Introduction
Gastric carcinoma is one of the most common types of
cancer worldwide, which may arise from any section of the stomach.
It accounts for ~800,000 mortalities worldwide annually, with a
poor prognosis (five-year survival rate, <5–15%). Adenocarcinoma
of the stomach is the most frequent type of gastric carcinoma,
representing 90–95% of cases. Certain drugs, including,
fluorouracil, cytarabine and tegafur, have been effective in the
inhibition of carcinoma growth. However, the toxicity of these
drugs is high and certain patients do not tolerate the side effects
well (1). Therefore, the
identification of novel drug molecules with low toxicity for the
treatment of gastric adenocarcinoma is required. Previous studies
have focused on the anticancer effects of natural products, which
are considered to exhibit reduced toxicity.
Bisdemethoxycurcumin (BDMC) is a demethoxy
derivative of curcumin, which is a natural substance found in the
turmeric root. The other two curcuminoids are demethoxycurcumin and
curcumin, which have been reported to suppress the proliferation of
numerous cancer cells (2). BDMC is
significantly more stable than curcumin in physiological media.
However, whether it arrests the cell cycle of gastric
adenocarcinoma and attenuates gastric adenocarcinoma growth remains
unclear. The current study investigated the effect of BDMC on the
growth inhibition of gastric adenocarcinoma in vivo and
in vitro. As mitochondria are critical for the growth of
tumors, the mitochondrial function of gastric adenocarcinoma cells
following BDMC treatment was analyzed.
Materials and methods
Tumor xenograft generation and size
detection
Human gastric adenocarcinoma tissue was obtained
from a 65 year old male patient (T2N1M0, stage IB) during radical
surgery at Lanzhou University Second Hospital (Lanzhou, China).
Written informed consent was obtained from the patient. The tissue
was cut into 2.5×2.5×2.5 mm sections, rinsed with RPMI-1640 medium
(Gibco-BRL, Carlsbad, CA, USA) and subcutaneously injected into the
right lower limbs of 24 male nude mice (age, 2 months old; weight,
20±2 g; Animal Research Center, Lanzhou University, Lanzhou,
China). The mice (tumor size, >150 mm3) were used 12
days following transplantation. In total, 12 mice were
symmetrically subcutaneously injected around the xenograft with 100
mg/kg/day BDMC dissolved in dimethyl sulfoxide (DMSO; Sigma-B6938,
Sigma-Aldrich, St. Louis, MO, USA) for three weeks. The tumor sizes
were measured every three days for 21 days and were calculated
using the following formula: Tumor size (v, mm3) = π/6 ×
ax b2 (a, major axis, mm; b, minor axis, mm2)
(3). This study was approved by the
ethics committee of Lanzhou University Second Hospital (Lanzhou,
China).
Pathology and apoptosis detection
Gastric adenocarcinomas transplanted into two groups
of nude mice were collected after 21 days and cut into 6 μm
sections transversely. Pathological analysis was conducted using
hematoxylin and eosin staining and apoptotic analysis was performed
using terminal deoxynucleotidyl transferase dUTP nick end labeling
(TUNEL) (QIA33 FragEL™ DNA Fragmentation Detection Kit, Merck KGaA,
Darmstadt, Germany) following the manufacturer’s instructions.
Immunohistochemistry (IHC) was performed to detect apoptotic
related factors. The primary antibodies used were anti-human
anti-B-cell lymphoma 2 (Bcl-2 )[B7025; Anbobio Biotechnology, Co.,
Ltd., San Francisco, CA, USA; rabbit immunoglobulin polyclonal
(Ig)G, 1:100], anti-Bcl-2-associated X protein (Bax) (C0132;
Anbobio Biotechnology, Co., Ltd.; rabbit polyclonal IgG, 1:100),
anti-caspase-3 (ab2302; Abcam, Cambridge, UK; Rabbit polyclonal
IgG, 1:200) while secondary antibody was goat anti-rabbit
polyclonal IgG (AP307P; horseradish peroxidase-conjugate;
Millipore, Billerica, MA, USA; 1:10,000). Images were captured
using a confocal microscope (Nikon TE2000; Nikon Corporation,
Tokyo, Japan).
Cytotoxicity and cell cycle analysis
Gastric cancer cells (cell line SGC 7901) were
seeded into a 96-well cell culture plate (Corning Inc., New York,
NY, USA) at a density of 3,000 cells/well and treated with final
concentration of 1, 5, 20, 50, 100 or 200 μM BDMC dissolved in
DMSO, for 72 h. Cytotoxicity was measured using an XTT cell
viability assay (Sigma-Aldrich) according to the manufacturer’s
instructions. Cell cycle analysis of SGC 7901 cells treated with
100 μM BDMC was performed using flow cytometry (FCM; BD FACSArray;
BD Biosciences, Franklin Lakes, NJ, USA).
