Introduction
Acute kidney injury (AKI), also termed acute renal
failure (ARF) (1), is mainly
caused by toxic and ischemic injury (2). The common characteristics of AKI are
functional and structural changes in the endothelium and proximal
tubule cells, followed by regeneration of damaged renal tubular
epithelial cells and rapidly progressive loss of kidney function
(3–5). In recent years, renal ischemic
disease research has progressed, however, there remains a
requirement for an effective therapy.
As previously reported, the kidney undergoes a
regenerative response and novel cells replace the damaged cells
(6). Previous experimental data
shows that mesenchymal stem cell treatment in improving
ischemia-related organ failure is safe and effective (7). The therapeutic potential of stem
cells, including bone marrow-derived mesenchymal stem cells,
hematopoietic stem/progenitor cells and adipose-derived mesenchymal
stem cells, have been extensively investigated in experimental
animals and patients with kidney disease. A number of experimental
studies (8–11) have shown that stem cells protect
glomerular and tubular structures, promote tubular epithelium
regeneration, increase paracrine and systemic secretory functions
and enhance peritubular capillary regeneration. However, the
underlying mechanisms in the improvement of renal function remains
unclear. Notably, a number of studies have demonstrated that the
contribution of extrarenal cells to the regenerative renal response
is minimal to none (6–12).
In the present study, the AKI rat model was
established by bilateral clamping of renal pedicles for 60 min, as
described previously, with some modifications (13). The allogeneic-bone marrow
mesenchymal stem cells (BMSCs) were implanted by intrarenal
injection into the ischaemia/reperfusion (I/R)-induced AKI rat
model. The purpose of this study was to investigate the protective
role of BMSCs on I/R-induced AKI and to discuss the potential
underlying mechanism.
Materials and methods
Animals
Male Sprague-Dawley (SD) rats weighing between 200
and 300 g were purchased from the Animal Experimental Center of the
Second Hospital of Shandong University, (Shandong, China). All
animals received human care in compliance with the Guide for the
Care and Use of Laboratory Animals published by the U.S. National
Institute of Health (NIH Publication no. 85–23, revised 1996). The
study was approved by the Ethics Committee of the Shandong
University, Jinan, China.
Isolation, culture, identification and
labelling of BMSCs
The cells were isolated from the bone of adult SD
rats using the methods described previously (14,15).
Briefly, cells were isolated by flushing the cavity of femurs and
tibias and transferred to a tissue culture dish 90 mm in diameter.
To separate BMSCs and other cells, the Ficoll (1.077) density
gradient centrifugation method was used with lymphocyte separation
medium (Boshide, Wuhan, China) and a low-speed tabletop centrifuge
(Thermo Electron, Waltham, MA, USA). The white coat, composed of
mononuclear cells from the upper layer and interface, was carefully
collected and washed three times. The isolated bone marrow cells
were cultured and expanded in low glucose culture containing
Dulbecco’s Modified Eagle’s medium with Ham’s nutrient mixture
F-12, supplemented with 10% fetal bovine serum, 100 U/ml penicillin
and 100 g/ml streptomycin. BMSCs were primarily cultured for 7–10
days and then subcultured for 3–5 days. Cells at the third culture
passage were transplanted. All culture experiments in this study
were maintained at 37°C and in 5% humidified CO2 and the
cultures were replenished with fresh medium every 3 days. The cells
were suspended with trypsin and 5×105 cells were washed
in PBS. Following centrifugation, the single-cell suspensions were
stained with mouse anti-human monoclonal antibodies against CD34,
CD44, CD45 and CD90 (Abcam, Cambridge, UK) for 30 min at 4°C on
ice. The second polyclonal antibody [fluorescein isothiocyanate
(FITC)-conjugated anti-mouse secondary antibodies (Santa Cruz
Biotechnology Inc., Santa Cruz, CA, USA)] was added and incubated
at 4°C for an additional 30 min in a dark room. An isotype-matched
irrelevant antibody of the same species was used as a negative
control. Following washing, cells were resuspended in
phosphate-buffered saline (PBS) for fluorescence-activated cell
sorting (FACS) analysis.
Prior to implantation, the cells were labelled with
the cross-linkable membrane dye CM-DiI (2 μg/ml, Invitrogen Life
Technologies, Carlsbad, CA, USA) according to the previously
described manufacturer’s instructions (16). Following labelling,
1×106 cells were resuspended in 100 μl culture medium
and maintained on ice prior to transplantation, the labelling
efficiency reached >85%.
