Introduction
Liver fibrogenesis is often present in various types
of chronic liver disease. Hepatic fibrosis, characterized by the
excessive generation of extracellular matrix constituents
(particularly collagens), is a precursor of cirrhosis (1). The mortality of patients with liver
fibrosis is gradually increasing (2) and long-term liver fibrosis
contributes to the development of hepatocellular carcinoma, which
is a malignancy of global importance that is difficult to treat
(3). Although numerous agents have
been used in the treatment of hepatic fibrosis, these are rarely
effective (4). Therefore, an
effective therapy is urgently required.
P2X7 is a member of the ionotropic purinergic
receptor family that is activated by ATP (5). P2X7 activity is demonstrated to be
readily detectable in cells of hemopoietic lineage, including
monocytes, macrophages, dendritic cells and lymphocytes (6,7). The
key involvement of P2X7 activation in inflammation and
interleukin-1β (IL-1β) maturation has been previously confirmed
(8). In addition, during chronic
inflammation, P2X7 is significantly upregulated in macrophages
(9,10). Studies have demonstrated that the
absence or inhibition of P2X7 is associated with less severe
chronic inflammation, indicating that P2X7 functions as an integral
component of an in vivo pro-inflammatory mechanism (11,12).
Notably, it has recently been shown that P2X7 is involved in lung
fibrosis by promoting IL-1β maturation (13). However, it remains to be determined
whether P2X7 contributes to hepatic fibrosis. The aim of the
present study was to investigate whether specific inhibition of
P2X7 attenuated the progression of chronic liver injury and
fibrosis in a mouse liver fibrotic model induced by carbon
tetrachloride (CCl4).
Materials and methods
Liver fibrosis induction and
treatment
Procedures were reviewed and approved by the Ethics
Committee of the Henan Provincial Cancer Hospital. Male C57BL/6
mice (age, 6 weeks) from Henan Provincial Animal Center were used
in this study. Mice were maintained under specific pathogen-free
conditions. Hepatic fibrosis was induced by subcutaneously
injecting a 1:1 solution of CCl4 in olive oil (Chemical
Agent Company of Shanghai, Shanghai, China). The solution was
administered three times a week for four weeks. At the end of the
first week, CCl4-injected mice were divided into two
groups: CCl4 alone and CCl4 plus A438079
(subcutaneous infusion) once daily. The usage of A438079 was based
on a previous study (14). In
brief, A-438079 was diluted at 3 mg/ml in vehicle solution
(saline). A-438079 (34.2 mg/kg) was then administered
subcutaneously to mice every 24 h.
The animals were divided into three groups (n=12 per
group): i) normal control with vehicle (saline/olive oil) alone
administered by subcutaneous injection; ii) CCl4
(dissolved in olive oil) alone, administered by subcutaneous
injection; iii) CCl4 + A438079, administered by
subcutaneous injection.
Histopathological analysis
Mice were sacrificed by carbon dioxide asphyxiation
at the end of the fourth week subsequent to the initial injection
(24 h following final injection). Blood was collected by cardiac
puncture, and serum was obtained by centrifugation of blood at 600
× g for 10 min, and stored at −20°C. Liver tissues were fixed in
10% phosphate-buffered formalin and embedded in paraffin. The
paraffin blocks were cut into 4-μm tissue sections. The sections
were stained with hematoxylin and eosin. The collagen that had
accumulated in the liver sections was stained with 0.1%
Picro-Sirius Red (Polysciences Inc., Warrington, PA, USA) and
quantified by an image analyzer (Leica Microsystems Ltd., Buffalo
Grove, IL, USA). The percentage area of the total quantity of
collagen was determined by the total of the areas of Sirius Red
positive stain divided by the reference field multiplied by 100.
The percentage positive area of the central veins was determined by
the total of the areas of Sirius Red positive stain in the central
veins divided by the reference field multiplied by 100. The
percentage positive of the perihepatic region was calculated by
subtracting the percentage positive of total area and the
percentage of the central vein.
Determination of serum alanine
aminotransferase (ALT) activity
Increased serum ALT level is a common indicator of
hepatic injury. The ALT assay was conducted using a commercially
available kit according to the manufacturer’s instructions (Wako
Pure Chemical, Tokyo, Japan).
RNA extraction and reverse
transcriptase-polymerase chain reaction (RT-PCR)
Total RNA was extracted from liver tissue using
TRIzol (Life Technologies, Carlsbad, CA, USA). The preparation of
the first-strand cDNA was performed by using the SuperScript™
First-Strand Synthesis System (Invitrogen Life Technologies,
Carlsbad, CA, USA), according to the manufacturer’s instructions.
The mRNA expression levels of P2X7 were measured by RT-PCR. The
primers forward: 5′-GTGCCATTCTGACCAGGGTTGTATAAA-3′ and reverse:
5′-GCCACCTCTGTAAAGTTCTCTCCGAT-3′ were used for P2X7; and
glyceraldehyde 3-phosphate dehydrogenase was amplified as an
internal control.
