Introduction
Gestational trophoblastic disease (GTD) is known to
be associated with increased maternal age and is more commonly
observed in Asia. GTD has been subdivided into partial hydatidiform
mole (PHM) with a fetal pole, often with triploidy, which refers to
the combination of a fetus with localized hydatidiform placenta,
and complete HM (CHM) without fetal tissue, which is typically
diploid but derived entirely from the paternal genome (1). CHM pregnancy is more common than PHM
(1). A few cases of CHM with twin
pregnancy have been reported (2–4), with
certain couples choosing to continue the pregnancy until 28 weeks
of gestation and deliver by Caesarean section, resulting in some
healthy babies (2–4). In this study, we present the first
reported case of a patient who was found to have PHM with a
co-existing twin following in vitro fertilization, resulting
in an invasive mole that progressed and metastasized to the
lung.
Case report
This study was approved by the Medical Ethics
Committee of Hubei University of Medicine. The patient provided
informed written consent. A 34-year-old, gravida 2, para 1, patient
was known to have pelvic inflammatory disease. The patient's first
pregnancy was a healthy girl delivered when the patient was 20
years old. The patient then divorced at 24 and remarried at 27. The
patient's husband also had a healthy girl from his first marriage.
No pregnancy occurred in the first 5 years of marriage, even though
the couple were not using any contraceptive methods. The patient
was then referred for assisted conception after she was diagnosed
with blockage of the fallopian tubes. Following ovarian suppression
with a gonadotropin-releasing hormone (GnRH) analogue, follicular
growth was stimulated using purified Gn and ovulation was induced
with human chorionic gonadotropin (hCG). Six oocytes were retrieved
under transvaginal ultrasound guidance and these oocytes were then
inseminated with spermatozoa at a concentration of 25,000 motile
spermatozoa per oocyte. Fertilization was confirmed the following
day, with 6 oocytes observed to be normally fertilized (2 pronuclei
observed). Two 4-cell stage embryos were transferred into the
uterus 48 h following egg collection. Progesterone intramuscular
injection was used for luteal support. Six weeks following embryo
transfer, 2 fetal poles, each with a positive fetal heart, were
observed. At 10 weeks the 2 fetuses appeared to have a human form,
as identified via transvaginal sonography (TVS).
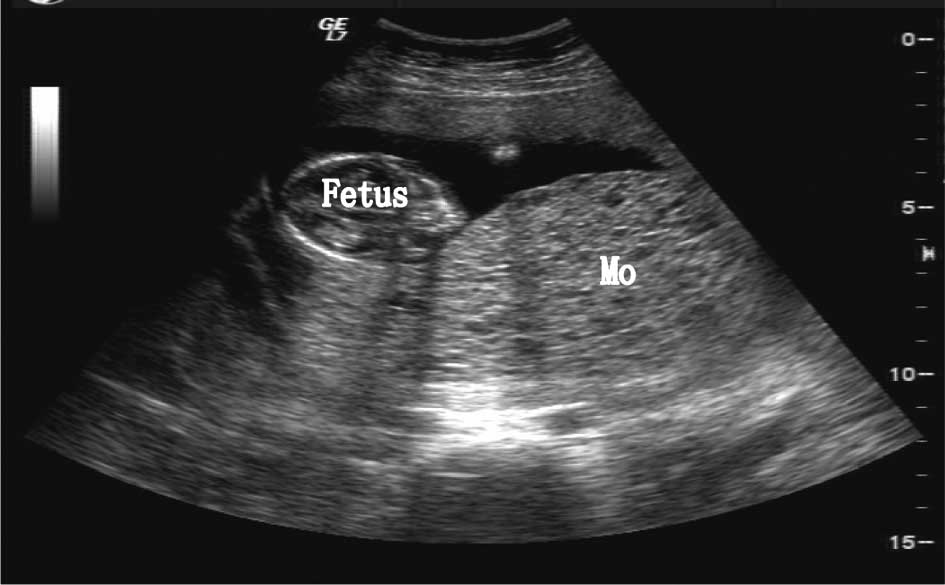
The patient experienced vaginal bleeding at 16
weeks, and repeated TVS revealed 2 viable fetuses; however, 1
placenta was found to be cystic (Fig.
