Introduction
The role of fine-needle aspiration biopsy (FNAB) has
been under debate worldwide in recent years (1–5).
Needle biopsy has gained popularity and is now considered the
standard biopsy method (1,2). On the other hand, it has been
discussed that the increase in the number of needle biopsies is not
entirely the result of evidence-based reasons (3). There are some advantages of FNAB over
needle biopsy for breast lesions: i) it is widely available, easy,
quick and inexpensive; ii) it is associated with a lower risk of
complications; and iii) it may be appropriate for small lesions,
symptomatic (palpable) lesions and for confirming benign lesions
(1,3,6–8).
Although there is an increasing need for preoperative evaluation of
hormone receptors and human epidermal growth factor receptor 2
(HER2/neu) status (1), a number of
physicians select needle biopsy in order to avoid obtaining
inadequate and/or indeterminate results from cytological analyses
(3–5). As a result, the cytological
examination has been omitted.
Although FNAB is highly accurate, it is not 100%
accurate (9–15). This point has not been well
understood, especially by the general public. However, breast
cancer screening is becoming more widespread, and the number of
people receiving breast examinations has been increasing. Under
these recent circumstances, it is considered that FNAB is an
appropriate diagnostic tool (6,7).
The Working Group on the Accuracy of Breast
Fine-Needle Aspiration Cytology of the Japanese Society of Clinical
Cytology was assembled to assess the current status of breast
cytology in Japan by conducting a large-scale survey on the
accuracy of breast FNAB in 12 cooperating facilities in Japan. Data
on 1,250 of the 10,890 subjects in the present study were presented
in our previous study as a pilot project (15). However, they were included in this
current study to provide a larger sample size, and the data were
re-analyzed as part of the larger sample. In the present study, we
analyzed and compared the data for cytological diagnosis and
individual variables at these 12 facilities, and further
investigated the discrepant cases between the cytological and
histological diagnoses (false-negative and false-positive cases).
It is considered that these data are important for doctors,
patients and those in medicolegal circles.
Patients and methods
We conducted a survey in 12 facilities in order to
determine the accuracy of breast FNAB. After reviewing the data
from 2009 and the preceding years, we conducted a survey over
several years (1–7 years, average 4.3 years) at 12 facilities that
dealt with a large number patients. Data were collected from each
institution or region, and cytological data confirmed by
histological findings obtained after surgery or needle biopsy were
included in the study. The manner of data collection is shown in
Table I (modified from our previous
report with permission) (15).
 | Table ICollection of data for assessing the
diagnostic accuracy of fine-needle aspiration biopsy of the
breast. |
Table I
Collection of data for assessing the
diagnostic accuracy of fine-needle aspiration biopsy of the
breast.
|
Cytologicalcategory | (No.) | Histology B3
(10,890) |
|---|
|
|---|
| By operation B1
(8,953) | By needle biopsy B2
(1,937) |
|---|
|
|
|---|
| Other than
malignancy | Malignancy | Other than
malignancy | Malignancy |
|---|
| Inadequate | A1 (5,465) | C1 (153) | D1 (403) | E1 (361) | F1 (52) |
| Normal or benign | A2 (14,538) | C2 (750) | D2 (269) | E2 (796) | F2 (32) |
| Indeterminate | A3 (2,068) | C3 (352) | D3 (673) | E3 (241) | F3 (58) |
| Malignancy
suspected | A4 (1,146) | C4 (54) | D4 (695) | E4 (24) | F4 (70) |
| Malignancy | A5 (7,138) | C5 (35) | D5 (5,569) | E5 (9) | F5 (294) |
| Total | A6 (30,535) | C6 (1,344) | D6 (7,609) | E6 (1,431) | F6 (506) |
Classification of cytological
samples
In accordance with the General Rules for Clinical
and Pathological Recording of Breast Cancer prepared by the
Japanese Breast Cancer Society in 2005 (16), individual cytological samples were
initially rated as ‘inadequate’ or ‘adequate’. Samples rated as
‘adequate’ were graded on a four-category scale (16): ‘normal/benign’; ‘indeterminate’
(difficult to distinguish between ‘benign’ and ‘malignant’);
‘malignancy suspected’; and ‘malignant’. Generally, the cytological
diagnostic procedure in Japan involves cytotechnologists initially
screening the samples (usually marking the findings on the slides),
and then the consultant pathologists making a diagnosis based on
the results. The samples are generally extracted from the patients
by surgeons, although sometimes by radiologists and/or general
practitioners.