Measurement of tumor necrosis factor-α
(TNF-α), IL-1β and CCL2 in the sera
The sera were collected for the measurement of
TNF-α, IL-1β and CCL2 by enzyme-linked immunosorbent assay (ELISA;
R&D Systems, Minneapolis, MN, USA), according to the
manufacturer’s instructions.
Western blot analysis
The liver tissues were lysed at 4°C in RIPA buffer
(Qiagen, Valencia, CA, USA) and extracts were clarified at 12,000 ×
g for 25 min. Following separation by sodium dodecyl
sulfate-polyacrylamide gel electrophoresis, proteins were assessed
using the bicinchoninic acid assay protein concentration assay kit
(Pierce Biochemicals, Rockford, IL, USA). Aliquots (60–90 μg
protein) were electrophoretically separated, transferred to
nitrocellulose membranes and incubated overnight with the following
specific primary antibodies: anti-TGF-β1, 1:2,000, anti-α-SMA
antibody, 1:1,000; anti-P2X7 antibody, 1:500; anti-β-tubulin
antibody, 1:5,000 (Abcam, Cambridge, MA, USA). Following three
washes, primary antibodies were detected with horseradish
peroxidase-labeled secondary antibody (1:5,000; Abcam). The
activity of nuclear factor-κB (NF-κB) was determined by the active
NF-κBp65 (1:2,000; Abcam) using western blot analysis. Histone H3
(1:3,000; Abcam) was used as nuclear internal control.
Immunoreactive proteins were visualized with an enhanced
chemiluminescence reagent (Amersham Biosciences, Piscataway, NJ,
USA) and quantitated by densitometry (GE Healthcare, Little
Chalfont, UK).
Statistical analysis
Data from each group are expressed as the mean ± SD.
Statistical comparisons between the groups were conducted using the
Kruskal-Wallis test followed by Dunn’s post hoc test to compare all
the groups. P<0.05 was considered to indicate a statistically
significant difference.
Results
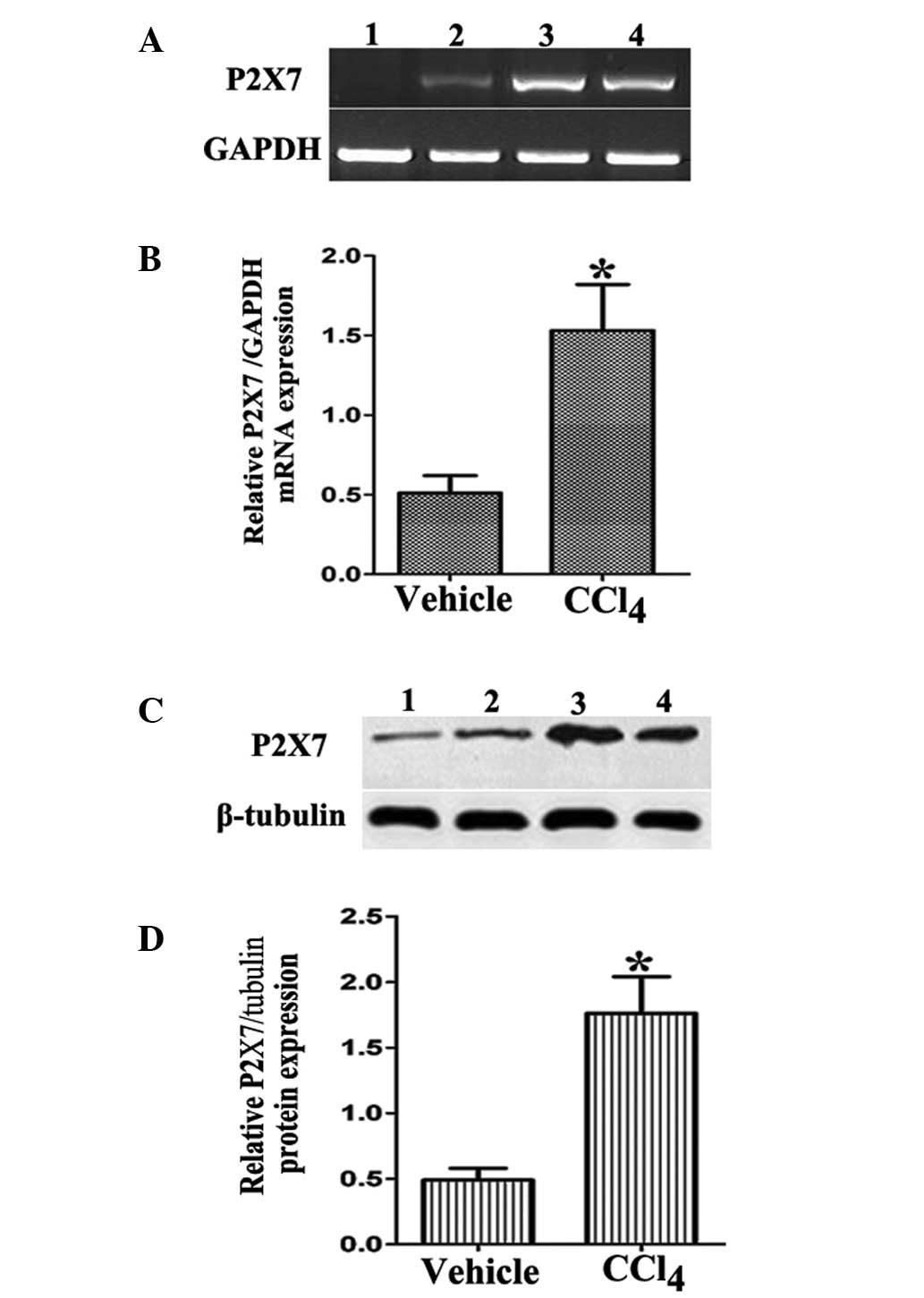
Enhanced P2X7 expression in
CCl4-induced liver fibrosis
The P2X7 expression in liver tissues from the
CCl4-induced liver fibrosis model and from
vehicle-treated normal controls was analyzed. As expected, livers
from CCl4-treated mice exhibited increased mRNA and
protein levels of P2X7 compared with vehicle-treated normal mice
(Fig. 1).
