Introduction
Lung cancer has a high mortality rate, accounting
for >12% of all newly diagnosed cases of cancer with a poor
prognosis, and is the leading cause of cancer-associated mortality
worldwide (1,2). Non-small-cell lung cancer (NSCLC)
accounts for ~80% of all lung cancer cases and has a five-year
overall survival rate of <15% (3,4). In
addition, ~40% of NSCLC patients have medically inoperable or
unresectable stage III disease at the time of diagnosis (5). Malignant pleural effusion or pleural
nodules are also common occurrences during NSCLC progression,
particularly in adenocarcinoma, due to its location in the
periphery of the lung allowing invasion into the pleural cavity
(6,7).
Visceral pleural invasion (VPI) is the most
significant prognostic factor in lung cancer, and is associated
with advanced tumor stages. The visceral pleura comprises two
elastic layers: The outer elastic layer, which is thick and lies
beneath the submesothelial layer; and the inner elastic layer,
which is thinner and located close to the pulmonary parenchyma. No
elastic fibers are present between the pleural surface and the
outer elastic layer; therefore, the outer elastic layer is more
easily recognized than the inner elastic layer (8,9). Based on
extensive evidence from previous reports, the International
Association for the Study of Lung Cancer (IASLC) proposed the 7th
revision of the tumor-node-metastasis (TNM) staging system for
NSCLC in 2009 (10,11). According to the TNM staging system,
VPI may be classified into the following stages: p10, lung tumor
with no pleural involvement beyond its elastic layer; p11, lung
tumor extending beyond the elastic layer of the visceral pleura,
without being exposed on the pleural surface; and p12, lung tumor
that is exposed on the pleural surface, without involvement of
adjacent anatomical structures (12,13).
Incomplete resection and the presence of nodal involvement have
been consistently reported to be poor prognostic factors in cases
of NSCLC with chest wall invasion (14). Additionally, the depth of chest wall
invasion may affect prognosis following lung cancer resection
(14,15).
Previous clinical studies have demonstrated that
cisplatin may be used as a primary chemotherapeutic agent for the
treatment of lung cancer (16,17).
Certain studies have also revealed that cisplatin is able to bind
to the N7 position of purine DNA bases, constructing cross-links
between adjacent guanines. The ability of cisplatin to react with
cellular DNA results in the formation of DNA adducts, accounting
for its cytotoxic activity and ability to induce apoptosis
(18,19). However, despite its clinical use in
the treatment of lung cancer, side effects and drug resistance have
been reported (20). Investigating
the disposition of cisplatin in the different visceral pleura
layers may provide further insight into the in vivo targets
of cisplatin in lung cancer therapy. A novel sensitive and specific
analytical method is required to facilitate such evaluation.
Tandem mass spectrometry (MS/MS) and electrospray
ionization (ESI) are novel techniques that, along with advances in
mass spectrometry (MS), may markedly improve the sensitivity,
selectivity and specificity of drug detection. In certain studies,
liquid chromatography (LC) coupled with MS/MS was used as the
primary method for identifying and quantifying the tissue
distribution of parent compounds (21,22).
Compared with gas chromatography and high-performance liquid
chromatography (HPLC), the LC-MS/MS method has a number of
advantages, including rapid analytical run-time, minimization of
the mobile phase and use of small sample volumes (23,24).
The aim of the present study was to investigate the
levels of cisplatin in different layers of the visceral pleura in
rats, following drug administration. A previously developed
LC-MS/MS method with rapid analysis and high sensitivity was
validated for use in the detection of cisplatin concentration in
different elastic layers of the visceral pleura in rats.
Materials and methods
Reagents
Cisplatin and apigenin were purchased from
Sigma-Aldrich (St. Louis, MO, USA). All other reagents were of
analytical grade or HPLC grade, and were obtained from DIMA
Technology, Inc. (Richmond Hill, GA, USA).