Rat AKI model and cell
transplantation
The I/R-induced AKI rat model was established by
bilateral clamping of renal pedicles for 60 min as previously
described (17). Adult male SD
rats were anaesthetised with Phenobarbital (50 mg/kg), following
separation of the renal capsule and exposure of left kidney under
sterile conditions, the bilateral renal pedicle was occluded with a
nontraumatic microaneurysm clamp (DW Medical Equipment Company,
Beijing, China) for 60 min and then the clamps were removed. The
kidney reperfusion was performed for 6 h prior to the sacrificing
of the animals by injection of excess pentobarbital sodium.
Following the successful establishment of the AKI model,
~1×106 BMSCs were administered by intra-renal injection.
The animals (n=40) were randomly assigned to four groups (n=10 in
each): 1 (control), 2 (I/R), 3 (I/R + culture medium only) and 4
(I/R + BMSCs).
Determination of renal function and
histopathology
Prior to sacrifice, blood was collected from animals
to determine serum levels of creatinine (Cr) and blood urea
nitrogen (BUN) in all groups pre-I/R injury (IRI), and 24 and 72 h
post-I/R injury, respectively, according to the manufacturer’s
instructions. The rats were euthanised and the kidney sections were
fixed with 10% formaldehyde prior to embedding in paraffin. Serial
sections (5 μm) of the embedded tissue were stained with
hematoxylin and eosin (H&E) and periodic acid-schiff stain
(PAS, Baso Biotechnology, Shenzhen, China). The average of the 10
high-power fields (hpf) was randomly selected and the number of the
positive cells in 100 cells in each field was counted at high
magnification (x200). The percentages of 50 sections were counted
by two examiners blinded to treatment assignments. The stained
sections were captured as digital images using Nikon imaging
software (Nikon, Tokyo, Japan).
Quantitative polymerase chain reaction
analysis (qPCR)
qPCR analysis was performed to detect the relative
expression levels of interleukin-6 (IL-6), tumour necrosis factor-α
(TNF-α) and vascular endothelial growth factor (VEGF). Total RNA
was extracted from kidney tissue homogenates with TRIzol Reagent
(Invitrogen Life Technologies). The RNA sample was dissolved in
RNase-free water and quantified spectrophotometrically. Primers
were designed using the Primer Express software package (Applied
Biosystems, Foster City, CA, USA) for the expression experiments in
kidney tissue homogenates. Data were analysed with the ABI Prism
7900 sequence detection system software (Applied Biosystems) and
Glyceraldehyde-3-phosphate dehydrogenase was used as an internal
control for input RNA.
Identification of the injected cells
Three days following cell transplantation, the rats
were euthanised and the renal parenchyma was inflated with optimal
cutting temperature compound (Sigma, St. Louis, MO, USA) and
rapidly frozen in liquid nitrogen and stored at −80°C. Sections
were cut into 5-μm slices and fixed in acetone for 10 min at −20°C.
Survival of the transplanted BMSCs was demonstrated by observing
the presence of CM-Dil-positive labelled cells by fluorescent
microscopy. Immunohistochemical staining with antibodies against
rabbit anti-human von Willebrand factor (vWF; 1:100) and VEGF
(1:200), according to the manufacturer’s instructions.
FITC-conjugated antiserum (1:200; Abcam) was used as a secondary
antibody. Following washing with PBS solution, the sections were
observed and images were captured by fluorescent microscopy.
Statistical analysis
All data are presented as the mean ± standard error
of the mean. Statistical analysis was performed by using the SPSS
version 13.0 statistical software (SPSS Inc., Chicago, IL, USA).
