Introduction
Breast cancer is the most common malignancy and is a
leading cause of cancer-related deaths among women, in developed
and developing countries (1).
Advanced and metastatic breast cancer is particularly difficult to
treat, causing the second-leading cause of cancer-related deaths in
women (2). In 2013, ~230,000 women
were estimated to be diagnosed with breast cancer and more than
40,000 succumbed to the disease in the USA (3). Although several types of treatment,
including surgery, radiotherapy, chemotherapy and hormone therapy,
have been designed to treat breast cancer, the success to date is
limited (4). Among the various
types of therapy, systemic chemotherapy is the main treatment for
cancer. However, chemotherapeutic drugs for breast cancer usually
have variable effectiveness with high toxicity to normal tissues,
and breast tumors often develop metastasis and drug resistance
(5). Therefore, searching for
effective regimens with minimal side effects remains the top
priority in breast cancer research.
It has been demonstrated that plant-derived
anticancer drugs are much more effective and have minimal side
effects when compared to synthetic drugs (6,7).
Classic examples of plant-derived anticancer drugs that are
currently in clinical use include vinblastine, vincristine,
paclitaxel, camptothecin, as well as Rhodiola rosea
(6–10).
Salidroside
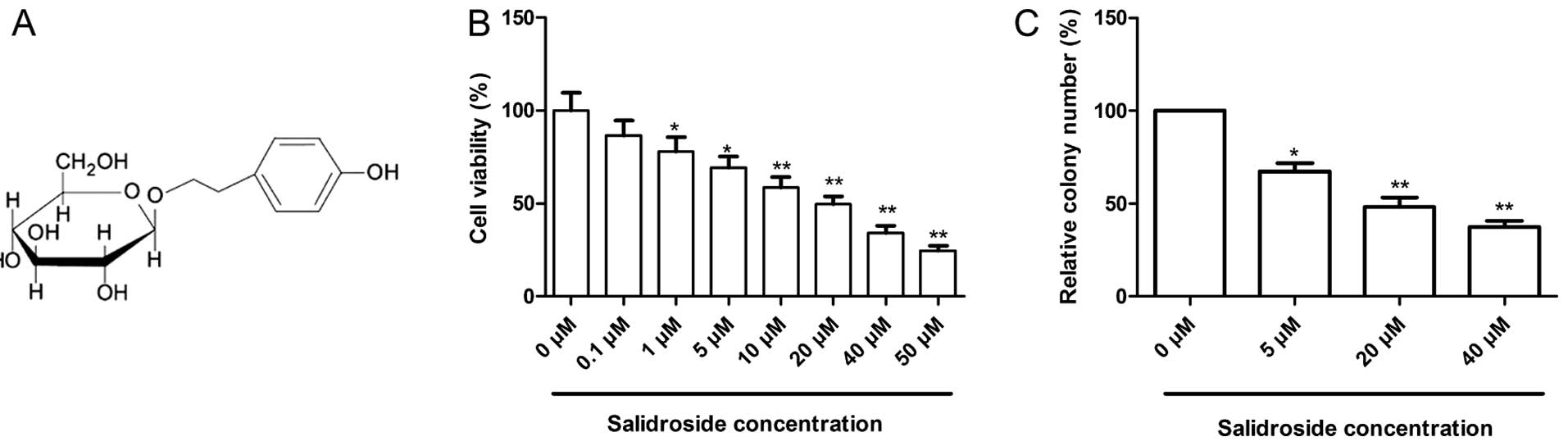
(p-hydroxyphenethyl-β-D-glucoside)(chemical structure shown
in Fig. 1A) is the main active
ingredient found in Rhodiola rosea L, and has displayed many
pharmacological properties including anti-aging, anti-fatigue,
antioxidant, antiviral and anti-inflammatory effects, as well as
neuroprotective and cardiovascular protective effects (11–18).
Recently, salidroside has been found to inhibit cell proliferation,
and induce cell apoptosis in lung (19) and bladder cancer (20), neuroblastoma (21) and glioma (22) in vitro. Regarding breast
cancer, despite a recent report showing that salidroside induces
cell cycle arrest and apoptosis in human breast cancer cells
(23), the potential role of
salidroside against breast cancer cell migration and invasion in
vitro, and tumor growth ability in vivo have not been
fully clarified. More significantly, the underlying mechanism of
the anticancer effect of salidroside remains largely unknown.
