Introduction
Squamous cell carcinoma of the oral cavity and
oropharynx is the sixth most frequent solid cancer worldwide
(1). Tongue squamous cell carcinoma
(TSCC) is the most common types of oral carcinoma and is well-known
for its rapid proliferation (2).
Prognostic assessment is critical for making better therapeutic
choices for patients, and the tumor-node-metastasis (TNM) staging
system is the key prognostic determinant for TSCC patients in
clinical practice (3). However,
conventional prognostic factors based on clinicopathological
features remain inadequate, and are unable to discriminate tumors
at the same clinical stage but with distinct clinical outcomes. The
lack of efficient diagnostic and prognostic biomarkers is
responsible for the high mortality rates (4). Therefore, it is necessary to identify
biomarkers that can provide additional prognostic information
beyond the standard clinical prognostic system for TSCC patients
(5).
microRNAs (miRNAs) are a group of endogenously
expressed small non-coding RNAs involved in the regulation of gene
expression at the post-transcriptional level (6). They show specific expression patterns
in cancers, which enable them to be biomarkers for cancer risk and
prognosis (7). Subsets of miRNAs,
including miR-195 (8), miR-21
(9), miR-200b and miR-15b (10), have been identified as potential
diagnostic and prognostic markers in TSCC patients. Other
tumor-related miRNAs may also have value for the prediction of
prognosis in TSCC patients.
Recent studies have shown that the expression of
miR-375 is associated with various clinicopathological parameters
in oral squamous cell carcinoma (OSCC) (11), and is correlated with clinical
outcomes in head and neck squamous cell carcinoma (HNSCC) (12). However, these studies included
heterogeneous groups of patients with cancers from different
subsites of the head and neck, including tongue, gingival, buccal,
lip, larynx and oropharynx carcinomas. Since the gene expression
patterns in HNSCC at different subsites may not be equally
associated with cancer prognosis (13,14), a
study focusing on a specific anatomical subsite, such as the
anterior body of the tongue, is likely to provide more accurate and
clinically useful information on the prognostic significance of
miR-375.
miR-375 frequently shows reduced expression in
various cancers, and it may act as a tumor suppressor by targeting
JAK-2 (15), PDK-1 (16), IGF1R (17) and ASCL-1 (18). However, the role of miR-375 and the
underlying mechanism in TSCC remain to be explored. Sp1 is a target
gene of miR-375 in cervical squamous cell cancer (19). Moreover, Sp1 upregulates cyclin D1
expression by binding to the cyclin D1 promoter (20) and is required for cell cycle
progression through the G1 phase (21). Thus, it was reasonable to test
whether miR-375 directly targets Sp1 and subsequently downregulates
cyclin D1 to induce cell cycle arrest in TSCC.
In the present study, we examined the expression of
miR-375 in 105 pairs of TSCC samples and matched adjacent normal
tissues, and the association of miR-375 expression with the overall
survival of TSCC patients. Moreover, we tested whether miR-375
downregulates Sp1 expression by targeting the Sp1 transcript and
subsequently downregulates cyclin D1 to induce cell cycle arrest in
TSCC cells. Our results suggest important roles for miR-375 in TSCC
pathogenesis and support its potential application in the
evaluation of patient prognosis.
Materials and methods
Clinical specimens
Paired primary TSCC samples and adjacent
histologically normal tissues were obtained from 105 patients who
were admitted to the Department of Oral and Maxillofacial Surgery
of the Peking University School of Stomatology (Beijing, China)
between 2008 and 2011. None of the patients received treatment
prior to radical surgical treatment. Tumor tissues and matched
nonmalignant tissues, at least 1.5 cm distal to the tumor margins,
were snap-frozen in liquid nitrogen and then stored at −80°C until
use. None of the TSCC patients had received adjuvant chemotherapy
or radiotherapy before surgery. The clinicopathological
characteristics of the patients are summarized in Table I. This study was approved by the
Ethics Committee of the Peking University School of Stomatology,
and all samples were obtained from patients who had signed informed
consent forms.
 | Table IRelationship between expression of
miR-375 and the clinicopathological factors in the 105 TSCC
patients. |
Table I
Relationship between expression of
miR-375 and the clinicopathological factors in the 105 TSCC
patients.
|
Characteristics | No. | miR-375 (T/N) (mean
± SD) | P-value |
|---|
| Gender | | | 0.132 |
| Male | 49 | 0.194±0.245 | |
| Female | 56 | 0.131±0.169 | |
| Age (years) | | | 0.424 |
| <60 | 65 | 0.172±0.243 | |
| ≥60 | 40 | 0.142±0.138 | |
| Tumor size | | | 0.041 |
|
T1–T2 | 67 | 0.189±0.232 | |
|
T3–T4 | 38 | 0.111±0.153 | |
|
Differentiation | | | 0.896 |
| Well | 45 | 0.168±0.257 | |
| Moderate | 47 | 0.159±0.178 | |
| Poor | 13 | 0.137±0.122 | |
| Clinical stage | | | 0.419 |
| I–II | 59 | 0.175±0.211 | |
| III–IV | 46 | 0.142±0.208 | |
| Node
metastasis | | | 0.266 |
| No | 62 | 0.178±0.247 | |
| Yes | 43 | 0.135±0.136 | |
| Status | | | 0.028 |
| Surviving | 60 | 0.197±0.235 | |
| Deceased | 45 | 0.111±0.158 | |
Cell lines and culture
Primary normal human oral keratinocyte (HOK) cells
were purchased and cultured in a keratinocyte growth medium
(ScienCell Research Laboratories, San Diego, CA, USA) according to
the manufacturer’s instructions. Human TSCC cell lines, SCC-15 and
CAL-27, were obtained from the American Type Culture Collection
(Manassas, VA, USA) and cultured in Dulbecco’s modified Eagle’s
medium supplemented with 10% fetal bovine serum and 1% antibiotics
at 37°C in a humidified atmosphere of 5% CO2 and 95%
air.
Vector construction
A 417-base pair (bp) fragment of the 3′ untranslated
region (UTR) of the human Sp1 transcript containing the miR-375
binding site (19) was amplified by
polymerase chain reaction (PCR). It was then cloned into a modified
version of pcDNA3.1(+) containing a firefly luciferase reporter
gene (a gift from Brigid L.M. Hogan, Duke University, Durham, NC,
USA) (22) and named wild-type Sp1
3′UTR. The following primers were used to clone the wild-type Sp1
3′UTR: sense, 5′-GAA TTC TTT TCC TTG TAT GTT CTT GGG T-3′ and
antisense, 5′-CTC GAG GCA GGT CTC TTT TAA TAT TGG C-3′.
Site-directed mutagenesis of the miR-375 binding site in the Sp1
3′UTR was performed using a Site-Directed Mutagenesis Kit (SBS
Genetech, Beijing, China). The product was named mutant Sp1 3′UTR.