Analytical procedures
Cisplatin was separated by HPLC using a Symmetry C18
column (50×2.1 mm; Waters Corporation, Beverly, MA, USA). The
elution conditions were as follows: Solvent A, 0.1% (v/v) aqueous
formic acid; solvent B, acetonitrile (100%); and flow rate, 0.2
ml/min with 25% solvent A and 75% solvent B. A high-throughput and
sensitive bioanalytical method was developed using LC/ESI-MS/MS.
The interface was used to generate positive ions,
[M-H]+, for the estimation of the cisplatin
concentration in tissues. Argon was used as the collision gas at a
rate of 0.2 ml/min, while nitrogen was used as the desolvation gas
at a rate of 450 ml/min. The electrospray capillary voltage was 3.0
kV, and the source temperature was 110°C. For quantification of
cisplatin, a previously described internal standard (IS) method
(25,26), using the peak area ratio, was used in
combination with multiple reaction monitoring (MRM) of the
deprotonation precursor ion and the associated product ion for
cisplatin. Collision-induced dissociation was achieved using argon.
The Masslynx Analyst software (version 4.1; Waters Corporation) was
selected for data acquisition.
Preparation of calibration curves
Stock solutions of cisplatin and IS (1.0 mg/ml) were
prepared by dissolving various amounts of the substances in
methanol. Tissue homogenates of the rat visceral pleura layer,
including cisplatin (0.5, 1.0, 10, 20, 50 and 100 ng/ml) and IS
working solution (50 ng/ml apigenin solution dissolved in methanol)
were combined and extracted using acetidin (Tianjin Kermel Chemical
Reagent Co., Ltd., Tianjin, China) (25,27).
Following vigorous vortexing for 1 min and centrifugation at 3,000
× g for 10 min, the supernatant was collected and evaporated to
dryness under nitrogen at 55°C. The residue was subsequently
dissolved in methanol (100 µl) and reconstituted prior to LC-MS/MS
analysis.
Recovery, precision and accuracy
The peak area ratio of cisplatin to the IS was
calculated, and the extract recovery was measured at concentrations
of 1.0, 5.0 and 50.0 ng/ml. Next, the accuracy [expressed as the
percentage of relative error (RE)] was determined by calculating
the values of quality control samples with various concentrations
of cisplatin. The inter-day accuracy and precision [expressed as
the percentage of relative standard deviation (RSD)] were
determined following daily analysis of the quality control samples
over a three-day period. The method was assessed for linearity,
recovery, accuracy and precision.
Sampling and sample preparation
Sprague-Dawley rats (n=66, age, 70–110 days; weight,
270–320 g) were obtained from the Experimental Animal Center of the
PLA General Hospital (Beijing, China). The rats were fasted
overnight with free access to water prior to the day of the
experiment. The experiments were approved by the Animal Ethics
Committee of the PLA General Hospital (Beijing, China). All the
measurements were performed in a coded and blinded manner.
The rats were anesthetized with diethyl ether at
0.17, 0.33, 0.5, 1, 2, 4, 8, 12, 16, 24 and 36 h after
administration of cisplatin, in order to collect blood samples from
the abdominal aorta and tissue samples from different visceral
pleura layers (n=6 in each group). Tissue samples were weighed
prior to homogenization. Based on the aforementioned sample
preparation methods, the tissue samples with cisplatin were
analyzed using LC-MS/MS.
Statistical analysis
Viability parameters were compared using the
Student's t-test for unpaired observations. P<0.05 was
considered to indicate a statistically significant difference. All
the pharmacokinetic parameters were analyzed using the 3P97
pharmacokinetics software version 97 (Chinese Academy of Sciences,
Beijing, China).
Results and Discussion
In the current experiments, the mass signals of
cisplatin were stronger in the positive ESI mode when compared with
the negative mode; therefore, the negative mode was used for
optimization of the mass. Following injection of the standard
solutions into a mass spectrometer, parent ions of cisplatin and IS
were obtained at a mass-to-charge ratio (m/z) of
302.7 and 268.9, respectively. After these parent ions collided
with argon in quadrupole (Q)2 of the MS/MS system, the
corresponding product ion with the most sensitive quantification
conditions was scanned in Q3. Therefore, an MRM transition from
m/z 302.7 to 253.4 for cisplatin and from
m/z 268.9 to 150.6 for apigenin (internal standard)
were used for quantification with the optimal mass condition. A
cone voltage of 30 V and collision energy of 25 eV were used for
cisplatin, whereas a cone voltage of 45 V and collision energy of
20 eV were used for apigenin.