Definitions of variables analyzed and
calculations of their diagnostic accuracy
Several terms used in this study merit precise
definition and are listed in Table
II (modified from Table II in
our previous report with permission) (15). These terms are: inadequate value;
indeterminate value; positive predictive value of ‘malignancy
suspected’ results; absolute sensitivity; complete sensitivity;
specificity; positive predictive value of ‘malignant’ cells;
negative predictive value of ‘normal or benign’ cells;
false-negative value; false-positive value, and accuracy rate. The
methods for calculating the diagnostic accuracy of these values
based on the recorded columns in Table
I are also shown in Table II
(15). The combined data for this
survey from each institution/region were simply added together. One
of the 12 ‘institutions’ in the present study was a region; data
were collected and combined from 7 local institutions within the
same geographical region, as we previously reported (15). For simplicity and based on the
arrangement of the working group conducting this study, these data
were considered as representative of 1 ‘institution’ together with
data from 11 other institutions. Thus, data obtained from 1,250
histologically confirmed cases from among 5,693 cytologically
diagnosed cases from the institutions/regions previously described
are included in the present study.
 | Table IIDefinitions of quality-assurance
parameters for cytological examination and calculations of
diagnostic accuracy [modified from Table II (15) with permission]. |
Table II
Definitions of quality-assurance
parameters for cytological examination and calculations of
diagnostic accuracy [modified from Table II (15) with permission].
| Term | Definition and
calculation |
|---|
| Inadequate
value | Percentage of cases
whose samples were rated as ‘inadequate’ among all cases who
underwent cell sampling
(A1/A6) ×100 |
| Indeterminate
value | Percentage of cases
rated as ‘indeterminate’ among all cases of ‘adequate’ samples
(cases of ‘inadequate’ samples subtracted from all cases having
received cell sampling)
{A3/(A6−A1)} ×100 |
| Positive predictive
value of ‘malignancy suspected’ results | Percentage of cases
other than false-positive cases among all cases cytologically rated
as ‘malignancy suspected’
[{A4− (C4+E4)}/A4] ×100 |
| Absolute
sensitivity | Percentage of cases
cytologically rated as ‘malignant’ (and confirmed as malignant by
histology) among all cases of ‘adequate’ samples histologically
rated as ‘malignant’
[(D5+F5)/{(D6+F6)−(D1+F1)}] ×100 |
| Complete
sensitivity | Percentage of cases
cytologically rated as ‘indeterminate’, ‘malignancy suspected’ or
‘malignant’ among all cases of ‘adequate’ samples histologically
rated as ‘malignant’
[{(D3+F3)+(D4+F4)+(D5+F5)}/{(D6+F6)−(D1+F1)}] ×100 |
| Specificity | Percentage of cases
cytologically rated as ‘normal or benign’ among all cases of
‘adequate’ samples histologically rated as
‘non-malignant’
[{A2− (D2+F2)}/[(A6−A1)−{(D6+F6)−(D1+F1)}]] ×100 |
| Positive predictive
value of ‘malignant’ cells | Percentage of cases
other than false-positive cases among all cases cytologically rated
as ‘malignant’
[{A5− (C5+E5)}/A5] ×100 |
| Negative predictive
value of ‘normal or benign’ cells | Percentage of cases
other than false-negative cases among all cases cytologically rated
as ‘normal or benign’
{A2− (D2+F2)}/A2} ×100 |
| False-negative
value | Percentage of
cytologically negative cases among all cases of ‘adequate’ samples
histologically rated as ‘malignant’
[(D2+F2)/{(D6+F6)−(D1+F1)}] ×100 |
| False-positive
value | Percentage of
cytologically positive cases among all cases of ‘adequate’ samples
histologically rated as ‘non-malignant’
[(C5+E5)/[(A6−A1)−{(D6+F6)−(D1+F1)}]] ×100 |
| Accuracy | Percentage of cases
cytologically rated as ‘normal or benign’ and confirmed as benign
by histology and cases cytologically rated as ‘indeterminate’,
‘malignancy suspected’ or ‘malignant’ and confirmed as malignant by
histology among all cases of ‘adequate’ samples histologically
rated as ‘non-malignant’ and ‘malignant’
[{A2− (D2+F2)+(D3+F3)+(D4+F4)+(D5+F5)}/(A6−A1)] ×100 |
Analyses of cases showing discrepancies
between the cytological and histological diagnoses
Any cases showing discrepancies (i.e.,
false-negative and false-positive) between the cytological and
histological diagnoses were re-analyzed based on histological type,
clinical information and tumor size. In addition, these cases were
re-evaluated and re-categorized in order to determine the possible
reasons for the discrepancies.