Student’s t-test was used to compare the data between two groups
and one-way analysis of variance was used to compare more than two
groups, followed by the Scheffé’s multiple-comparison test.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Characterization and FACS of cultured
BMSCs
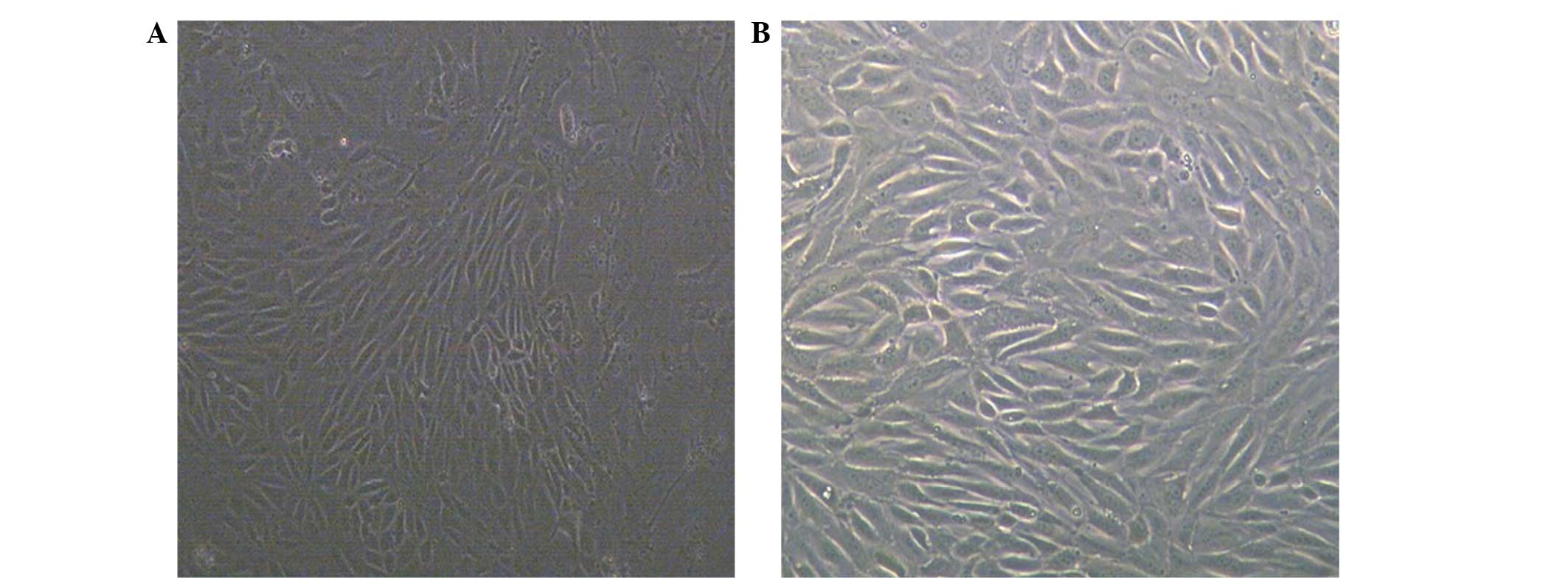
The cells were monomorphic with a typical
spindle-shaped appearance and attached to the tissue culture dishes
(Fig. 1A). Following subculturing,
the cells adhered tightly to the culture dish and proliferated
rapidly in the culture medium (Fig.
1B). In the present study, FACS showed that the cultured cells
were positive for CD90 and CD44 and negative for CD45 and CD34.
Serum levels of Cr and BUN and
histopathology analysis following IRI
The expression levels of serum Cr and BUN were
determined at three time points, pre-IRI, and 24 and 72 h post-IRI.
The levels of BUN and Cr were markedly higher in groups 2 and 3
compared with the control group (P<0.05), however, levels were
notably lower in group 4 compared with groups 2 and 3 at 24 and 72
h post-IRI (P<0.05). The levels of BUN and Cr were not
significantly different between groups 2 and 3 (P>0.05; Fig. 2A and B).
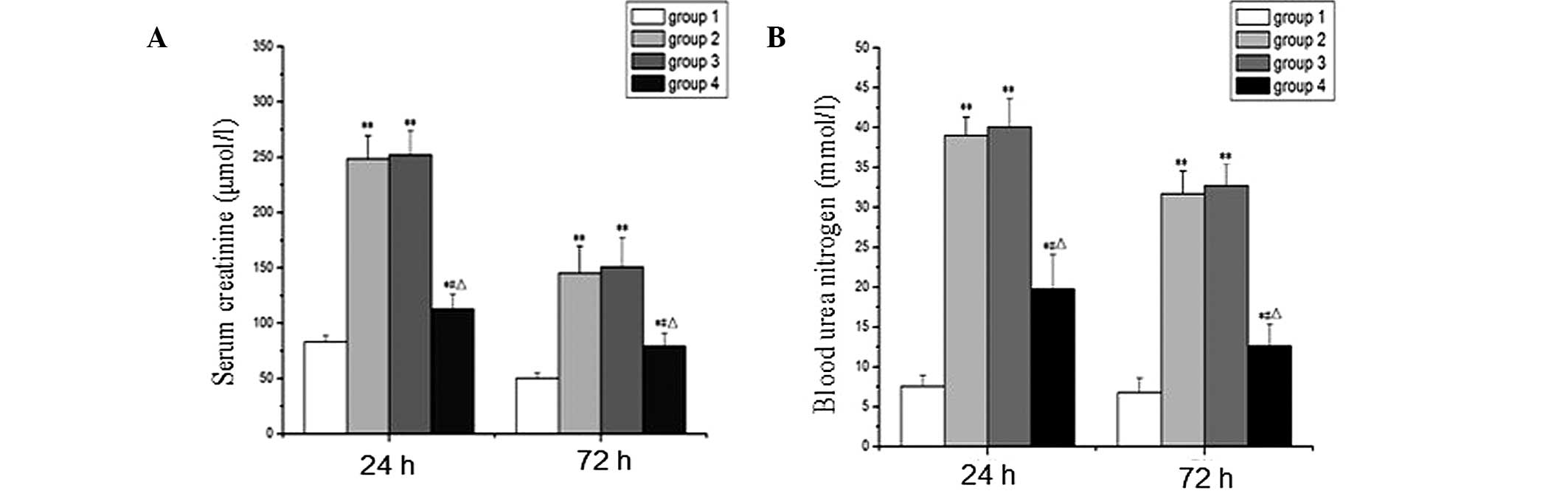 | Figure 2Expression levels of serum levels of
Cr and BUN in all groups. The level of (A) Cr and (B) BUN was
markedly higher in groups 2 and 3 compared with that in control
group, but notably lower in group 4 at 24 and 72 h post-IRI. Group
1, control; group 2, I/R; group 3, I/R + culture medium; group 4,
I/R + BMSCs. Data are presented as the mean ± standard error
(n=10). CR, creatinine; BUN, blood urea nitrogen; BMSCs, bone
marrow mesenchymal stem cells; IRI, ischaemia/reperfusion injury.