With regard to the chromatographic conditions, the
analytical column and mobile phase were optimized following several
trials, in order to obtain the shortest run-time, optimum
separation, highest sensitivity and symmetric peak shapes for
cisplatin and apigenin. The retention times for cisplatin and
apigenin were 1.18 and 2.25 min, respectively.
Standard curves were developed for the
quantification of cisplatin following the analysis of tissue
samples with different concentrations of cisplatin (0.5, 1, 10, 20,
50 and 100 ng/ml). After calculating the peak area ratio of
cisplatin (y-axis) or IS against the analyte concentration
(x-axis), linear regression analysis of cisplatin was conducted.
Using the LC/ESI-MS/MS method, the standard curves demonstrated a
good linear response (r2>0.9986; n=6) for tissue
samples with concentrations between 0.5 and 100.0 ng/ml, while the
regression equation was y = 0.927x − 0.025.
The lower limit of quantification (LLOQ) represented
the lowest concentration that can be confirmed with sufficient
accuracy (RE≤20%) and precision (RSD≤20%). In order to obtain the
lower detection limit in the current study, the biological samples
were treated with ethyl acetate to achieve efficient cleanup and
reduce the matrix effects, which influence the ionization and
detection of cisplatin. The LLOQ was determined to be 0.1 ng/ml at
a signal-to-noise ratio of >10, with RE<13.5% and
RSD<11.3% (n=6). The chromatograms of cisplatin and apigenin,
without endogenous interfering peaks, exhibited no clear matrix
effects in the rat tissue samples.
In the present study, a liquid-liquid extraction
