Introduction
Irritable bowel syndrome (IBS) is a common chronic
gastrointestinal disorder (1–9). It
occurs primarily at a young age, with a female predominance and
causes lifelong illness (2,10–21).
IBS considerably reduces the quality of life of the patients due to
the uncomfortable symptoms associated with the syndrome, which are
often found embarrassing and humiliating and thus interfere with
education, working ability and social interactions. Furthermore,
IBS represents an economic burden to society caused by
overconsumption of healthcare resources and lower work productivity
of IBS patients (9,13,20,22–24).
The diagnosis of IBS is based primarily on symptom
assessment, including the Rome III criteria (25,26).
Patients with IBS suffer from abdominal discomfort or pain, altered
bowel habits and bloating/abdominal distension (1,2).
Based on the stool pattern, patients with IBS are subdivided,
according to Rome III criteria, into four subtypes, namely
diarrhoea-predominant (IBS-D), constipation-predominant (IBS-C),
mixed diarrhoea and constipation (M-IBS) and unclassified IBS
(U-IBS) (25,26).
Serotonin producing cells occur in large numbers in
the mucosa of the majority of segments of the gastrointestinal
tract and represent the main source of serotonin in the human
(27–29). Serotonin acts upon sensory and
motor neurons in the submucosal and myenteric ganglia as well as in
the spinal cord (27). This, in
turn, stimulates motor, secretory and vasodilatory reflexes as well
as afferent signals to the central nervous system (27). Serotonin, following acting on its
receptors, is transported by serotonin-selective reuptake
transporter (SERT) into gut epithelial cells, where it is degraded
(27,30). The gut mucosa has a high capacity
for the production of SERT as all the epithelial cells lining the
luminal surface of the gut express SERT (29,31).
Serotonin cell densities in the ileum and colon were reduced in IBS
patients compared with those in the duodenum and rectum which were
unchanged (32–36). However, SERT immunoreactivity
intensity has been observed to be high in the rectum (36,37).
In the present study the immunoreactivity intensity
of serotonin and SERT was investigated in the ileum of the same
cohort of IBS patients where low density of ileal serotonin cells
were found (32).
Materials and methods
Patients and controls
Ninety-eight patients (77 females and 21 males; mean
age, 35 years; range, 18–66 years), which fulfilled Rome III
Criteria for IBS were included in the study (25,38).
The IBS subtypes were distributed as follows: 35 patients with
IBS-D, 31 patients with IBS-M and 32 patients with IBS-C. Symptoms
had been present in all patients for a number of years and the
onset of IBS symptoms were not associated with any events, in
particular gastrointestinal infections. All patients underwent a
complete physical examination and were investigated using the
following blood tests: Full blood count, electrolytes, inflammatory
markers, liver tests and thyroid function tests. Patients also
underwent further gastroscopy with duodenal biopsies, which were
used to exclude celiac disease.
A total of 27 subjects who underwent colonoscopy (16
females and 11 males; mean age, 52 years; range, 20–69 years) were
used as controls. These subjects underwent a colonoscopy for the
following reasons: Gastrointestinal bleeding, where the source of
bleeding was identified as haemorrhoids (n=18) or angiodysplasia
(n=2) and health worries resulting from a relative being diagnosed
with colon carcinoma (n=7).
The study was performed in accordance with the
Declaration of Helsinki and was approved by the Regional Committee
for Medical and Health Research Ethics, Health West, University of
Bergen (Bergen, Norway). All subjects provided oral and written
consent.
Colonoscopy, histopathology and
immunohistochemistry
Colonoscopy was performed on the patients and
controls, segmental biopsy specimens were taken from the colon and
rectum and four biopsy samples were extracted from the ileum of
each subject. These biopsy samples were fixed overnight in 4%
buffered paraformaldehyde, embedded in paraffin and sliced into
5-μm sections. The sections were stained with hematoxylin and eosin
(H&E) and immunostained by the avidin-biotin complex (ABC)
method using the Vectastain ABC kit (Vector Laboratories,
Burlingame, CA, USA). The sections were hydrated and immersed in
phosphate-buffered saline buffer (PBS; pH 7.4) and were incubated
with the primary antibodies at room temperature for 2 h. The
sections were washed in PBS buffer and incubated with biotinylated
swine anti-mouse IgG (Vector Laboratories) diluted 1:100 for 30 min
at room temperature. Following washing the slides in PBS buffer,
the sections were incubated for 30 min with
avidin-biotin-peroxidase complex diluted 1:100 and immersed in
3,3′-diaminobenzidine (DAB) peroxidase substrate (Vector
Laboratories), followed by counterstaining in hematoxylin. The
primary antibodies used were monoclonal mouse anti-serotonin (code
no. 5HT-209; Dako, Carpinteria, CA, USA) and mouse anti-human
serotonin transporter (code no. GR120164-1; Abcam, Cambridge, MA,
USA). The antibodies were used at dilutions of 1:1,500 and 1:500,
respectively.
