Introduction
CD9, a 24- to 27-kDa cell surface glycoprotein, is a
member of the tetraspanin superfamily. It is expressed in numerous
normal tissues and plays a critical role in various types of cell
processes, such as cell adhesion, motility and various signaling
pathways involving integrins. In malignancies, its expression
usually suppresses tumor progression and metastasis by inhibition
of tumor proliferation and survival (1,2).
Although converse functions have also been reported in certain
tumors, downregulation of CD9 correlates well with tumor
progression or metastasis in bladder, breast, lung and colon
cancers (2). An in vivo
study using administration of the CD9 antibody to mice bearing
human gastric cancer xenografts showed inhibition of tumor
progression via anti-proliferative, pro-apoptotic and
anti-angiogenetic effects (3),
suggesting its potential for the molecular-targeted therapy of
human malignancies. Moreover, we previously identified CD9, along
with side population, CD24 and CD26 cells, as a cancer stem cell
marker of mesothelioma, thus demonstrating its potential for cancer
stem cell-targeted therapy in the future (4).
Malignant mesothelioma is an aggressive cancer with
few patients surviving beyond 2 years following diagnosis. The
median survival of patients without any treatment barely exceeds 1
year. A large population-based study reported 6-month, 1-year and
5-year overall survival rates of 55, 33 and 5% in mesothelioma
(5). In Japanese patients, the
median survival of mesothelioma has been reported to be 9–10 months
from the date of diagnosis (6,7).
The clinical predictors for poor survival in
patients with mesothelioma are reported to include sarcomatoid
histology, older age, advanced IMIG stage, patients without
palliative surgery or chemotherapy. Other biological prognostic
factors such as serum and tumor EGFR expression (8,9),
pleural effusion VEGF level (10),
angiopoietin-1 expression (11),
ER-β expression (12), methylation
profile (6,13) and miRNA signatures (14) have also been reported. In this
study, we identified CD9 as an independent predictor of survival,
and loss of expression showed biological aggressive behavior in
mesothelioma cells.
Materials and methods
Cell line
Mesothelioma cell lines, MSTO-211H [derived from
biphasic mesothelioma (BM)] and TUM1 (4) were maintained in RPMI-1640 medium
(Gibco-BRL; Invitrogen Life Technologies, Grand Island, NY)
supplemented with 10% fetal calf serum (FCS), 100 U/ml penicillin
and 100 mg/ml streptomycin. The cells were maintained as monolayers
in 10-cm diameter cell culture dish at 37°C in a humidified
atmosphere of 5% CO2 in air.
shRNA lentiviral transfection
CD9-targeted shRNA lentiviral plasmid (Mission;
Sigma-Aldrich, target sequence: ccgggctg
ttcggatttaacttcatctcgagatgaagttaaatccgaacagctttttg) and
non-targeting control plasmid (pLKO.1-puro) were transfected with
ViraPower™ Lentiviral packaging mix to cell lines using
Lipofectamine 2000 (Invitrogen Life Technologies). The cells were
transfected with the shRNA-expressing lentivirus, and stable cell
lines were generated by selection with puromycin. Knockdown of CD9
was confirmed by FACS analysis with the FITC mouse anti-human CD9
antibody (BD Pharmingen).
In vitro migration assay
Migration assay was performed using a 24-well Boyden
chamber with a non-coated 8-mm pore size filter in the insert
chamber (BD BioCoat). Cells (CD9 shRNA- and control
shRNA-transfected MSTO-211H) (5×104) were suspended in
0.5 ml RPMI-1640 media containing 0.1% FCS and seeded into the
insert chamber. Cells were allowed to migrate for 48 h into the
bottom chamber containing 1 ml of RPMI-1640 media containing 10%
FCS in a humidified incubator at 37°C in 5% CO2.
Migrated cells which had attached to the outside of the filter were
visualized by staining with Diff-Quik (International Reagents Co.)
and counted.
Patients and tissue specimens
One hundred and twelve cases of malignant pleural
mesothelioma were retrieved from the archival pathology files of
the Department of Pathology, Graduate School of Biomedical
Sciences, Hiroshima University. Small biopsy specimens were not
included in this study. The histological diagnosis of mesothelioma
was previously carried out by three independent pathologists
(V.J.A., Y.T., K.I.) based on WHO criteria (15) and were confirmed in all instances by
clinical, histological and immunohistochemical findings.
Epithelioid mesothelioma (EM) was further subdivided into two
subtypes, i.e., ‘differentiated’ type (EM-D) and
‘less-differentiated’ type (EM-LD) based on the morphology of
‘papillo-tubular structures’ as an indicator of differentiation
(16). Thus, EM-D were EMs showing
a papillo-tubular pattern, micropapillary pattern and/or
microcystic pattern and EM-LD were EMs showing solid nest,
trabecular pattern, signet-ring cell-like appearance and/or
single-cell infiltration pattern. The histological classification
was carried out prior to this study. The clinical data of the
patients were retrieved from the hospital records. This study was
carried out in accordance with the Ethical Guidelines for
Epidemiological Research enacted by the Japanese Government as
tissue specimens were collected and carried out strictly to protect
personal identity after approval by the Institutional Review Board
at Hiroshima University.