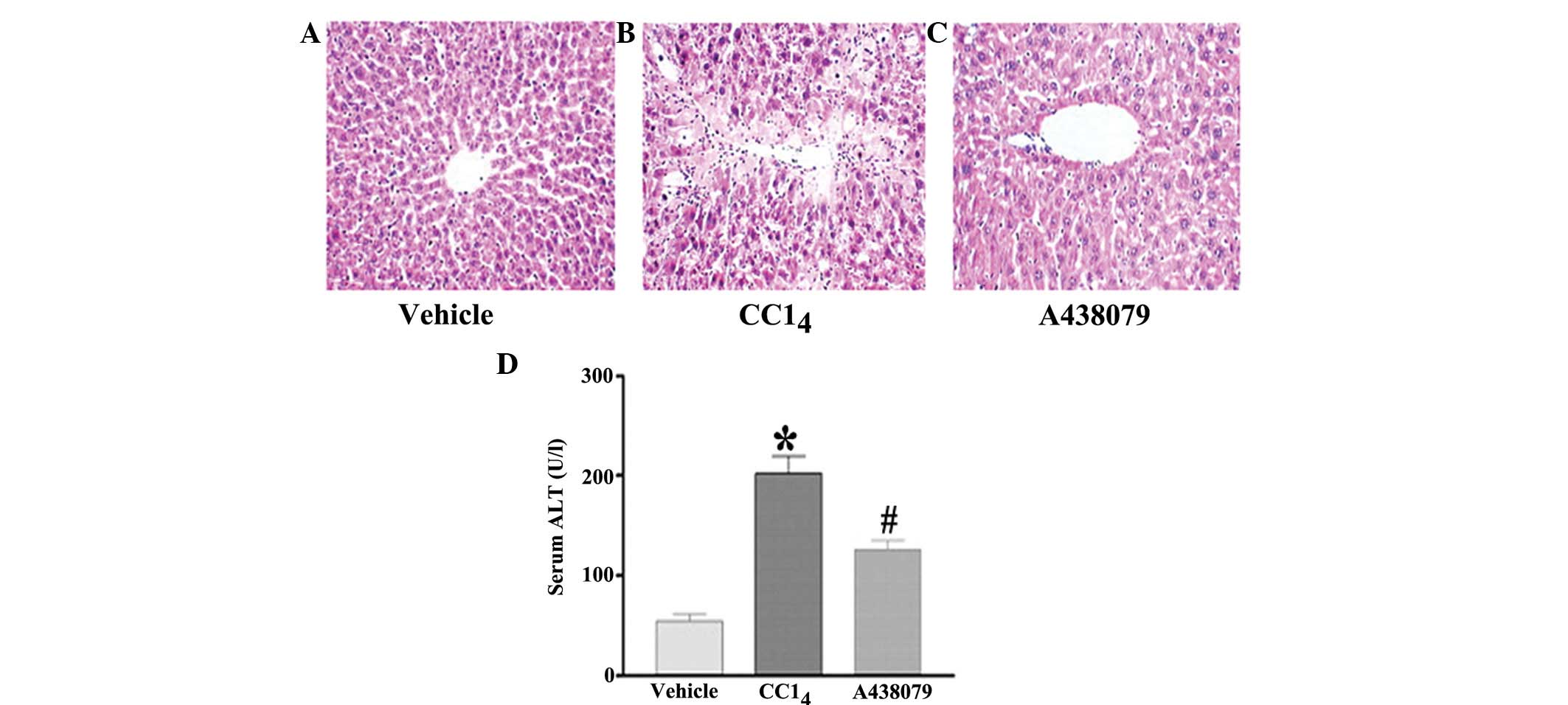
A438079 reduces CCl4-induced
necrosis, inflammatory infiltration and cell injury
It was identified that CCl4 treatment was
found to induce the formation of necrosis in the liver with
inflammatory infiltration surrounding the centrilobular veins
(Fig. 2B). The A438079 treatment
significantly attenuated the severity of necrosis (Fig. 2C). CCl4 also increased
the level of serum alanine aminotransferase (ALT), indicative of
cellular injury compared with the normal control group (P<0.01)
(Fig. 2D). Mice treated with
A438079 and CCl4 showed a significant reduction in serum
ALT and histopathological damage compared with the
CCl4-treated group (P<0.01).