Computerised image analysis
The immunoreactivity intensities of serotonin and
SERT were measured using Olympus cellSens imaging software (version
1.7). A ×20 magnification was used, for which each frame (field)
displayed on the monitor represented an area of 0.14 mm2
of the tissue. In each individual, serotonin and SERT were measured
in five randomly selected fields. Immunostained sections from IBS
patients and controls were coded and mixed and all measurements
were assessed by the same person without knowledge of the identity
of the sections.
Statistical analysis
Differences between controls, all IBS patients
(IBS-total) and IBS-D, IBS-M and IBS-C patients were analysed by
the Kruskal-Wallis nonparametric test with Dunn’s test as a
post-test. The data are presented as the mean ± standard error of
the mean values and P<0.05 was considered to indicate a
statistically significant difference.
Results
Endoscopy and histopathology
The ileum, colon and rectum of the patients and the
control subjects were macroscopically normal. The results of
histopathological examinations of the ileum, colon and rectum were
normal in the patients and controls.
Immunohistochemistry and computerised
image analysis
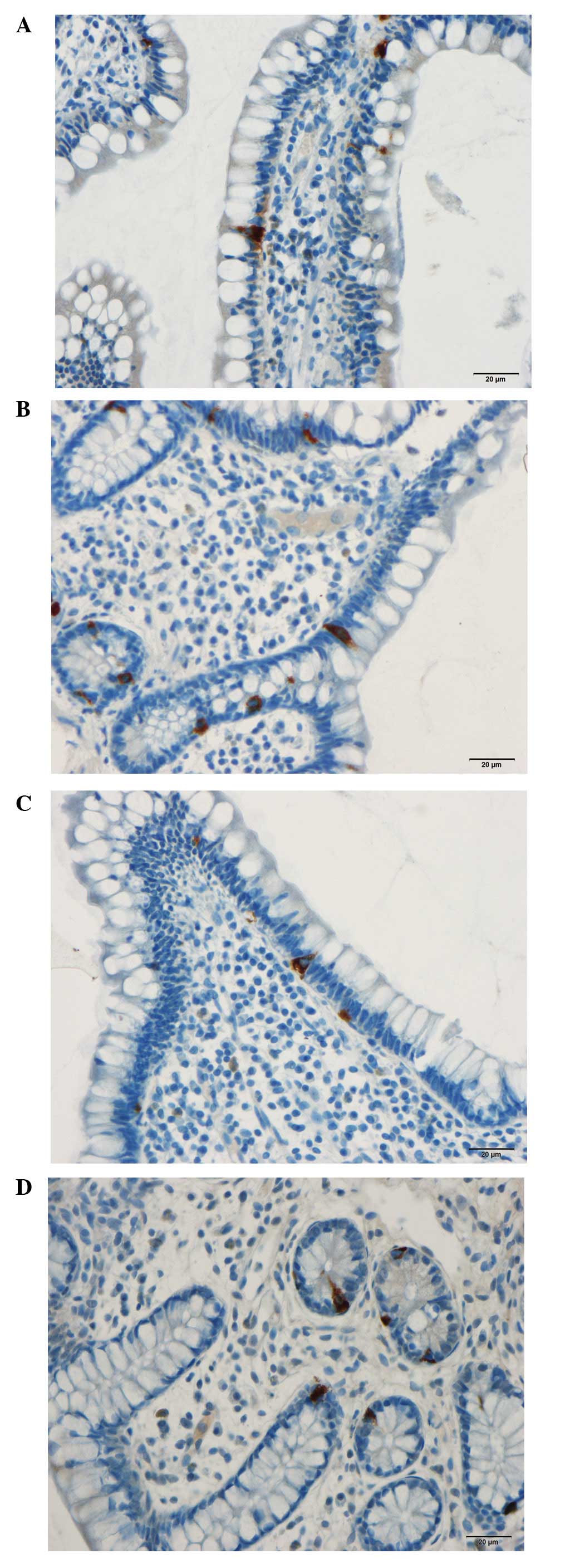
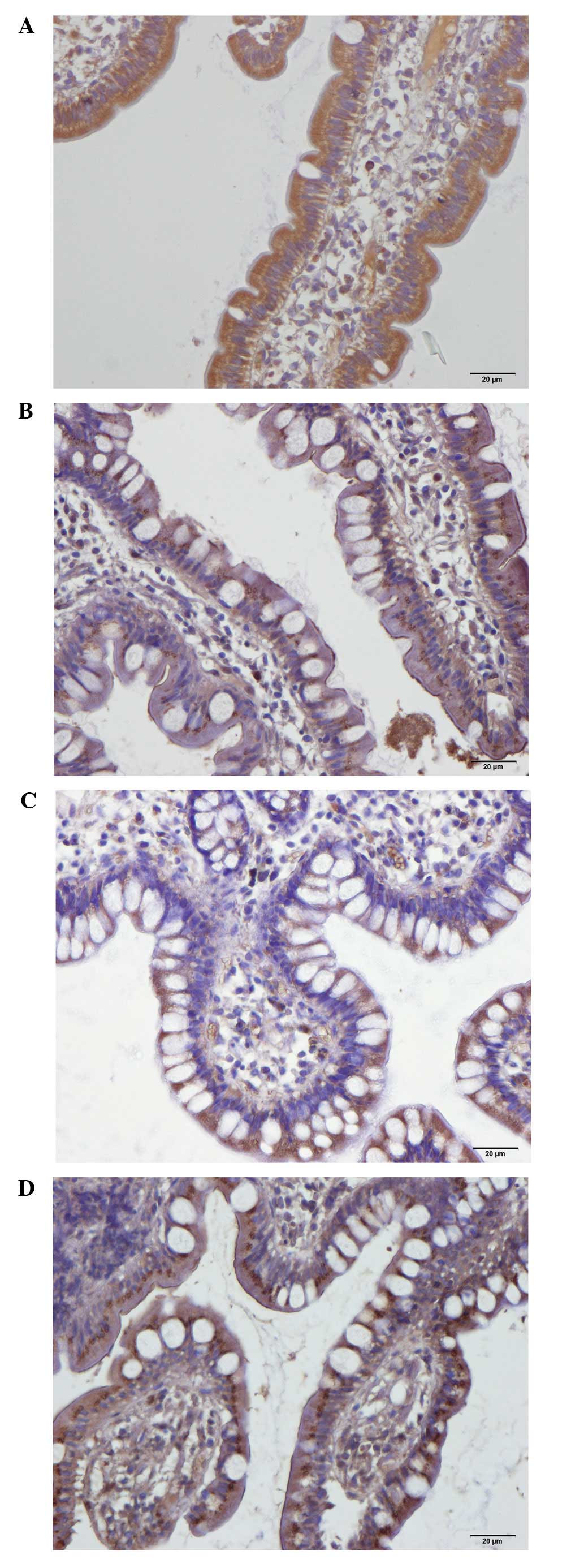
In the patients and control subjects,
serotonin-immunoreactive cells were found primarily in the
intestinal crypts. These cells were basket- or flask-shaped. SERT
immunoreactivity was observed in the lining epithelium of the ileum
in the patients and controls.