Effect of BDMC on mitochondrial
function
Cells were seeded into six-well plates (Corning,
Inc.) and treated with 100 μM BDMC for 24 h. Cells were then washed
with phosphate-buffered saline (PBS), and incubated in PBS
containing 10 μM fluorescent probe, dichlorodihydrofluorescein
diacetate (Sigma-Aldrich) for 30 min at 37°C prior to determination
of reactive oxygen species (ROS) levels and then analyzed by FCM.
JC-1 was used to determine the inner mitochondrial membrane
potential (Δψm). Cells were incubated in RPMI-1640 (Gibco-BRL)
containing cationic carbocyanine dye, JC-1 (Sigma-Aldrich; 5
μg/ml), for 30 min at 37°C then analyzed by FCM. The Δψm was
calculated as the red/green fluorescent ratio. The adenosine
triphosphate (ATP) concentration in the mitochondrial fraction and
the cytochrome c (Cyt c) levels in the mitochondrial
and cytosolic fractions of the cells were measured using
reverse-phase high-performance liquid chromatography (HPLC 1100;
Agilent Technologies, Palo Alto, CA, USA). The translocation of Cyt
c was assessed using its ratio in the mitochondrial and
cytosolic fractions following the manufacturer’s instructions.
Data analysis
Each experiment was performed at least in
triplicate. Data was analyzed using SPSS version 12.0 (SPSS, Inc.,
Chicago, IL, USA) and presented as the mean ± standard deviation.
The Student’s t test was used to evaluate two groups and
one-way analysis of variance was used for multi-groups. P<0.05
was considered to indicate a statistically significant
difference.
Results
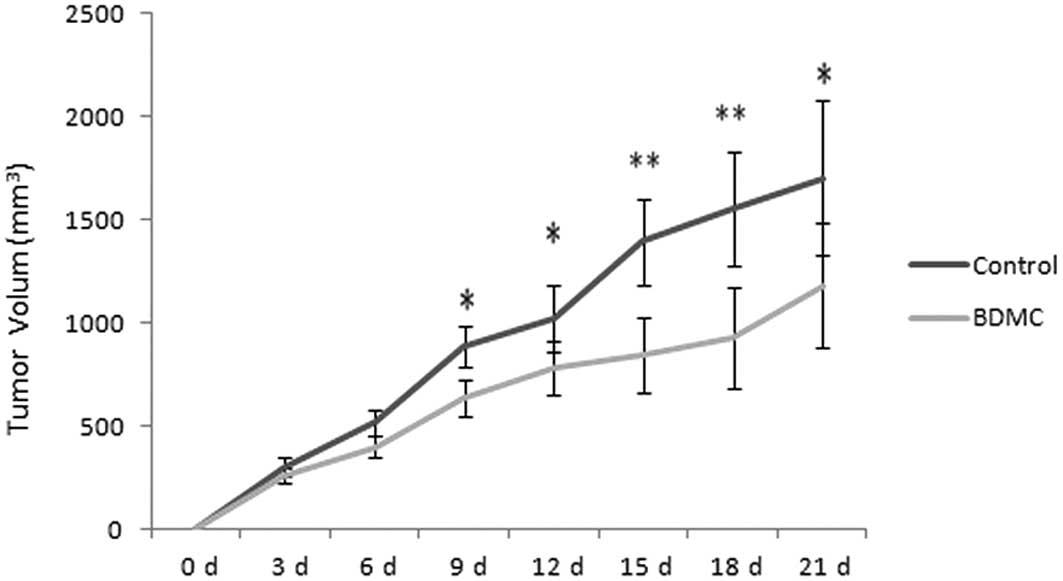
Analysis of tumor size and pathology
The xenograft model was generated successfully in
nude mice and the tumors grew rapidly without treatment. However,
the tumor size was decreased in the BDMC group when compared with
the control group (Fig. 1).