The following primers were used to clone the mutant Sp1 3′UTR:
sense, 5′-GAA TGA TAG CCC AGT TGT TAA AGA AAT CTT GT-3′ and
antisense, 5′-ACA AGA TTT CTT TAA CAA CTG GGC TAT CAT TC-3′. A
1,934-bp fragment of human cyclin D1 promoter (−1,696-bp to +238-bp
relative to the transcription start site) was amplified from the
genomic DNA of HeLa cells and cloned into the PGL3-Enhancer vector
(Promega, Madison, WI, USA). The primers used to clone the cyclin
D1 promoter were as follows: sense, 5′-GCG GTA CCG CTA GCC AGC TGG
GCC GCC CTT GT-3′ and antisense, 5′-ATC CAT GGA AGC TTT GGG GCT CTT
CCT GGG CA-3′. All constructs were confirmed by DNA sequencing.
RNA oligoribonucleotide
A chemically modified double-stranded miR-375 mimic
and the corresponding miRNA mimic control were designed and
purchased from RiboBio Co. (Guangzhou, China). A small interfering
RNA (siRNA) targeting the human Sp1 transcript (siSp1) and the
corresponding scrambled control were purchased from Integrated
Biotech Solutions Co. (Shanghai, China). The miRNA and siRNA
sequences are listed in Table
II.
 | Table IISequences of RNA and DNA
oligonucleotides. |
Table II
Sequences of RNA and DNA
oligonucleotides.
| Name | Sense strand/sense
primer (5′-3′) | Antisense
strand/antisense primer (5′-3′) |
|---|
| Primers for
qRT-PCR |
| miR-375 RT
primer |
GTCGTATCCAGTGCAGGGTCCGAGGTAT
TCGCACTGGATACGACTCACGC | |
| miR-375 |
GTGCAGGGTCCGAGGT |
AGCCGTTTGTTCGTTCGGCT |
| U6 |
CTCGCTTCGGCAGCACA |
AACGCTTCACGAATTTGCGT |
| Sp1 |
ACCAGAATAAGAAGGGAGG |
GGTGGTAATAAGGGCTGAA |
| Cyclin D1 |
GTGCTGCGAAGTGGAAACC |
ATCCAGGTGGCGACGATCT |
| β-actin | CGG GAA ATC GTG CGT
GAC | CAG GCA GCT CGT AGC
TCT T |
| miRNA mimic |
| miR-375 mimic |
UUUGUUCGUUCGGCUCGCGUGA |
AAACAAGCAAGCCGAGCGCACU |
| miRNA mimic
control |
UUUGUACUACACAAAAGUACUG |
AAACAUGAUGUGUUUUCAUGAC |
| siRNA duplexes |
| Sp1 siRNA |
UGUAGAGUCUGCCAACUGACCUGUCTT |
GACAGGUCAGUUGGCAGACUCUACATT |
| siRNA control
(scramble) | UUC UCC GAA CGU GUC
ACG UTT | ACG UGA CAC GUU CGG
AGA ATT |
Transient transfection
Cells were plated into 6-well plates before
transfection. After reaching 80% confluency, the cells were
transfected with 100 nM miRNA mimic or siRNA using Lipofectamine
2000 (Invitrogen, Carlsbad, CA, USA) according to the
manufacturer’s procedure. The cells were harvested 48 h after
transfection.
RNA isolation and quantitative
reverse-transcription (qRT)-PCR
Total RNA was extracted using the TRIzol reagent
(Invitrogen) according to the manufacturer’s procedure, and then
reverse-transcribed into complementary DNA (cDNA) using a cDNA
reverse transcription kit (Applied Biosystems, Foster City, CA,
USA). Quantitative PCR was conducted with the ABI Prism 7500
real-time PCR system (Applied Biosystems). The following thermal
settings were used: 95°C for 10 min followed by 40 cycles of 95°C
for 15 sec and 60°C for 1 min. The primers used for miR-375, Sp1,
cyclin D1, U6 (internal control for miRNAs) and β-actin (internal
control for mRNAs and lncRNAs) are listed in Table II. The data was analyzed using the
2−ΔΔCt relative expression quantity as described
previously (8).
Cell proliferation assays
Cell proliferation was determined using the Cell
Counting Kit-8 (CCK-8, Dojindo, Kumamoto, Japan). SCC-15 and CAL27
cells were plated into 96-well plates (2×103
cells/well). After transfection with miR-375 mimic or the scrambled
control, CCK-8 (10 μl) was added to each well at 24, 48, and 72 h,
and the plates were incubated at 37°C for 3 h. The absorbance at
450 nm was measured using a microplate spectrophotometer (Bio-Tek
Instruments).
Colony formation assay
At 24 h after transfection, the cells were plated
into 60-mm dishes at an initial density of 500 cells/dish and
cultured until colonies were visible (10 days). Cell colonies were
fixed with cold methanol for 20 min and stained with 0.25% crystal
violet for 30 min. The number of cells that formed a clone was
calculated.
Cell cycle analysis
At 48 h post-transfection, CAL27 cells were
harvested by trypsinization and washed with phosphate-buffered
saline. The cell cycle was analyzed with fluorescence-activated
cell sorting (FACS) using FACSCalibur flow cytometry (Becton
Dickinson, Bedford, MA, USA) as described previously (8).
Dual luciferase reporter assay
Luciferase assays were performed as described
previously (23). Briefly, SCC-15
and CAL27 cells grown in a 48-well plate were cotransfected with
the miR-375 mimic or the miRNA mimic control (100 nM), the
luciferase reporter plasmid (40 ng/well) and pRL-TK, a plasmid
expressing Renilla luciferase (4 ng/well; Promega).
Luciferase activity was measured 24 h after transfection using the
Dual-luciferase reporter assay system (Promega).
Western blot analysis
Western blotting was performed as described
previously (8). Primary antibodies
against Sp1 (Cell Signaling Technology, Beverly, MA, USA), cyclin
D1 and β-actin (both from Santa Cruz Biotechnology, Santa Cruz, CA,
USA) were diluted at 1:1,000. The intensities of the bands obtained
by western blotting were quantified using ImageJ software
(http://rsb.info.nih.gov/ij). The
background was subtracted, and the signal of the target bands was
normalized to that of the β-actin band.
Statistical analysis
Statistical analyses were performed using SPSS for
Windows ver. 16.0 (IBM, Armonk, NY, USA). All data are expressed as
the mean ± standard deviation (SD). Differences between groups were
analyzed by the Student’s t-test. A one-way ANOVA was used to
analyze the relationship between miR-375 expression and the
clinicopathological characteristics. Survival curves were
constructed by the Kaplan-Meier method and were compared using the
log-rank test. The Cox regression model was applied to
simultaneously adjust all potential prognostic variables.
Experiments with cell cultures were conducted at least in
triplicate. A two-tailed value of P<0.05 was considered to
indicate a statistically significant difference.
