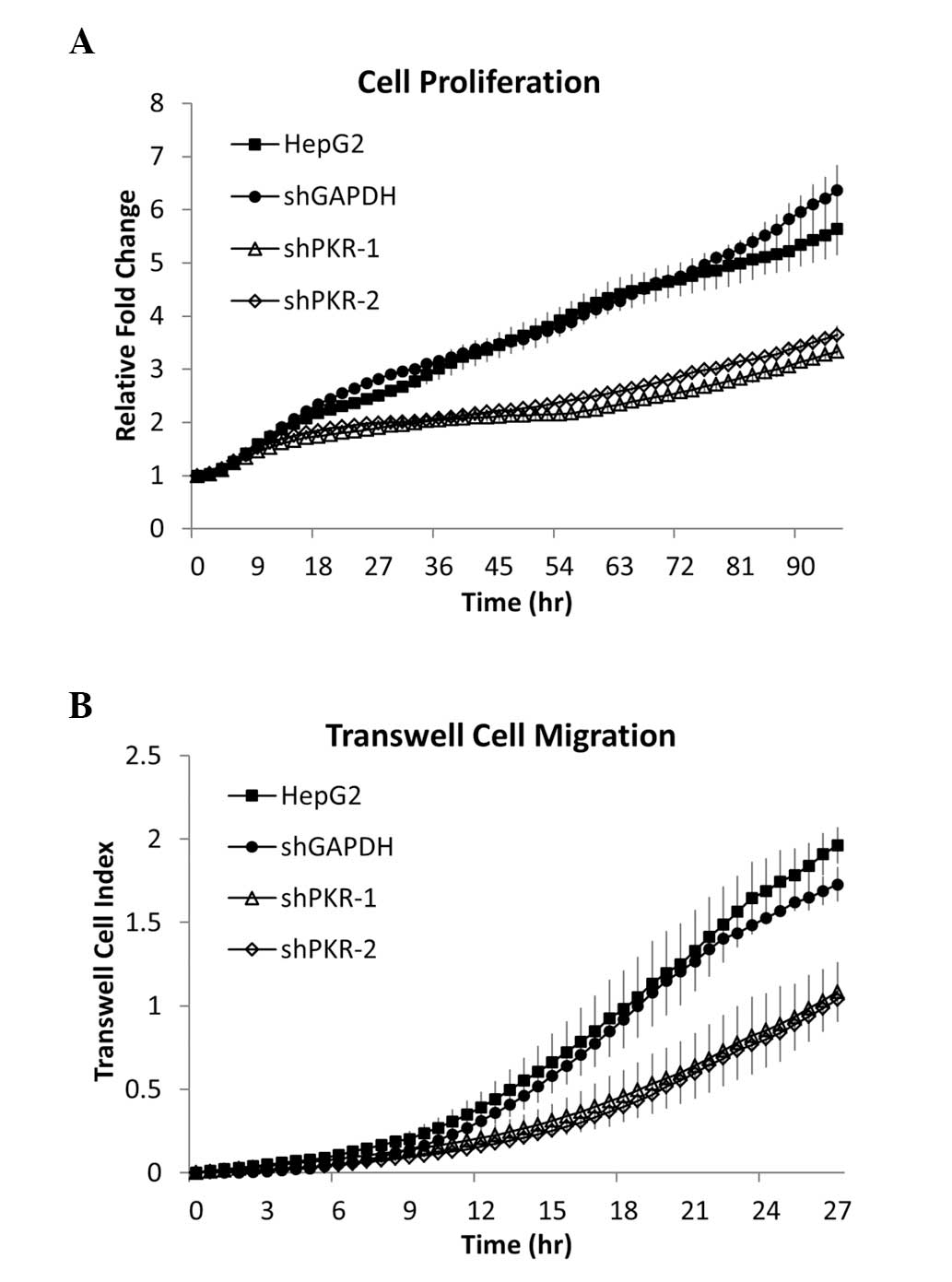
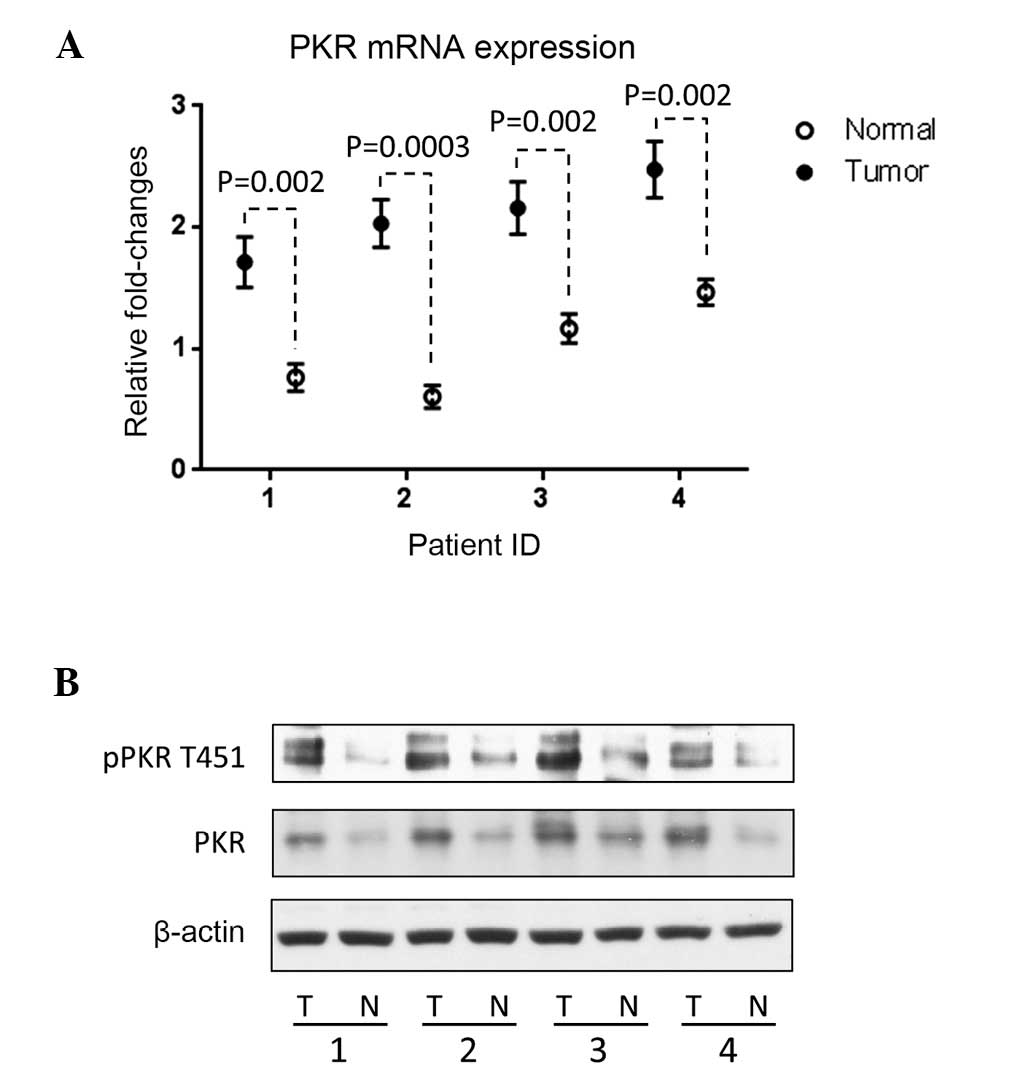
