Introduction
Hepatocellular carcinoma (HCC) is the fifth most
common malignant tumor worldwide, and the third most common cause
of cancer-related mortality (1–4).
Chemotherapy, as a treatment for HCC patients has proven to be
toxic and not very effective. The most effective therapy to date is
still surgical resection; however, less than 15% of patients
benefit from this treatment due to the presence of multiple tumor
nodules. This situation is slowly being improved with a better
understanding of the molecular mechanisms involved in the
pathogenesis of HCC. It is imperative to develop novel strategies,
such as suicide gene therapy, in which nucleic acids encoding
specific therapeutic genes are used as anti-tumor agents.
The best characterized suicide gene therapy system
is the herpes simplex virus type 1 thymidine kinase (TK) combined
with the guanosine analog, ganciclovir (GCV), which is
phosphorylated with the aid of TK into a GCV monophosphate and
further by cellular kinases into di- and triphosphate forms
(5). GCV triphosphate is
incorporated into DNA during cell division, causing single-strand
DNA breaks and the inhibition of DNA polymerase (6–8), thus
causing DNA chain termination, eventually leading to programmed
cell death. Moreover, nearby unmodified tumor cells are known to
also be killed by the ‘bystander effect’ in this form of therapy.
Both the transfection efficiency and ‘bystander effect’ are
essential factors for the success of the anti-tumor efficacy of
HSV-TK and prodrug GCV suicide gene therapy system. The potential
of the HIV-1 transactivator protein transduction domain (TAT PTD)
as a carrier to deliver various macromolecules into cells has been
established in a number of recent studies. In the majority of the
studies, the TAT peptide has been utilized to cargo different
proteins and peptides mostly in cultured cells but also in animal
models (9). In the field of gene
therapy, the TAT peptide has been harnessed to, for example,
enhance viral vector transduction efficiency (10,11)
and gene-transfer efficiency of liposomes (12).
However, the clinical efficacy of gene therapy is
not satisfactory due to the lack of targeting (13). Survivin, a member of the inhibitor
of apoptosis (IAP) family, has attracted considerable attention as
an ideal target for cancer treatment since it is highly and
uniquely expressed in the majority of human tumors and plays a
critical role in the control of cell division and the inhibition of
apoptosis (14–18). A high level of survivin expression
has been observed in HCC (19). In
the present study, we constructed the TK and TAT-TK gene expression
vector with the promoter of the survivin gene and examined its
effect on the inhibition of proliferation and the induction of
apoptosis in the HCC cell line, HepG2.
Materials and methods
Cell culture and DNA preparation
The L02 human liver cell line and the HepG2 human
HCC cell line were purchased from KeyGen. Cells were cultured in
DMEM (Gibco, Carlsbad, CA, USA) supplemented with 10% newborn calf
serum (Gibco) at 37°C in a humidified atmosphere containing 5%
CO2.
Construction of expression vector
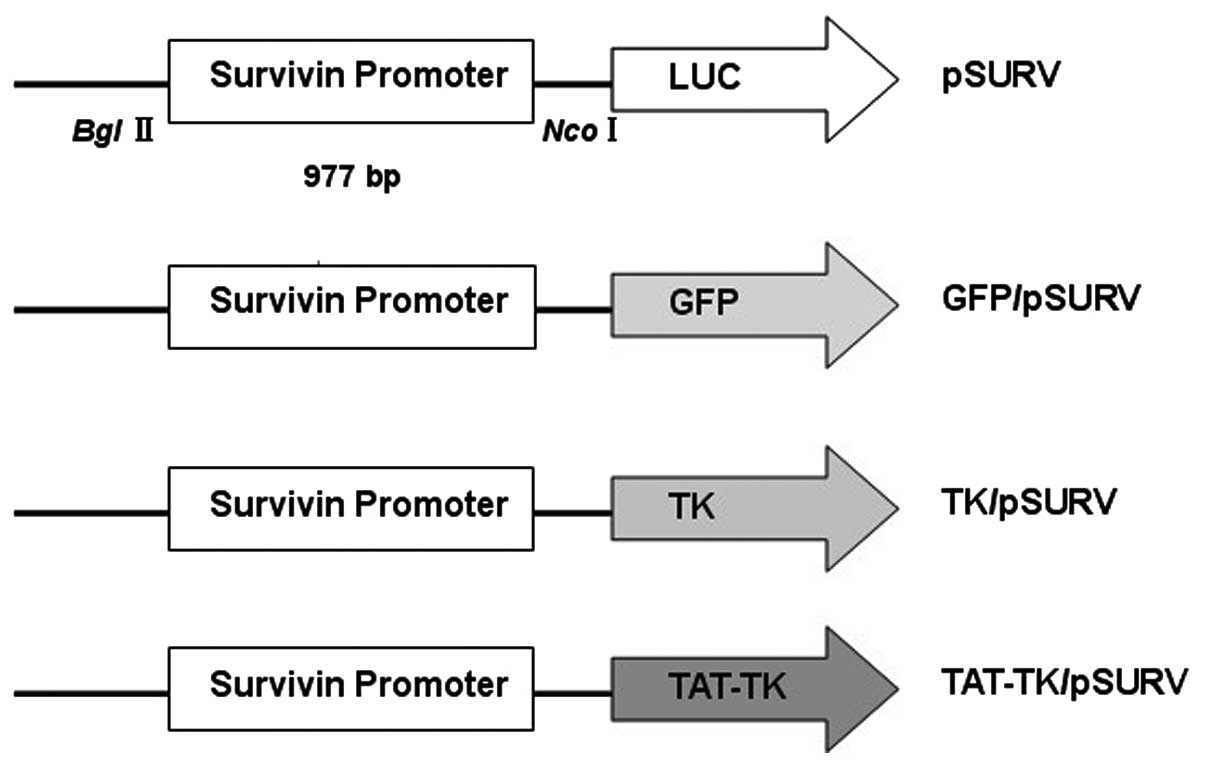
Four different plasmids were constructed (Fig. 1). In order to create the luciferase
expression plasmid under the control of the survivin promoter
(pSURV-Luc), we generated a 977-bp fragment of the human survivin
gene promoter (nucleotides 1824–2800, GenBank Accession no. U75285)
by polymerase chain reaction (PCR) of human bacterial artificial
chromosome libraries (RPC 1–11, 0219G17; ResGen, Inc., San Leandro,
CA, USA) as a template. The sequences of the oligonucleotides
primers were: forward primer, 5′-ATACGAGATCTGCCATAGAACCA-3′ and
reverse primer, 5′-ATGTAAAGCTTCCACCTCTGCCA-3′ (20). After restriction enzyme digestion
and purification, the fragment was inserted into the luciferase
vector, pGL3-basic (Promega Corp., Madison, WI, USA), at the