#P<0.05 and **P<0.01, vs. the control
group, #P<0.05, vs. group 2, ΔP<0.05,
vs. group 3. |
Microscopic analysis of PAS staining showed that
kidney tissue exhibited severe pathological damage in groups 2 and
3, including tubular necrosis, loss of brush border, cast formation
and tubular dilatation, compared with complete microstructure and
normal morphology in the sham control group. Furthermore, this
pathological damage of kidney was lower in group 4 compared with
groups 2 and 3, three days following surgery. The H&E stain
score was applied based on the aforementioned damage in 10 randomly
selected, non-overlapping fields (7) (magnification, ×200) and were as
follows: 0 (none), 1, <10%; 2, 46–75%; 3, 48–76%; and 4
(36–50%). These results also indicated that the AKI rat model was
successful induced by I/R.
mRNA expression of inflammation
factors
Three days after surgery, there was a significant
increase of IL-6 and TNF-α and decreased VEGF mRNA expression in
groups 2, 3 and 4 compared with the control. Notably, the mRNA
levels of IL-6 and TNF-α were significantly lower and VEGF was
significantly higher in group 4 compared with groups 2 and 3
(P<0.05; Fig. 3A and B). No
significant differences were identified between groups 2 and 3
(P>0.05).
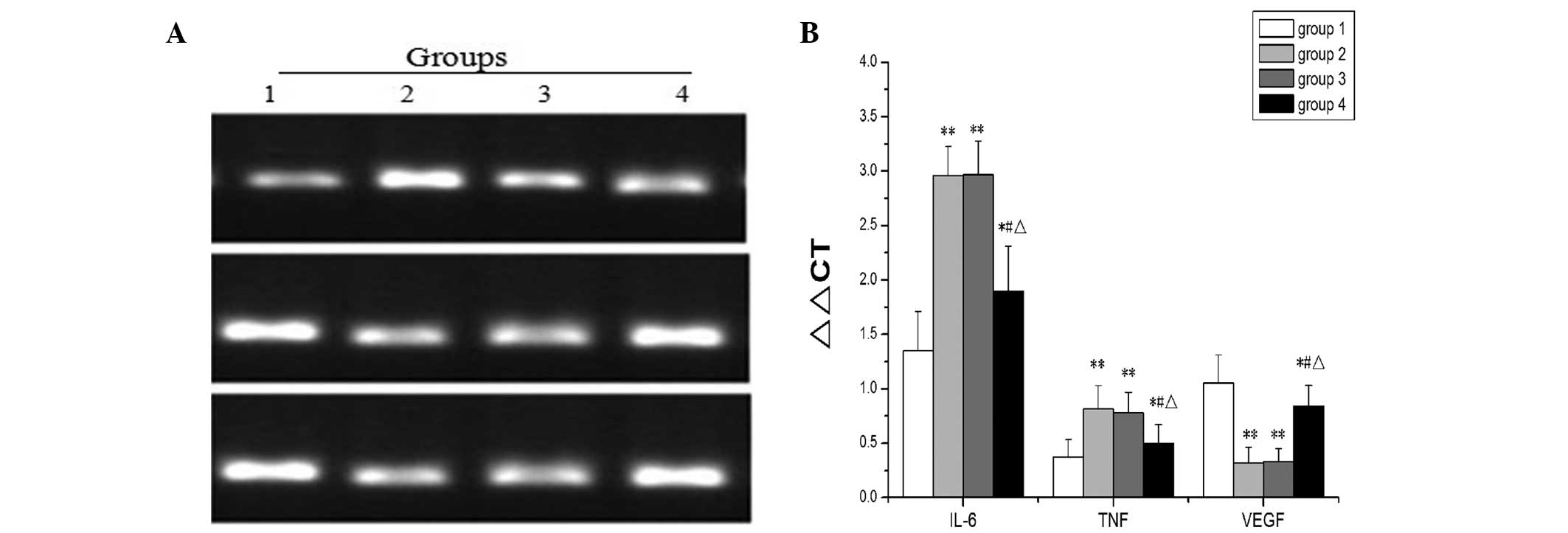 | Figure 3qPCR analysis. The mRNA expression of