Results
miR-375 expression is reduced in TSCC and
is correlated with cancer progression
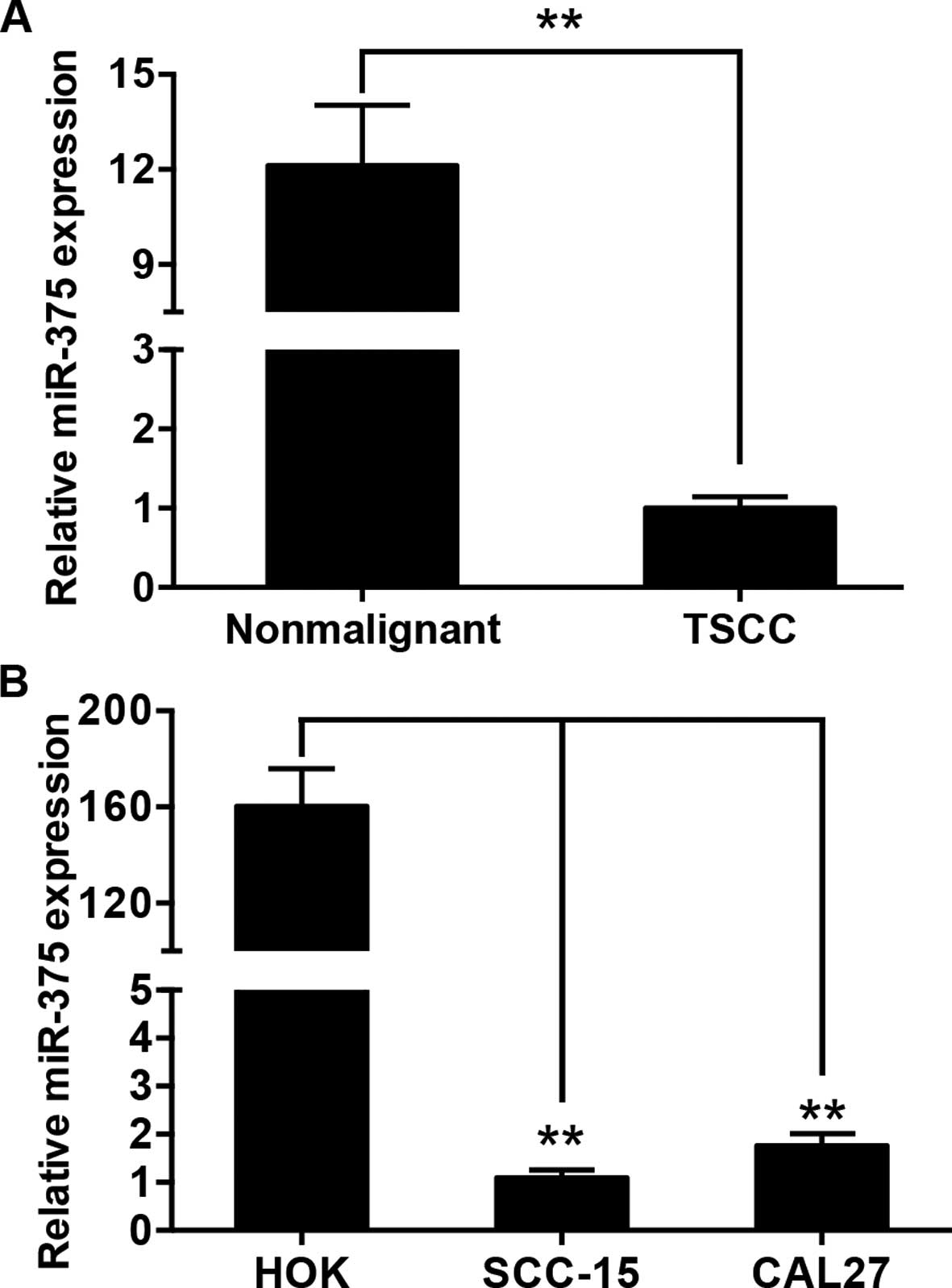
The expression of miR-375 was examined in 105 pairs
of tongue cancer tissues and matched adjacent histologically normal
tissues by qRT-PCR. The average expression level of miR-375 was
significantly decreased in the tumor tissues when compared with the
level in the normal tissues (Fig.
1A). Moreover, miR-375 expression was substantially reduced in
the TSCC cell lines, SCC-15 and CAL27, compared with that in the
HOK cells (Fig. 1B). The
relationship between miR-375 expression and the clinicopathological
factors of TSCC are presented in Table
I. By normalizing the miR-375 expression levels in the tumor
tissues to those in the adjacent nonmalignant tissues
(Tumor/Nonmalignant, T/N), we observed that miR-375 expression
(T/N) was significantly correlated with tumor size (P=0.041) and
patient mortality (P=0.028).
Decreased miR-375 expression is
associated with poor overall survival in the TSCC patients
With the mean fold change of miR-375 expression
(T/N) chosen as the cut-off point, the patients were divided into
high and low expression groups. The patients with high miR-375
expression survived significantly longer than those with low
miR-375 expression (Fig. 2).
Moreover, we conducted multivariable Cox proportional hazards
analysis to identify whether miR-375 is an independent prognostic
covariate for TSCC patients. Our results showed that low miR-375
expression in TSCC was associated with a poor prognosis in terms of
overall survival (P=0.044, relative risk =0.449), independent of
the other clinical covariates (Table
III ).
 | Table IIIMultivariate analysis of various
prognostic variables in the TSCC patients using Cox’s regression
analysis. |
Table III
Multivariate analysis of various
prognostic variables in the TSCC patients using Cox’s regression
analysis.