Immunohistochemistry
Immunohistochemical stainings were performed on 3-μm
paraffin sections using the monoclonal anti-CD9 antibody. Tissue
sections were deparaffinized, hydrated and endogeneous peroxide was
quenched using 0.3% hydrogen peroxidase for 30 min. Sections were
incubated in a humidified chamber with mouse monoclonal anti-CD9
antibody (diluted 1:100, clone 72F6, NB110–41534; Novus
Biologicals, Littleton, CO, USA) overnight at 4°C. The reaction was
visualized using the Histofine Simple Stain kit (Nichirei
Biosciences, Tokyo, Japan) with diaminobenzidine as a chromogen and
nuclear counterstaining with Mayer’s hematoxylin. A similar
immunohistochemical procedure was carried out with the omission of
the primary antibody as a negative control. Endothelial cells in
and around the tumor tissue were considered as the internal
positive control for validation of the immunohistochemistry. The
membranous staining of CD9 was scored as 0, no staining;
1+, 1–10%; 2+, 10–50%; and 3+,
>50% of tumor cells immunostained in the tissue sections.
Immunohistochemical scoring was carried out by two pathologists
(V.J.A., Y.T.) independently without knowledge of the
clinicopathologic or disease outcome variables. Multiple sections
from different paraffin blocks were analyzed for CD9 expression to
confirm negative CD9 expression.
Statistical analysis
Fisher’s exact test and Pearson Chi-square test were
used for association analyses of the clinicopathological parameters
with CD9 expression. Univariate analysis and multivariate Cox
proportional hazards regression analysis for overall survival were
performed with the regressors: CD9 expression, age, gender,
clinical IMIG staging, histological type, differentiation,
therapeutic regimen, extrapleural pneumonectomy and chemotherapy
status. The overall survival curves of patients with follow-up data
were estimated using the Kaplan-Meier method. Multivariate Cox
proportional hazard ratio (HR) was separately calculated for all
mesotheliomas and EMs for patients with clinical data for all
parameters including age, histology (in analyzing all
cases)/differentiation (in analyzing EM alone), IMIG staging and
therapeutic regimen. Statistical analysis of the migration assay
was performed by the two-tailed t-test. All of the statistical
analyses were carried out using JMP 9.0 software. P-value <0.05
was considered to indicate a statistically significant result.
Results
Effect of shRNA knockdown of CD9 on cell
migration
Immunohistochemical analysis of CD9 expression in
the patient mesothelioma samples indicated that CD9 expression was
a favorable prognostic factor. Therefore, we investigated the
effects of CD9 on the cell migration of mesothelioma cell lines
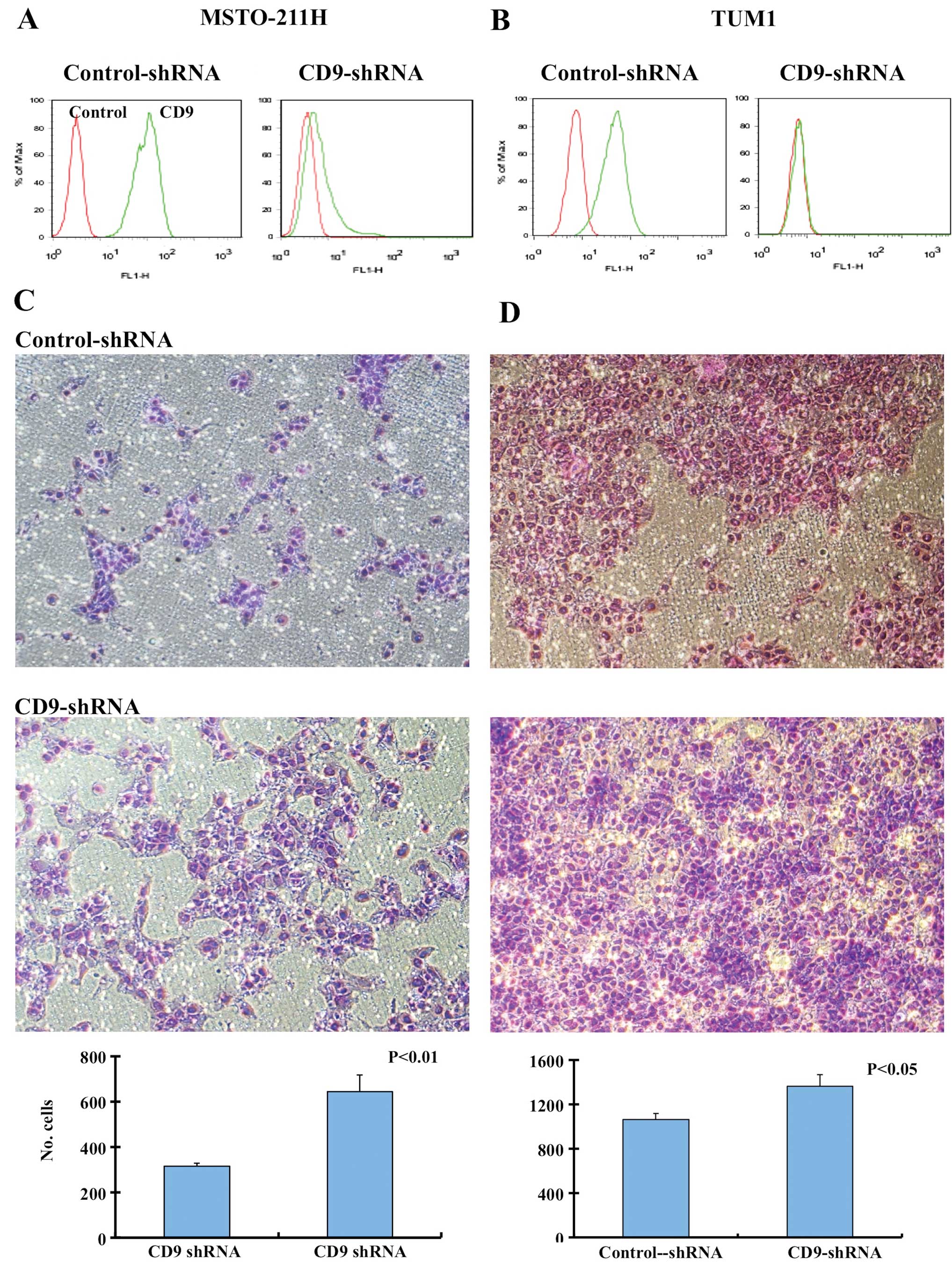
using shRNA-mediated CD9 knockdown. Knockdown of CD9 in the
MSTO-211H and TUM1 cell lines was confirmed by FACS analysis, and
the migration of cells was analyzed using Boyden chamber assay
(Fig. 1). CD9 shRNA-transfected and