IL-6, TNF-α and VEGF 3 days following surgery. (A) There was
significantly increased of mRNA expression of IL-6 and TNF-α in
groups 2, 3 and 4 compared with the control group, but
significantly lower in group 4 than that in groups 2 and 3; the
level of VEGF in groups 2 and 3 compared with control group, but
significantly higher in group 4 compared with groups 2 and 3. (B)
Data are expressed as the mean of 2−ΔΔCT ± standard
error (n=10). Group 1, control; group 2, I/R; group 3, I/R +
culture medium; group 4, I/R + BMSCs. qPCR, quantitative polymerase
chain reaction; IL-6 interleukin-6; TNF-α, tumour necrosis
factor-α; VEGF, vascular endothelial growth factor; BMSCs, bone
marrow mesenchymal stem cells; IRI, ischaemia/reperfusion injury.
#P<0.05 and **P<0.01, vs. control
group, #P<0.05, vs. group 2, ΔP<0.05,
vs. group 3. |
Identification of the transplanted
BMSCs
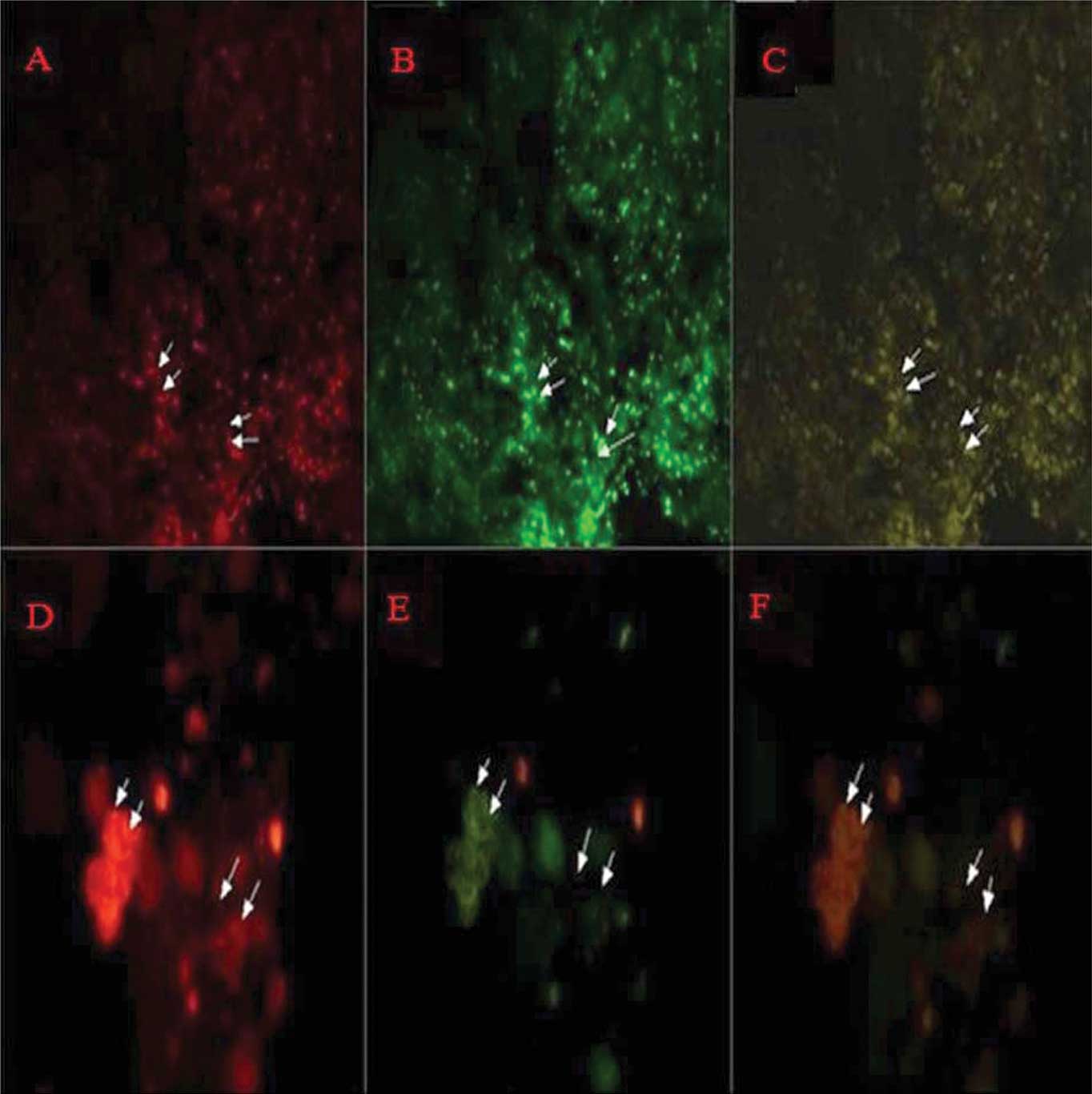
Three days following cell transplantation,
CM-DiI-labelled cells were observed at the transplanted area in
group 4. Immunofluorescence demonstrated that in a number of
regions the CM-DiI-positive BMSCs were observed to coincide with
the green fluorescence spots of vascular endothelial cell specific
markers vWF and VEGF, which suggested that BMSCs may have a
potential angiogenesis effect (Fig.