BglII and HindIII sites. To create the green
fluorescent protein (GFP), TK and TAT-TK constructs, termed
GFP/pSURV, TK/pSURV and TAT-TK/pSURV, we replaced the luciferase
gene of pSURV-Luc with GFP, TK and TAT-TK, respectively by
NcoI and XbaI digestion. The GFP fragment was
previously preserved in our laboratory. The TK fragment from the TK
gene-containing plasmid, r-pAs16Dr (Professor A. Söling,
Martin-Luther-University Halle-Wittenberg, Halle, Germany), was
amplified using the following primers: forward,
5′-ATCCATGGATGGCTTCGT ACCCCT-3′; and reverse,
5′-ATCTCGAGTCAAGCCTCCC CC-3′. The TAT-TK fragment from the TAT-TK
gene-containing plasmid, pGEX2T-Tat11-TK [Professor M. Zoppè,
International Center for Genetic Engineering and Biotechnology
(ICGEB), Trieste, Italy], was amplified using the forward primer,
5′-ATC CATGGGCATGTATGGCAGGAAG-3′; and the reverse primer,
5′-GCTCTAGACGGAGGACAGTCCTCCGGAGA
CCGGAGGACAGTCCTCCGTCAGTTAGCCTCC-3′. These plasmids were confirmed
by sequence analysis.
Transient transfection and GFP reporter
assays
Cells were plated in six-well plates at a density of
3×105 cells per well and incubated overnight. Cells were
transfected with GFP/pSURV using X-tremeGENE HP DNA Transfection
Reagent (Roche, USA) according to the manufacturer’s instructions.
After 48 h of transfection, the GFP/pSURV tansfection efficiency of
HepG2 and L02 cells were detected by fluorescence microscopy and
flow cytometry (FCM).
Stable transfection
After transfection for 48 h, HepG2 cells were
diluted to 1:10 for passage and neomycin-resistant clones were
selected in the medium containing 400 μg/ml G418 (Gibco) for two
weeks. The positive clones were selected and expanded to establish
cell lines. The stably transfected cell clones, designated as
HepG2/pSURV, HepG2/TK/pSURV and HepG2/TAT-TK/pSURV, were verified
by quantitative real-time RT-PCR and western blot analysis.
Quantitative real-time reverse
transcriptase (RT)-PCR
Total cellular RNA was extracted using TRIzol
reagent (Invitrogen, Carlsbad, CA, USA). RNA integrity was
confirmed by electrophoresis on ethidium bromide-stained 1% agarose
gels. The primer sequences used for TK were: sense, 5′-TGGCCAAA
CGCCTCCGTTCC-3′ and antisense, 5′-GTGCGCGCCA GGTCACATA-3′; GAPDH:
sense, 5′-TAAATTGAGCCCG CAGCCTCCC-3′ and antisense,
5′-GACCAAATCCGTT GACTCCGACCT-3′. The mRNA level for TK was analyzed
by one-step real-time RT-PRC with RNA-direct™ SYBR-Green Real-time
PCR Master mix (Toyobo, Osaka, Japan) according to the
manufacturer’s instructions. Cycling conditions were: 90°C for 30
sec, 61°C for 20 min, 95°C for 60 sec, then 40 cycles at 95°C for
15 sec and 60°C for 1 min. The amplification was monitored on an
ABI Prism 7500 Real-time PCR apparatus (Applied Biosystems, USA).
The products were detected on a 1% agarose gel.
Western blot analysis
Cells were harvested from flasks, and lysed with
ice-cold lysis buffer (50 mM Tris-HCl, pH 7.4, 150 mM NaCl, 1 mM
MgCl2, 100 μg/ml PMSF and 1% Triton X-100 ) for 30 min
on ice. Cell lysates were then collected after centrifugation at
12,000 rpm for 5 min at 4°C. Equal amounts (40 μg) of lysate
proteins were separated on 10% SDS-PAGE gels, and transblotted onto
PVDF membranes (Pall Corp., Ann Arbor, MI, USA). After blocking
with 5% non-fat dry milk in TBST buffer (10 mM Tris, pH 7.5, 150 mM
NaCl, and 0.05% Tween-20) for 2 h at room temperature, the
membranes were probed with 1:500 dilution of anti-TK (Santa Cruz
Biotechnology, Inc., Santa Cruz, CA, USA) or anti-β-actin (Sigma,
St. Louis, MO, USA) antibodies at 4°C overnight, followed by
incubation in a 1:1,000 dilution of secondary antibodies conjugated
to horseradish peroxidase (the membranes were probed with a 1:500
dilution of anti-TK (Santa Cruz Biotechnology, Inc.) or
anti-β-actin (Sigma) for 1 h at room temperature. Protein bands
were detected using the ECL detection system. All the western blot
analyses were performed at least three times.