control shRNA-transfected MSTO-211H and TUM1 cells were allowed to
migrate toward medium containing 10% FCS as a chemoattractant. CD9
shRNA-transfected MSTO-211H and TUM1 cells showed increased
migration in comparison to the control shRNA-transfected cells
(Fig. 1).
Clinicopathological characteristics of
the malignant mesothelioma patients
This study consisted of 103 male patients and 9
female patients (M:F ratio 11:1), with a mean age of 65.8 years
with standard deviation of 9.4 (range 42–88 years). Twenty-five
cases were IMIG stage I, 40 cases were stage II, 25 cases were
stage III and 21 cases were stage IV. Histologically, 71 cases were
EMs (63.4%) 21 cases were sarcomatoid mesothelioma (SM) (18.75%)
and 20 cases were BM (17.85%). Thirty-three cases of EM were
differentiated type (EM-D) and 38 cases were less differentiated
type (EM-LD).
Clinically, 30 patients received best supportive
care alone, 42 patients were treated with chemotherapy alone and 40
cases underwent extrapleural pneumonectomy with or without
chemotherapy and/or radiotherapy. Other surgical procedures were
not included in this study as their numbers were limited for the
analysis. The chemotherapy regimen consisted of various
combinations: pemetrexed plus cisplatin regimen (39 cases),
pemetrexed plus carboplatin (5 case), pemetrexed-containing regimen
(44 cases) and chemotherapy regimens without pemetrexed (30 cases).
The mean follow-up period was 16.5 months (from 10 days to 79
months) with 94 patients succumbing to the disease and 18 alive
with disease at the time of the study.
CD9 expression in malignant
mesothelioma
Positive immunoreactivity for CD9 was observed in
the membrane and cytoplasm of the tumor cells in 76 of 112
malignant mesothelioma cases. Histologically, CD9 immunoreactivity
was observed in 62 of 71 epithelioid mesotheliomas, 13 of 20
biphasic mesotheliomas and only 1 of 21 SMs. Among EMs, all cases
of EM-D were CD9-positive and showed higher immunohistochemical
score for CD9 expression compared to cases of EM-LD (Fig. 2) (Table
I).
 | Table IImmunohistochemical scoring of CD9
expression in malignant mesothelioma. |
Table I
Immunohistochemical scoring of CD9
expression in malignant mesothelioma.
| | CD9
immunohistochemical scoring |
|---|
| |
|
|---|
| Histology | Total cases | 0 | 1 | 2 | 3 |
|---|
| Epithelioid
mesothelioma | 71 | 9 | 21 | 25 | 16 |
| Differentiated
type | 33 | 0 | 6 | 15 | 12 |
| Less-differentiated
type | 38 | 9 | 15 | 10 | 4 |
| Sarcomatoid
mesothelioma | 21 | 20 | 0 | 1 | 0 |
| Biphasic
mesothelioma | 20 | 7 | 4 | 6 | 3 |
Association between CD9 expression and
clinicopathological parameters
To determine the statistical significance of CD9
expression in malignant mesothelioma, all cases were divided into
two groups based on their CD9 expression: a CD9-positive (n=76,
67.9%) and a CD9-negative (n=36, 32.1%) group. The association
between CD9 expression and various clinicopathological parameters
is listed in Table II.