1). Serum hCG levels were found to be elevated at 800,842
mIU/ml. Following careful consideration, the couple decided to
terminate the pregnancy and delivery was induced with rivanol.
After 37 h, 2 fetuses (230 and 249 g), a normal placenta (200 g)
and a partial cystic placenta (290 g) were delivered. Histological
examination confirmed the clinical impression of 1 normal placenta
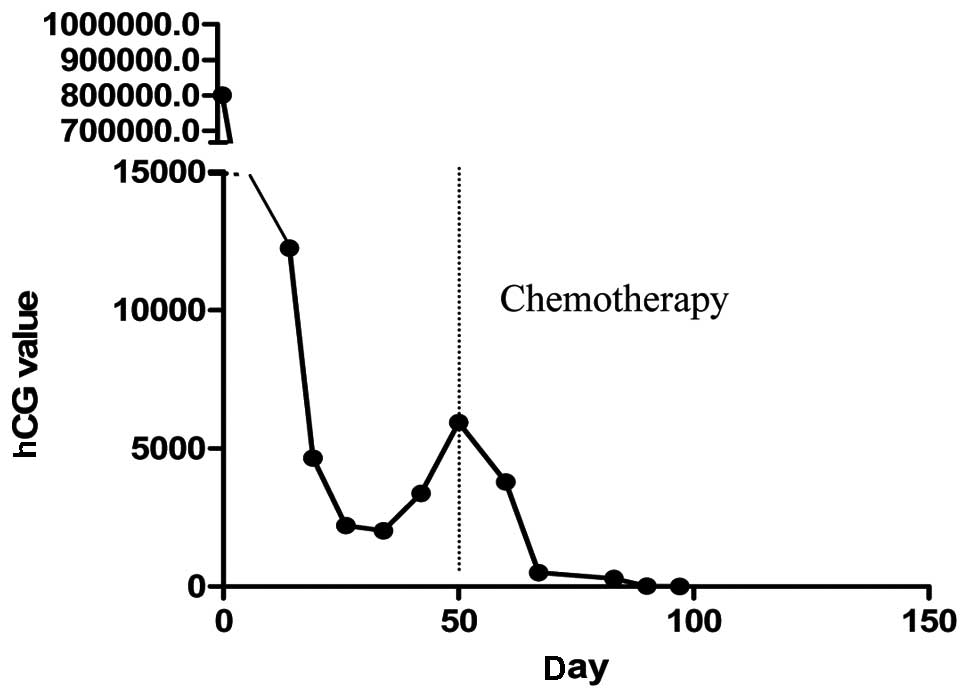
and a second partial HM (69, XXY). Three weeks following curettage,
serial serum hCG concentrations increased continually, and TVS
indicated that the trophoblast had invaded the uterine myometrium.
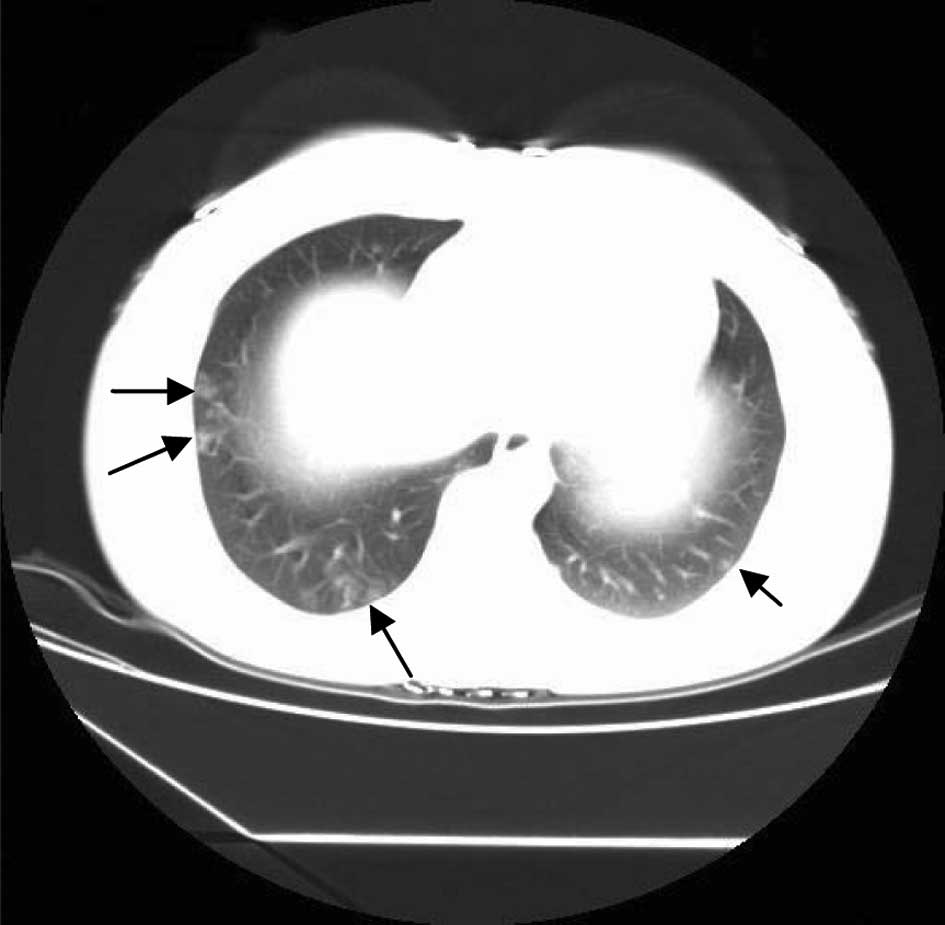
An X-ray computed tomography (CT) chest scan indicated that the
trophoblast had metastasized (Fig.
2). Following 3 cycles of chemotherapy consisting of
fluorouracil (5-Fu) (26 mg/kg/day, 8 day/cycle) and dactinomycin
(KSM) (6 ug/kg/day, 8 day/cycle), the metastases disappeared and
hCG levels became negative, remaining normal for 1 year (Fig. 3).
Discussion
HM results in abnormal pregnancy with excessive
proliferation of placental villi but severely stunted or absent
embryonic development. HMs should be regarded as premalignant
lesions since 15–20% of CHMs and 1% of PHMs undergo malignant
transformation into invasive moles, choriocarcinomas or, in rare
cases, placental-site trophoblastic tumors (PSTTs) (5). Metastases in gestational trophoblastic
neoplasia (GTN) develop in approximately 4% of cases following the
evacuation of CHMs, while fewer than 5% of PHMs are likely to
develop postmolar GTN. Metastases rarely occur and a