Cell proliferation assay
Cell proliferation was analyzed with 3-(4,
5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT;
Sigma). Briefly, 8,000 cells from each group were plated per well
in three 96-well microplates in 200 μl of medium and replaced with
0, 5, 10, 20, 40, 60, 80 and 100 μg/ml GCV the following day. After
48 h, 50 μl of MTT substrate were added to each well, and the
plates were returned to standard tissue incubator conditions for an
additional 4 h. The medium was then removed, the cells were
solubilized in 150 μl of dimethyl sulfoxide, and colorimetric
analysis was performed (wavelength, 490 nm). The inhibition rate
was calculated as [1-(OD value of the transfection/OD value of
untreated HepG2 cells)] ×100%. Each experiment was performed in
triplicate.
‘Bystander effect’ analysis
The modified cells, HepG2/TK/pSURV and
HepG2/TAT-TK/pSURV, incorporated with HepG2 unmodified cells at
different ratios (0, 10, 20, 40, 80 and 100%), adjusted to a
density of 8,000 cells/well, were co-cultured in 96-well plates at
37°C for 48 h in the presence of 20 μg/ml of GCV. The ‘bystander
effect’ was determined using the MTT method, as described
above.
Apoptosis detection by FCM
The apoptotic cells were differentiated from viable
or necrotic ones by combined application of Annexin V-FITC and
propidium iodide (PI) (BD Biosciences, Franklin Lakes, NJ, USA).
The samples were washed twice and adjusted to a concentration of
5×105 cells/ml with PBS. Suspensions (200 μl) was added
to each labeled tube, 5 μl of Annexin V-FITC and 10 μl PI (20
μg/ml) were added into the labeled tube, incubated for at least 10
min at room temperature in the dark, then 200 μl of PBS binding
buffer were added to each tube without washing and analyzed using
FCM (BD Biosciences) as soon as possible (within 30 min). Apoptotic
cells were defined as the population that were PI-negative
(indicating an intact plasma membrane) and Annexin V-FITC-positive.
This assay was performed in triplicate.
DNA fragmentation analysis
Cells (1×104 cells) were centrifuged,
resuspended in 200 μl PBS, incubated with 4 μl RNase A for 0.5 h at
37°C, followed by digestion with 20 μl proteinase K for 1 h at
37°C, and then lysed in 200 μl of lysis buffer [1 ml of 1 M
Tris-HCl buffer at pH 7.4, 0.2 ml of 0.5 M
ethylenediaminetetraacetic acid (EDTA)], and 0.5 ml 10% Triton
X-100). Lysed cells were held at 70°C for 10 min. The solution was
mixed with 5 M NaCl (20 μl) and isopropanol (120 μl), and the
mixture was incubated at −20°C for 24 h, followed by centrifugation
at 15,000 × g for 20 min. The precipitated DNA was dissolved in
Tris-EDTA (5 μl) buffer and subjected to electrophoresis using a 1%
agarose gel and Tris-acetate-EDTA buffer at 90 V. The DNA
fragmentation pattern was visualized with a UV
transilluminator.
Detection of apoptosis and proliferation
signaling by western blot analysis
Western blot analysis were performed as described
above. Rabbit polyclonal antibodies against cleaved caspase-3 (Cell
Signaling, USA) and proliferating cell nuclear antigen (PCNA)
(Santa Cruz Biotechnology, Inc.) were employed for antigen
detection while rabbit polyclonal antibody against GAPDH (Xianzhi
Bio, Hangzhou, China) was used as the control. Immunodetection was
carried out with HRP-coupled secondary antibodies to mouse (Sigma)
or rabbit (Santa Cruz Biotechnology, Inc.) antibodies.
Statistical analysis
SPSS16.0 software was used. Each assay was performed
at least three times. The data are expressed as the means ± SD. The
Student’s t-test and ANOVA were used to determine the significance
of differences in multiple comparisons. P<0.05 was considered to
indicate a statistically significant difference.