Additionally, during animal feeding, on observation the physical
and mental capacity of the mice in the BDMC group appeared to
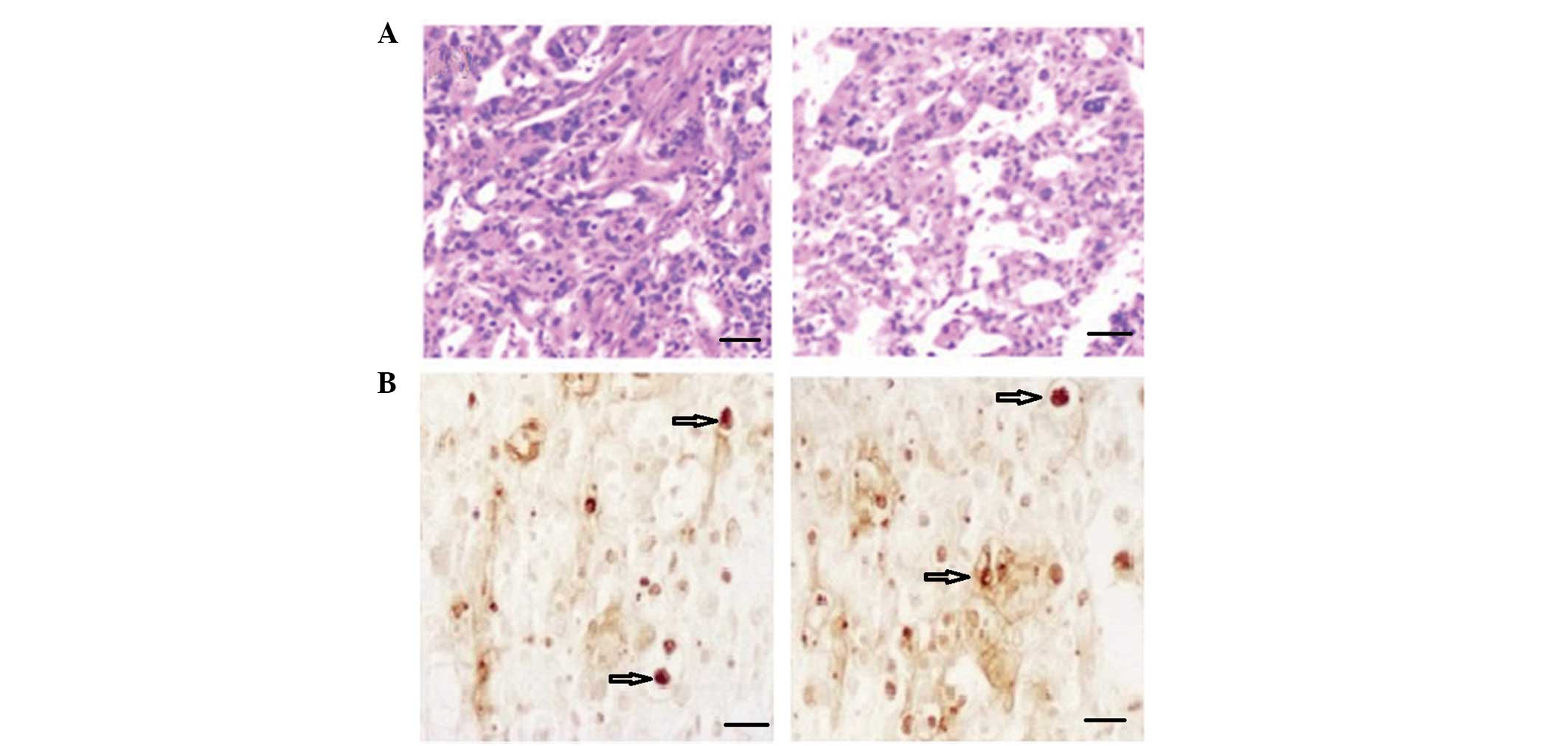
improve. Pathological analysis indicated increased hyperplasia in
the tumors with high density cell nuclei and abundant cytoplasm in
the control group. Low density infiltration and smaller cell nuclei
were identified in the BDMC group, in addition to low activity in
the tumor tissues (Fig. 2A). BDMC
suppressed the growth and activity of the tumor cells.