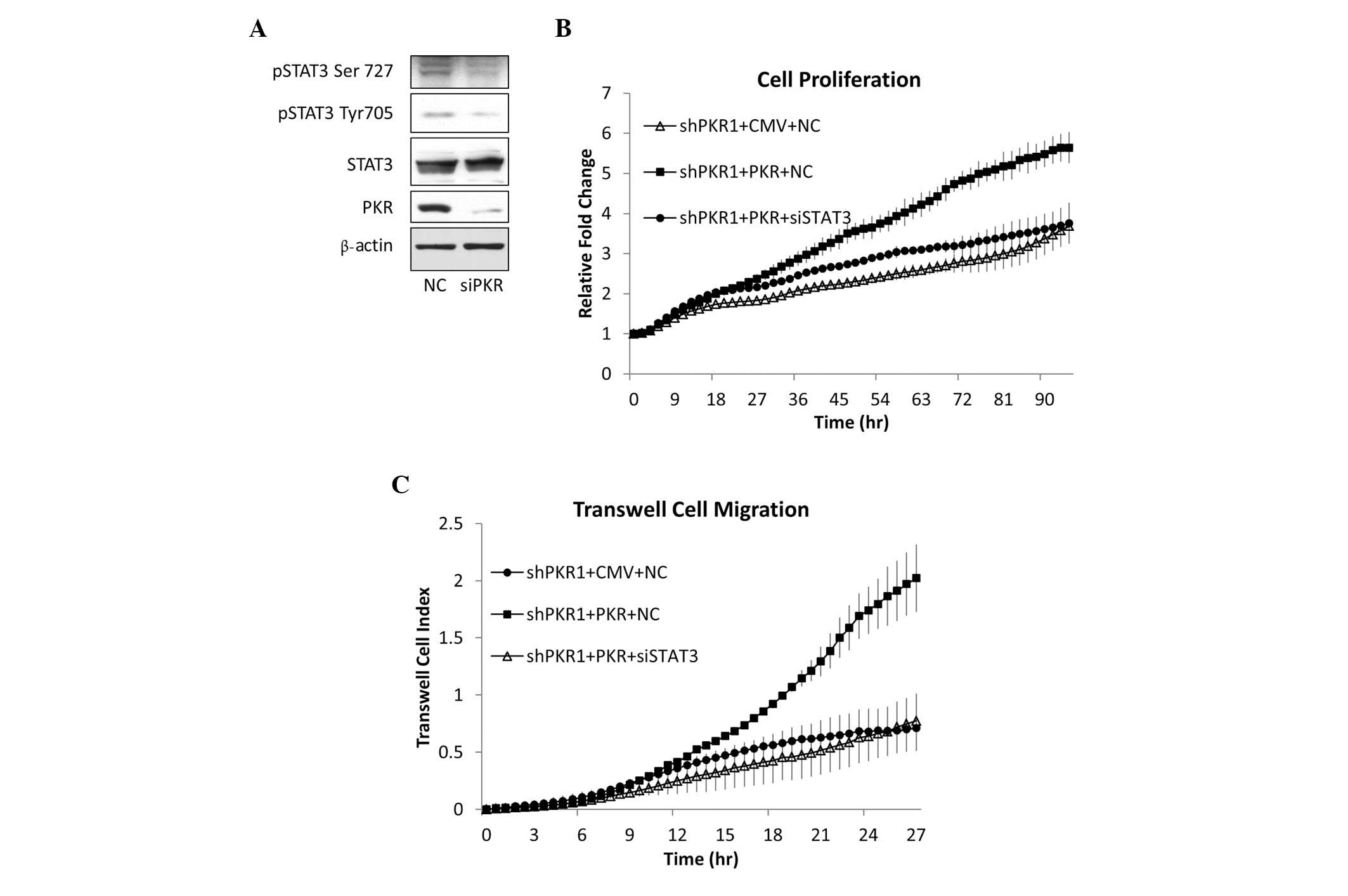