Results
Data from the survey
The data obtained from the individual
facilities/regions were summed and calculated. In total, data from
30,535 cases were collected. The cytological diagnosis was
established by histological means in 10,890 (35.7%) of these cases,
and this formed the basis for determining the diagnostic accuracy
(Table I).
The data were as follows: inadequate rate, 17.7%;
indeterminate rate, 7.8%; positive predictive value of ‘malignancy
suspected’ cells, 92.4%; absolute sensitivity, 76.7%; complete
sensitivity, 96.7%; specificity, 84.3%; negative predictive value
of ‘normal/benign’ cells, 98.2%; positive predictive value of
‘malignant’ cells, 99.5%; false-negative value, 3.31%; and
false-positive value, 0.25%. The accuracy rate of breast FNAB was
88.0%.
Analyses of cases showing discrepancies
between the cytological and histological diagnoses
False-negative cases
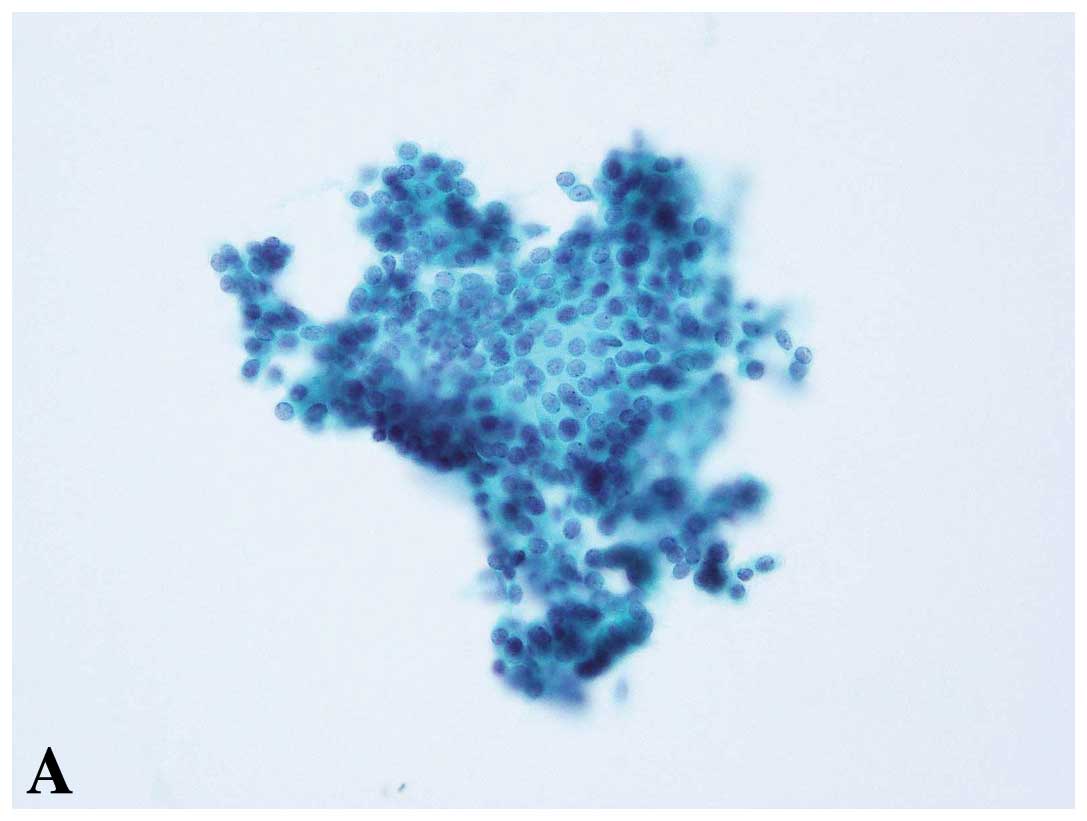
There were 301 false-negative cases. Four cases
could not be evaluated because we were unable to collect the
slides, and therefore a total of 297 cases from the institutions
were re-evaluated (including 52 cases from the previous study
(17). Histologically, these 297
cases consisted of 94 cases (31.6%) of invasive ductal carcinoma
(IDC), scirrhous-growing type (SIDC) (16) (Figs.