Apoptotic and relative factor analysis of
the tumor
An increased number of apoptotic cells (Fig 2; arrow indicated) were detected using
TUNEL in the BDMC group. The results indicated that tumor apoptosis
was enhanced following treatment with BDMC. Images are shown in
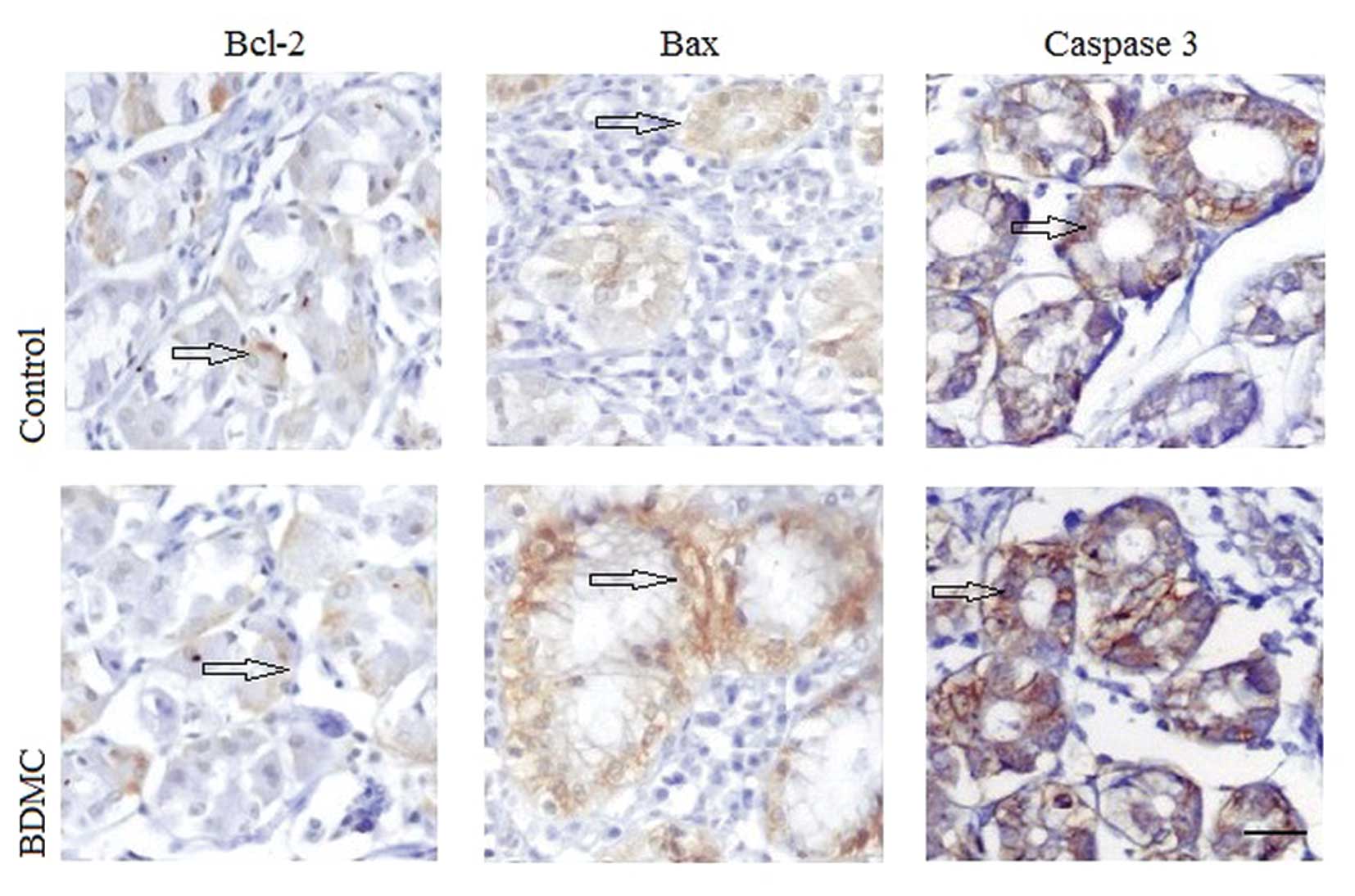
Fig. 2B. IHC was used to detect the
apoptotic factors in situ. As shown in Fig. 3, cytoplasmic Bcl-2 levels were
decreased, while cytoplasmic Bax and nuclear caspase-3 levels were
increased in the BDMC group when compared with that of the control
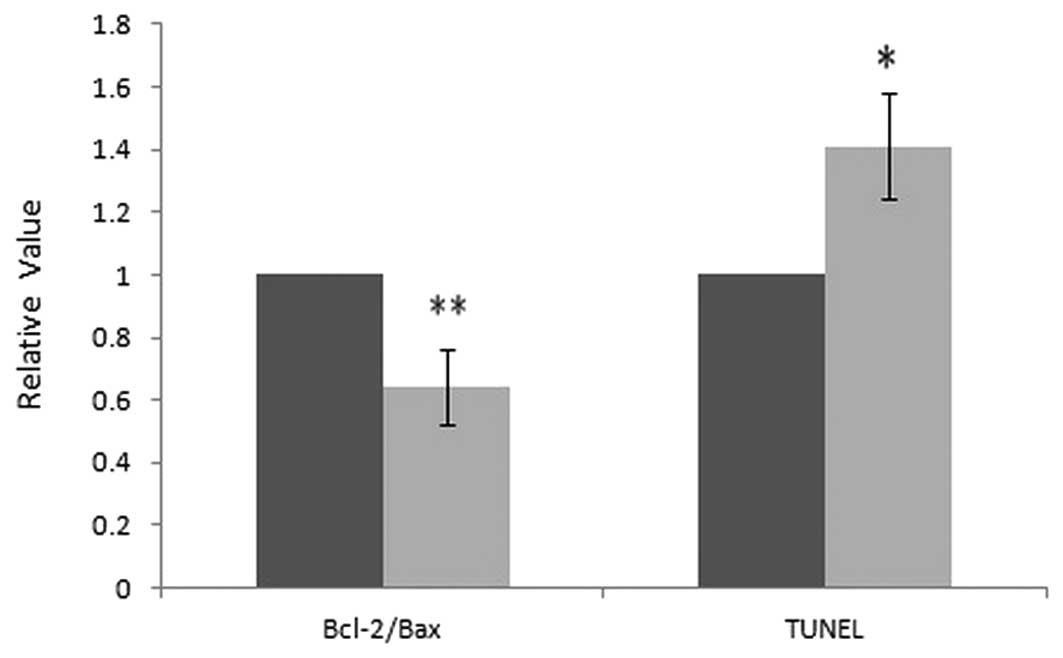
group. The relative values of TUNEL positive cells and the relative
ratio of Bcl-2/Bax was analyzed, where the control group was set as
1, and the relative values in the BDMC group were compared with the
control, as shown in Fig. 4. These