Results
Cancer-specific transgene expression of
the survivin promoter in vitro
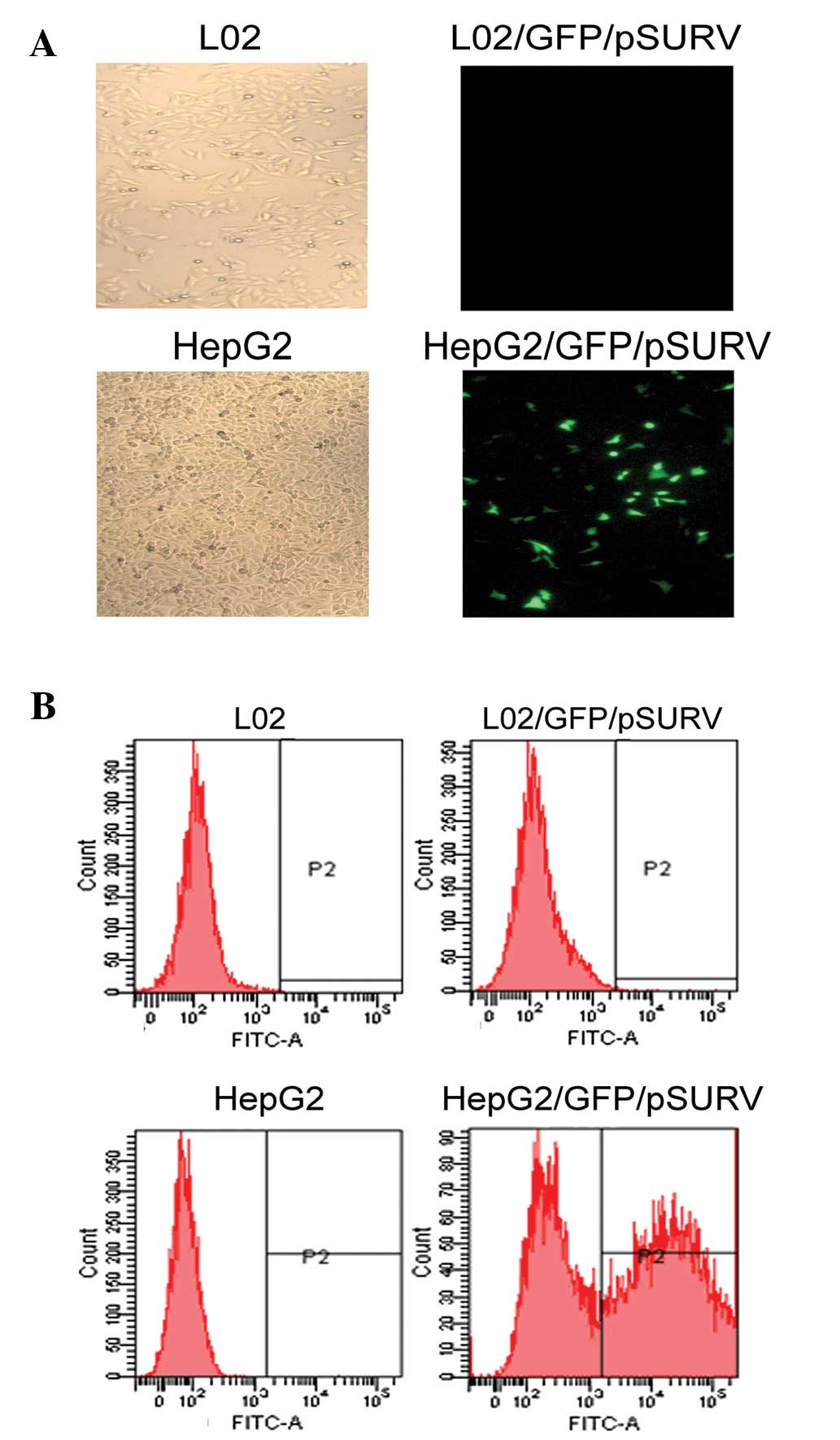
To assess whether the activation of the survivin
promoter is cancer-specific, we used GFP/pSURV to determine the
activity of the survivin promoter in the HepG2 cancer cell line and
in the L02 normal hepatic cell line. As shown in Fig. 2, a stronger fluorescence signal was
observed in the HepG2 cells, 48 h after transfection with
GFP/pSURV, indicating that the survivin promoter was more
specifically activated in the HCC cells than in the normal
cells.
Survivin promoter-driven expression of
TK
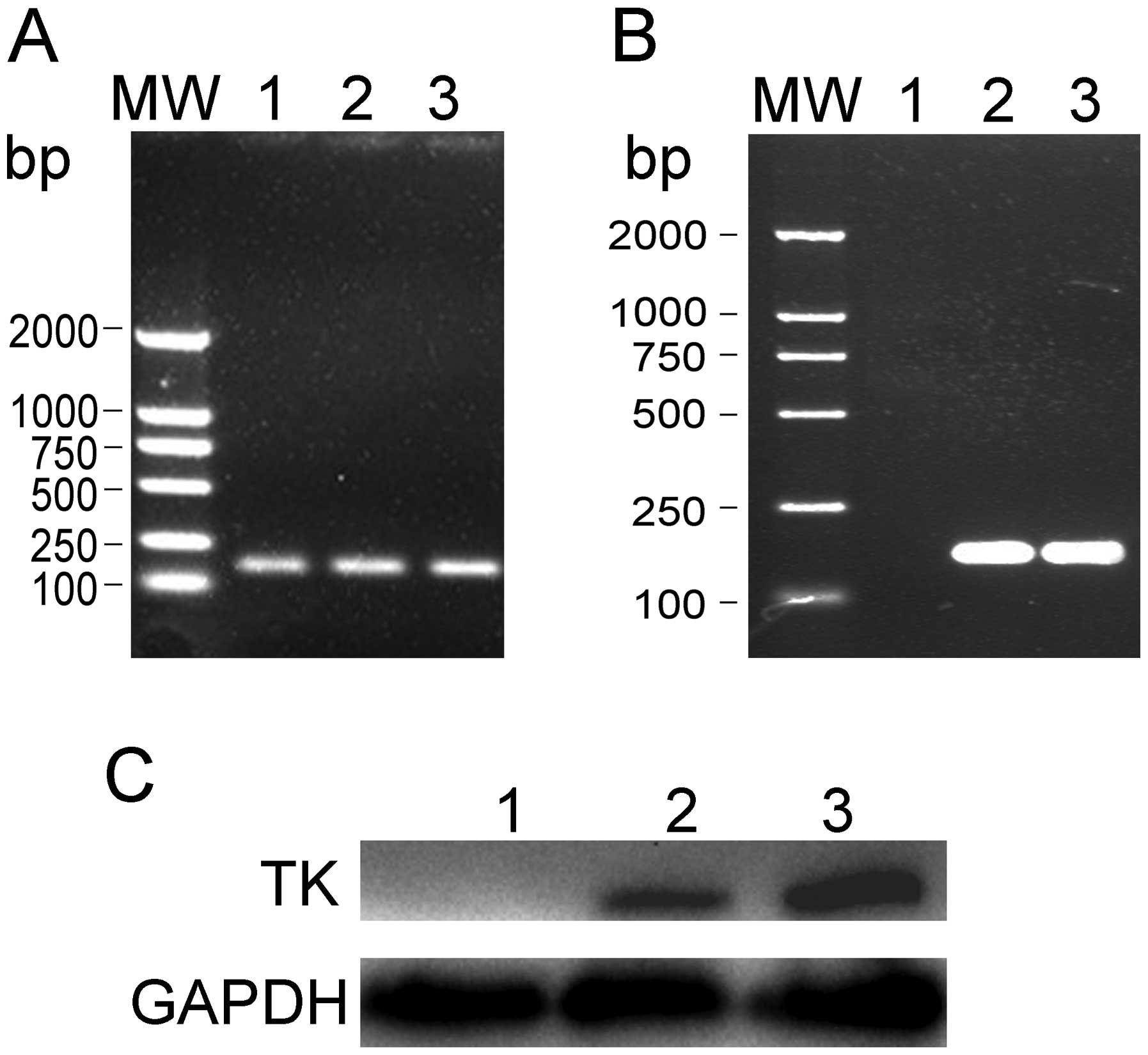
To determine whether the recombinant plasmids,
TK/pSURV and TAT-TK/pSURV, can be successfully transferred into
HepG2 cells and whether the TK expression can be achieved, total
HepG2 cellular RNA and protein were extracted from the different
transfected groups. The expression of TK was detected by RT-PCR and
western blot analysis. As shown in Fig.