1 and 2), 70 cases (23.7%) of
ductal carcinoma in situ (DCIS), 40 cases (13.5%) of IDC,
papillotubular type (16), 22 cases
(7.4%) of IDC, solid-tubular type (16), 14 cases (4.7%) of invasive lobular
carcinoma and several other cases (57 cases including special
types, i.e., mucinous carcinoma, apocrine carcinoma).
Regarding the clinical information, there were 59
cases of breast cancer (palpable) (19.9%), 47 of breast tumor
(palpable, but uncertain if benign or malignant) (15.8%), 38
lacking information (12.8%) and 30 with abnormal image findings
(mammography and/or ultrasound (US) BI-RADS >3b including
non-palpable lesions) (10.1%). In addition, the false-negative rate
for cases with a tumor size or hypoechoic area of US ≤1 cm was
15.5% (46/297).
After re-evaluating the false-negative cases, the
classifications were 20 ‘inadequate’ (6.7%), 212 ‘normal/benign’
(71.4%), 49 ‘indeterminate’ (16.5%), 11 ‘malignancy suspected’
(3.7%) and 5 ‘malignant’ cases (1.7%).
The reasons for re-categorization into a new
category upon re-evaluation were as follows: ‘inadequate’ (the
small number and/or poor quality of cells made re-evaluation
difficult in this category), there were only small clusters in 14
of 20 cases (70%), and the other 6 cases (30%) were composed of
small clusters with drying or degeneration; ‘normal/benign’, 175 of
212 cases (82.5%) were benign and/or normal epithelial cells (not
atypical, benign small clusters with myoepithelial cells), and the
other 37 cases (17.5%) showed other benign findings (i.e., apocrine
metaplasia, foamy cells, fat cells, fibroadenoma-like findings);
‘indeterminate’, 19 of 50 cases (38.8%) had clusters of unclear
myoepithelial cells, 9 cases (18.7%) with a small number of
atypical cells presented in the specimens, 5 cases (10.2%) showed
abundant papillary clusters, 3 cases (6.1%) showed mild atypia and
14 cases (26.6%) were for other reasons; ‘malignancy suspected’, in
2 of the 11 cases (18.2%), atypical cells were present but unclear,
atypical cells were present in clusters of unclear myoepithelial
cells, and a small number of small atypical cells were present, and
7 cases (45.5%) were for other reasons (i.e., the presence of
abundant cells, atypical apocrine cells); ‘malignancy’,
myoepithelial cells were absent in clusters in 3 of 5 cases (60%),
1 case (20%) showed cribriform structures and 1 case (20%) had
isolated atypical cells.
False-positive cases
There were 26 false-positive cases. Three cases
could not be evaluated because we were unable to collect the
slides; therefore, a total of 22 cases were re-evaluated among all
the institutions (including 3 cases from the previous study)
(17). Histologically, there were 3
cases each (13.4%) of papilloma, fibroadenoma, fibrocystic disease
(mastopathy) and adenomyoepithelioma (Figs. 3 and 4) and 10 (43.5%) other cases (i.e., ductal
adenoma, epidermal cyst).
Regarding the clinical information, the cases
included 6 with abnormal image findings (mammography and/or US
BI-RADS >3b with non-palpable lesions) (26.1%), 5 with breast
cancer (palpable) (21.7%), 5 with breast tumor (palpable, but
uncertainty whether benign or malignant) (21.7%), 2 with breast
cancer, suspected (8.7%) and 5 others (21.7%). In addition, the
false-positive rate for cases with a tumor size or hypoechoic area
of US >2 cm was 39.1% (9/23).