result indicate that BDMC may promote apoptosis in gastric
adenocarcinoma via mitochondria mediated Bcl-2 family and caspase
pathways.
Cell viability and cell cycle
analysis
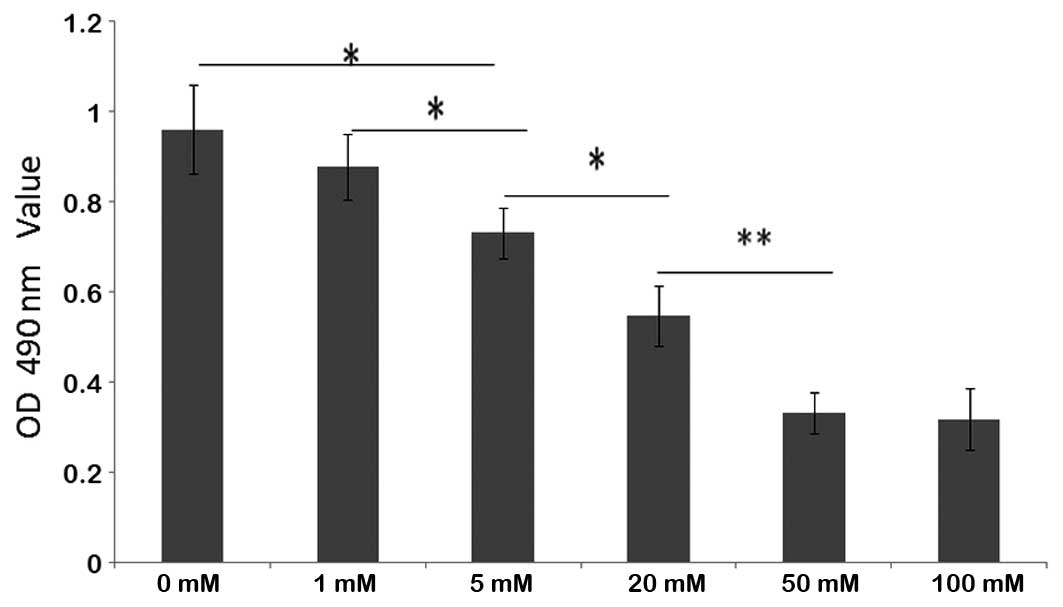
BDMC exhibited dose-dependent cytotoxic effects on
the growth of SGC 7901 cells. The viability of SGC 7901 cells
treated with BDMC at concentrations of >5 μM was significantly
inhibited. No evident increase in inhibition was identified in the
groups treated with >100 μM (Fig.
5). A dose of 100 μM BDMC was determined to be the most
efficacious concentration. Following treatment with 100 μM BDMC,
the percentage of cells at G1 phase in the BDMC group
(62.9±5.5%) was increased significantly when compared with that of
the control group (53.4±6.0%) (P<0.05). The results indicated
that BDMC may inhibit cell growth and arrest the cell cycle at
G1 phase.