method, which is able to reduce matrix effects, was used to prevent
contamination or interference in the mass spectra of the tissue
samples, as described in previous reports (28). This extraction procedure may improve
the peak shape, as well as the assay sensitivity, precision or
accuracy, by removing protein precipitates and minimizing ion
suppression. In addition, intra- and inter-day precision was found
to vary by ≤12.7%. Furthermore, control samples treated with
cisplatin were analyzed as described. Low, middle and high
concentrations were selected to evaluate the recovery of cisplatin
and apigenin during extraction. Recovery was determined to be
85.4±2.7%, 87.3±1.6% and 89.5±2.3% (n=6) for the low, middle and
high concentrations, respectively, which demonstrates the robust
efficiency of this method.
In addition, the aforementioned LC-MS/MS method was
validated and used to quantify the distribution of cisplatin in the
different visceral pleura layers following administration of
cisplatin to rats. When cisplatin (5 mg/kg) was injected into the
visceral pleura layers of the rats, the maximum plasma
concentration of drug after administration (Cmax) values
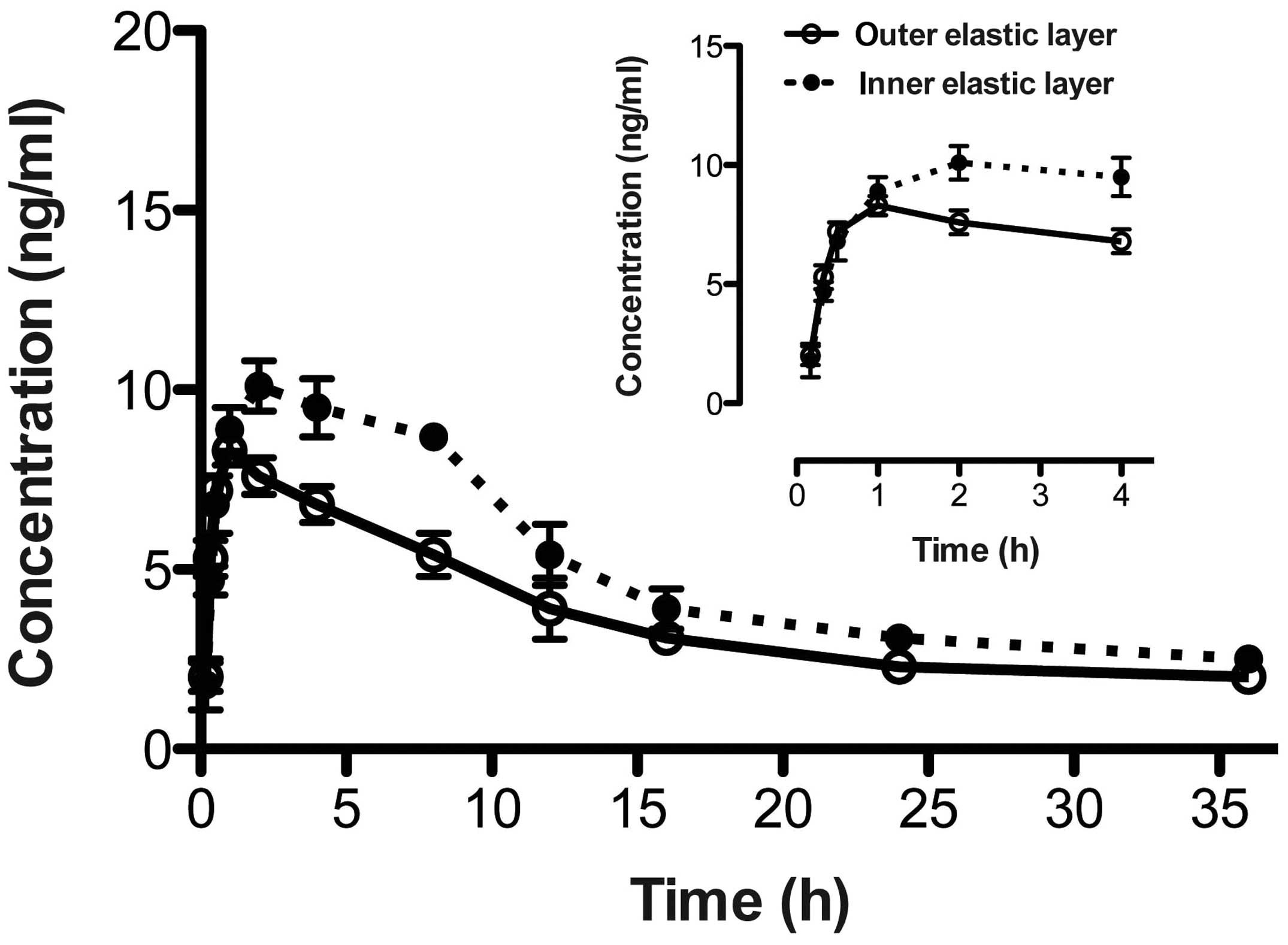
of cisplatin were 8.3±0.4 ng/ml (at 1.0 h post-injection) and
10.1±0.7 ng/ml (at 2.0 h post-injection; n=6) in the outer and
inner elastic layers, respectively. The changes in cisplatin
disposition in the different visceral pleura layer are shown in
Fig. 1, while the pharmacokinetic
parameters are listed in Table I.
 | Table I.Pharmacokinetics of cisplatin
following injection in visceral pleura. |
Table I.