Mesothelioma patients with a younger age (P=0.0083), epithelioid
histology (P<0.0001), differentiated type EMs (P=0.0027), who
did not receive best supportive care (P=0.0469), who underwent EPP
and chemotherapy (P=0.0195) and who received chemotherapy with
inclusion of pemetrexed (P=0.0434) showed statistically
significantly a high frequency of positive CD9 expression.
 | Table IIClinicopathological characteristics of
patients with mesothelioma and its correlation with CD9
expression. |
Table II
Clinicopathological characteristics of
patients with mesothelioma and its correlation with CD9
expression.
| CD9 expression | |
|---|
|
| |
|---|
| Clinicopathological
parameters | Total cases | Positive | Negative | P-valueb |
|---|
| Age (years) |
| <60 | 35 | 30 | 5 | 0.0083 |
| ≥60 | 77 | 46 | 31 | |
| Gender |
| Male | 103 | 68 | 35 | 0.2672 |
| Female | 9 | 8 | 1 | |
| IMIG staging |
| Stage I/II | 65 | 48 | 17 | 0.1511 |
| Stage III/IV | 47 | 28 | 19 | |
| Histology |
| Epithelioid | 71 | 62 | 9 | <0.0001c |
| Sarcomatoid | 21 | 1 | 20 | |
| Biphasic | 20 | 13 | 7 | |
| Differentiation in
epithelioid mesothelioma |
|
Differentiated | 33 | 33 | 0 | 0.0027 |
|
Less-differentiated | 38 | 29 | 9 | |
| Therapeutic
regimen |
| BSC alone | 30 | 18 | 12 | 0.0469c |
| Chemotherapy
alone | 42 | 25 | 17 | |
| Extrapleural
pneumonectomya | 40 | 33 | 7 | |
| Extrapleural
pneumonectomy (EPP) |
| With EPP | 40 | 33 | 7 | 0.0195 |
| No EPP | 72 | 43 | 29 | |
| Chemotherapy |
| With/without EPP
and/or RT | 74 | 50 | 24 | 1.0000 |
| No
chemotherapy | 38 | 26 | 12 | |
| Chemotherapy |
| With
pemetrexed | 44 | 34 | 10 | 0.0434 |
| Without
pemetrexed | 30 | 16 | 14 | |
Association between CD9 expression and
patient prognosis
The median survival period for the CD9-positive
group was 15.1 months and that for the CD9-negative group was 9
months. The difference between the two groups was statistically
significant (Wilcoxon; P=0.0025) (Fig.
3A). However, when CD9 expression was stratified according to
immunohistochemical scores, no significant association was noted
between CD9 expression score and patient survival (Fig. 3B). The CD9-positive group showed
higher 1- and 2-year survival rates (63.2 and 25.0%) compared to