| Variables | No. of cases | P-value | Regression
coefficient | Relative risk | 95% CI |
|---|
| Tumor size | | 0.177 | 0.322 | 1.545 | 0.822–2.906 |
|
T1–T2 | 67 | | | | |
|
T3–T4 | 38 | | | | |
|
Differentiation | | 0.723 | 0.307 | 1.115 | 0.611–2.035 |
| Well | 45 | | | | |
| Moderate and
poor | 60 | | | | |
| Clinical stage | | 0.259 | 0.341 | 1.469 | 0.754–2.865 |
| I–II | 59 | | | | |
| III–IV | 46 | | | | |
| Node
metastasis | | 0.212 | 0.334 | 1.517 | 0.789–2.919 |
| No | 62 | | | | |
| Yes | 43 | | | | |
| miR-375
expression | | 0.044 | 0.396 | 0.449 | 0.207–0.978 |
| High | 36 | | | | |
| Low | 69 | | | | |
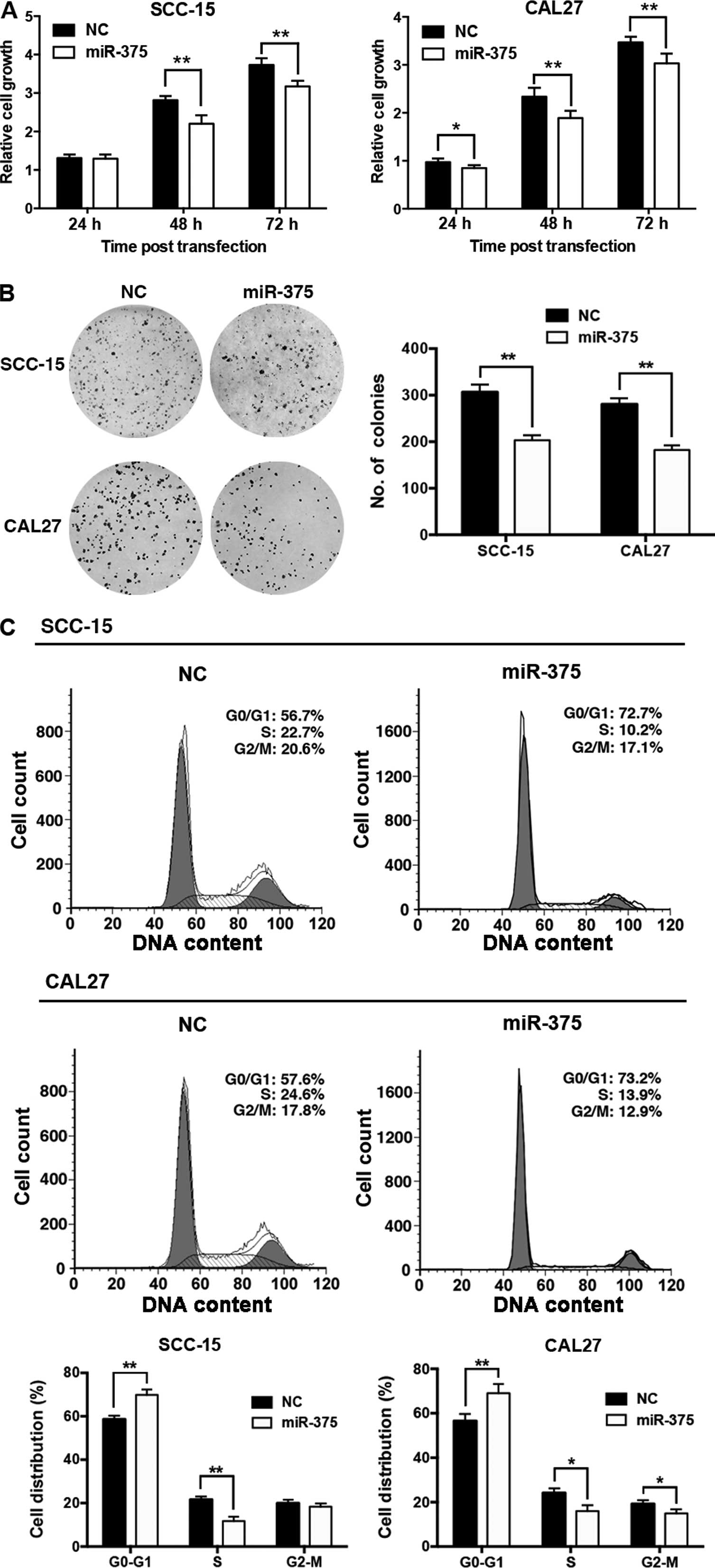
Overexpression of miR-375 inhibits cell
proliferation and cell cycle progression in TSCC cell lines
To investigate the biological function of miR-375 in
TSCC, we transfected the miR-375 mimic into TSCC cells, and
confirmed that miR-375 expression was significantly increased in
the SCC-15 and CAL27 cells at 48 h post-transfection (data not
shown). CCK-8 and colony formation assays showed that
overexpression of miR-375 significantly inhibited the proliferation
of the SCC-15 and CAL27 cells (Fig. 3A
and B), and resulted in substantial accumulation of cells in
the G1-phase (Fig.
3C).
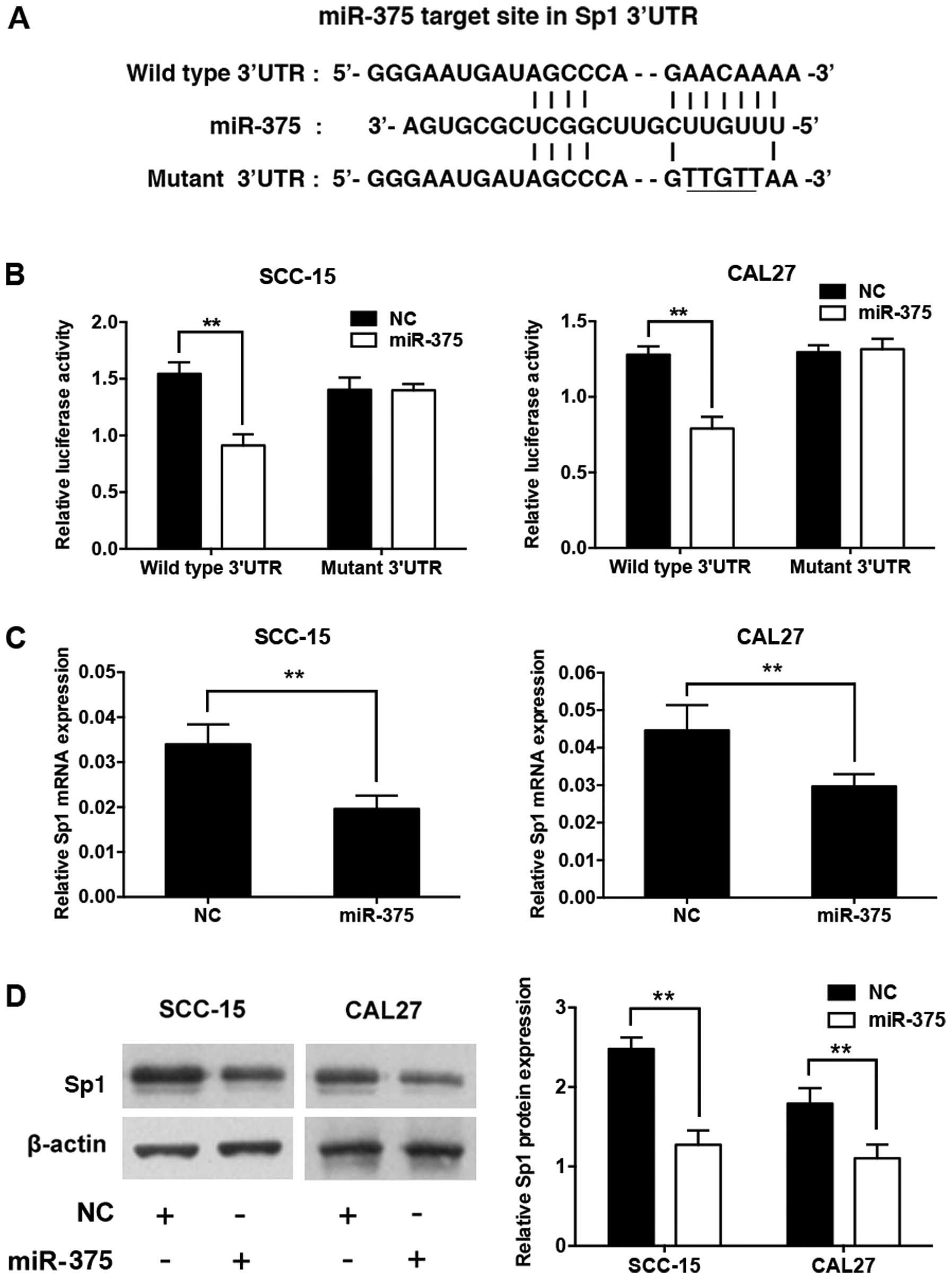
miR-375 inhibits Sp1 expression by
targeting its 3′UTR
According to a previous study, the human Sp1
transcript contains a putative miR-375 target site in its 3′UTR
(19) (Fig. 4A). We constructed a luciferase
reporter plasmid containing the human Sp1 3′UTR with the miR-375
target site intact (wild-type) or mutated. SCC-15 and CAL27 cells
were co-transfected with the miR-375 mimic, the miRNA mimic
control, and the reporter plasmid. Ectopic expression of miR-375
significantly suppressed the luciferase activity of the reporter
with the wild-type Sp1 3′UTR, but not that of the reporter with the
mutant Sp1 3′UTR, at 24 h post-transfection (Fig. 4B). Meanwhile, overexpression of
miR-375 significantly decreased the mRNA and protein levels of
endogenous Sp1 at 48 h post-transfection compared to the negative
control (Fig. 4C and D).