4).
Discussion
The present study, to the best of our knowledge
showed, for the first time, that intrarenal administration of
allogeneic BMSCs may improve kidney function during acute injury
via an anti-inflammatory effect and a paracrine mechanism.
IRI is a major cause of AKI and acute renal failure
(18–20) and is associated with prolonged
hospitalization and high mortality rates (21). Previous reports (7,22,23)
have shown that there are functional and structural changes in
I/R-induced AKI, including acute damage to the tubular-epithelium,
loss of peri-tubular microvasculature, inflammation and leukocyte
infiltration. Several studies have demonstrated that the
inflammatory reaction is important in the pathophysiological
changes resulting from ischemia (8,24,25),
The primary mechanism is associated with endothelial cells,
leukocytes and tubular cells produce inflammatory cytokines
following ischemic injury. In previous years, a large number of
studies regarding AKI have been performed; however, there remains a
high mortality rate and therefore, it is important to perform
further investigations. Novel research has shown that extra-renal
stem cell administration is an effective treatment modality for AKI
and IRI (9,11,26–28).
The mechanism is highly complex and includes anti-oxidative
activity, anti-inflammatory reactions, angiogenesis, stem cell
homing and immuno-modulation effects (29).
A number of studies have indicated that the
protective effect is attributed to the differentiation of stem
cells for the replacement of damaged cells. Novel tubular cells are
required to replace damaged cells, three possible sources of which
are: i) Adjacent less-damaged tubular cells; ii) extra-renal cells,
presumably of bone marrow origin, that home to the injured kidney
or iii) resident renal stem cells (6). A recent study showed that only a
small proportion of the tubular cells are derived from stem cells,
suggesting that stem cells have a potential paracrine/endocrine
effects (30). The underlying
mechanisms of stem cells improving kidney function are complicated
and remain unclear. Notably, a number of studies have shown that
the contribution of extra-renal cells to the regenerative renal
response is minimal to none (6,12).
Therefore, allogeneic BMSCs were administered by intra-renal
injection into the I/R induced AKI rat model and BMSCs were
hypothesised to protect the kidney against inflammatory stimuli
during acute renal injury.
CM-DiI-labelled allogeneic BMSCs (1×106)
were injected intrarenally into the AKI injury rat model and the
results showed that the expression levels of serum Cr and BUN were
significantly decreased and the pathological damage to the kidney
was significantly lower in the stem cell group compared with the
AKI group. IL-6 and TNF-α are significant mediators in renal I/R
injury (17). TNF-α was observed
to reduce glomerular blood flow and the glomerular filtration rate
by stimulating mesangial cells to produce a variety of
vasoconstrictive mediators, including platelet activating factor,
endothelin-1 and prostaglandins, and recruit neutrophils and
monocytes to the kidney (31). The
current study also demonstrates that the mRNA levels of IL-6 and
TNF-α in I/R kidney were significantly lower three days following
cell injection.
The paracrine/endocrine system is the main mechanism
in stem cell therapy for ischemic organ dysfunction (32–34).