After re-evaluating the false-positive cases, the
classifications were 9 ‘normal/benign’ cases (39.1%), 10
‘indeterminate’ cases (43.5%) and 4 ‘malignancy suspected’ cases
(17.4%). There were no re-classifications as ‘inadequate’ and
‘malignant’ cases.
The findings (reasons) for re-categorization into a
new category upon re-evaluation were as follows: ‘normal/benign’, 4
of 9 cases (44.4%) showed that there were clusters with some
myoepithelial cells, 2 cases (22.2%) showed degenerative chromatic
nuclear cells, 2 cases were re-categorized as ‘normal/benign’
because the background of degenerative findings was similar to
necrotic findings and in 1 case the investigator had insufficient
experience in breast disease (normal ductal cells were present);
‘indeterminate’, 4 of 10 cases (40%) showed clusters of unclear
myoepithelial cells and loose connections between cells, 2 cases
each (20%) showed loosely connected papillary lesions and
cribriform-like structures, 1 case showed atypical apocrine cells,
and 1 case showed hyperplastic cells; ‘malignancy suspected’ in
each of the 4 cases (25%), necrosis-like findings, atypical
apocrine cells, a small number of intracytoplasmic lumina, and
low-grade DCIS-like cells were observed. In total, there were no
high-grade cells in the false-positive cases.
Discussion
To the best of our knowledge, this survey collected
data from the largest number of cases for breast FNAB. The role of
FNAB has been debated recently, as to whether FNAB has been
replaced with the core needle (or vacuum) biopsy (2). Consequently, many reports concerning
the accuracy of breast cytology have already been discussed, mainly
in the 1990s (9–12,18).
Although there are a number of advantages and disadvantages for
both needle biopsy and FNAB (1),
usage of FNAB has been decreasing in many countries over the last
decade. Based on our preliminary survey, a number of hospitals and
clinics in Japan primarily use FNAB (unpublished data). Therefore,
the current accuracy rate of FNAB must be determined and doctors,
paramedics, patients as well as medical lawyers must be informed. A
cytological examination for cancer screening in Japan and in
several other countries continues to play an important role. For
example, cytological examination plays a major role in the
diagnosis of both palpable and non-palpable breast masses in Egypt
because it is cost-effective (7).
We compared the combined data from the present study
with previously reported data (9–14). The
data from the present study were compared with the goals of
assessment of diagnostic accuracy reported in the UK (9,10)
(absolute sensitivity, >60%; complete sensitivity, >80%;
specificity, >60%; positive predictive value, >95%;
false-negative rate, <5%; false-positive rate, <1%;
inadequate rate, <25%; suspicious rate, <20%), and were also
compared with studies from several other countries, including a
large-scale study (10,571 cases) in North America (the values had
ranges of 75.8–98.7% for sensitivity, 92–100% for specificity,
0.6–2.5% for false-positive rate and 2.5–17.9% for false-negative
rate) (11,14). The combined data in our survey were
within these ranges, except for the specificity (84.3%) and the
inadequate rate (17.7%) which were >10% (the goal of the
Japanese criteria) (16).
Therefore, the accuracy of a cytological examination in our survey
is considered to be generally useful during clinical practice.
However, we previously reported several differences among the
institutions in our regions (15),
discussed some of the reasons for this situation and speculated on
the poor communication between the clinical and pathological sides
(15). The present study provided
improved results in many categories compared to the previous pilot
study. Likely reasons for this are that the previous study included
general hospitals and a reference laboratory (15), whereas the present study included
mainly larger university-level hospitals with specialized breast
disease departments.
In addition, we investigated the data with a focus
on false-negative and false-positive cases. These values are
important because if cases were omitted by the histological
diagnosis, false-positive cases would proceed directly to breast
cancer surgery, while many false-negative cases may not be
re-evaluated. Such results may lead to legal issues. The present
data showed that FNAB was not 100% accurate, and doctors, patients
as well as lawyers should be informed. On the other hand, the
accuracy of a cytological examination used in order for a diagnosis
to be made, was relatively high.