The aim of the present study was to evaluate the
potency of salidroside in inhibiting breast cancer cell
proliferation, colony formation, migration and invasion in
vitro and to reveal the underlying molecular mechanisms
involved in the anticancer effects. In addition, tumor growth
ability in nude mice was detected to define the salidroside
treatment effect on tumorigenesis in vivo.
Materials and methods
Reagents and antibodies
Salidroside (purity, >99%) was purchased from the
National Institute of Pharmaceutical and Biological Products
(Beijing, China). Salidroside was dissolved in water and filtered
through a 0.22-μm filter before use. Propidium iodide (PI)
and 3-(4,5-dimethylthiazol-2-yl)-2,5 diphenyltetrazolium bromide
(MTT) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Stock
solutions of PI and MTT were prepared by dissolving 1 mg of each
compound in 1 ml of phosphate-buffered saline (PBS). The solution
was protected from light, stored at 4°C, and used within 1
month.
For western blot analysis, the following antibodies
were used: mouse monoclonal anti-human β-actin (Sigma-Aldrich),
mouse monoclonal anti-human Bcl2, mouse monoclonal anti-human Bax,
mouse monoclonal anti-human p21, mouse monoclonal anti-human cyclin
D1, mouse monoclonal anti-human cyclin D3, mouse monoclonal
anti-human matrix metalloproteinase (MMP)-2, mouse monoclonal
anti-human MMP-9, mouse monoclonal anti-human p38MAPK, mouse
monoclonal anti-human phosphorylated (p)-p38MAPK, mouse monoclonal
anti-human c-Jun N-terminal kinase (JNK), mouse monoclonal
anti-human p-JNK, mouse monoclonal anti-human ERK1/2 and mouse
monoclonal anti-human p-ERK1/2 (all from Santa Cruz Biotechnology,
Santa Cruz, CA, USA). Secondary antibody HRP-conjugated goat
anti-mouse IgG was purchased from Amersham Biosciences (Uppsala,
Sweden).
Cell lines and culture
Human breast cancer cell line, MCF-7, was purchased
from the Cell Bank of the Type Culture Collection of the Chinese
Academy of Sciences, Shanghai Institute of Cell Biology, Chinese
Academy of Sciences, (Shanghai, China) and cultured in Dulbecco’s
modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine
serum (FBS) (both from Gibco-BRL, Gaithersburg, MD, USA), 100 U/ml
of penicillin and 0.1 mg/ml of streptomycin at 37°C in a humidified
atmosphere of 5% CO2.
MTT assay
Cell proliferation was determined by MTT assay as
previously described (24).
Briefly, 1×104 cells/well were cultivated in 100
μl of culture medium in 96-well flat-bottomed plates
(Corning Inc., Corning, NY, USA) and incubated with different
concentrations of salidroside (0.01–50 μM) for 72 h,
followed by the addition of 10 μl of MTT solution (5 mg/ml,
dissolved in PBS; Sigma-Aldrich). After a 4-h incubation, 100
μl of SDS (10%, w/v, dissolved in 0.01 M HCl; Sigma-Aldrich)
was added and mixed thoroughly to dissolve the formazan crystals at
37°C. After shaking the plates for 10 min, the absorbance was read
at 570 nm in an ELISA plate reader (Molecular Devices Corp.,
Sunnyvale, CA, USA).
Colony formation assay
Cells were seeded in 6-well culture plates at a
density of 1×104 cells/well. After being cultured for 24
h, the cells were treated with different concentrations of
salidroside (0, 5, 20 and 40 μM). After 14 days, the cells
were washed, fixed by paraformaldehyde, and stained with Giemsa for
10 min. Then extra Giemsa was washed 3 times by ddH2O,
and the colonies were photographed using a digital camera. The
visible colonies in each group were counted.