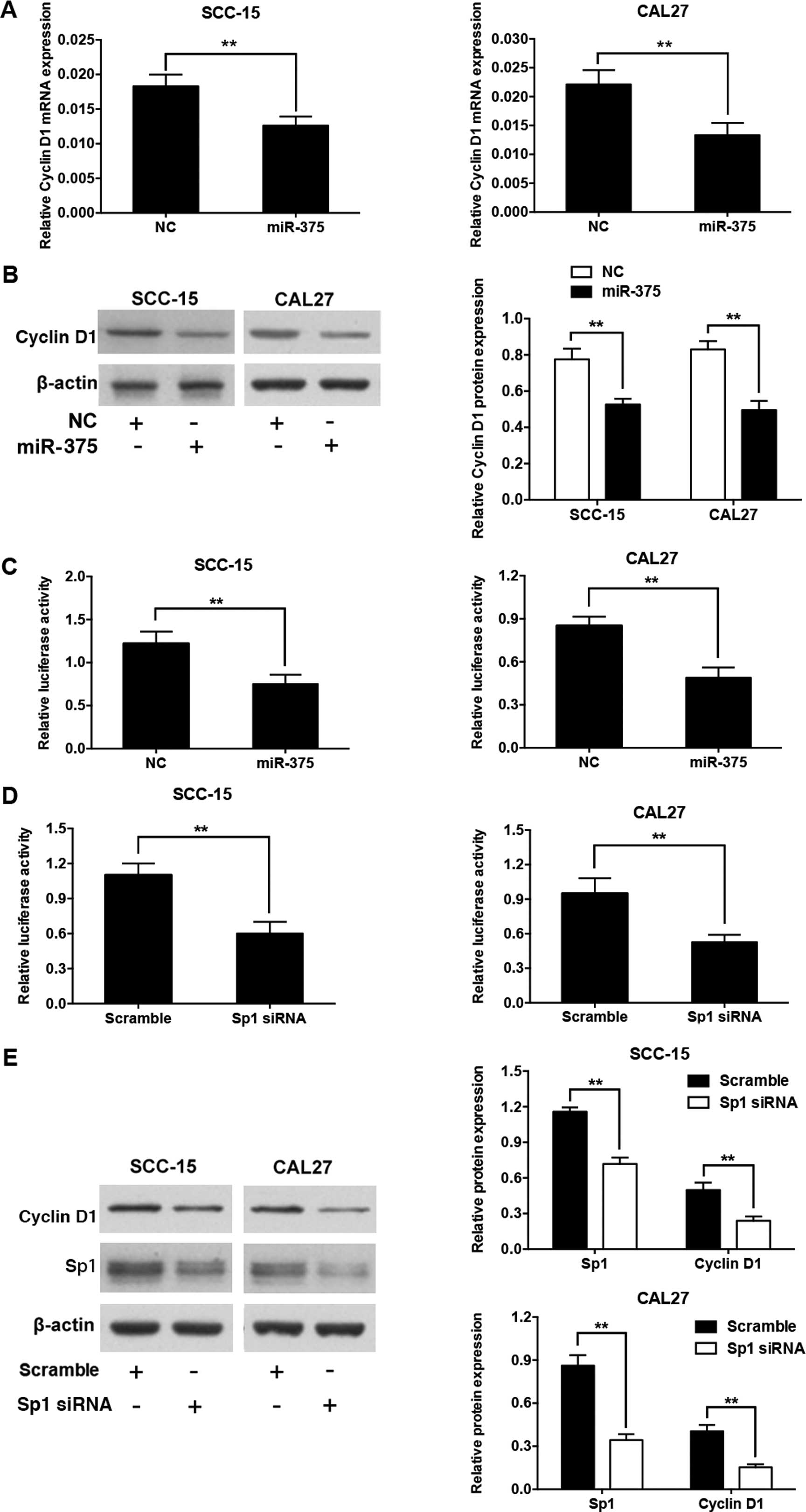
Overexpression of miR-375 downregulates
cyclin D1 expression by targeting Sp1
To explore whether miR-375 also regulates the
expression of cyclin D1, we measured its mRNA and protein levels in
SCC-15 and CAL27 cells transfected with the miR-375 mimic and the
miRNA mimic control. Overexpression of miR-375 significantly
downregulated the mRNA and protein levels of cyclin D1 (Fig. 5A and B). A previous study reported
that Sp1 binding sites in the cyclin D1 promoter are involved in
transcriptional activation of the gene (20). We constructed a luciferase reporter
plasmid containing the cyclin D1 promoter with the Sp1 binding
sites. When SCC-15 or CAL27 cells were co-transfected by the
miR-375 mimic and the cyclin D1 promoter luciferase reporter
plasmid, the luciferase activity level was significantly reduced
compared with the control (Fig.
5C). Moreover, luciferase activity was markedly suppressed by
the introduction of siRNA against the Sp1 transcript into the
SCC-15 and CAL27 cells, which mimicked the effects of miR-375
overexpression (Fig. 5D). In
addition, the transfection of Sp1 siRNA in the SCC-15 or CAL27
cells significantly reduced the protein expression levels of both
Sp1 and cyclin D1, as expected (Fig.
5E).