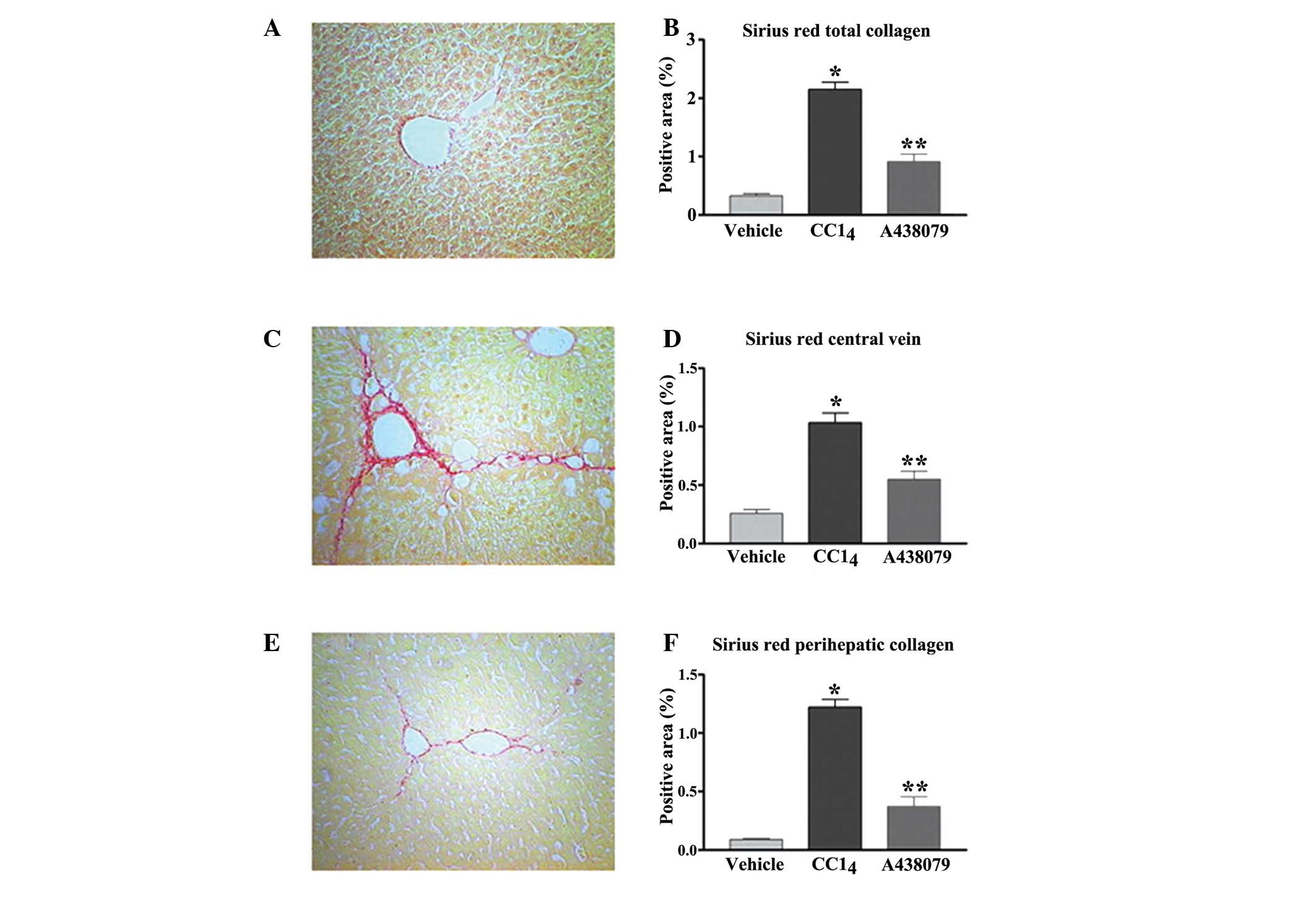
A438079 reduces CCl4-induced
collagen accumulation in the liver
Chronic CCl4 treatment showed a
significant increase in collagen accumulation in the liver
(Fig. 3B). The deposition of
collagen in the pericellular area and along the central vein was
significantly elevated, respectively (P<0.01) (Fig. 3D and F). Overall, the total
quantity of collagen was markedly increased in the
CCl4-treated mice. Treatment with A438079 resulted in a
significant reduction of collagen accumulation within the liver
compared with the CCl4-treated group (P<0.01).