histopathological diagnosis of choriocarcinoma has not previously
been confirmed following a partial mole (5). A total of 10–17% of HMs result in an
invasive mole, and approximately 15% of these metastasize to the
lungs or vagina (6).
Although recent advances in TVS may help to diagnose
a twin molar pregnancy commencing at the end of the first
trimester, diagnosis of PHM based on morphological observation is
sometimes difficult and inaccurate (5–6).
However, in the present case, the TVS scan clearly revealed a
normal placenta and sharply defined molar tissue. An invasive mole
is most often clinically diagnosed rather than pathologically,
based on persistent hCG elevation following molar evacuation, and
is frequently treated with chemotherapy without a histopathological
diagnosis (5). In China,
combination chemotherapy consisting of 5-FU and KSM is used as an
effective treatment for GTN (7).
Assisted reproductive technology has enhanced the
fertility of older females, which may increase the proportion of
GTD cases in this age group. This increase is of concern since
malignant sequelae occur more frequently in older patients
(8). However, it is most likely
that certain clinical factors (advanced maternal/paternal age
and/or poor oocyte quality) that precipitate in vitro
fertilization (IVF) treatment also predispose these patients to
GTD. For instance, if 2 spermatozoa or a diploid sperm enter an
oocyte, the result is triploid tissue. It has been established that
some spermatozoa remain diploid, particularly as paternal age
increases.