3, TK was strongly expressed in the HepG2/TK/pSURV and
HepG2/TAT-TK/pSURV groups, but not in the HepG2 and HepG2/pSURV
groups.
Expression of TK driven by the survivin
promoter inhibits cancer cell growth in vitro
To investigate the biological effect induced by
these novel suicide gene systems (TK/pSURV and TAT-TK/pSURV), the
level of cell proliferation was assessed by MTT assay. As shown by
the cell growth inhibition rate curve (Fig. 4), the cell growth inhibition rates
in the HepG2/TK/pSURV and HepG2/TAT-TK/pSURV groups gradually
increased and the cell growth inhibition rate in the
HepG2/TAT-TK/pSURV group was higher than that in the HepG2/TK/pSURV
group under the same experimental conditions (P<0.05). No
difference in cell proliferation was observed between the HepG2 and
HepG2/pSURV groups.
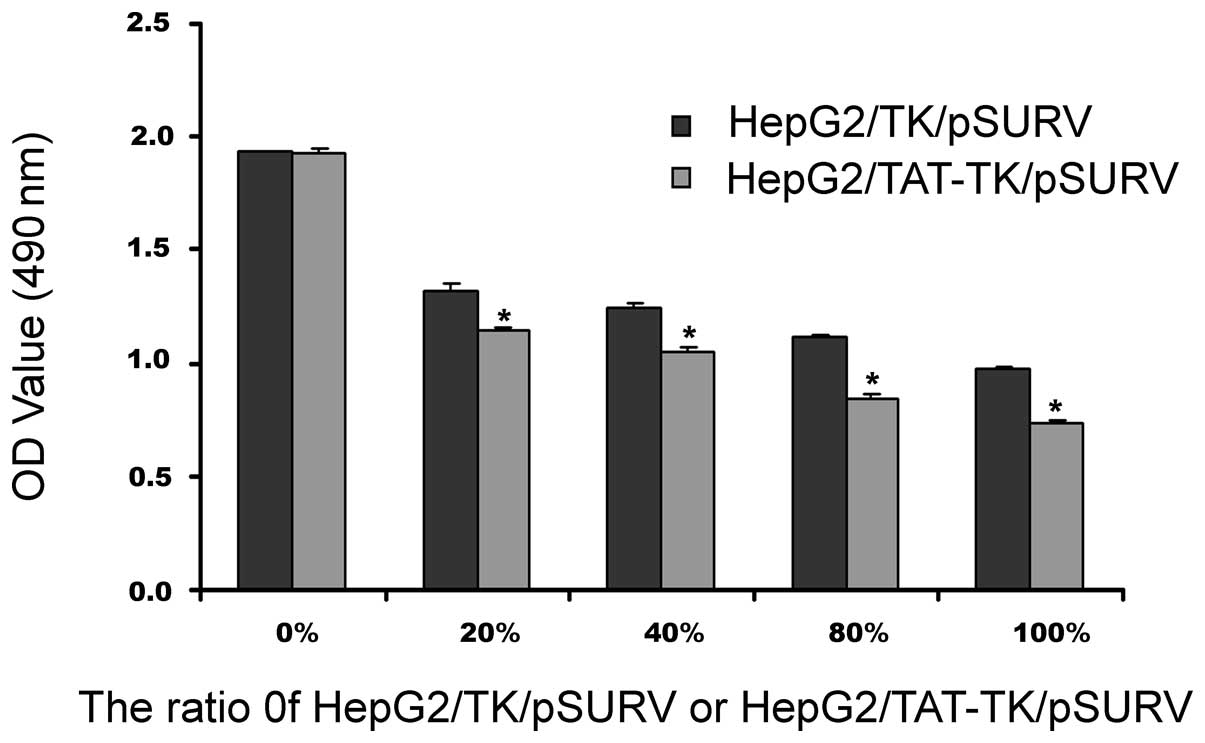
‘Bystander effect’ in the presence of
TK/pSURV/GCV and TAT-TK/pSURV/GCV
As shown in Fig. 5,
with the increase in the percentage of TK/pSURV/GCV- or
TAT-TK/pSURV/GCV-transfected cells co-cultured with unmodifed HepG2
cells, cell growth inhibition rates were progressively elevated,
indicative of a marked ‘bystander effect’. However, the most
prominent ‘bystander effect’ was observed in the HepG2/TAT-TK/pSURV
group.