the CD9-negative group (38.9 and 11.1%) (Table III).
 | Table IIIUnivariate analysis of overall
survival in patients with malignant mesothelioma. |
Table III
Univariate analysis of overall
survival in patients with malignant mesothelioma.
| Survival in
months | | | |
|---|
|
| | | |
|---|
| Clinicopathological
parameters | Median | Mean | 1-year
survival | 2-year
survival | P-value |
|---|
| Age (years) |
| <60 | 22.0 | 25.4 | 77.1% | 42.9% | 0.0003 |
| ≥60 | 11.6 | 12.6 | 45.5% | 10.4% | |
| Gender |
| Male | 12.2 | 15.3 | 53.4% | 18.5% | 0.0789 |
| Female | 18.0 | 30.7 | 77.8% | 44.4% | |
| IMIG staging |
| Stage I/II | 16.3 | 20.4 | 63.5% | 30.2% | 0.0001 |
| Stage III/IV | 9.0 | 9.9 | 42.4% | 3.1% | |
| Histology |
| Epithelioid | 15.7 | 19.5 | 63.4% | 29.6% | <0.0001 |
| Sarcomatoid | 3.8 | 6.1 | 14.3% | 0% | |
| Biphasic | 13.9 | 17.0 | 70.0% | 10.0% | |
| Differentiation in
epithelioid mesothelioma |
|
Differentiated | 18.6 | 22.5 | 72.7% | 36.4% | 0.0301 |
| Less
differentiated | 14.0 | 17.0 | 55.3% | 23.7% | |
| Therapeutic
regimen |
| BSC | 5.1 | 7.7 | 23.3% | 3.3% | <0.0001 |
| Chemotherapy
alone | 14.2 | 15.3 | 57.1% | 11.9% | |
| Extrapleural
pneumonectomy | 18.9 | 24.5 | 77.5% | 42.5% | |
| Extrapleural
pneumonectomy |
| With EPP | 18.9 | 24.5 | 77.5% | 42.5% | <0.0001 |
| Without EPP | 11.0 | 12.1 | 43.1% | 8.3% | |
| Chemotherapy |
| With/without EPP
and/or RT | 16.5 | 19.7 | 70.3% | 27.0% | <0.0001 |
| No
chemotherapy | 6.4 | 11.0 | 26.3% | 7.9% | |
| Chemotherapy |
| With
pemetrexed | 18.4 | 21.2 | 81.8% | 29.6% | 0.0715 |
| Without
pemetrexed | 13.4 | 17.4 | 53.3% | 23.3% | |
| CD9 expression |
| Positive | 15.1 | 18.4 | 63.2% | 25.0% | 0.0025 |
| Negative | 9 | 12.6 | 38.9% | 11.1% | |
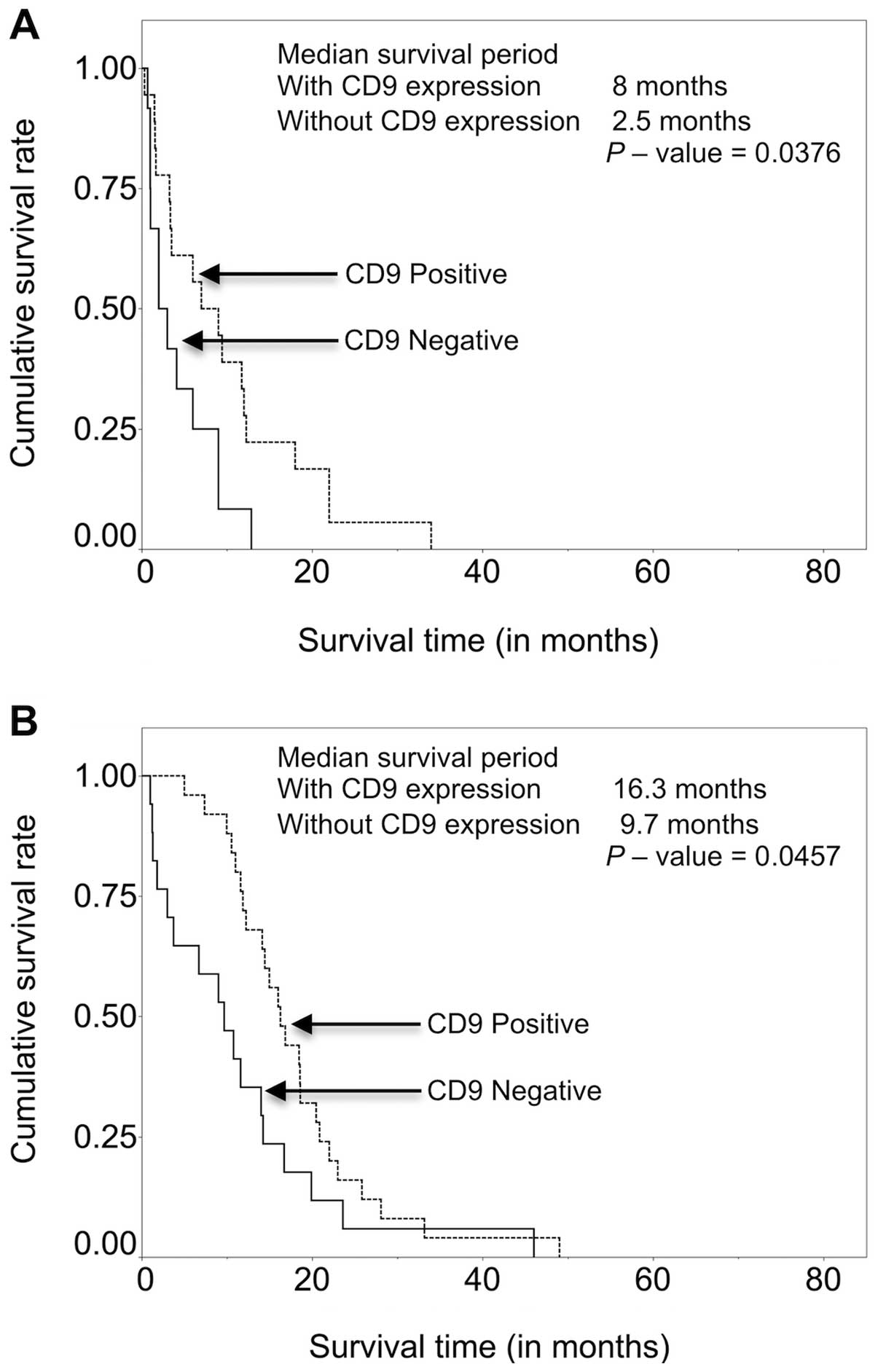
Among the patients receiving best supportive care,
patients with CD9 expression had higher survival (mean survival
time 8 months) compared to those without CD9 expression (mean
survival time, 2.5 months) (P=0.0376). A similar result was found
among the patients treated with chemotherapy alone with a mean
survival time of 16.2 months for patients with CD9 expression and
9.7 months for patients without CD9 expression (P=0.0037) (Fig. 4).