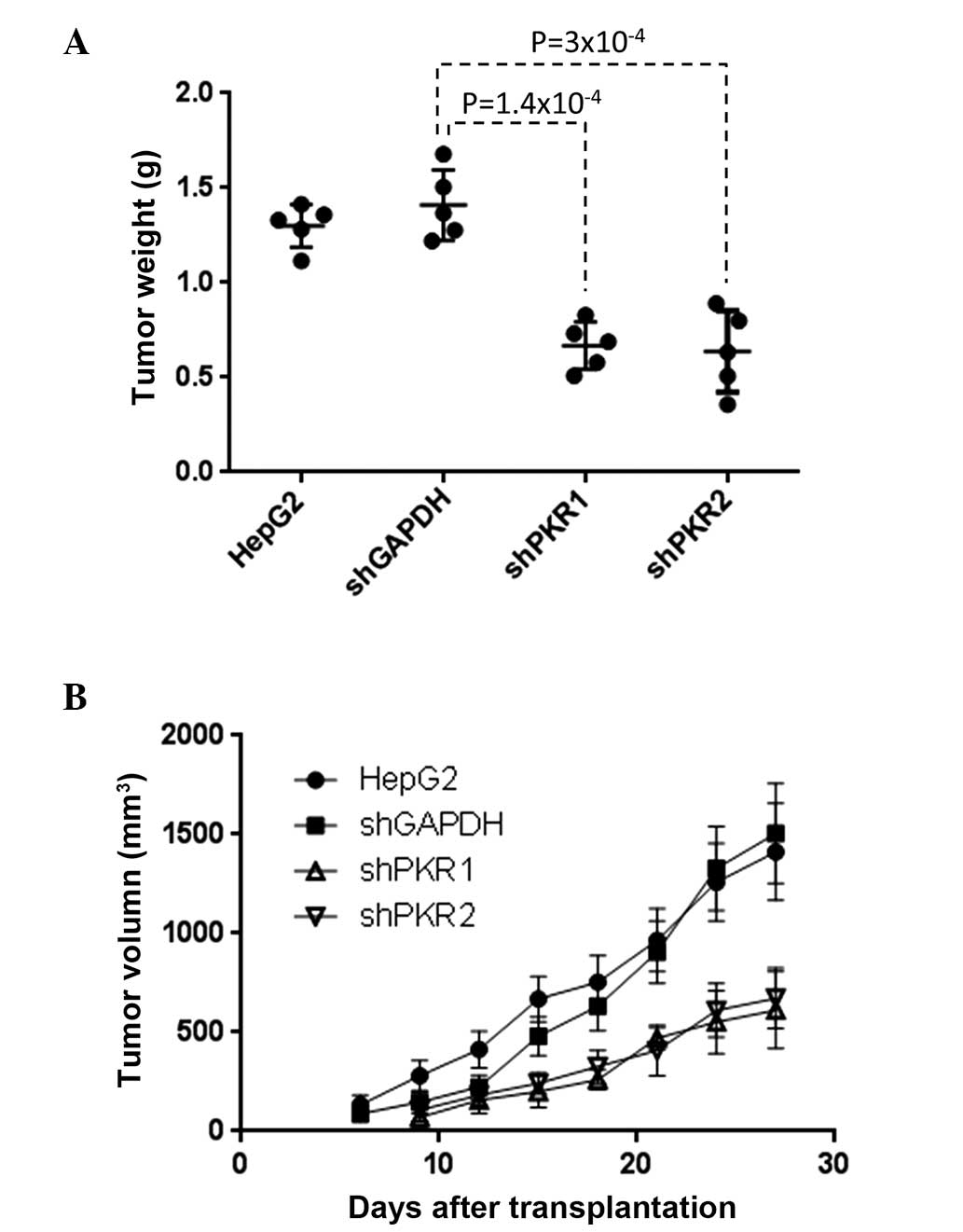
|
1
|
Barber GN: The dsRNA-dependent protein
kinase, PKR and cell death. Cell Death Differ. 12:563–570.