Regarding the 297 false-negative cases, the
histology of most cases was SIDC, followed by DCIS. These types of
carcinoma are usually detected as small atypical cells in the
cytological diagnosis (17,18). After re-evaluation of the 297
false-negative cases, the category of over 90% cases was changed to
‘inadequate’, ‘normal/benign’ or ‘indeterminate’. However, the
category of 11 other cases was changed to ‘malignancy suspected’
and that of 5 cases was changed to ‘malignancy’. These histological
types were mainly SIDC (data not shown; 5/11 cases and 2/5 cases,
respectively) and the cells of small clusters showed mild atypia.
SIDC showed a tendency toward inadequate and false-negative
cytological findings based on their histological characteristics;
that is, they are usually accompanied by thick fibrous tissues
(18). Therefore, when this
histological subtype which is simple to detect by imaging (i.e.,
mammography or ultrasound) is detected, needle biopsy rather than
FNAB is recommended (17). Due to
the lack of communication between the clinical and pathological
sides, clinical information for 15% of the cases was not available
(15). The rate of tumor size under
1 cm was approximately 15%; thus, the small tumor size was not the
main reason for the false-negative cases.
Regarding false-positive cases, the histology of
most cases was papilloma, fibroadenoma, fibrocystic disease
(mastopathy) or adenomyoepithelioma. Papilloma is one of the most
difficult histologic types for cytological diagnosis (19), while fibroadenoma is often misread
for hyperplastic epithelial cells (20). Interestingly, adenomyoepithelioma
showed the same rate of false-positive cases, for which the
histological type has benign to malignant potential and is
generally considered a benign lesion (21). This type is comparatively rare, and
the frequency of encountering this type is low. However, we should
be aware of the type, and of the characteristic cytological
findings, including a loosely cohesive fragment of ductal
epithelium surrounding fibrous and myxomatous cores (22). The clinical information was based
mainly on the abnormal image findings and breast cancer (tumor),
and therefore, most cases were suspected of malignancy on the
clinical side. The pathological side may have been affected by this
information. The rate of tumor size over 2 cm was more than
one-third, and thus the tumor size was often larger. Even after
re-evaluation, the category of 4 of 23 cases was still ‘malignancy
suspected’, and these signs were similar to malignant findings.
Knowledge of these findings (i.e., apocrine atypia) must be taken
into consideration when making a diagnosis.
Although the present study did not always coincide
with imaging data, a triple approach (clinical, pathological and
radiological) is necessary for accuracy (1,3).
Moreover, when the suspected histological type displays
difficulties in cytological diagnosis using imaging analysis, which
method to select (needle biopsy or cytological examination) depends
on individual cases (1). In the
future, FNAB should be used for non-palpable lesions with imaging
guidance (especially ultrasound) (3).
In conclusion, the accuracy of a cytological
examination in Japan is as high as those in other countries, such
as the UK and US (9–11). On the other hand, the study
demonstrated that there were some false-negative and false-positive
cases. Japan may also need to establish a national quality
assurance program, similar to the one in the UK (9,10).
Furthermore, we must continue to improve accuracy based on the
learning points in the present study, so that FNAB can become a
more useful and reliable examination in clinical practice.
Acknowledgements
The authors are indebted to the pathologists and all
staff members of the facilities who cooperated in this study. The
present study was carried out by the Working Group on the Accuracy
of Breast Fine-Needle Aspiration Cytology of the Japanese Society
of Clinical Cytology, and the authors would like to thank the
members of this highly respected society for their assistance in
conducting the study.