Other clinicopathological parameters that correlated
significantly with overall survival according to univariate
analysis (Table III) included age
(P=0.0003), IMIG staging (P=0.0001), histology (P<0.0001),
differentiation (P=0.0301), therapeutic regimen (P=<0.0001),
extrapleural pneumonectomy (P<0.0001) and chemotherapy
(P<0.0001). Chemotherapy with inclusion of pemetrexed showed a
tendency for better survival, but did not achieve statistical
significance (P=0.0715). Multivariate analysis using the Cox
proportional hazards model of mesothelioma patients showed loss of
CD9 expression as an independent predictor of overall survival in
patients with malignant mesothelioma with an HR 1.99 (P=0.0261) in
addition to age, IMIG staging, histology and therapeutic regimen
(Table IV). As the CD9 expression
in EMs showed a significant difference in the differentiation type,
we also analyzed multivariate analysis using Cox proportional
hazards model of 71 EMs. CD9 expression was again an independent
predictor of overall survival with an HR of 2.60 (P=0.0376) along
with other factors; age (P=0.0023) therapeutic regimens,
chemotherapy (P=0.0113) and extrapleural pneumonectomy (P=0.0014),
but not IMIG staging (P=0.1336) and differentiation (P=0.1337)
(Table V).
 | Table IVMultivariate analysis of overall
survival in malignant mesothelioma (Cox proportional hazards
model). |
Table IV
Multivariate analysis of overall
survival in malignant mesothelioma (Cox proportional hazards
model).
| | 95% confidence
interval | |
|---|
| |
| |
|---|
| Prognostic
factors | Hazard ratio | Lower | Upper | P-value |
|---|
| CD9 expression | 1.99 | 1.08 | 3.82 | 0.0261 |
| Age, 60 years or
more | 2.10 | 1.24 | 3.66 | 0.0053 |
| IMIG stage
III/IV | 2.04 | 1.23 | 3.37 | 0.0059 |
| Histology against
epithelioid mesothelioma |
| Biphasic
mesothelioma | 2.13 | 1.15 | 3.87 | 0.0171 |
| Sarcomatoid
mesothelioma | 6.65 | 2.91 | 15.22 | <0.0001 |
| Therapeutic regimen
against BSC |
| Chemotherapy
alone | 0.37 | 0.21 | 0.67 | 0.0011 |
| Extrapleural
pneumonectomy | 0.26 | 0.14 | 0.50 | <0.0001 |
 | Table VMultivariate analysis of overall
survival in epithelioid mesothelioma (Cox proportional hazards
model). |
Table V
Multivariate analysis of overall
survival in epithelioid mesothelioma (Cox proportional hazards
model).
| | 95% confidence
interval | |
|---|
| |
| |
|---|
| Prognostic
factors | Hazard ratio | Lower | Upper | P-value |
|---|
| CD9 expression | 2.60 | 1.05 | 7.37 | 0.0376 |
| Age, 60 years or
more | 2.62 | 1.40 | 5.12 | 0.0023 |
| IMIG stage
III/IV | 1.64 | 0.85 | 3.11 | 0.1336 |
| Less-differentiated
type | 1.54 | 0.87 | 2.71 | 0.1337 |
| Therapeutic regimen
against BSC |
| Chemotherapy
alone | 0.37 | 0.18 | 0.79 | 0.0113 |
| Extrapleural
pneumonectomy | 0.27 | 0.12 | 0.59 | 0.0014 |
Discussion
Disruption of cell adhesion and alteration in cell
motility play an important role in cancer cell invasion and
metastasis. The tetraspanin superfamily proteins (TM4SF) mainly
CD9, CD63, CD82, CD151 and CD81 have been implicated in cell
migration, proliferation and tumor cell metastasis (17,18).
CD9 is to date the best characterized member of the TM4SF proteins
and is involved in cell growth, adhesion and motility. Moreover,
CD9 has been recently reported as a prognostic factor in
adenocarcinoma of the lung (19),
colon (20), breast (21), pancreas (22), prostate (23) and SCC of the esophagus (24) and oral cavity (25). We found increased cell migration in
CD9-knockdown mesothelioma cell lines. In this migration assay
experiment using MSTO-211H cells, we found a decrease in CD9
expression after CD9-shRNA transfection which led to increased cell
migration compared to control-shRNA-transfected cells.
These data suggest the importance of CD9 in
determining the aggressive behavior of malignant mesothelioma.
Recently, Nakamoto et al(3)
investigated the antitumor effect of the anti-CD9 monoclonal
antibody (ALB6) in human gastric cancer cell xenografts. They found
a profound effect on tumor progression by anti-proliferative,
pro-apoptotic and anti-angiogenetic effects. Moreover, we
previously identified CD9 along with side population CD24 and CD26
cells to be markers of cancer stem cells in mesothelioma. We also
demonstrated that CD9-positive cell lines had a clear tendency to
generate larger tumors in mice (4).