Effects of BDMC on mitochondrial
function
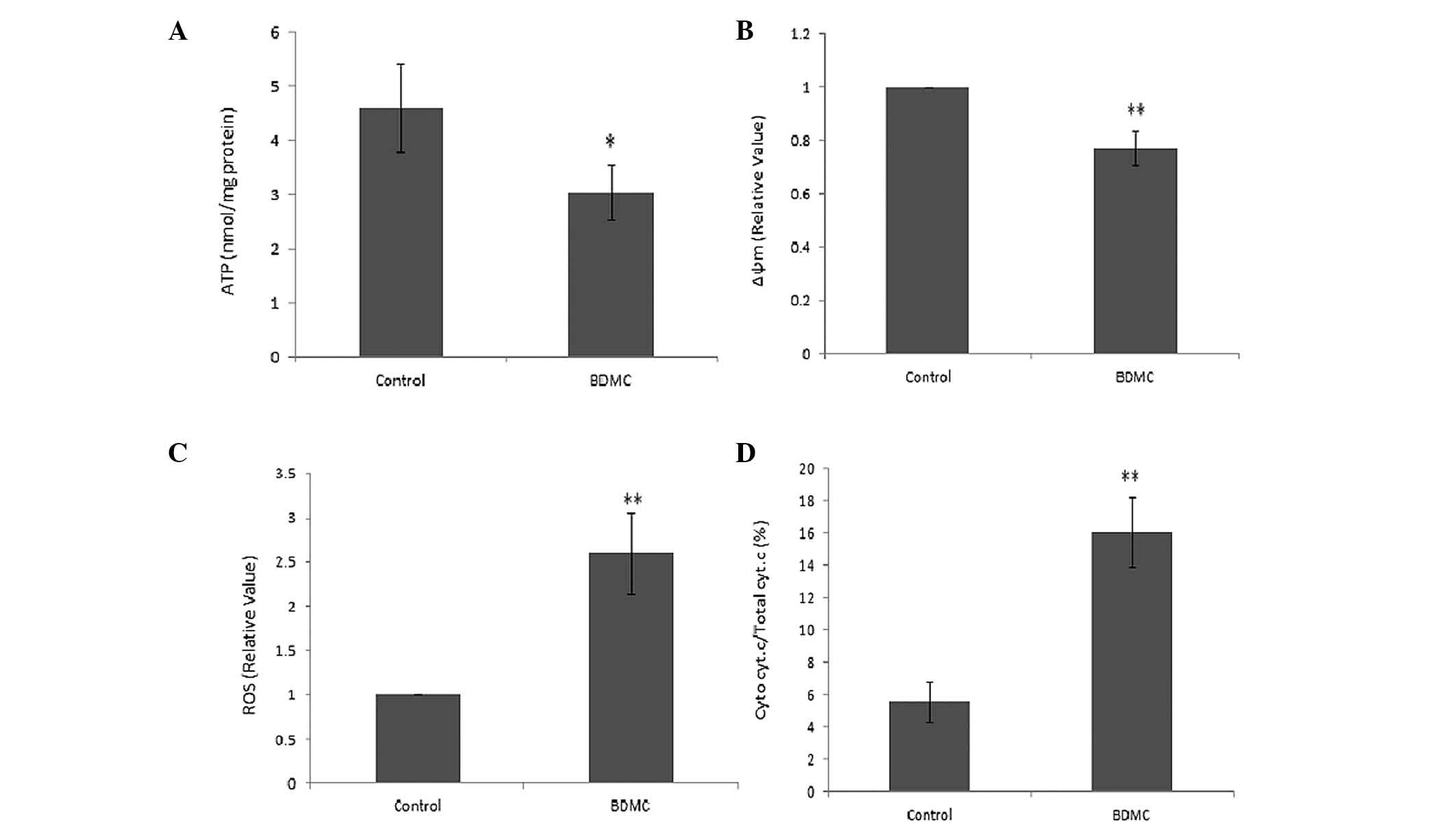
The generation of ATP was markedly decreased in SGC
7901 cells following treatment with BDMC (Fig. 6A). The mitochondrial potential
sensor, JC-1, was used to detect the Δψm, where a ~25% reduction in
the ratio of red to green fluorescence was identified in the BDMC
group, indicating that BDMC causes a reduction in Δψm (Fig. 6B). Rapid increases in ROS generation
in SGC 7901 cells were identified following treatment with BDMC
(Fig. 6C). Furthermore, the release
of Cyt c due to mitochondrial damage caused by BMDC, was
observed in the BDMC group, which resulted in an increased ratio of
Cyt c in the mitochondrial and cytosolic fractions (Fig. 6D). These results indicated the
effect of BDMC on mitochondrial dysfunction.
Discussion
BDMC, together with curcumin and demethoxycurcumin,
are the three predominant active compounds derived from the
turmeric root. Previous studies have shown that curcuminoids
exhibit a variety of therapeutic effects, including neointima
formation (4),
anti-neurodegenerative (5),
anti-cancer (6) and
anit-inflammatory (7) effects.