Following the diagnosis of HM and co-existing fetus,
the decision to terminate or continue the pregnancy was a difficult
one. However, a previous study has demonstrated that only three
patients out of 14 (21%) delivered a healthy child in those
circumstances, and that these patients potentially face a higher
risk of GTN than CHM (4). For
patients with PHM with a co-existing fetus, few have full-term
infants (4). It has been suggested
that medical induction of labor and hysterotomy are not recommended
for molar evacuation since these methods increase maternal
morbidity, including blood loss, incomplete evacuation requiring
dilation and curettage, and the requirement for Caesarean delivery
in subsequent pregnancies. These treatments also increase
trophoblastic dissemination and the development of postmolar GTN
requiring chemotherapy (9–10). However, since bones of the fetus
have formed, a bone spur may penetrate the uterus or cervix and
cause severe and/or heavy bleeding during clamping. These
possibilities should also be considered. We hypothesize that
rivanol induction contributes to patient progression into invasive
mole and metastasis of the lung. Therefore, determination of the
best method for terminating these pregnancies requires further
investigation.
Twin pregnancies with a molar pregnancy and
co-existing viable fetus have been described (4,11–13).
The majority of pregnancies were terminated once the diagnosis was
made. However, a few reports have shown that these patients
completed a successful pregnancy following CHM diagnosis. It
remains unclear as to whether a greater risk of postmolar disease
is associated with more aggressive behavior of the molar tissue or
with delayed delivery. Certain reports suggest that prolonging
gestation does not appear to increase HM malignant changes
(5). Since PHM results in milder
clinical symptoms and a reduced incidence of malignant
transformation than CHM, we suggest that it may be safe to continue
these pregnancies, provided that the fetal karyotype is normal and
maternal complications are controlled.
References
|
1
|
Vassilakos P and Kajii T: Letter:
Hydatidiform mole: two entities. Lancet. 1:2591976. View Article : Google Scholar
|
|
2
|
Montes-de-Oca-Valero F, Macara L and
Shaker A: Twin pregnancy with a complete hydatidiform mole and
co-existing fetus following in-vitro fertilization: case report.
Hum Reprod. 14:2905–2907. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lee SW, Kim MY, Chung JH, Yang JH, Lee YH
and Chun YK: Clinical findings of multiple pregnancy with a
complete hydatidiform mole and coexisting fetus. J Ultrasound Med.
29:271–280. 2010.PubMed/NCBI
|
|
4
|
Massardier J, Golfier F, Journet D, et al:
Twin pregnancy with complete hydatidiform mole and coexistent
fetus: obstetrical and oncological outcomes in a series of 14
cases. Eur J Obstet Gynecol Reprod Biol. 143:84–87. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Seckl MJ, Sebire NJ and Berkowitz RS:
Gestational trophoblastic disease. Lancet. 376:717–729. 2010.
View Article : Google Scholar
|
|
6
|
Lurain JR: Gestational trophoblastic
disease I: epidemiology, pathology, clinical presentation and
diagnosis of gestational trophoblastic disease, and management of
hydatidiform mole. Am J Obstet Gynecol. 203:531–539. 2010.
View Article : Google Scholar
|
|
7
|
Zhao Y, Zhang W and Duan W: Management of
gestational trophoblastic neoplasia with 5-fluorouracil and
actinomycin D in northern China. J Reprod Med. 54:88–94.
2009.PubMed/NCBI
|
|
8
|
Tsukamoto N, Iwasaka T, Kashimura Y,
Uchino H, Kashimura M and Matsuyama T: Gestational trophoblastic
disease in women aged 50 or more. Gynecol Oncol. 20:53–61. 1985.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Soper JT: Surgical therapy for gestational
trophoblastic disease. J Reprod Med. 39:168–174. 1994.PubMed/NCBI
|
|
10
|
Tidy JA, Gillespie AM, Bright N, Radstone
CR, Coleman RE and Hancock BW: Gestational trophoblastic disease: a
study of mode of evacuation and subsequent need for treatment with
chemotherapy. Gynecol Oncol. 78:309–312. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kajii T and Ohama K: Androgenetic origin
of hydatidiform mole. Nature. 268:633–634. 1977. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kim CH, Kim YH, Kim JW, et al: Triplet
pregnancy with partial hydatidiform mole coexisting with two
fetuses: a case report. J Obstet Gynaecol Res. 34:641–644. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Niemann I, Bolund L and Sunde L: Twin
pregnancies with diploid hydatidiform mole and co-existing normal
fetus may originate from one oocyte. Hum Reprod. 23:2031–2035.
2008. View Article : Google Scholar : PubMed/NCBI
|