Thus, CD9 may be a potential candidate as a molecular target in the
treatment of mesothelioma.
Loss of CD9 expression correlates with poor
prognosis in bladder carcinoma (26) and esophageal squamous cell carcinoma
(24), small and non-small cell
lung cancers (27,28) and prostatic carcinoma (23). This study is the first to analyze
CD9 expression in human mesothelioma tissue and to correlate its
expression with survival with other clinicopathological parameters.
CD9 expression was noted more frequently in younger patients, IMIG
stage I–II and epithelioid histology compared to older patients,
IMIG stage III–IV and sarcomatoid histology.
The present study found that the loss of CD9
expression in mesothelioma is related to a shorter overall survival
(medial survival 9 months, 1-year survival 38.9% and 2-year
survival 11.1%) compared to patients with CD9 expression (median
survival 15.1 months, 1-year survival, 63.2% and 2-year survival
25%). When CD9 expression in mesothelioma was stratified according
to score (1–3) did not show a statistically significant
association with overall survival rates (Fig. 3B), suggesting that the complete loss
of CD9 expression has more significance than the extent of CD9
expression. Age, IMIG staging, histology, differentiation of
epithelioid mesothelioma, therapeutic regimen, status of
extrapleural pneumonectomy and status of chemotherapy all had a
statistically significant association with overall survival.
Patients with CD9 expression had higher survival compared to those
without CD9 expression in patients receiving best supportive care
or patients treated with chemotherapy. This suggests the importance
of CD9 expression as an indicator for patients receiving
chemotherapy in mesothelioma patients.
The multivariate analysis showed CD9 expression is
an independent predictor of survival of mesothelioma patients with
an HR of 1.99 (P=0.0261) as well as older age (HR, 2.10), IMIG
stage III/IV (HR, 2.04), sarcomatoid histology (HR, 6.65), patients
treated with chemotherapy alone (HR, 0.37) and patients treated
with extrapleural pneumonectomy (HR, 0.26) (Table IV). As CD9 expression was observed
in only one case of sarcomatoid mesothelioma and sarcomatoid
histology was itself a strong independent predictor of survival of
mesothelioma patients, multivariate analysis excluding sarcomatoid
mesothelioma is necessary to evaluate the importance of CD9
expression as an independent predictor of mesothelioma survival.
Hence, we performed multivariate analysis of patients with
epithelioid mesothelioma alone and we again found that CD9
expression was a predictor of survival of mesothelioma patients
with an HR of 2.60 (P=0.0376) (Table
V), suggesting the independent prognostic value of CD9
expression in mesothelioma.
In the present study, sarcomatoid mesothelioma did
not show CD9 expression. It may thus be hypothesized that the loss
of CD9 expression in epithelioid mesothelioma leads to loss of
epithelioid differentiation and ultimately transition into
sarcomatoid mesothelioma. This is supported, in part, by the fact
that histologically less-differentiated epithelioid mesotheliomas
showed lower CD9 expression. In contrast, loss of differentiation
or epithelial-mesenchymal transition by other molecular pathways
leading to loss of CD9 expression may also be postulated. The
biological significance of loss of CD9 expression in sarcomatoid
mesothelioma requires further investigation.
In conclusion, CD9 expression is a favorable
prognostic marker in patients with mesothelioma. Our in
vitro study demonstrated increased cell migration after CD9
knockdown, suggesting loss of CD9 as a predictor of more aggressive
behavior. CD9 expression may be also an indicator of
epithelial-mesenchymal transition from epithelioid mesothelioma to
sarcomatoid mesothelioma.
Acknowledgements
The authors thank Yuka Fukushima for her excellent
technical assistance and Keiko Honda and Naomi Fukuhara for their
administrative assistance. This research was supported by the
Program for the Promotion of Fundamental Studies in Health Sciences
of the National Institute of Biomedical Innovation.
References
|
1
|
Tarrant JM, Robb L, van Spriel AB and
Wright MD: Tetraspanins: molecular organisers of the leukocyte
surface. Trends Immunol. 24:610–617. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zoller M: Tetraspanins: push and pull in
suppressing and promoting metastasis. Nat Rev Cancer. 9:40–55.
2009. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Nakamoto T, Murayama Y, Oritani K, et al:
A novel therapeutic strategy with anti-CD9 antibody in gastric
cancers. J Gastroenterol. 44:889–896. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ghani FI, Yamazaki H, Iwata S, et al:
Identification of cancer stem cell markers in human malignant
mesothelioma cells. Biochem Biophys Res Commun. 404:735–742. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Milano MT and Zhang H: Malignant pleural
mesothelioma: a population-based study of survival. J Thorac Oncol.