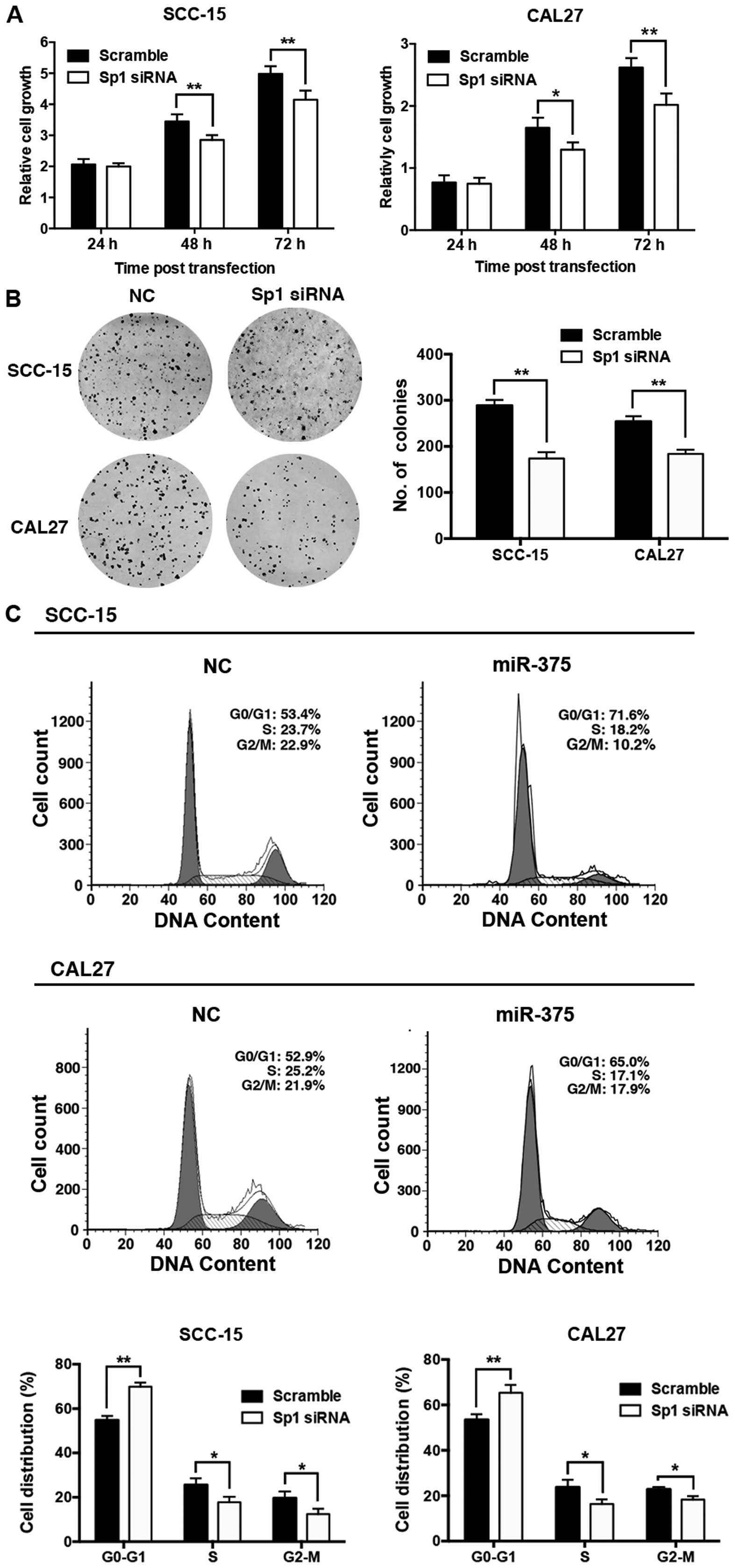
Knockdown of Sp1 inhibits cell
proliferation and cell cycle progression in TSCC cell lines
To test whether the downregulation of Sp1 is
involved in the antitumor actions of miR-375, we knocked down Sp1
expression in the SCC-15 and CAL27 cells. The knockdown of Sp1 in
the TSCC cells suppressed cell proliferation (Fig. 6A) and colony formation (Fig. 6B), and induced a G1 phase
arrest (Fig. 6C).
Discussion
In the present study, we demonstrated that miR-375
expression was reduced in TSCC specimens and cell lines, and was
correlated with tumor size and patient mortality. Moreover, our
results suggest that low expression of miR-375 was correlated with
shorter survival of TSCC patients, and served as a prognostic
factor independent of other clinicopathological factors. Similarly,
a study of 123 HNSCC patients, including patients with cancers of
the oral cavity, oropharynx, and larynx, showed that a low level of
miR-375 was a potential prognostic marker of poor outcome in
patients. However, this study found no significant correlation
between miR-375 and any of the clinicopathological parameters
studied (12). By contrast, a
recent study of 26 OSCC patients, including patients with cancers
of the tongue, gingival and buccal tissue, revealed that miR-375
expression was reduced and was correlated with clinical stage and
tumor size (11). However, the
prognostic implication of miR-375 in oral carcinoma was not
analyzed. Variation in correlation with clinicopathological
parameters and the prognostic significance of miR-375 in previous
studies may be attributable to differences in sample size and the
inclusion of tumors from different subsites of the head and neck.
Thus, our study of 105 patients with HNSCC at a single anatomical
site, the anterior tongue, is valuable for confirming the
prognostic value of miR-375 in TSCC patients.
Previous reports have shown that low miR-375
expression is also related to poor survival and a poor therapeutic
outcome in gastric cancer (24),
esophageal squamous cell carcinoma (17,25),
non-small cell lung cancer (26,27)
and glioma (28). Thus, as a tumor
suppressor, miR-375 seems to be a useful prognostic factor for
various types of malignant tumors. However, upregulation of miR-375
was reported to correlate with a poor prognosis in pediatric acute
myeloid leukemia (29). Variation
in the prognostic significance of miR-375 in previous studies may
be attributable to differences in cancer type.
The present study also demonstrated that
overexpression of miR-375 in TSCC cell lines inhibited cell
proliferation and cell cycle progression, consistent with previous
studies of avian leucosis (30) and
colorectal cancer (31). We focused
on the biological effects of miR-375 on TSCC growth since miR-375
was significantly correlated with tumor size but showed no
relationship with other clinicopathological parameters.
Furthermore, we confirmed that Sp1 is a direct target gene of
miR-375, consistent with a previous study of squamous cervical
cancer (19). Since Sp1 is an
important transcription factor in cellular processes (32–34),
it is reasonable that the downregulation of Sp1 contributes to the
inhibition of TSCC cell growth.