However, the effects of BDMC have rarely been reported. Thus, the
present study investigates the effects of BDMC on gastric
carcinoma. Gastric carcinoma is the second most common type of
cancer worldwide (8). It may be
classified into several types, including adenocarcinoma of the
stomach (90–95% of cases), which develops from the epithelial
tissue of glandular origin and/or glandular characteristics,
lymphoma of the stomach (4% of gastric carcinomas),
gastrointestinal stromal tumor, neuroendocrine tumors, squamous
cell carcinoma, leiomyosarcoma and small cell carcinoma. Treatment
of adenocarcinoma of the stomach is difficult as few drugs are
effective with few side effects.
In the current study, a human gastric adenocarcinoma
tumor xenograft model was generated in nude mice, which were then
treated with BDMC. BDMC was observed to inhibit the tumor growth
and caused an increase in body weight. Pathological analysis
indicated that the number of positively stained cells was reduced,
indicating that BDMC reduced the rapid growth of cancer cells and
decreased activity in the tumor cells. This was also observed in
vitro. BDMC inhibits the growth of gastric cancer SGC 7901
cells in a dose-dependent manner. The most efficacious
concentration for cell growth inhibition was determined as 100 mM
BDMC. Cell growth is controlled by the cell cycle, which is divided
into G1, S, G2 and M phase. This study
revealed that BDMC arrests the cell growth in G1 phase
and thus, cells could not reach S phase and complete the cycle.
Apoptosis and inhibition of cell growth were observed in the tumor
following treatment with BDMC. The apoptosis regulators, the Bcl-2
family, governs mitochondrial outer membrane permeabilization
(9). The level of proapoptotic
factor Bax, was increased and anti-apoptotic Bcl-2 was decreased
following treatment with BDMC. Caspase-3 was also increased
following treatment with BDMC. Caspase-3 is activated in apoptotic
cells by mitochondrial pathways, in which Cyt c from the
mitochondria works in combination with caspase-9,
apoptosis-activating factor 1 (Apaf-1) and ATP to process
procaspase-3 and subsequently activate caspase-3 (10–12).
The results of this study indicated that BDMC may cause
mitochondrial dysfunction in tumors.
Cancer is characterized uncontrolled cell growth.
Considering that mitochondria is involved in cell proliferation and
division, mitochondrial dysfunction is serious in cells and may
cause cytotoxicity and cell cycle arrest (13). The mitochondrion is a membrane-bound
organelle in eukaryotic cells, which generates ATP as chemical
energy via respiration and the regulation of cellular metabolism.
Due to their rapid growth, cancer cells require a large amount of
ATP to synthesize bioactive compounds for rapid cell proliferation
mainly via the oxidative phosphorylation pathway (14,15).
However, following treatment with BDMC, ATP generation was
decreased significantly and thus, the energy supply for rapid cell
proliferation was insufficient, leading to the inhibition of cell
growth. The Δψm is an important parameter of mitochondrial function
and may be used as an indicator of cell growth (16). A high Δψm has been observed in
rapidly proliferating cells with complex form called J-aggregate,
while a low Δψm has been observed in apoptotic cells with a
monomeric form of JC-1 (17). In
the current study, a significant decrease in Δψm was detected
following treatment with BDMC for 24 h. ROS are chemically reactive
molecules, which at low levels, facilitate cancer cell survival by
cell-cycle promotion; however, at higher levels may suppress tumor
growth via the activation of cell-cycle inhibitors and the
induction of cell death (18).
Following treatment with BMDC for 4 and 24 h, the levels of ROS in
the SGC 7901 gastric cancer cells increased significantly. The
augmentation of ROS production may cause fatal injury to the cancer
cells (19). Cyt c is an
essential component of the electron transport chain, capable of
undergoing oxidation and reduction, which transfers electrons
between Complexes III (Coenzyme Q-Cyt c reductase) and IV
(Cyt c oxidase), indicating its significance in cancers
(20). Cyt c is also
involved in the initiation of apoptosis, by binding to Apaf-1
(21). This study revealed
increased levels of Cyt c and caspase-3 following treatment
with BDMC, compared with the control groups. These results
indicated that BMDC attenuated gastric adenocarcinoma growth by
inducing mitochondrial dysfunction. Therefore, BDMC may present as
a potential drug for gastric adenocarcinoma. However, further
preclinical studies regarding BMDC pharmacodynamics, pharmacology
and side-effects are required to identify whether BDMC may be used
as an anti-cancer reatment.
Acknowledgements
This study was supported by the National Natural
Science Foundation of China (grant no. 81172295) and the Chongqing
Medical University and Lanzhou University Second Hospital.