5:1841–1848. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kobayashi N, Toyooka S, Yanai H, et al:
Frequent p16 inactivation by homozygous deletion or methylation is
associated with a poor prognosis in Japanese patients with pleural
mesothelioma. Lung Cancer. 62:120–125. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nojiri S, Gemba K, Aoe K, et al: Survival
and prognostic factors in malignant pleural mesothelioma: a
retrospective study of 314 patients in the west part of Japan. Jpn
J Clin Oncol. 41:32–39. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gaafar R, Bahnassy A, Abdelsalam I, et al:
Tissue and serum EGFR as prognostic factors in malignant pleural
mesothelioma. Lung Cancer. 70:43–50. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Rena O, Boldorini LR, Gaudino E and
Casadio C: Epidermal growth factor receptor overexpression in
malignant pleural mesothelioma: prognostic correlations. J Surg
Oncol. 104:701–705. 2011. View Article : Google Scholar
|
|
10
|
Hirayama N, Tabata C, Tabata R, et al:
Pleural effusion VEGF levels as a prognostic factor of malignant
pleural mesothelioma. Respir Med. 105:137–142. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tabata C, Hirayama N, Tabata R, et al: A
novel clinical role for angiopoietin-1 in malignant pleural
mesothelioma. Eur Respir J. 36:1099–1105. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Pinton G, Brunelli E, Murer B, et al:
Estrogen receptor-beta affects the prognosis of human malignant
mesothelioma. Cancer Res. 69:4598–4604. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Fischer JR, Ohnmacht U, Rieger N, et al:
Promoter methylation of RASSF1A, RARbeta and DAPK predict poor
prognosis of patients with malignant mesothelioma. Lung Cancer.
54:109–116. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Pass HI, Goparaju C, Ivanov S, et al:
hsa-miR-29c* is linked to the prognosis of malignant
pleural mesothelioma. Cancer Res. 70:1916–1924. 2010.
|
|
15
|
Travis W, Brambilla E, Müller-Hermelink H
and Harris C: World Health Organization Classification of Tumours:
Pathology & Genetics. Tumours of the Lung, Pleura, Thymus and
Heart. IARC Press; Lyon: 2004
|
|
16
|
Amatya VJ, Takeshima Y, Kushitani K,
Yamada T, Morimoto C and Inai K: Overexpression of CD26/DPPIV in
mesothelioma tissue and mesothelioma. Oncol Rep. 26:1369–1375.
2011.PubMed/NCBI
|
|
17
|
Ikeyama S, Koyama M, Yamaoko M, Sasada R
and Miyake M: Suppression of cell motility and metastasis by
transfection with human motility-related protein (MRP-1/CD9) DNA. J
Exp Med. 177:1231–1237. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Miyake M, Koyama M, Seno M and Ikeyama S:
Identification of the motility-related protein (MRP-1), recognized
by monoclonal antibody M31–15 which inhibits cell motility. J Exp
Med. 174:1347–1354. 1991.PubMed/NCBI
|
|
19
|
Higashiyama M, Doi O, Kodama K, et al:
Immunohistochemically detected expression of motility-related
protein-1 (MRP-1/CD9) in lung adenocarcinoma and its relation to
prognosis. Int J Cancer. 74:205–211. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Mori M, Mimori K, Shiraishi T, et al:
Motility related protein 1 (MRP1/CD9) expression in colon cancer.
Clin Cancer Res. 4:1507–1510. 1998.PubMed/NCBI
|
|
21
|
Jamil F, Peston D and Shousha S: CD9
immunohistochemical staining of breast carcinoma: unlikely to
provide useful prognostic information for routine use.
Histopathology. 39:572–577. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sho M, Adachi M, Taki T, et al:
Transmembrane 4 superfamily as a prognostic factor in pancreatic
cancer. Int J Cancer. 79:509–516. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang JC, Begin LR, Berube NG, et al:
Down-regulation of CD9 expression during prostate carcinoma
progression is associated with CD9 mRNA modifications. Clin Cancer
Res. 13:2354–2361. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Okochi H, Mine T, Nashiro K, Suzuki J,
Fujita T and Furue M: Expression of tetraspans transmembrane family
in the epithelium of the gastrointestinal tract. J Clin
Gastroenterol. 29:63–67. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Buim ME, Lourenco SV, Carvalho KC, et al:
Downregulation of CD9 protein expression is associated with
aggressive behavior of oral squamous cell carcinoma. Oral Oncol.
46:166–171. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Mhawech P, Herrmann F, Coassin M, Guillou
L and Iselin CE: Motility-related protein 1 (MRP-1/CD9) expression
in urothelial bladder carcinoma and its relation to tumor
recurrence and progression. Cancer. 98:1649–1657. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Higashiyama M, Taki T, Ieki Y, et al:
Reduced motility related protein-1 (MRP-1/CD9) gene expression as a
factor of poor prognosis in non-small cell lung cancer. Cancer Res.
55:6040–6044. 1995.PubMed/NCBI
|
|
28
|
Kohmo S, Kijima T, Otani Y, et al: Cell
surface tetraspanin CD9 mediates chemoresistance in small cell lung
cancer. Cancer Res. 70:8025–8035. 2010. View Article : Google Scholar : PubMed/NCBI
|