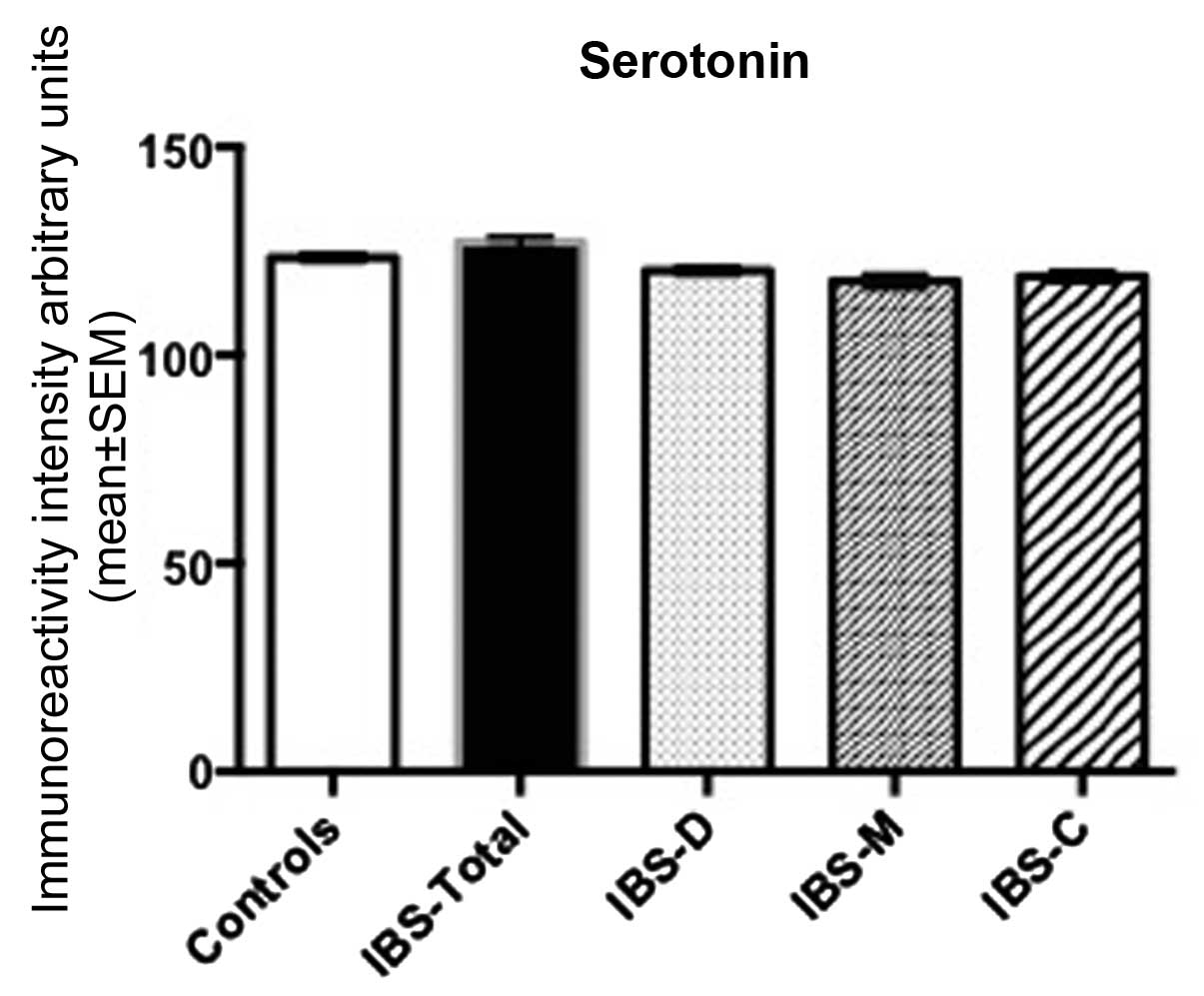
Serotonin immunoreactivity intensity
The immunoreactivity intensity of serotonin in the
controls was 121.7±0.8. The corresponding figures for IBS-total,
IBS-D, IBS-M and IBS-C were 122.9±2.0, 120.3±1.1, 117.8±1.7 and
118.9±1.3, respectively. There was no statistical difference in
multiple comparisons between controls, IBS-total, IBS-D and IBS-C
(P=0.3). The post-test did not reveal any statistical differences
between the four groups (P<0.05 in all; Figs. 1 and 2).
SERT immunoreactivity intensity
The immunoreactivity intensity of SERT in controls,
IBS-total, IBS-D, IBS-M and IBS-C were 115.4±3.3, 126.1±1.9,
127.3±1.6, 125.1±3.2 and 129.1±2.1, respectively (Figs. 3 and 4). There was a significant statistical
difference in multiple comparisons between controls, IBS-total,
IBS-D, IBS-M and IBS-C (P=0.0005). SERT immunoreactivity intensity
of IBS-total, IBS-D, IBS-M and IBS-C differed significantly from
that of controls (P<0.01, P<0.05, P <0.05 and P <0.001,
respectively).