2005.
|
|
2
|
Zhang F, Romano PR, Nagamura-Inoue T, et
al: Binding of double-stranded RNA to protein kinase PKR is
required for dimerization and promotes critical autophosphorylation
events in the activation loop. J Biol Chem. 276:24946–24958.
2001.
|
|
3
|
Taylor SS, Haste NM and Ghosh G: PKR and
eIF2alpha: integration of kinase dimerization, activation, and
substrate docking. Cell. 122:823–825. 2005.
|
|
4
|
Williams BR: Signal integration via PKR.
Sci STKE. 2001:re22001.
|
|
5
|
Stark GR, Kerr IM, Williams BR, Silverman
RH and Schreiber RD: How cells respond to interferons. Annu Rev
Biochem. 67:227–264. 1998.
|
|
6
|
Koromilas AE, Roy S, Barber GN, Katze MG
and Sonenberg N: Malignant transformation by a mutant of the
IFN-inducible dsRNA-dependent protein kinase. Science.
257:1685–1689. 1992.
|
|
7
|
Meurs EF, Galabru J, Barber GN, Katze MG
and Hovanessian AG: Tumor suppressor function of the
interferon-induced double-stranded RNA-activated protein kinase.
Proc Natl Acad Sci USA. 90:232–236. 1993.
|
|
8
|
Roh V, Laemmle A, Von Holzen U, et al:
Dual induction of PKR with E2F-1 and IFN-alpha to enhance gene
therapy against hepatocellular carcinoma. Cancer Gene Ther.
15:636–644. 2008.
|
|
9
|
Hiasa Y, Kamegaya Y, Nuriya H, et al:
Protein kinase R is increased and is functional in hepatitis C
virus-related hepatocellular carcinoma. Am J Gastroenterol.
98:2528–2534. 2003.
|
|
10
|
Delhem N, Sabile A, Gajardo R, et al:
Activation of the interferon-inducible protein kinase PKR by
hepatocellular carcinoma derived-hepatitis C virus core protein.
Oncogene. 20:5836–5845. 2001.
|
|
11
|
Mohamed AA, Nada OH and El Desouky MA:
Implication of protein kinase R gene quantification in hepatitis C
virus genotype 4 induced hepatocarcinogenesis. Diagn Pathol.
7:1032012.
|
|
12
|
Kim SH, Forman AP, Mathews MB and Gunnery
S: Human breast cancer cells contain elevated levels and activity
of the protein kinase, PKR. Oncogene. 19:3086–3094. 2000.
|
|
13
|
Kim SH, Gunnery S, Choe JK and Mathews MB:
Neoplastic progression in melanoma and colon cancer is associated
with increased expression and activity of the interferon-inducible
protein kinase, PKR. Oncogene. 21:8741–8748. 2002.
|
|
14
|
Delgado André N and De Lucca FL: Knockdown
of PKR expression by RNAi reduces pulmonary metastatic potential of
B16-F10 melanoma cells in mice: Possible role of NF-kappaB. Cancer
Lett. 258:118–125. 2007.
|
|
15
|
Donzé O, Deng J, Curran J, et al: The
protein kinase PKR: a molecular clock that sequentially activates
survival and death programs. EMBO J. 23:564–571. 2004.
|
|
16
|
Yang X and Chan C: Repression of PKR
mediates palmitate-induced apoptosis in HepG2 cells through
regulation of Bcl-2. Cell Res. 19:469–486. 2009.
|
|
17
|
Kumar A, Yang YL, Flati V, et al:
Deficient cytokine signaling in mouse embryo fibroblasts with a
targeted deletion in the PKR gene: role of IRF-1 and NF-kappaB.
EMBO J. 16:406–416. 1997.
|
|
18
|
Cuddihy AR, Li S, Tam NW, et al:
Double-stranded-RNA-activated protein kinase PKR enhances
transcriptional activation by tumor suppressor p53. Mol Cell Biol.