References
|
1
|
Hayashi Y, Blum MA and Ajani JA: Advanced
gastroesophageal carcinoma: an update on the current therapeutic
landscape. Onkologie. 35:204–209. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Esatbeyoglu T, Huebbe P, Ernst IM, Chin D,
Wagner AE and Rimbach G: Curcumin - from molecule to biological
function. Angew Chem Int Ed Engl. 51:5308–5332. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhang Y, Yao Y, Wang H, Guo Y, Zhang H and
Chen L: Effects of salidroside on glioma formation and growth
inhibition together with improvement of tumor microenvironment.
Chin J Cancer Res. 25:520–526. 2013.PubMed/NCBI
|
|
4
|
Sheu MJ, Lin HY, Yang YH, Chou CJ, Chien
YC, Wu TS and Wu CH: Demethoxycurcumin, a major active curcuminoid
from Curcuma longa, suppresses balloon injury induced vascular
smooth muscle cell migration and neointima formation: an in vitro
and in vivo study. Mol Nutr Food Res. 57:1586–1597. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kim DS, Kim JY and Han Y: Curcuminoids in
neurodegenerative diseases. Recent Pat CNS Drug Discov. 7:184–204.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chen MJ, Cheng YM, Lai PH, Wu JF and Hsu
YC: In vitro biocompatibility of thermally gelling liquid
mucoadhesive loaded curcuminoids in colorectal cancer
chemoprevention. Int J Colorectal Dis. 27:869–878. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ramadan G, Al-Kahtani MA and El-Sayed WM:
Anti-inflammatory and anti-oxidant properties of Curcuma longa
(turmeric) versus Zingiber officinale (ginger) rhizomes in rat
adjuvant-induced arthritis. Inflammation. 34:291–301. 2011.
View Article : Google Scholar
|
|
8
|
Cidón EU: Gastric cancer and the search
for a good prognostic classification: a challenge. Clin Exp
Gastroenterol. 3:113–116. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shamas-Din A, Kale J, Leber B and Andrews
DW: Mechanisms of action of Bcl-2 family proteins. Cold Spring Harb
Perspect Biol. 5:a0087142013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Robertson JD and Orrenius S: Molecular
mechanisms of apoptosis induced by cytotoxic chemicals. Crit Rev
Toxicol. 30:609–627. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Garrido C, Galluzzi L, Brunet M, Puig PE,
Didelot C and Kroemer G: Mechanisms of cytochrome c release from
mitochondria. Cell Death Differ. 13:1423–1433. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cain K: Chemical-induced apoptosis:
formation of the Apaf-1 apoptosome. Drug Metab Rev. 35:337–363.
2003. View Article : Google Scholar
|
|
13
|
Gutierrez J, Ballinger SW, Darley-Usmar VM
and Landar A: Free radicals, mitochondria, and oxidized lipids: the
emerging role in signal transduction in vascular cells. Circ Res.
99:924–932. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhou F, Shen Q and Claret FX: Novel roles
of reactive oxygen species in the pathogenesis of acute myeloid
leukemia. J Leukoc Biol. 94:423–429. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gasparre G, Porcelli AM, Lenaz G and Romeo
G: Relevance of mitochondrial genetics and metabolism in cancer
development. Cold Spring Harb Perspect Biol. 5:2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Smaili SS, Hsu YT, Carvalho AC, Rosenstoc
TR, Sharpe JC and Youle RJ: Mitochondria, calcium and pro-apoptotic
proteins as mediators in cell death signaling. Braz J Med Biol Res.
36:183–190. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chazotte B: Labeling mitochondria with
MitoTracker dyes. Cold Spring Harb Protoc. 9:990–992. 2011.
|
|
18
|
Marullo R, Werner E, Degtyareva N, Moore
B, Altavilla G, Ramalingam SS and Doetsch PW: Cisplatin induces a
mitochondrial-ROS response that contributes to cytotoxicity
depending on mitochondrial redox status and bioenergetic functions.
PLoS One. 8:e811622013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Shimada K, Fujii T, Anai S, Fujimoto K and
Konishi N: ROS generation via NOX4 and its utility in the
cytological diagnosis of urothelial carcinoma of the urinary
bladder. BMC Urol. 11:222011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yadav N and Chandra D: Mitochondrial and
postmitochondrial survival signaling in cancer. Mitochondrion.
16:18–25. 2014. View Article : Google Scholar
|
|
21
|
Pérez-Payá E, Orzáez M, Mondragón L, Wolan
D, Wells JA, Messeguer A and Vicent MJ: Molecules that modulate
Apaf-1 activity. Med Res Rev. 31:649–675. 2011. View Article : Google Scholar
|