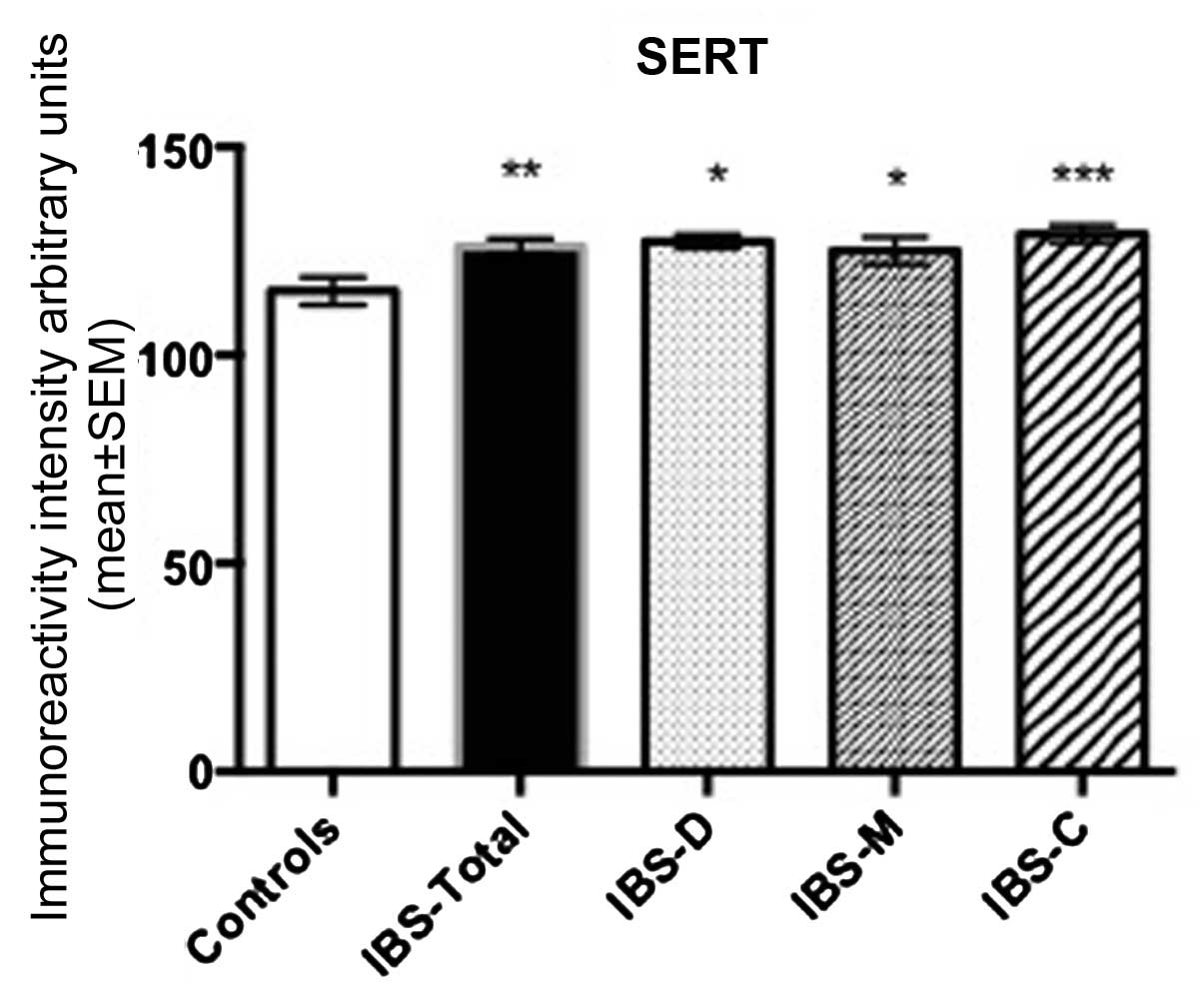 | Figure 4SERT immunoreactivity intensity in
ileum epithelial cells of control subjects, IBS-total, IBS-D, IBS-M
and IBS-C. *P<0,05, **P<0.01 and
***P<0.0001. IBS, irritable bowel syndrome. SERT,
Serotonin-selective reuptake transporter; IBS-D,
diarrhoea-predominant; IBS-C, constipation-predominant; M-IBS,
mixed diarrhoea and constipation. |
Discussion
The rapid advances in the software for computer
image analysis has rendered it possible to measure the intensity of
the colour product of immunohistochemical staining for a specific
intracellular substance. This intensity reflects the cellular
contents of this particular substance. These measurements are
useful in comparing similar tissue from different groups treated in
the same conditions during immunostaining.