19:2475–2484. 1999.
|
|
19
|
Deb A, Zamanian-Daryoush M, Xu Z, Kadereit
S and Williams BR: Protein kinase PKR is required for
platelet-derived growth factor signaling of c-fos gene expression
via Erks and Stat3. EMBO J. 20:2487–2496. 2001.
|
|
20
|
Wang S, Raven JF, Baltzis D, et al: The
catalytic activity of the eukaryotic initiation factor-2alpha
kinase PKR is required to negatively regulate Stat1 and Stat3 via
activation of the T-cell protein-tyrosine phosphatase. J Biol Chem.
281:9439–9449. 2006.
|
|
21
|
Calvisi DF, Ladu S, Gorden A, et al:
Ubiquitous activation of Ras and Jak/Stat pathways in human HCC.
Gastroenterology. 130:1117–1128. 2006.
|
|
22
|
He G, Yu GY, Temkin V, et al: Hepatocyte
IKKbeta/NF-kappaB inhibits tumor promotion and progression by
preventing oxidative stress-driven STAT3 activation. Cancer Cell.
17:286–297. 2010.
|
|
23
|
Carro MS, Lim WK, Alvarez MJ, et al: The
transcriptional network for mesenchymal transformation of brain
tumours. Nature. 463:318–325. 2010.
|
|
24
|
Maeda S, Kamata H, Luo JL, Leffert H and
Karin M: IKKbeta couples hepatocyte death to cytokine-driven
compensatory proliferation that promotes chemical
hepatocarcinogenesis. Cell. 121:977–990. 2005.
|
|
25
|
Maeda S, Hikiba Y, Sakamoto K, et al:
Ikappa B kinasebeta/nuclear factor-kappaB activation controls the
development of liver metastasis by way of interleukin-6 expression.
Hepatology. 50:1851–1860. 2009.
|
|
26
|
He G and Karin M: NF-kappaB and STAT3 -
key players in liver inflammation and cancer. Cell Res. 21:159–168.
2011.
|
|
27
|
Papadakis AI, Paraskeva E, Peidis P, et
al: eIF2α Kinase PKR Modulates the Hypoxic Response by
Stat3-Dependent Transcriptional Suppression of HIF-1α. Cancer Res.
70:7820–7829. 2010.
|
|
28
|
Proud CG: PKR: a new name and new roles.
Trends Biochem Sci. 20:241–246. 1995.
|
|
29
|
Kumar A, Haque J, Lacoste J, Hiscott J and
Williams BR: Double-stranded RNA-dependent protein kinase activates
transcription factor NF-kappa B by phosphorylating I kappa B. Proc
Natl Acad Sci USA. 91:6288–6292. 1994.
|
|
30
|
Gil J, Alcamí J and Esteban M: Activation
of NF-kappa B by the dsRNA-dependent protein kinase, PKR involves
the I kappa B kinase complex. Oncogene. 19:1369–1378. 2000.
|
|
31
|
Zhou HR, Lau AS and Pestka JJ: Role of
double-stranded RNA-activated protein kinase R (PKR) in
deoxynivalenol-induced ribotoxic stress response. Toxicol Sci.
74:335–344. 2003.
|
|
32
|
Xu Z and Williams BR: The B56alpha
regulatory subunit of protein phosphatase 2A is a target for
regulation by double-stranded RNA-dependent protein kinase PKR. Mol
Cell Biol. 20:5285–5299. 2000.
|
|
33
|
Donzé O, Jagus R, Koromilas AE, Hershey JW
and Sonenberg N: Abrogation of translation initiation factor eIF-2
phosphorylation causes malignant transformation of NIH 3T3 cells.
EMBO J. 14:3828–3834. 1995.
|
|
34
|
Watanabe T, Hiasa Y, Tokumoto Y, et al:
Protein kinase R modulates c-Fos and c-Jun signaling to promote
proliferation of hepatocellular carcinoma with hepatitis C virus
infection. PLoS One. 8:e677502013.
|