Quantitative analysis of apoptotic cells
by Annexin V/PI staining
Apoptotic cell death induced by salidroside was
quantified by flow cytometry using the Annexin V-fluorescein
isothiocyanate (FITC) kit following the manufacturer’s
instructions. Briefly, the cells were plated at a density of
3×105 cells/well in a 6-well plate and incubated with
different concentrations of salidroside (0, 5, 20 and 40 μM)
for 48 h. Floating cells as well as residual attached cells were
collected and washed twice with PBS. The cell pellets were
resuspended in 500 μl of 1X binding buffer at a
concentration of 1×106 cells/ml. Five microliters of
Annexin V-FITC and PI was added to the cell suspension for 10 min
at room temperature, stained samples were examined using a Coulter
Epics XL flow cytometer (Beckman Coulter, Miami, FL, USA), and the
data were analyzed using CellQuest software (BD Biosciences, San
Jose, CA, USA). Experiments were performed in triplicate. In
addition, we also detected Bax and Bcl-2 expression by western
blotting as an additional indicator of apoptosis.
Measurement of caspase activity
Caspase-3, -8 and -9 activity was measured using the
Caspase Colorimetric Assay kit (Millipore Corporation, Billerica,
MA, USA) according to the manufacturer’s instructions. Briefly, the
cells after treatment for 48 h were harvested and lysed in lysis
buffer on ice for 10 min and then centrifuged at 10,000 × g for 1
min. After centrifugation, the supernatants were incubated with
caspase-3, -8 and -9 substrates in reaction buffer. Samples were
seeded into a 96-well flat-bottom microplate at 37°C for 1 h.
Samples were analyzed at 405 nm in a microplate reader (Thermo
Fisher Scientific Inc., Waltham, MA, USA). The relative caspase
activity of the untreated group (0 μM salidroside treatment)
was taken as 100. Each assay was conducted in triplicate.
Cell cycle analysis
To determine the cell cycle distribution, the cells
were plated in 60-mm dishes and treated with different
concentrations of salidroside (0, 5, 20 and 40 μM) for 48 h.
After treatment, the cells were collected by trypsinization, fixed
in 70% ethanol, and kept at −20°C overnight for fixation. Cells
were washed twice with PBS, and then resuspended in 1 ml of PBS
containing RNase (100 μg/ml) and PI (40 μg/ml) in the
dark for 30 min at room temperature. The distribution of cells in
the cell cycle phases was analyzed from the DNA histogram with a
FACS Caliber flow cytometer (Becton-Dickinson, San Jose, CA, USA).
The data were analyzed using CellQuest software (BD Biosciences).
Furthermore, we also detected p21, cyclin D1 and cyclin D3
expression by western blotting as an additional indicator of cell
cycle arrest.
Transwell migration and invasion
assays
To assess the effect of salidroside on cell
migration and invasion, the migration and invasion assays were
performed using Transwell insert chambers (Corning Inc.). For the
migration assay, the cells were incubated with different
concentrations of salidroside (0, 5, 20 and 40 μM) for 48 h.
After treatment, a total of 1×105 cells were plated into
the upper chamber in serum-free DMEM. Medium containing 20% FBS in
the lower chamber served as chemoattractant. After being cultured
for 24 h, the media were removed from the upper chamber by wiping
with a cotton swab and cells that migrated to the lower surface of
the filter were fixed in 70% ethanol for 30 min followed by
staining with 0.2% crystal violet for 10 min. Cell migration was
counted by counting five random fields per filter under a light
microscope (Olympus, Tokyo, Japan).
For the invasion assay, after treatment,
3×105 cells were incubated in the upper chambers
pre-coated with Matrigel (BD Biosciences) in serum-free DMEM, and
the subsequent steps were consistent with the migration assay. The
number of cells invading the Matrigel was counted in five randomly
selected fields using a light microscope (Olympus). All experiments
were performed in triplicate.
Measurement of intracellular reactive
oxygen species (ROS)
Intracellular changes in ROS were determined using
the MGT Live Cell Fluorescent ROS Detection kit (MGT, Inc., USA) as
described previously (24).
Briefly, cells were plated in a 96-well plate (25×103
cells/well) and were treated with different concentrations of
salidroside (0, 5, 20 and 40 μM) for 48 h. After treatment,
the cells were further incubated with 20 μM
2′,7′-dichlorofluorescein diacetate in Hank’s balanced salt
solution (HBSS) at 37°C for 30 min in the dark. Subsequently, the
cells were harvested and washed with HBSS and analyzed for DCF
fluorescence using a Synergy HT Multi-Mode microplate reader
(BioTek Instruments, San Jose, CA, USA).