The present study showed that whereas the
immunoreactivity intensity of serotonin in the ileum of IBS
patients did not differ from that in control subjects, the
immunoreactivity intensity of SERT increased regardless of the IBS
subtypes. These observations were noted in the same cohort of IBS
patients, where the ileal serotonin cells were lower compared with
the control subjects, regardless of the IBS subtype (32). The normal cellular content of
serotonin, as reflected by the immunoreactivity intensity and the
reduction in the number of cells in the ileum of IBS patients
implicates a decrease in the mucosal serotonin. The increase in
ileal epithelial content of SERT, as indicated by increased
immunoreactivity intensity, may increase the intracellular uptake
of serotonin and its degradation in the gut epithelial cells and
consequently decrease the availability of serotonin within the gut
mucosa (39,40). The present findings and the earlier
observations (32) indicate that
there is a decrease in the production of serotonin and a rapid
degradation resulting in the low availability of serotonin at its
receptors in the ileum of IBS patients.
The low availability of serotonin at it receptors in
the ileum of IBS patients appear to occur in all IBS subtypes. This
may indicate that this abnormality is associated with a common
symptom in all IBS subtypes, which is abdominal pain/discomfort. It
is possible that the abnormality in the ileum serotonin signalling
system observed in the present study is responsible for the
development of this common symptom in IBS patients. In agreement
with this hypothesis, serotonin acts upon sensory neurons in the
submucosal and myenteric ganglia as well as in spinal cord
(27). The abnormality observed in
this study in SERT may be genetic, as genetic abnormalities
concerning SERT have been observed in IBS patients (41–50).
The present observations may have an impact in the use of serotonin
agonists and antagonists in the treatment of IBS patients.
Acknowledgements
The authors would like to thank Hans-Olav Fadnes for
his support during the course of the study and for reading and
commenting on the manuscript. This study was supported by a grant
from Helse-Fonna.
References
|
1
|
Thompson WG: A world view of IBS.
Irritable Bowel Syndrome. Camilleri M and Spiller RC: Saunders;
Philadelphia and London: pp. 17–26. 2002
|
|
2
|
Drossman DA, Li Z, Andruzzi E, et al: U.S.
householder survey of functional gastrointestinal disorders.
Prevalence, sociodemography, and health impact. Dig Dis Sci.
38:1569–1580. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
El-Salhy M, Gundersen D, Hatlebakk JG and
Hausken T: Irritable Bowel Syndrome: Diagnosis, Pathogenesis and
Treatment Options. Nova Science Publishers; New York: 2012
|
|
4
|
Ford AC and Vandvik PO: Irritable bowel
syndrome. Clin Evid (Online). 2012:04102012.
|
|
5
|
Lovell RM and Ford AC: Global prevalence
of and risk factors for irritable bowel syndrome: a meta-analysis.
Clin Gastroenterol Hepatol. 10:712–721. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Quigley EM, Locke GR, Mueller-Lissner S,
et al: Prevalence and management of abdominal cramping and pain: a
multinational survey. Aliment Pharmacol Ther. 24:411–419. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Vandvik PO, Lydersen S and Farup PG:
Prevalence, comorbidity and impact of irritable bowel syndrome in
Norway. Scand J Gastroenterol. 41:650–656. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Saito YA, Schoenfeld P and Locke GR III:
The epidemiology of irritable bowel syndrome in North America: a
systematic review. Am J Gastroenterol. 97:1910–1915.
2002.PubMed/NCBI
|
|
9
|
Whitehead WE, Burnett CK, Cook EW III and
Taub E: Impact of irritable bowel syndrome on quality of life. Dig
Dis Sci. 41:2248–2253. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Agréus L, Svärdsudd K, Nyrén O and Tibblin
G: Irritable bowel syndrome and dyspepsia in the general
population: overlap and lack of stability over time.