References
|
1
|
Tse GM and Tan PH: Diagnosing breast
lesions by fine needle aspiration cytology or core biopsy: which is
better? Breast Cancer Res Treat. 123:1–8. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lieske B, Ravichandran D and Wright D:
Role of fine-needle aspiration cytology and core biopsy in the
preoperative diagnosis of screen-detected breast carcinoma. Br J
Cancer. 95:62–66. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kocjan G, Bourgain C, Fassina A, et al:
The role of breast FNAC in diagnosis and clinical management: a
survey of current practice. Cytopathology. 19:271–278. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kocjan G: Fine needle aspiration cytology
(Review). Cytopathology. 14:307–308. 2003. View Article : Google Scholar
|
|
5
|
Levine T: Breast cytology: is there still
a role? Cytopathology. 15:293–296. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Nasuti JF, Gupta PK and Baloch ZW:
Diagnostic value and cost-effectiveness of on-site evaluation of
fine-needle aspiration specimens: review of 5,688 cases. Diagn
Cytopathol. 27:1–4. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Abdel-Hadi M, Abdel-Hamid GF, Abdel-Razek
N and Fawzy RK: Should fine-needle aspiration cytology be the first
choice diagnostic modality for assessment of all nonpalpable breast
lesions? The experience of a breast cancer screening center in
Alexandria, Egypt. Diagn Cytopathol. 38:880–889. 2010. View Article : Google Scholar
|
|
8
|
Rosa M, Mohammadi A and Masood S: The
value of fine needle aspiration biopsy in the diagnosis and
prognostic assessment of palpable breast lesions. Diagn Cytopathol.
40:26–34. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wells CA, Ellis IO, Zakhour HD and Wilson
AR: Guidelines for cytology procedures and reporting on fine needle
aspirates of the breast. Cytology Subgroup of the National
Coordinating Committee for Breast Cancer Screening Pathology.
Cytopathology. 5:316–334. 1994. View Article : Google Scholar
|
|
10
|
Singh N and Wells A: Assessment of
accuracy in breast cytology. Cytopathology. 12:211–218. 2001.
View Article : Google Scholar
|
|
11
|
Zarbo RJ, Howanitz PJ and Bachner P:
Interinstitutional comparison of performance in breast fine-needle
aspiration cytology. A Q-probe quality indicator study. Arch Pathol
Lab Med. 115:743–750. 1991.PubMed/NCBI
|
|
12
|
Ciatto S, Bonardi R and Cariaggi MP:
Performance of fine-needle aspiration cytology of the
breast-multicenter study of 23,063 aspirates in ten Italian
laboratories. Tumori. 81:13–17. 1995.PubMed/NCBI
|
|
13
|
Akiu N, Endoh M, Isawa M, et al: Fine
needle aspiration cytology of the breast-review of 15470 cases. J
Jpn Soc Clin Cytol. 46:323–331. 2007.(In Japanese, abstract in
English).
|
|
14
|
Zagorianakou P, Fiaccavento S,
Zagorianakou N, Makrydimas G, Stefanou D and Agnantis NJ: FNAC: its
role, limitations and perspective in the preoperative diagnosis of
breast cancer (Review). Eur J Gynaecol Oncol. 26:143–149.
2005.PubMed/NCBI
|
|
15
|
Yamaguchi R, Tsuchiya S, Koshikawa T, et
al: Comparison of the accuracy of breast cytological diagnosis at
seven institutions in southern Fukuoka Prefecture, Japan. Jpn J
Clin Oncol. 42:21–28. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sakamoto G, Inaji H, Akiyama F, et al:
General rules for clinical and pathological recording of breast
cancer. Breast Cancer. 12(Suppl): S10–S11. 2005.PubMed/NCBI
|
|
17
|
Yamaguchi R, Tsuchiya S, Koshikawa T, et
al: Evaluation of inadequate, indeterminate, false-negative and
false-positive cases in cytological examination for breast cancer
according to histological type. Diagn Pathol. 7:532012. View Article : Google Scholar
|
|
18
|
Park IA and Ham EK: Fine needle aspiration
cytology of palpable breast lesions. Histologic subtype in false
negative cases. Acta Cytol. 41:1131–1138. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Field A and Mak A: A prospective study of
the diagnostic accuracy of cytological criteria in the FNAB
diagnosis of breast papillomas. Diagn Cytopathol. 35:465–475. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yamaguchi R, Tanaka M, Yokoyama T, et al:
Cytological features of myxomatous fibroadenoma of the breast.
Diagn Cytopathol. 40:316–320. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Tavassoli FA and Soares J: Myoepithelial
lesions. Pathology and Genetics of Tumours of the Breast and Female
Genital Organs. World Health Organization Classfication of Tumours.
Tavassoli FA and Devilli P: IARC Press; Lyon: pp. 86–88. 2003
|
|
22
|
Ali SZ and Parwani AV: Benign and
borderline lesions. Breast Cytopathology. Rosenthal DL: Springer;
New York: pp. 57–84. 2007
|