Pharmacokinetics of cisplatin
following injection in visceral pleura.
| Disposition | K,
h−1 | T1/2Ke,
h | Tpeak,
h | Cmax,
ng/ml | AUC, ng × h/ml |
|---|
| Outer | 0.2±0.1 | 3.3±0.1 | 1.0 | 8.3±0.4 | 71.3±6.7 |
| Inner | 0.1±0.1 | 3.9±0.1 | 2.0 | 10.1±0.7 | 89.6±5.3 |
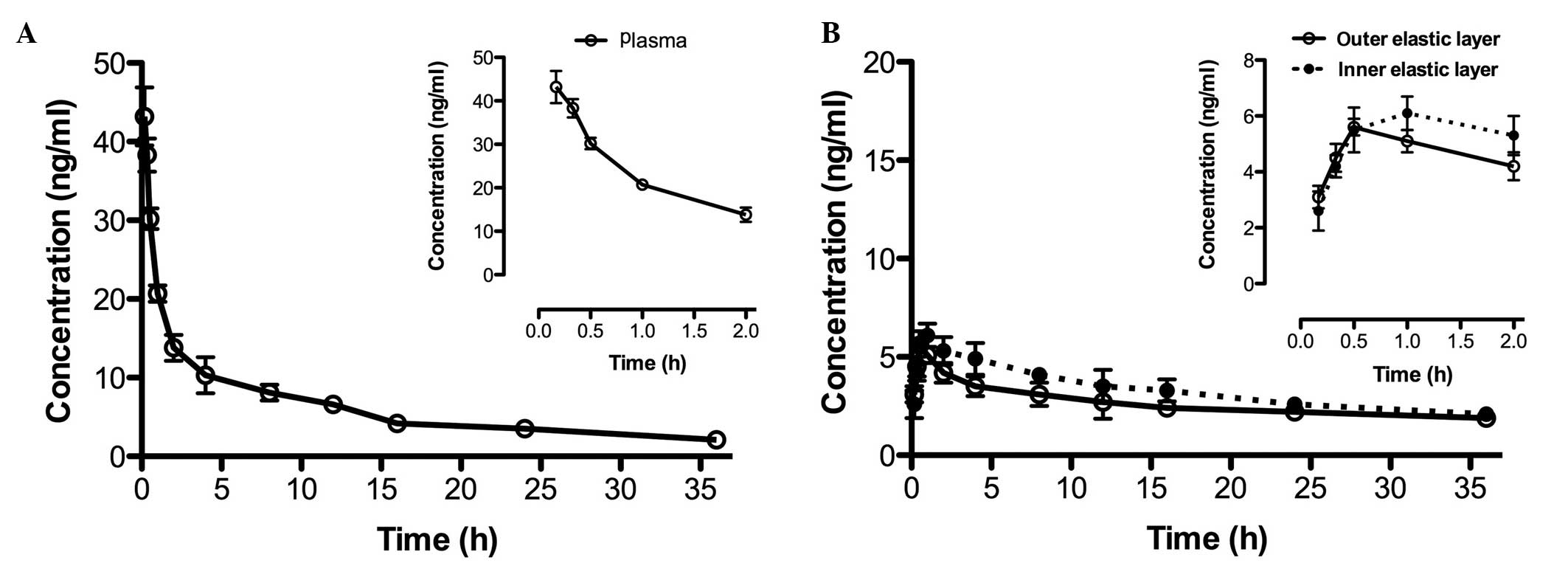
Following injection of cisplatin (5 mg/kg) through
the caudal vein, the Cmax value of cisplatin was
43.2±3.7 ng/ml in the rat plasma (Fig.
2A) and was reached at 0.17 h post-injection. In addition, the
area under the curve was determined to be 312.5±18.3 ng × h/ml. In
the outer and inner elastic layers, the Cmax values were
5.6±0.3 ng/ml and 6.1±0.6 ng/ml (n=6), at 0.5 h and 1.0 h,
respectively (Fig. 2B; Table II).
 | Table II.Pharmacokinetics of cisplatin
following caudal vein injection. |
Table II.