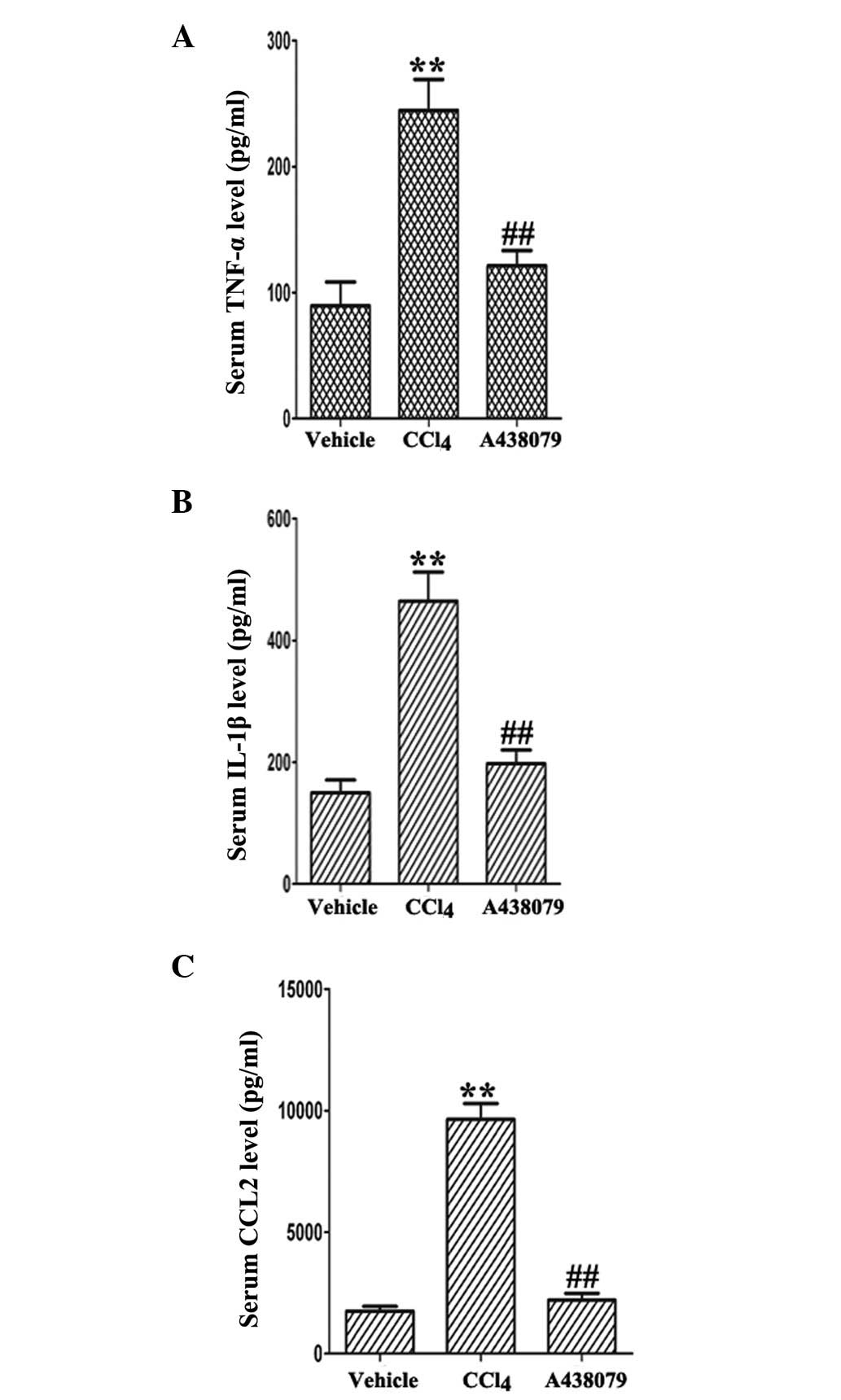
A438079 treatment reduces the production
of pro-inflammatory cytokines
Serum levels of TNF-α, IL-1β and CCL2 were
significantly elevated by >4-fold, respectively, compared with
the vehicle-treated control group (P<0.01; Fig. 4). Notably, the A438079-treated
group showed markedly reduced serum levels of TNF-α, IL-1β and CCL2
(all P<0.01), as indicated by ELISA. These data indicated that
A438079 treatment also significantly reduced the
CCl4-induced inflammatory response.