A number of growth factors, including insulin-like growth factor-I,
hepatocyte growth factor, angiopoietin-1, keratinocyte growth
factor and VEGF exhibit a renal-protective function. Previous
studies have shown that angiogenesis/vasculogenesis are key in the
improvement of ischemic organ dysfunction following stem cell
therapy and the efficacy of BMSCs was reduced significantly when
VEGF was knocked down by small interfering RNA (siRNA). In the case
of the present study, the mRNA level of VEGF was significantly
higher in the stem cell group compared with the AKI model rats. By
contrast, immunofluorescence demonstrated that three days following
cell transplantation, the CM-DiI-positive BMSCs were observed to
coincide with the green fluorescence spots of vascular endothelial
cell specific markers vWF, which suggested that BMSCs may have the
potential to differentiate into endothelial cells in vivo
and exhibit an angiogenesis effect. These results indicated that
injected BMSCs by angiogenesis and vasculogenesis mechanisms may
improve endothelial function.
In conclusion, BMSCs may protect the kidney against
the I/R-induced AKI model via anti-inflammatory effects and a
paracrine mechanism, these results provide sufficient evidence for
potential use in clinical trials.
Acknowledgements
This study was supported by grants from the Natural
Science Foundation of Shandong Province (grant no. ZR2010HM125) and
the Jinan Science and Technology Development Projects (grant no.
201218005).
References
|
1
|
Webb S and Dobb G: ARF, ATN or AKI? It’s
now acute kidney injury. Anaesth Intensive Care. 35:843–844.
2007.
|
|
2
|
Garcia-Tsao G, Parikh CR and Viola A:
Acute kidney injury in cirrhosis. Hepatology. 48:2064–2077. 2008.
View Article : Google Scholar
|
|
3
|
Sáenz-Morales D, Escribese MM, Stamatakis
K, García-Martos M, Alegre L, Conde E, Pérez-Sala D, Mampaso F and
García-Bermejo ML: Requirements for proximal tubule epithelial cell
detachment in response to ischemia: role of oxidative stress. Exp
Cell Res. 312:3711–3727. 2006.PubMed/NCBI
|
|
4
|
Thurman JM: Triggers of inflammation after
renal ischemia/reperfusion. Clin Immunol. 123:7–13. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Boros P and Bromberg JS: New cellular and
molecular immune pathways in ischemia/reperfusion injury. Am J
Transplant. 6:652–658. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gupta S, Verfaillie C, Chmielewski D, et
al: Isolation and characterization of kidney-derived stem cells. J
Am Soc Nephrol. 17:3028–3040. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chen YT, Sun CK, Lin YC, et al:
Adipose-derived mesenchymal stem cell protects kidneys against
ischemia-reperfusion injury through suppressing oxidative stress
and inflammatory reaction. J Transl Med. 9:512011. View Article : Google Scholar
|
|
8
|
Li B, Cohen A, Hudson TE, Motlagh D,
Amrani DL and Duffield JS: Mobilized human hematopoietic
stem/progenitor cells promote kidney repair after
ischemia/reperfusion injury. Circulation. 121:2211–2220. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bi B, Schmitt R, Israilova M, Nishio H and
Cantley LG: Stromal cells protect against acute tubular injury via
an endocrine effect. J Am Soc Nephrol. 18:2486–2496. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yip HK, Chang LT, Wu CJ, Sheu JJ, Youssef
AA, Pei SN, Lee FY and Sun CK: Autologous bone marrow-derived
mononuclear cell therapy prevents the damage of viable myocardium
and improves rat heart function following acute anterior myocardial
infarction. Circ J. 72:1336–1345. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Dekel B, Shezen E, Even-Tov-Friedman S,
Katchman H, Margalit R, Nagler A and Reisner Y: Transplantation of
human hematopoietic stem cells into ischemic and growing kidneys
suggests a role in vasculogenesis but not tubulogenesis. Stem
Cells. 24:1185–1193. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Szczypka MS, Westover AJ, Clouthier SG,
Ferrara JL and Humes HD: Rare incorporation of bone marrow-derived
cells into kidney after folic acid-induced injury. Stem Cells.
23:44–54. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sutton TA, Kelly KJ, Mang HE, Plotkin Z,
Sandoval RM and Dagher PC: Minocycline reduces renal microvascular
leakage in a rat model of ischemic renal injury. Am J Physiol Renal
Physiol. 288:F91–F97. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gao J, Dennis JE, Muzic RF, Lundberg M and
Caplan AI: The dynamic in vivo distribution of bone marrow-derived
mesenchymal stem cells after infusion. Cells Tissues Organs.
169:12–20. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nagaya N, Fujii T, Iwase T, Ohgushi H,
Itoh T, Uematsu M, Yamagishi M, Mori H, Kangawa K and Kitamura S:
Intravenous administration of mesenchymal stem cells improves
cardiac function in rats with acute myocardial infarction through
angiogenesis and myogenesis. Am J Physiol Heart Circ Physiol.