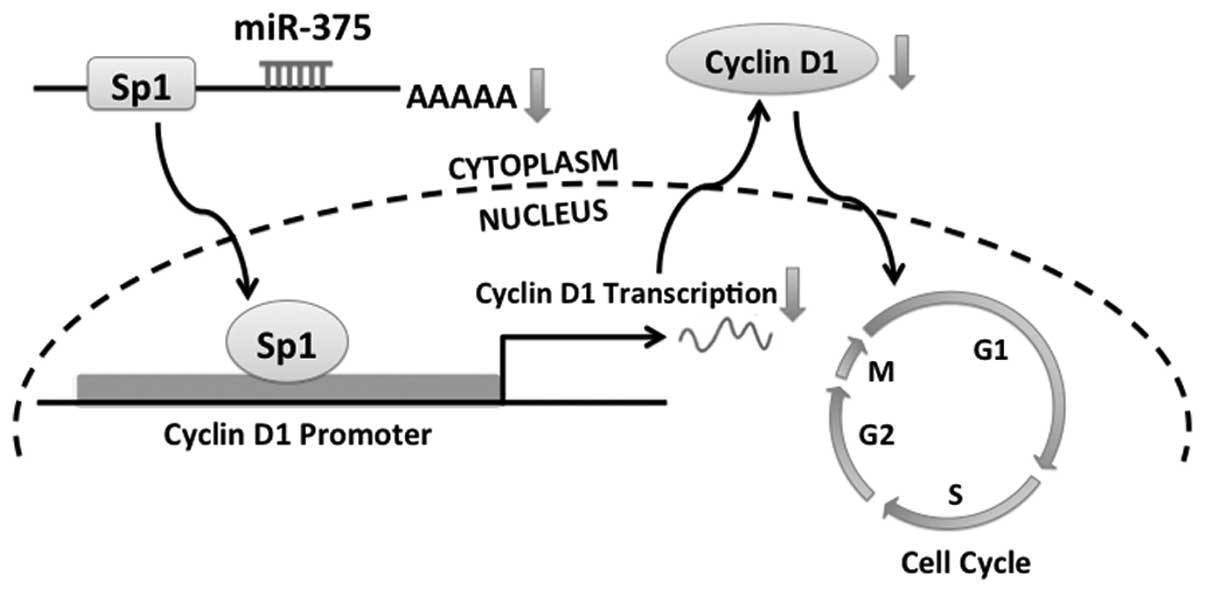
We further demonstrated that overexpression of
miR-375 in TSCC cells downregulated cyclin D1 via Sp1, leading to
cell cycle arrest (Fig. 7). Cyclin
D1 is a key protein involved in cell cycle control and is essential
for G1 to S transition (35,36),
and is regulated by Sp1 (20). More
importantly, several studies have shown that the dysregulation of
cyclin D1 contributes to HNSCC development (37–39)
and our recent study also demonstrated that cyclin D1 plays an
important role in cell growth and cell cycle progression in TSCC
cell lines (8). Therefore, the fact
that miR-375 inhibited cell proliferation and blocked G1
to S transition can at least partially be explained by the
Sp1-dependent downregulation of cyclin D1.
However, as a ubiquitous transcription factor, Sp1
is also reported to regulate cell cycle progression by interaction
with many other genes, including those encoding retinoblastoma
protein (40,41), retinoblastoma-related protein p107
(42), the transcription factor E2F
(43), p53 (44) and mdm2 (45). We cannot exclude the possibility
that miR-375 inhibits TSCC growth by simultaneously regulating
other cell cycle regulatory proteins via Sp1. Further study is
needed to identify other possible signaling pathways downstream of
miR-375 in the suppression of TSCC growth.
In conclusion, miR-375 expression was reduced in
TSCC and its downregulation was correlated with a poor prognosis in
TSCC patients. Overexpression of miR-375 in TSCC cells inhibited
proliferation and induced cell cycle arrest through an Sp1-cyclin
D1 signaling pathway. These results suggest that miR-375 inhibits
cell growth and is correlated with clinical outcomes in TSCC.
Acknowledgements
This study was supported by the National Natural
Science Foundation of China (81402235, 81472527), and the
Foundation of the Peking University School and Hospital of
Stomatology (PKUSS20140104).
References
|
1
|
Leemans CR, Braakhuis BJ and Brakenhoff
RH: The molecular biology of head and neck cancer. Nat Rev Cancer.
11:9–22. 2011. View
Article : Google Scholar
|
|
2
|
Greenlee RT, Hill-Harmon MB, Murray T and
Thun M: Cancer statistics, 2001. CA Cancer J Clin. 51:15–36. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Patel SG and Shah JP: TNM staging of
cancers of the head and neck: striving for uniformity among
diversity. CA Cancer J Clin. 55:242–258. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Iorio MV and Croce CM: MicroRNA
dysregulation in cancer: diagnostics, monitoring and therapeutics.
A comprehensive review. EMBO Mol Med. 4:143–159. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Shah NG, Trivedi TI, Tankshali RA, et al:
Prognostic significance of molecular markers in oral squamous cell
carcinoma: a multivariate analysis. Head Neck. 31:1544–1556. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Inui M, Martello G and Piccolo S: MicroRNA
control of signal transduction. Nat Rev Mol Cell Biol. 11:252–263.
2010. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bartels CL and Tsongalis GJ: MicroRNAs:
novel biomarkers for human cancer. Clin Chem. 55:623–631. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Jia LF, Wei SB, Gong K, Gan YH and Yu GY:
Prognostic implications of micoRNA miR-195 expression in human
tongue squamous cell carcinoma. PLoS One. 8:e566342013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li J, Huang H, Sun L, et al: MiR-21
indicates poor prognosis in tongue squamous cell carcinomas as an
apoptosis inhibitor. Clin Cancer Res. 15:3998–4008. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sun L, Yao Y, Liu B, et al: MiR-200b and
miR-15b regulate chemotherapy-induced epithelial-mesenchymal
transition in human tongue cancer cells by targeting BMI1.
Oncogene. 31:432–445. 2012. View Article : Google Scholar
|
|
11
|
Siow M, Ng LK, Chong VV, et al:
Dysregulation of miR-31 and miR-375 expression is associated with
clinical outcomes in oral carcinoma. Oral Dis. 20:345–351. 2014.
View Article : Google Scholar
|
|
12
|
Harris T, Jimenez L, Kawachi N, et al:
Low-level expression of miR-375 correlates with poor outcome and
metastasis while altering the invasive properties of head and neck
squamous cell carcinomas. Am J Pathol. 180:917–928. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bell RB, Kademani D, Homer L, Dierks EJ
and Potter BE: Tongue cancer: Is there a difference in survival
compared with other subsites in the oral cavity. J Oral Maxillofac
Surg. 65:229–236. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Shaw RJ, McGlashan G, Woolgar JA, et al:
Prognostic importance of site in squamous cell carcinoma of the
buccal mucosa. Br J Oral Maxillofac Surg. 47:356–359. 2009.
View Article : Google Scholar
|
|
15
|
Ding L, Xu Y, Zhang W, et al: MiR-375
frequently downregulated in gastric cancer inhibits cell
proliferation by targeting JAK2. Cell Res. 20:784–793. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhou J, Song S, He S, et al: MicroRNA-375
targets PDK1 in pancreatic carcinoma and suppresses cell growth
through the Akt signaling pathway. Int J Mol Med. 33:950–956.