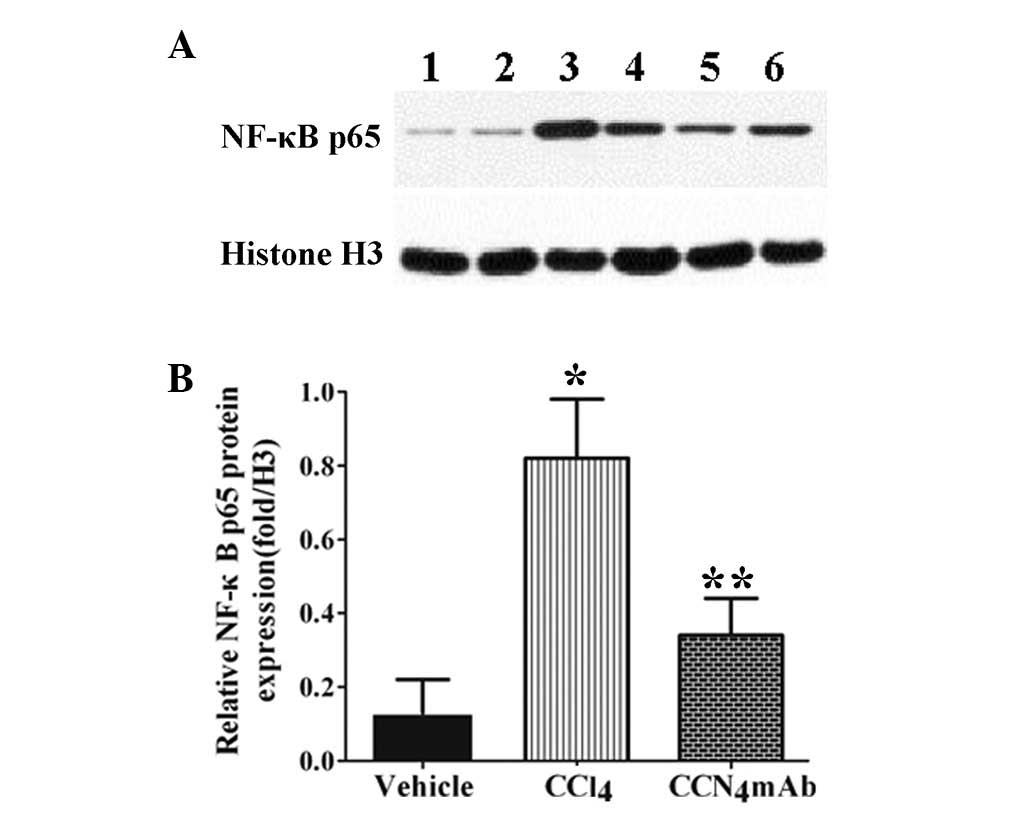
A438079 inhibits the activity of NF-κB
induced by CCl4
Chronic CCl4 administration increased the
activity of NF-κB to 4-fold relative to that of the normal control
(P<0.01) (Fig. 5). The activity
level of NF-κB was significantly reduced via treatment with
A438079, compared with that in the CCl4-treated group
(P<0.01).
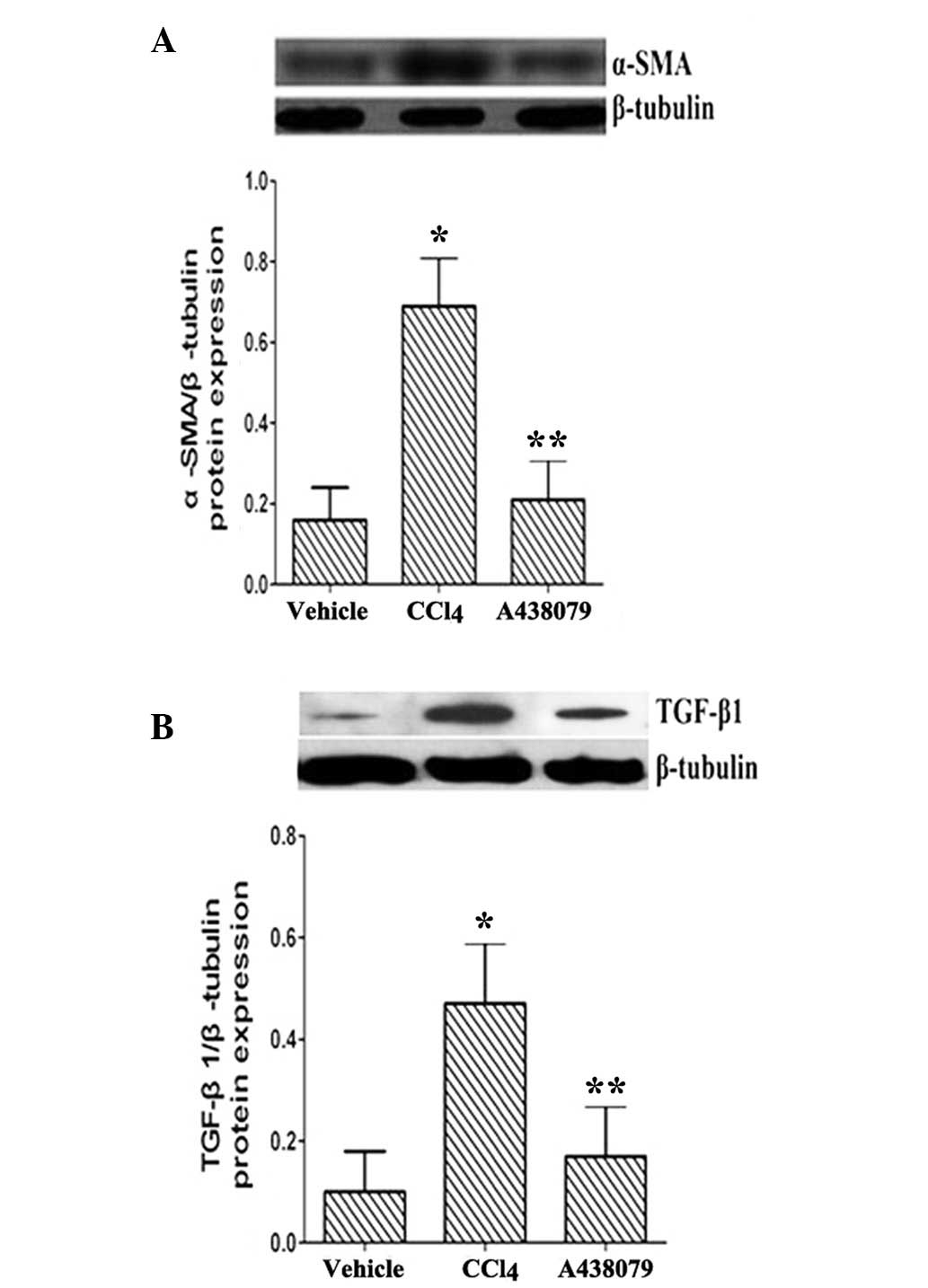
A438079 inhibits the protein expression
of pro-fibrotic factors
Chronic treatment with CCl4 enhanced the
protein expression levels of α-SMA and TGF-β1 compared with the
normal control (P<0.01; Fig.
6). A438079 treatment significantly downregulated the
expression levels of these pro-fibrotic markers (P<0.01).
Discussion
In the present study, it was demonstrated that
chronic CCl4 treatment resulted in necrosis, increased
accumulation of collagen in the liver and a marked increase in the
production of various pro-inflammatory and pro-fibrotic mediators,
indicating the successful fibrosis model induction. Furthermore,
the effects of P2X7 inhibition on liver injury and fibrosis were
investigated using the specific P2X7 inhibitor, A438079. As a
result, A438079 administration effectively attenuated the
CCl4-induced liver injury and fibrosis.
Results of recent studies have shown that P2X7 may
contribute to organ fibrosis (13,15,16).
P2X7 promotes macrophage infiltration and collagen deposition
contributing to the inflammation and fibrosis of unilateral
ureteral obstruction in mice (15). P2X7-deficient mice exhibited
markedly reduced lung inflammation, with reduced fibrosis (13). Subsequent data demonstrated that
P2X7 may be a potential target for the treatment of pancreatic
fibrosis (16). In accordance, the
results suggested that P2X7 activity was present in
CCl4-induced liver injury and fibrosis and contributed
to fibrogenesis.
Hepatic inflammation was correlated with liver
fibrosis (17). NF-κB is a nuclear
transcriptional activator that is central in stress responses and
inflammation. Activation of NF-κB promotes the production of