Gastroenterology. 109:671–680. 1995.PubMed/NCBI
|
|
11
|
Thompson WG and Heaton KW: Functional
bowel disorders in apparently healthy people. Gastroenterology.
79:283–288. 1980.PubMed/NCBI
|
|
12
|
Kennedy TM, Jones RH, Hungin AP,
O’flanagan H and Kelly P: Irritable bowel syndrome,
gastro-oesophageal reflux, and bronchial hyper-responsiveness in
the general population. Gut. 43:770–774. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Talley NJ, Gabriel SE, Harmsen WS,
Zinsmeister AR and Evans RW: Medical costs in community subjects
with irritable bowel syndrome. Gastroenterology. 109:1736–1741.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hungin AP, Whorwell PJ, Tack J and Mearin
F: The prevalence, patterns and impact of irritable bowel syndrome:
an international survey of 40,000 subjects. Aliment Pharmacol Ther.
17:643–650. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jones R and Lydeard S: Irritable bowel
syndrome in the general population. BMJ. 304:87–90. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bordie AK: Functional disorders of the
colon. J Indian Med Assoc. 58:451–456. 1972.PubMed/NCBI
|
|
17
|
O’Keefe EA, Talley NJ, Zinsmeister AR and
Jacobsen SJ: Bowel disorders impair functional status and quality
of life in the elderly: a population-based study. J Gerontol A Biol
Sci Med Sci. 50:M184–M189. 1995.PubMed/NCBI
|
|
18
|
Everhart JE and Renault PF: Irritable
bowel syndrome in office-based practice in the United States.
Gastroenterology. 100:998–1005. 1991.PubMed/NCBI
|
|
19
|
Wilson S, Roberts L, Roalfe A, Bridge P
and Singh S: Prevalence of irritable bowel syndrome: a community
survey. Br J Gen Pract. 54:495–502. 2004.PubMed/NCBI
|
|
20
|
Harvey RF, Salih SY and Read AE: Organic
and functional disorders in 2000 gastroenterology outpatients.
Lancet. 1:632–634. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Spiegel BM: The burden of IBS: looking at
metrics. Curr Gastroenterol Rep. 11:265–269. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gralnek IM, Hays RD, Kilbourne A, Naliboff
B and Mayer EA: The impact of irritable bowel syndrome on
health-related quality of life. Gastroenterology. 119:654–660.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Drossman DA, Morris CB, Schneck S, et al:
International survey of patients with IBS: symptom features and
their severity, health status, treatments, and risk taking to
achieve clinical benefit. J Clin Gastroenterol. 43:541–550. 2009.
View Article : Google Scholar
|
|
24
|
Sandler RS, Everhart JE, Donowitz M, et
al: The burden of selected digestive diseases in the United States.
Gastroenterology. 122:1500–1511. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Longstreth GF, Thompson WG, Chey WD,
Houghton LA, Mearin F and Spiller RC: Functional bowel disorders.
Gastroenterology. 130:1480–1491. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Drossman DA: Rome III: the new criteria.
Chin J Dig Dis. 7:181–185. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mawe GM, Coates MD and Moses PL: Review
article: intestinal serotonin signalling in irritable bowel
syndrome. Aliment Pharmacol Ther. 23:1067–1076. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Erspamer V: Occurrence and distribution of
5-hydroxytryptamine (enteramine) in the living organism. Z Vitam
Horm Fermentforsch. 9:74–96. 1957.PubMed/NCBI
|
|
29
|
Chen JX, Pan H, Rothman TP, Wade PR and
Gershon MD: Guinea pig 5-HT transporter: cloning, expression,
distribution, and function in intestinal sensory reception. Am J
Physiol. 275:G433–G448. 1998.PubMed/NCBI
|
|
30
|
Spiller R and Lam C: An update on
post-infectious irritable bowel syndrome: role of genetics, immune
activation, serotonin and altered microbiome. J Neurogastroenterol
Motil. 18:258–268. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Coates MD, Mahoney CR, Linden DR, et al:
Molecular defects in mucosal serotonin content and decreased
serotonin reuptake transporter in ulcerative colitis and irritable
bowel syndrome. Gastroenterology. 126:1657–1664. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
El-Salhy M, Gundersen D, Hatlebakk JG and
Hausken T: Abnormal endocrine cells in the ileum of patients with
irritable bowel syndrome. World J Gastroenterol. 2013.(In
press).
