. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hagemann UB, Mason JM, Müller KM and Arndt
KM: Selectional and mutational scope of peptides sequestering the
Jun-Fos coiled-coil domain. J Mol Biol. 381:73–88. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Luo X, Li Z, Li X, et al: hSav1 interacts
with HAX1 and attenuates its anti-apoptotic effects in MCF-7 breast
cancer cells. Int J Mol Med. 28:349–355. 2011.PubMed/NCBI
|
|
22
|
Fay FS, Taneja KL, Shenoy S, Lifshitz L
and Singer RH: Quantitative digital analysis of diffuse and
concentrated nuclear distributions of nascent transcripts, SC35 and
poly(A). Exp Cell Res. 231:27–37. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hachet-Haas M, Converset N, Marchal O, et
al: FRET and colocalization analyzer - a method to validate
measurements of sensitized emission FRET acquired by confocal
microscopy and available as an ImageJ Plug-in. Microsc Res Tech.
69:941–956. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Nobes CD and Hall A: Rho, rac, and cdc42
GTPases regulate the assembly of multimolecular focal complexes
associated with actin stress fibers, lamellipodia, and filopodia.
Cell. 81:53–62. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Faruqi TR, Gomez D, Bustelo XR, Bar-Sagi D
and Reich NC: Rac1 mediates STAT3 activation by autocrine IL-6.
Proc Natl Acad Sci USA. 98:9014–9019. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Albertinazzi C, Cattelino A and de Curtis
I: Rac GTPases localize at sites of actin reorganization during
dynamic remodeling of the cytoskeleton of normal embryonic
fibroblasts. J Cell Sci. 112:3821–3831. 1999.PubMed/NCBI
|
|
27
|
Hoang MV, Nagy JA and Senger DR: Active
Rac1 improves pathologic VEGF neovessel architecture and reduces
vascular leak: mechanistic similarities with angiopoietin-1. Blood.
117:1751–1760. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Migeotte I, Omelchenko T, Hall A and
Anderson KV: Rac1-dependent collective cell migration is required
for specification of the anterior-posterior body axis of the mouse.
PLoS Biol. 8:e10004422010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sehgal PB: Paradigm shifts in the cell
biology of STAT signaling. Semin Cell Dev Biol. 19:329–340. 2008.
View Article : Google Scholar : PubMed/NCBI
|