287:H2670–H2676. 2004. View Article : Google Scholar
|
|
16
|
Luan Y, Zhang ZH, Wei DE, Lu Y and Wang
YB: Effects of autologous bone marrow mononuclear cells
implantation in canine model of pulmonary hypertension. Circ J.
76:977–985. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Semedo P, Palasio CG, Oliveira CD, et al:
Early modulation of inflammation by mesenchymal stem cell after
acute kidney injury. Int Immunopharmacol. 9:677–682. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Infanger M, Kossmehl P, Shakibaei M, et
al: Induction of three-dimensional assembly and increase in
apoptosis of human endothelial cells by simulated microgravity:
Impact of vascular endothelial growth factor. Apoptosis.
11:749–764. 2006. View Article : Google Scholar
|
|
19
|
Sementilli A and Franco M: Renal acute
cellular rejection: correlation between the immunophenotype and
cytokine expression of the inflammatory cells in acute
glomerulitis, arterial intimitis, and tubulointerstitial nephritis.
Transplant Proc. 42:1671–1676. 2010. View Article : Google Scholar
|
|
20
|
Ali T, Khan I, Simpson W, Prescott G,
Townend J, Smith W and Macleod A: Incidence and outcomes in acute
kidney injury: a comprehensive population-based study. J Am Soc
Nephrol. 18:1292–1298. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Grossini E, Molinari C, Pollesello P,
Bellomo G, Valente G, Mary D, Vacca G and Caimmi P: Levosimendan
protection against kidney ischemia/reperfusion injuries in
anesthetized pigs. J Pharmacol Exp Ther. 342:376–388. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lafrance JP and Miller DR: Acute kidney
injury associates with increased long-term mortality. J Am Soc
Nephrol. 21:345–352. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
da Silva LB, Palma PV, Cury PM and Bueno
V: Evaluation of stem cell administration in a model of kidney
ischemia-reperfusion injury. Int Immunopharmacol. 15:1609–1616.
2007.PubMed/NCBI
|
|
24
|
Bonventre JV and Zuk A: Ischemic acute
renal failure: an inflammatory disease? Kidney Int. 66:480–485.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li H and Nord EP: CD40 ligation stimulates
MCP-1 and IL-8 production, TRAF6 recruitment, and MAPK activation
in proximal tubule cells. Am J Physiol Renal Physiol.
282:F1020–F1033. 2002.PubMed/NCBI
|
|
26
|
Deng J, Kohda Y, Chiao H, Wang Y, Hu X,
Hewitt SM, Miyaji T, McLeroy P, Nibhanupudy B, Li S and Star RA:
Interleukin-10 inhibits ischemic and cisplatin-induced acute renal
injury. Kidney Int. 60:2118–2128. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Imberti B, Morigi M, Tomasoni S, et al:
Insulin-like growth factor-1 sustains stem cell mediated renal
repair. J Am Soc Nephrol. 18:2921–2928. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Tögel F, Weiss K, Yang Y, Hu Z, Zhang P
and Westenfelder C: Vasculotropic, paracrine actions of infused
mesenchymal stem cells are important to the recovery from acute
kidney injury. Am J Physiol Renal Physiol. 292:F1626–F1635.
2007.PubMed/NCBI
|
|
29
|
Bussolati B, Tetta C and Camussi G:
Contribution of stem cells to kidney repair. Am J Nephrol.
28:813–822. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yuan L, Wu MJ, Sun HY, Xiong J, Zhang Y,
Liu CY, Fu LL, Liu DM, Liu HQ and Mei CL: VEGF-modified human
embryonic mesenchymal stem cell implantation enhances protection
against cisplatin-induced acute kidney injury. Am J Physiol Renal
Physiol. 300:F207–F218. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hiroyoshi T, Tsuchida M, Uchiyama K,
Fujikawa K, Komatsu T, Kanaoka Y and Matsuyama H: Splenectomy
protects the kidneys against ischemic reperfusion injury in the
rat. Transpl Immunol. 27:8–11. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Morigi M, Introna M, Imberti B, et al:
Human bone marrow mesenchymal stem cells accelerate recovery of
acute renal injury and prolong survival in mice. Stem Cells.
26:2075–2082. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hagiwara M, Shen B, Chao L and Chao J:
Kallikrein-modified mesenchymal stem cell implantation provides
enhanced protection against acute ischemic kidney injury by
inhibiting apoptosis and inflammation. Hum Gene Ther. 19:807–819.
2008. View Article : Google Scholar
|
|
34
|
Hoste EA and Schurgers M: Epidemiology of
acute kidney injury: how big is the problem? Crit Care Med.
36(Suppl 4): S146–S151. 2008. View Article : Google Scholar : PubMed/NCBI
|