Suicide gene system induces apoptosis in
HepG2 cells
Based on Annexin V-FITC and PI staining, FCM was
applied to quantify the extent of apoptosis. As demonstrated in
Fig. 6A, the apoptotic rates in the
HepG2/TK/pSURV and HepG2/TAT-TK/pSURV groups were 20.51 and 55.82%,
respectively, which were significantly higher compared to the other
groups (P<0.05). The maximal apoptotic rate in the
HepG2/TAT-TK/pSURV group was significantly higher than that in the
HepG2/TK/pSURV group (P<0.05). The findings observed by FCM were
corroborated by DNA fragmentation analysis (Fig. 6B).
Suicide gene system modifies cell
proliferation and apoptosis signaling activity
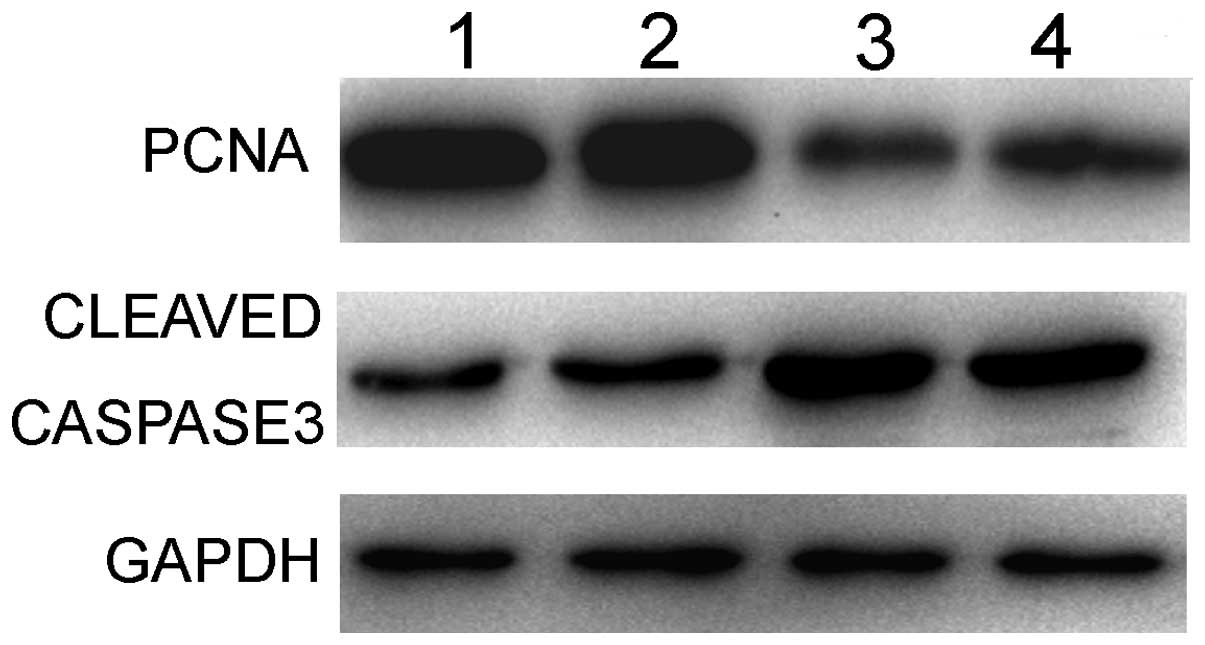
The PCNA protein levels and active caspase-3 were
subsequently detected. As shown in Fig.
7, the expression of PCNA in the HepG2/TK/pSURV and
HepG2/TAT-TK/pSURV groups was lower compared to the HepG2 and
HepG2/pSURV groups, while that of caspase-3 was higher compared to
the other groups. These findings strongly suggest that the
TAT-TK/GCV gene therapy system mechanistically upregulates
caspase-3 directed apoptotic signaling and represses PCNA-mediated
cell proliferation in HepG2 cells.
Discussion
Despite other recent advances in anticancer
therapeutics, tumor-specific suicide gene therapy using a
tissue-specific promoter is a rational therapeutic strategy for
HCC. Suicide gene therapy using the herpes simplex virus thymidine
kinase/ganciclovir (HSV-TK/GCV) system is a well-characterized tool
used in cancer gene therapy (21–25).
However, thus far, this suicide gene system (HSV-TK/GCV) has
demonstrated little efficacy in clinical practice, mostly due to
low targeting, absence of the ‘bystander effect’ and poor
gene-transfer efficiency in tumors.
Survivin, a member of the IAP family is
overexpressed in most types of cancer cells and embryonic tissue;
however, it is only slightly detected in terminally differentiated
normal tissue (26). Transcription
experiments have indicated that the protein expression of survivin
in cancer tissue appears to be regulated, at least in part,
transcriptionally (27,28). Since the increased survivin activity
is controlled transcriptionally, it has been suggested that the
survivin promoter may control the transgene expression in a
cancer-specific manner (28). Chen
et al demonstrated that the survivin promoter can drive the
expression of BikDD in lung cancer cells and inhibit cancer cell
growth in vitro and in vivo(20). A high level of survivin expression
has been observed in HCC (19). In
the present study, four different plasmids were constructed with
the survivin promoter. Following transfection with GFP/pSURV, a
stronger fluorescence signal was observed in the HepG2 cells, but
not in the L02 cells and TK gene expression observed in the HepG2
cells, but not in the L02 cells following transfection with
TK/pSURV and TAT-TK/pSURV. These results suggest that when the
survivin promoter-driven suicide gene is systemically administered,
the systemic toxicity can be significantly reduced.