Pharmacokinetics of cisplatin
following caudal vein injection.
| Disposition | K,
h−1 | T1/2Ke,
h | Tpeak,
h | Cmax,
ng/ml | AUC, ng × h/ml |
|---|
| Plasma | 0.5±0.1 | 1.2±0.1 | 0.17 | 43.2±3.7 | 312.5±18.3 |
| Outer | 0.3±0.1 | 3.7±0.2 | 0.5 | 5.6±0.3 | 46.4±4.7 |
| Inner | 0.2±0.1 | 4.3±0.1 | 1.0 | 6.1±0.6 | 52.1±3.2 |
Furthermore, after the injection of cisplatin
through the caudal vein, the plasma concentration rapidly reached
Cmax; however, in the visceral pleura layer, no
sufficient therapeutic concentration of cisplatin was obtained in
the outer or inner elastic layers. For the visceral pleural
invasion of lung cancer, chest wall invasion has been found to
occur in the majority of stages (29,30). The
results of the current study indicate that standard cisplatin
chemotherapy for the treatment of lung cancer with VPI may not
produce the desired therapeutic effects due to insufficient drug
concentration in the target region. Therefore, chemotherapy only
may not be a viable treatment strategy for these patients. In
addition, injection of cisplatin into the visceral pleura layers
demonstrated that a sufficient therapeutic concentration of
cisplatin was not reached in the outer or inner elastic layers.
Based on the results of the present study, it is
hypothesized that surgical intervention in combination with
chemotherapy for lung cancer with VPI may be a more effective
treatment strategy compared with chemotherapy only. Clinical
research must be performed to acquire additional data and further
understanding to establish an optimal treatment for this
disease.
References
|
1
|
Hinojosa de la Garza OR, Sanín LH, Montero
Cabrera ME, et al: Lung cancer mortality and radon concentration in
a chronically exposed neighborhood in Chihuahua, Mexico: a
geospatial analysis. Scientific World Journal. 2014:9353802014.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Torre LA, Bray F, Siegel RL, et al: Global
cancer statistics. 2012. CA Cancer J Clin. Feb 4–2015.(Epub ahead
of print).
|
|
3
|
Fidias P and Novello S: Strategies for
prolonged therapy in patients with advanced non-small-cell lung
cancer. J Clin Oncol. 28:5116–5123. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Cohen MH, Cortazar P, Justice R and Pazdur
R: Approval summary: Pemetrexed maintenance therapy of
advanced/metastatic nonsquamous, non-small cell lung cancer
(NSCLC). Oncologist. 15:1352–1358. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bareschino MA, Schettino C, Rossi A, et
al: Treatment of advanced non small cell lung cancer. J Thorac Dis.
3:122–133. 2011.PubMed/NCBI
|
|
6
|
Leighl NB: Treatment paradigms for
patients with metastatic non-small-cell lung cancer: First-,
second-, and third-line. Curr Oncol. 19:S52–S58. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sangha R, Price J and Butts CA: Adjuvant
therapy in non-small cell lung cancer: Current and future
directions. Oncologist. 15:862–872. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ost D, Goldberg J, Rolnitzky L and Rom WN:
Survival after surgery in stage IA and IB non-small cell lung
cancer. Am J Respir Crit Care Med. 177:516–523. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Riquet M, Arame A and Mordant P: Visceral
pleural invasion: a prognostic factor beginning with small size and
increasing with the progression of lung cancer. Ann Thorac Surg.
97:3832014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
van Meerbeeck JP and Janssens A: The
seventh tumour-node-metastasis staging system for lung cancer:
Sequel or prequel? Europ J Cancer. Suppl 11:150–158. 2013.
View Article : Google Scholar
|
|
11
|
Detterbeck FC, Boffa DJ and Tanoue LT: The
new lung cancer staging system. Chest. 136:260–271. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cho S, Park TI, Lee EB and Son SA: Poor
Prognostic Factors in Surgically Resected Stage I Non-small Cell
Lung Cancer: Histopathologic and Immunohistochemical Analysis.
Korean J Thorac Cardiovasc Surg. 45:101–109. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Park SY, Lee JG, Kim J, et al: Efficacy of
platinum-based adjuvant chemotherapy in T2aN0 stage IB non-small
cell lung cancer. J Cardiothorac Surg. 8:1512013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kawase A, Yoshida J, Miyaoka E, et al:
Visceral pleural invasion classification in non-small-cell lung
cancer in the 7th edition of the tumor, node, metastasis
classification for lung cancer: validation analysis based on a
large-scale nationwide database. J Thorac Oncol. 8:606–611.