|
|
33
|
El-Salhy M, Gundersen D, Ostgaard H,
Lomholt-Beck B, Hatlebakk JG and Hausken T: Low densities of
serotonin and peptide YY cells in the colon of patients with
irritable bowel syndrome. Dig Dis Sci. 57:873–878. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
El-Salhy M, Vaali K, Dizdar V and Hausken
T: Abnormal small-intestinal endocrine cells in patients with
irritable bowel syndrome. Dig Dis Sci. 55:3508–3513. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
El-Salhy M, Gundersen D, Hatlebakk JG and
Hausken T: Abnormal rectal endocrine cells in patients with
irritable bowel syndrome. Regul Pept. 2013.(In press).
|
|
36
|
Coates MD, Johnson AC, Greenwood-Van
Meerveld B and Mawe GM: Effects of serotonin transporter inhibition
on gastrointestinal motility and colonic sensitivity in the mouse.
Neurogastroenterol Motil. 18:464–471. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
El-Salhy M, Wendelbo I and Gundersen D:
Serotonin and serotonin transporter in the rectum of patients with
irritable bowel disease. Mol Med Rep. 8:451–455. 2013.PubMed/NCBI
|
|
38
|
Spiller R, Aziz Q, Creed F, et al:
Guidelines on the irritable bowel syndrome: mechanisms and
practical management. Gut. 56:1770–1798. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Keating C, Beyak M, Foley S, et al:
Afferent hypersensitivity in a mouse model of post-inflammatory gut
dysfunction: role of altered serotonin metabolism. J Physiol.
586:4517–4530. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Coleman NS, Foley S, Dunlop SP, et al:
Abnormalities of serotonin metabolism and their relation to
symptoms in untreated celiac disease. Clin Gastroenterol Hepatol.
4:874–881. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Camilleri M, Andrews CN, Bharucha AE, et
al: Alterations in expression of p11 and SERT in mucosal biopsy
specimens of patients with irritable bowel syndrome.
Gastroenterology. 132:17–25. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Camilleri M, Busciglio I, Carlson P, et
al: Candidate genes and sensory functions in health and irritable
bowel syndrome. Am J Physiol Gastrointest Liver Physiol.
295:G219–G225. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Kumar S, Ranjan P, Mittal B and Ghoshal
UC: Serotonin transporter gene (SLC6A4) polymorphism in patients
with irritable bowel syndrome and healthy controls. J
Gastrointestin Liver Dis. 21:31–38. 2012.PubMed/NCBI
|
|
44
|
Park JM, Choi MG, Park JA, et al:
Serotonin transporter gene polymorphism and irritable bowel
syndrome. Neurogastroenterol Motil. 18:995–1000. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Saito YA, Larson JJ, Atkinson EJ, et al:
The role of 5-HTT LPR and GNβ3 825C>T polymorphisms and
gene-environment interactions in irritable bowel syndrome (IBS).
Dig Dis Sci. 57:2650–2657. 2012.
|
|
46
|
Colucci R, Blandizzi C, Bellini M, Ghisu
N, Tonini M and Del Tacca M: The genetics of the serotonin
transporter and irritable bowel syndrome. Trends Mol Med.
14:295–304. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Colucci R, Gambaccini D, Ghisu N, et al:
Influence of the serotonin transporter 5HTTLPR polymorphism on
symptom severity in irritable bowel syndrome. PloS One.
8:e548312013. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Faure C, Patey N, Gauthier C, Brooks EM
and Mawe GM: Serotonin signaling is altered in irritable bowel
syndrome with diarrhea but not in functional dyspepsia in pediatric
age patients. Gastroenterology. 139:249–258. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Kim HJ, Camilleri M, Carlson PJ, et al:
Association of distinct alpha(2) adrenoceptor and serotonin
transporter polymorphisms with constipation and somatic symptoms in
functional gastrointestinal disorders. Gut. 53:829–837. 2004.
View Article : Google Scholar
|
|
50
|
Park CS and Uhm JH: Polymorphisms of the
serotonin transporter gene and G-protein β3 subunit gene in Korean
children with irritable bowel syndrome and functional dyspepsia.
Gut Liver. 6:223–228. 2012.
|