Besides the advantage of the survivin promoter, the
HSV-TK/GCV suicide gene therapy system has another superiority: the
‘bystander effect’. The ‘bystander effect’ occurs when
non-transfected or genetically unmodified cells are killed as the
result of enzyme/prodrug activation during the death of genetically
modified tumor cells transfected with a suicide gene. The
‘bystander effect’ greatly amplifies the efficacy of HSV-TK/GCV
gene therapy for cancer in which only a fraction of the cells are
targeted. Suicide gene therapy has become more effective with the
enhancement of the ‘bystander effect’ (29). This phenomenon is thought to occur
due to the transfer of toxic GCV metabolites from HSV-TK-modified
cells to non-transfected tumor cells through gap junctions
(30–32).
In this study, when fused with HIV-1 TAT, the
‘suicide gene’ product TK induced cell death with a significant
‘bystander effect’. Our results also indicated that induced
apoptosis increased when the ‘suicide gene’ product TK was fused
with HIV-1 TAT. It has been previously demonstrated that the
cytotoxicity of GCV can be enhanced by fusing the HSV-TK with the
cell penetrating peptide from the HIV-1 transactivator protein
transduction domain (TAT PTD) (33). The mechanism involved however,
remains unclear. Previous studies have focused on the intracellular
transduction of TAT, as the GRKKR sequence of TAT can act as a
nuclear localization signal (34).
A substantial amount of research has been carried out using a 10–12
amino acid domain derived from the HIV TAT protein (35). This peptide enters cells by a
non-receptor-dependent, non-energy-dependent mechanism that
presumably involves passive permeation across the plasma membrane
(34). This peptide also contains
nuclear localization signals (NLS) that help to overcome the
nuclear membrane bottleneck (36).
Cao et al found that when the suicide gene TK was fused with
TAT, the ‘suicide gene’ product TK bound to the plasma membrane and
entered the hepatoma cells. Moreover, it was targeted to the
nucleus and the internalized TK was stable (37).
In this study, we demonstrate the efficacy of the
suicide gene therapy system driven by the survivin promoter in
suppressing HCC cell growth and inducing apoptosis when the
‘suicide gene’ product TK was fused with HIV-1 TAT. Apoptosis, also
known as programmed cell death, refers to certain physiological or
pathological conditions in which the end of active life is
regulated by the activation of a set of apoptotic factors. We
detected the level of PCNA and cleaved caspase-3. In normal cells,
apoptosis and proliferation co-exist and maintain a dynamic
equilibrium. When the TK/GCV suicide gene targeting system was
delivered into tumor cells, a significant inhibition of cell growth
was observed, as well as enhanced apoptosis via the increased
caspase-3 protein activation. Therefore, our findings strongly
suggest that the survivin promoter-mediated tumor-targeting suicide
gene therapy system may represent a novel therapy for HCC.
Acknowledgments
This study was supported by the National Natural
Science Foundation of China (no. 30972913) and Natural Science
Foundation of Jiangsu province of China (no. BK2009451).
References
|
1
|
Parkin DM, Pisani P and Ferlay J: Global
cancer statistics. CA Cancer J Clin. 49:33–64. 1999. View Article : Google Scholar
|
|
2
|
Okuda K: New trends in hepatocellular
carcinoma. Int J Clin Lab Res. 23:173–178. 1993. View Article : Google Scholar
|
|
3
|
Bruix J, Boix L, Sala M and Llovet JM:
Focus on hepatocellular carcinoma. Cancer Cell. 5:215–219. 2004.
View Article : Google Scholar
|
|
4
|
Llovet JM, Burroughs A and Bruix J:
Hepatocellular carcinoma. Lancet. 362:1907–1917. 2003. View Article : Google Scholar
|
|
5
|
Moolten FL: Tumor chemosensitivity
conferred by inserted herpes thymidine kinase genes: paradigm for a
prospective cancer control strategy. Cancer Res. 46:5276–5281.
1986.
|
|
6
|
Springer CJ and Niculescu-Duvaz I:
Prodrug-activating systems in suicide gene therapy. J Clin Invest.
105:1161–1167. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Denny WA: Prodrugs for Gene-Directed
Enzyme-Prodrug Therapy (Suicide Gene Therapy). J Biomed Biotechnol.
2003:48–70. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Schepelmann S and Springer CJ: Viral
vectors for gene-directed enzyme prodrug therapy. Curr Gene Ther.
6:647–670. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Dietz GP and Bahr M: Delivery of bioactive
molecules into the cell: the Trojan horse approach. Mol Cell
Neurosci. 27:85–131. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gratton JP, Yu J, Griffith JW, et al:
Cell-permeable peptides improve cellular uptake and therapeutic
gene delivery of replication-deficient viruses in cells and in
vivo. Nat Med. 9:357–362. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lehmusvaara S, Rautsi O, Hakkarainen T and
Wahifors J: Utility of cell-permeable peptides for enhancement of
virus-mediated gene transfer to human tumor cells. Biotechniques.
40:573–576. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Torchilin VP, Levchenko TS, Rammohan R,
Volodina N, Papahadjopoulos-Sternberg B and D’Souza GG: Cell
transfection in vitro and in vivo with nontoxic TAT
peptide-liposome-DNA complexes. Proc Natl Acad Sci USA.
100:1972–1977. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li SD and Huang L: Targeted delivery of
siRNA by nonviral vectors: lessons learned from recent advances.