collagen and inflammatory chemokines in the process of liver
fibrosis (18). In addition, it is
indicated that monocyte infiltration into the liver is a
predominant pathogenic factor for chronic hepatic inflammation and
fibrosis (19). In the present
study, administration of A438079 reduced the
CCl4-induced expression of pro-inflammatory mediators,
such as TNF-α, IL-1β and CCL2. In addition, A438079 reduced the
activity of NF-κB during inflammation. These data demonstrated the
pro-inflammatory effect of P2X7. It is suggested that the
anti-inflammatory effect of A438079 may in part be regulated by
NF-κB, TNF-α, IL-1β and CCL2, which are involved in the
inflammatory process.
TGF-β1 is a well-established fibrogenic cytokine,
predominantly produced by α-SMA-positive myofibroblasts (20). It is now known that subsequent to
binding to its receptors, TGF-β1 activates its downstream signaling
pathway to mediate fibrosis, which is negatively regulated via the
ubiquitin-proteasome degradation mechanism (21). Studies of liver biopsy samples have
shown that α-SMA may be a valuable marker in the evaluation of
fibrosis progression and an early indicator of the development of
fibrosis (22). In the present
study, it was demonstrated that the CCl4-induced
increase in the expression levels of TGF-β1 and α-SMA was reduced
by A438079. Thus, it is suggested that the protective effect of
A438079 is a result of decreased activation of various pro-fibrotic
factors and the accumulation of collagen.
The results offer novel insights into the potential
importance of P2X7 in hepatic fibrosis. It was demonstrated that
P2X7 was upregulated in the injured livers. Furthermore, P2X7
blockade via A438079 exhibits protective effects, attenuating
CCl4-induced liver injury and fibrosis. In conclusion,
the protective effect of A438079 may be due to its ability to
modulate the inflammatory process. However, the precise mechanism
underlying the involvement of P2X7 in liver fibrosis remains
unclear and requires further investigation.