2013.PubMed/NCBI
|
|
15
|
Hattori A, Suzuki K, Matsunaga T, et al:
Visceral pleural invasion is not a significant prognostic factor in
patients with a part-solid lung cancer. Ann Thorac Surg.
98:433–438. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
O'Byrne KJ, Barr MP and Gray SG: The role
of epigenetics in resistance to Cisplatin chemotherapy in lung
cancer. Cancers Basel. 3:1426–1453. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wangpaichitr M, Wu C, You M, et al: N',
N'-Dimethyl-N', N'-bis(phenylcarbonothioyl) Propanedihydrazide
(Elesclomol) Selectively Kills Cisplatin Resistant Lung Cancer
Cells through Reactive Oxygen Species (ROS). Cancers Basel.
1:23–38. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wu XX and Kakehi Y: Enhancement of
lexatumumab-induced apoptosis in human solid cancer cells by
Cisplatin in caspase-dependent manner. Clin Cancer Res.
15:2039–2047. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sanborn RE: Cisplatin versus carboplatin
in NSCLC: Is there one “best” answer. Curr Treat Options Oncol.
9:326–342. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chang A: Chemotherapy, chemoresistance and
the changing treatment landscape for NSCLC. Lung Cancer. 71:3–10.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Mudiam MK, Chauhan A, Jain R, et al:
Molecularly imprinted polymer coupled with dispersive liquid-liquid
microextraction and injector port silylation: a novel approach for
the determination of 3-phenoxybenzoic acid in complex biological
samples using gas chromatographytandem mass spectrometry. J
Chromatogr B Analyt Technol Biomed Life Sci. 945–946:23–30. 2014.
View Article : Google Scholar
|
|
22
|
Hoofnagle AN and Roth MY: Clinical review:
Improving the measurement of serum thyroglobulin with mass
spectrometry. J Clin Endocrinol Metab. 98:1343–1352. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Gosetti F, Mazzucco E, Gennaro MC and
Marengo E: Ultra high performance liquid chromatography tandem mass
spectrometry determination and profiling of prohibited steroids in
human biological matrices. A review. J Chromatogr B Analyt Technol
Biomed Life Sci. 927:22–36. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gouveia MJ, Brindley PJ, Santos LL, et al:
Mass spectrometry techniques in the survey of steroid metabolites
as potential disease biomarkers: A review. Metabolism.
62:1206–1217. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bielawski J, Szulc ZM, Hannun YA and
Bielawska A: Simultaneous quantitative analysis of bioactive
sphingolipids by high-performance liquid chromatographytandem mass
spectrometry. Methods. 39:82–91. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liu X, Teng Z, Zhang Y, et al: High
performance liquid chromatographytandem mass spectrometric
determination of resveratrol and its metabolites in rat tissues.
Anal Lett. 43:557–569. 2010. View Article : Google Scholar
|
|
27
|
Chow TW, Szeitz A, Rurak DW and Riggs KW:
A validated enantioselective assay for the simultaneous
quantitation of (R)-, (S)-fluoxetine and (R)-, (S)-norfluoxetine in
ovine plasma using liquid chromatography with tandem mass
spectrometry (LC/MS/MS). J Chromatogr B Analyt Technol Biomed Life
Sci. 879:349–358. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Nordberg H, Jerndal G and Thompson RA:
Direct injection of lipophilic compounds in the organic phase from
liquid-liquid extracted plasma samples onto a reversed-phase
column. Bioanalysis. 3:1963–1973. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Haam SJ, Park IK, Paik HC, et al: T-stage
of non-small cell lung cancer directly invading an adjacent lobe.
Eur J Cardiothorac Surg. 42:807–810. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Torii I, Hashimoto M, Terada T, et al:
Well-differentiated papillary mesothelioma with invasion to the
chest wall. Lung Cancer. 67:244–247. 2010. View Article : Google Scholar : PubMed/NCBI
|