Curr Opin Investig Drugs. 9:1317–1323. 2008.PubMed/NCBI
|
|
14
|
Ambrosini G, Adida C, Sirugo G and Altieri
DC: Induction of apoptosis and inhibition of cell proliferation by
survivin gene targeting. J Biol Chem. 273:11177–11182. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Altieri DC: Survivin, versatile modulation
of cell division and apoptosis in cancer. Oncogene. 22:8581–8589.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ryan BM, O’Donovan N and Duffy MJ:
Survivin: a new target for anti-cancer therapy. Cancer Treat Rev.
35:553–562. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Blanc-Brude OP, Mesri M, Wall NR, Plescia
J, Dohi T and Altieri DC: Therapeutic targeting of the survivin
pathway in cancer: initiation of mitochondrial apoptosis and
suppression of tumor-associated angiogenesis. Clin Cancer Res.
9:2683–2692. 2003.PubMed/NCBI
|
|
18
|
Sah NK, Khan Z, Khan GJ and Bisen PS:
Structural, functional and therapeutic biology of survivin. Cancer
Lett. 244:164–171. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ikeguchi M, Hirooka Y and Kaibara N:
Quantitative analysis of apoptosis-related gene expression in
hepatocellular carcinoma. Cancer. 95:1938–1945. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chen JS, Liu JC, Shen L, et al:
Cancer-specific activation of the survivin promoter and its
potential use in gene therapy. Cancer Gene Ther. 11:740–747. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen SH, Kosai K, Xu B, et al: Combination
suicide and cytokine gene therapy for hepatic metastases of colon
carcinoma: sustained antitumor immunity prolongs animal survival.
Cancer Res. 56:3758–3762. 1996.PubMed/NCBI
|
|
22
|
Kieback DG, Fischer DC, Engehausen DG, et
al: Intraperitoneal adenovirus-mediated suicide gene therapy in
combination with either topotecan or paclitaxel in nude mice with
human ovarian cancer. Cancer Gene Ther. 9:478–481. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Qiu Z, Harms JS, Zhu J and Splitter GA:
Bovine herpesvirus tegument protein VP22 enhances thymidine
kinase/ganciclovir suicide gene therapy for neuroblastomas compared
to herpes simplex virus VP22. J Virol. 78:4224–4233. 2004.
View Article : Google Scholar
|
|
24
|
Mizuguchi H and Hayakawa T: Enhanced
antitumor effect and reduced vector dissemination with
fiber-modified adenovirus vectors expressing herpes simplex virus
thymidine kinase. Cancer Gene Ther. 9:236–242. 2002. View Article : Google Scholar
|
|
25
|
Kagaya T, Nakamoto Y, Sakai Y, et al:
Monocyte chemoattractant protein-1 gene delivery enhances antitumor
effects of herpes simplex virus thymidine kinase/ganciclovir system
in a model of colon cancer. Cancer Gene Ther. 13:357–366. 2006.
View Article : Google Scholar
|
|
26
|
Altieri DC: Validating survivin as a
cancer therapeutic target. Nat Rev Cancer. 3:46–54. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li F and Altieri DC: Transcriptional
analysis of human survivin gene expression. Biochem J. 344:305–311.
1999. View Article : Google Scholar
|
|
28
|
Bao R, Connolly DC, Murphy M, et al:
Activation of cancer-specific gene expression by the survivin
promoter. J Natl Cancer Inst. 94:522–528. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Huang Q, Liu XZ, Kang CS, Wang GX, Zhong Y
and Pu PY: The anti-glioma effect of suicide gene therapy using
BMSC expressing HSV/TK combined with overexpression of Cx43 in
glioma cells. Cancer Gene Ther. 17:192–202. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Culver KW, Ram Z, Wallbridge S, Ishii H,
Oldfield EH and Blaese RM: In vivo gene transfer with retroviral
vector-producer cells for treatment of experimental brain tumors.
Science. 256:1550–1552. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Freeman SM, Abboud CN, Whartenby KA, et
al: The ‘bystander effect’: tumor regression when a fraction of the
tumor mass is genetically modified. Cancer Res. 53:5274–5283.
1993.
|
|
32
|
Touraine RL, Ishii-Morita H, Ramsey WJ and
Blaese RM: The bystander effect in the HSVtk/ganciclovir system and
its relationship to gap junctional communication. Gene Ther.
5:1705–1711. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Merilainen O, Hakkarainen T, Wahlfors T,
Pellinen R and Wahlfors J: HIV-1 TAT protein transduction domain
mediates enhancement of enzyme prodrug cancer gene therapy in
vitro: a study with TAT-TK-GFP triple fusion construct. Int J
Oncol. 27:203–208. 2005.PubMed/NCBI
|
|
34
|
Vives E, Brodin P and Lebleu B: A
truncated HIV-1 Tat protein basic domain rapidly translocates
through the plasma membrane and accumulates in the cell nucleus. J
Biol Chem. 272:16010–16017. 1997. View Article : Google Scholar
|
|
35
|
Schwarze SR and Dowdy SF: In vivo protein
transduction: intracellular delivery of biologically active
proteins, compounds and DNA. Trends Pharmacol Sci. 21:45–48. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Bremner KH, Seymour LW and Pouton CW:
Harnessing nuclear localization pathways for transgene delivery.
Curr Opin Mol Ther. 3:170–177. 2001.PubMed/NCBI
|
|
37
|
Cao L, Si J, Wang W, et al: Intracellular
localization and sustained prodrug cell killing activity of
TAT-HSVTK fusion protein in hepatocelullar carcinoma cells. Mol
Cells. 21:104–111. 2006.PubMed/NCBI
|