References
|
1
|
Poli G: Pathogenesis of liver fibrosis:
role of oxidative stress. Mol Aspects Med. 21:49–98. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Brenner DA: Molecular pathogenesis of
liver fibrosis. Trans Am Clin Climatol Assoc. 120:361–368.
2009.PubMed/NCBI
|
|
3
|
Cabibbo G, Maida M, Genco C, Antonucci M
and Cammà C: Causes of and prevention strategies for hepatocellular
carcinoma. Semin Oncol. 39:374–383. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rosenbloom J, Castro SV and Jimenez SA:
Narrative review: fibrotic diseases: cellular and molecular
mechanisms and novel therapies. Ann Intern Med. 152:159–166. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Surprenant A, Rassendren F, Kawashima E,
North RA and Buell G: The cytolytic P2Z receptor for extracellular
ATP identified as a P2X receptor (P2X7). Science. 272:735–738.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Di Virgilio F, Chiozzi P, Ferrari D, et
al: Nucleotide receptors: an emerging family of regulatory
molecules in blood cells. Blood. 97:587–600. 2001.PubMed/NCBI
|
|
7
|
Coutinho-Silva R, Persechini PM, Bisaggio
RC, et al: P2Z/P2X7 receptor-dependent apoptosis of dendritic
cells. Am J Physiol. 276:C1139–C1147. 1999.PubMed/NCBI
|
|
8
|
Pelegrin P: Targeting interleukin-1
signaling in chronic inflammation: focus on P2X(7) receptor and
Pannexin-1. Drug News Perspect. 21:424–433. 2008.PubMed/NCBI
|
|
9
|
Colomar A, Marty V, Médina C, et al:
Maturation and release of interleukin-1beta by
lipopolysaccharide-primed mouse Schwann cells require the
stimulation of P2X7 receptors. J Biol Chem. 278:30732–30740. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Derks R and Beaman K: Regeneration and
tolerance factor modulates the effect of adenosine
triphosphate-induced interleukin 1 beta secretion in human
macrophages. Hum Immunol. 65:676–682. 2004. View Article : Google Scholar
|
|
11
|
Labasi JM, Petrushova N, Donovan C, et al:
Absence of the P2X7 receptor alters leukocyte function and
attenuates an inflammatory response. J Immunol. 168:6436–6445.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chessell IP, Hatcher JP, Bountra C, et al:
Disruption of the P2X7 purinoceptor gene abolishes chronic
inflammatory and neuropathic pain. Pain. 114:386–396. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Riteau N, Gasse P, Fauconnier L, et al:
Extracellular ATP is a danger signal activating P2X7 receptor in
lung inflammation and fibrosis. Am J Respir Crit Care Med.
182:774–783. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Díaz-Hernández M, Díez-Zaera M,
Sánchez-Nogueiro J, et al: Altered P2X7-receptor level and function
in mouse models of Huntington’s disease and therapeutic efficacy of
antagonist administration. FASEB J. 23:1893–1906. 2009.PubMed/NCBI
|
|
15
|
Gonçalves RG, Gabrich L, Rosário A Jr, et
al: The role of purinergic P2X7 receptors in the inflammation and
fibrosis of unilateral ureteral obstruction in mice. Kidney Int.
70:1599–1606. 2006.PubMed/NCBI
|
|
16
|
Haanes KA, Schwab A and Novak I: The P2X7
receptor supports both life and death in fibrogenic pancreatic
stellate cells. PLoS One. 7:e511642012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zimmermann HW, Seidler S, Nattermann J, et
al: Functional contribution of elevated circulating and hepatic
non-classical CD14CD16 monocytes to inflammation and human liver
fibrosis. PLoS One. 5:e110492010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yang H, Zhao LF, Zhao ZF, et al: Heme
oxygenase-1 prevents liver fibrosis in rats by regulating the
expression of PPARγ and NF-κB. World J Gastroenterol. 18:1680–1688.
2010.PubMed/NCBI
|
|
19
|
Wasmuth HE, Tacke F and Trautwein C:
Chemokines in liver inflammation and fibrosis. Semin Liver Dis.
30:215–225. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ueberham E, Löw R, Ueberham U, et al:
Conditional tetracycline-regulated expression of TGF-beta1 in liver
of transgenic mice leads to reversible intermediary fibrosis.
Hepatology. 37:1067–1078. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Derynck R and Zhang YE: Smad-dependent and
Smad-independent pathways in TGF-beta family signalling. Nature.
425:577–584. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Akpolat N, Yahsi S, Godekmerdan A, Yalniz
M and Demirbag K: The value of alpha-SMA in the evaluation of
hepatic fibrosis severity in hepatitis B infection and cirrhosis
development: a histopathological and immunohistochemical study.
Histopathology. 47:276–280. 2005. View Article : Google Scholar : PubMed/NCBI
|