2014.PubMed/NCBI
|
|
17
|
Kong KL, Kwong DL, Chan TH, et al:
MicroRNA-375 inhibits tumour growth and metastasis in oesophageal
squamous cell carcinoma through repressing insulin-like growth
factor 1 receptor. Gut. 61:33–42. 2012. View Article : Google Scholar
|
|
18
|
Zhao H, Zhu L, Jin Y, Ji H, Yan X and Zhu
X: miR-375 is highly expressed and possibly transactivated by
achaete-scute complex homolog 1 in small-cell lung cancer cells.
Acta Biochim Biophys Sin. 44:177–182. 2012. View Article : Google Scholar
|
|
19
|
Wang F, Li Y, Zhou J, et al: miR-375 is
down-regulated in squamous cervical cancer and inhibits cell
migration and invasion via targeting transcription factor SP1. Am J
Pathol. 179:2580–2588. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Nagata D, Suzuki E, Nishimatsu H, et al:
Transcriptional activation of the cyclin D1 gene is mediated by
multiple cis-elements, including SP1 sites and a cAMP-responsive
element in vascular endothelial cells. J Biol Chem. 276:662–669.
2001. View Article : Google Scholar
|
|
21
|
Grinstein E, Jundt F, Weinert I, Wernet P
and Royer HD: Sp1 as G1 cell cycle phase specific transcription
factor in epithelial cells. Oncogene. 21:1485–1492. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lu Y, Thomson JM, Wong HY, Hammond SM and
Hogan BL: Transgenic over-expression of the microRNA miR-17-92
cluster promotes proliferation and inhibits differentiation of lung
epithelial progenitor cells. Dev Biol. 310:442–453. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jia LF, Wei SB, Gan YH, et al: Expression,
regulation and roles of miR-26a and MEG3 in tongue squamous cell
carcinoma. Int J Cancer. 135:2282–2293. 2014. View Article : Google Scholar
|
|
24
|
Zhang WH, Gui JH, Wang CZ, et al: The
identification of miR-375 as a potential biomarker in distal
gastric adenocarcinoma. Oncol Res. 20:139–147. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li J, Li X, Li Y, et al: Cell-specific
detection of miR-375 downregulation for predicting the prognosis of
esophageal squamous cell carcinoma by miRNA in situ hybridization.
PLoS One. 8:e535822013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Li Y, Jiang Q, Xia N, Yang H and Hu C:
Decreased expression of microRNA-375 in nonsmall cell lung cancer
and its clinical significance. J Int Med Res. 40:1662–1669. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yu H, Jiang L, Sun C, et al: Decreased
circulating miR-375: a potential biomarker for patients with
non-small-cell lung cancer. Gene. 534:60–65. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chang C, Shi H, Wang C, et al: Correlation
of microRNA-375 downregulation with unfavorable clinical outcome of
patients with glioma. Neurosci Lett. 531:204–208. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang Z, Hong Z, Gao F and Feng W:
Upregulation of microRNA-375 is associated with poor prognosis in
pediatric acute myeloid leukemia. Mol Cell Biochem. 383:59–65.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li H, Shang H, Shu D, et al: gga-miR-375
plays a key role in tumorigenesis post subgroup J avian leukosis
virus infection. PLoS One. 9:e908782014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wang Y, Tang Q, Li M, Jiang S and Wang X:
MicroRNA-375 inhibits colorectal cancer growth by targeting PIK3CA.
Biochem Biophys Res Commun. 444:199–204. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Cawley S, Bekiranov S, Ng HH, et al:
Unbiased mapping of transcription factor binding sites along human
chromosomes 21 and 22 points to widespread regulation of noncoding
RNAs. Cell. 116:499–509. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Michinaga S, Ishida A, Takeuchi R and
Koyama Y: Endothelin-1 stimulates cyclin D1 expression in rat
cultured astrocytes via activation of Sp1. Neurochem Int. 63:25–34.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Cram EJ, Liu BD, Bjeldanes LF and
Firestone GL: Indole-3-carbinol inhibits CDK6 expression in human
MCF-7 breast cancer cells by disrupting Sp1 transcription factor
interactions with a composite element in the CDK6 gene promoter. J
Biol Chem. 276:22332–22340. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Baldin V, Lukas J, Marcote MJ, Pagano M
and Draetta G: Cyclin D1 is a nuclear protein required for cell
cycle progression in G1. Genes Dev. 7:812–821. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Motokura T, Keyomarsi K, Kronenberg HM and
Arnold A: Cloning and characterization of human cyclin D3, a cDNA
closely related in sequence to the PRAD1/cyclin D1 proto-oncogene.
J Biol Chem. 267:20412–20415. 1992.PubMed/NCBI
|
|
37
|
Bova RJ, Quinn DI, Nankervis JS, et al:
Cyclin D1 and p16INK4A expression predict reduced survival in
carcinoma of the anterior tongue. Clin Cancer Res. 5:2810–2819.
1999.PubMed/NCBI
|
|
38
|
Michalides R, van Veelen N, Hart A, Loftus
B, Wientjens E and Balm A: Overexpression of cyclin D1 correlates
with recurrence in a group of forty-seven operable squamous cell
carcinomas of the head and neck. Cancer Res. 55:975–978.
1995.PubMed/NCBI
|
|
39
|
Huang SF, Cheng SD, Chuang WY, et al:
Cyclin D1 overexpression and poor clinical outcomes in Taiwanese
oral cavity squamous cell carcinoma. World J Surg Oncol. 10:402012.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chang YC, Illenye S and Heintz NH:
Cooperation of E2F-p130 and Sp1-pRb complexes in repression of the
Chinese hamster dhfr gene. Mol Cell Biol. 21:1121–1131. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kim SJ, Onwuta US, Lee YI, Li R, Botchan
MR and Robbins PD: The retinoblastoma gene product regulates
Sp1-mediated transcription. Mol Cell Biol. 12:2455–2463.
1992.PubMed/NCBI
|
|
42
|
Datta PK, Raychaudhuri P and Bagchi S:
Association of p107 with Sp1: genetically separable regions of p107
are involved in regulation of E2F- and Sp1-dependent transcription.
Mol Cell Biol. 15:5444–5452. 1995.PubMed/NCBI
|
|
43
|
Karlseder J, Rotheneder H and
Wintersberger E: Interaction of Sp1 with the growth- and cell
cycle-regulated transcription factor E2F. Mol Cell Biol.
16:1659–1667. 1996.PubMed/NCBI
|
|
44
|
Gualberto A and Baldwin AS Jr: p53 and Sp1
interact and cooperate in the tumor necrosis factor-induced
transcriptional activation of the HIV-1 long terminal repeat. J
Biol Chem. 270:19680–19683. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Johnson-Pais T, Degnin C and Thayer MJ:
pRB induces Sp1 activity by relieving inhibition mediated by MDM2.
Proc Natl Acad Sci USA. 98:2211–2216. 2001. View Article : Google Scholar : PubMed/NCBI
|