Western blot analysis
The cells were collected by trypsinization and lysed
in radioimmunoprecipitation assay lysis buffer (Sigma-Aldrich) with
the addition of protease inhibitors (Roche) and phosphatase
inhibitors (Sigma-Aldrich) for 30 min on ice. Then the homogenates
were centrifuged at 14,000 rpm at 4°C for 30 min to remove
insoluble material, and the supernatants were collected for protein
concentration determination using the BCA protein assay kit
(Sigma-Aldrich). The cell extracts (20 μg of protein) were
separated on a sodium dodecyl sulfate-polyacrylamide
electrophoretic gel (SDS-PAGE) and transferred to nitrocellulose
membranes. The membranes were blocked with 5% dry milk in PBS and
incubated with the primary antibodies. After incubation with
primary antibodies, the membranes were washed in PBS and incubated
with secondary horseradish peroxidase-coupled goat anti-mouse
antibodies. The proteins were detected and protein bands were
visualized with enhanced chemiluminescence reagent (ECL;
Sigma-Aldrich). The integrated density value (IDV) was analyzed
with a computerized image analysis system (Fluor Chen 2.0) and
normalized with that of β-actin.
Tumor xenografts in nude mice
Female BALB/c nude mice at 6–7 weeks of age were
obtained from the Experimental Animal Center of Jilin University
(Changchun, China) and were housed under standard conditions. MCF-7
cells were trypsinized and washed with PBS and suspended in
DMEM-free serum. A total of 2×106 cells were injected
into the flanks of nude mice. Tumor growth was measured every 7
days, and tumor volume was estimated as length × width × height ×
0.5236. When tumors grew to an average volume of 100
mm3, the mice were divided randomly into 2 groups (10
mice/group). The control group received 1% PBS in deionized water.
The treatment group was treated with salidroside (50 mg/kg body
weight) intraperitoneally on alternate days for 3 weeks. The doses
were selected based on a previous experiment (22). The tumor size was measured using a
caliper on days 7, 14 and 21 of treatment. On day 21, the animals
were sacrificed using chloroform, and tumor tissues were isolated
and weighed. In addition, spleen tissues were collected and
cultured for a splenocyte surveillance study to assay splenocyte
proliferation as previously described (25). All procedures were in agreement with
Jilin University Guide for the Care and Use of Laboratory Animals
and were approved by the Animal Care and Use Committee, Jilin
University (Changchun, China).
Statistical analysis
All experiments were performed at least three
independent times, and the results are expressed as the mean ±
standard deviation (SD). For statistical comparison of quantitative
data between groups, analysis of variance (ANOVA) or Student’s
t-test was performed. All statistical analyses were performed using
the GraphPad Prism version 5.01 (GraphPad Software, San Diego, CA,
USA) and the SPSS software (version 16.0; SPSS Inc., Chicago, IL,
USA) for Windows®. P-values <0.05 were considered to
be statistically significant.
Results
Salidroside inhibits proliferation and
colony formation in MCF-7 cells
To determine the cytotoxic effects of salidroside on
MCF-7 cells, MTT assay was carried out. It was found that
salidroside significantly inhibited the viability of the MCF-7
cells in a dose-dependent manner (Fig.
1B). In the MCF-7 cells, as shown in Fig. 1B, a significant inhibitory effect
was observed at 1 μM, which reached a maximum level at 50
μM. The IC50 value (the effective dose that
inhibits 50% of growth) for the treatment of MCF-7 cells by
salidroside was 19.48 (P<0.05). Based on the results, we chose
concentrations of 5, 20 (IC50) and 40 μM (2x
IC50) salidroside for further treatments throughout the
study.
Next, the effect of salidroside on the cell colony
formation of MCF-7 cells was also analyzed. As shown in Fig. 1C, salidroside significantly
inhibited the colony formation of MCF-7 cells in a dose-dependent
manner (P<0.05).
Salidroside induces apoptosis in MCF-7
cells
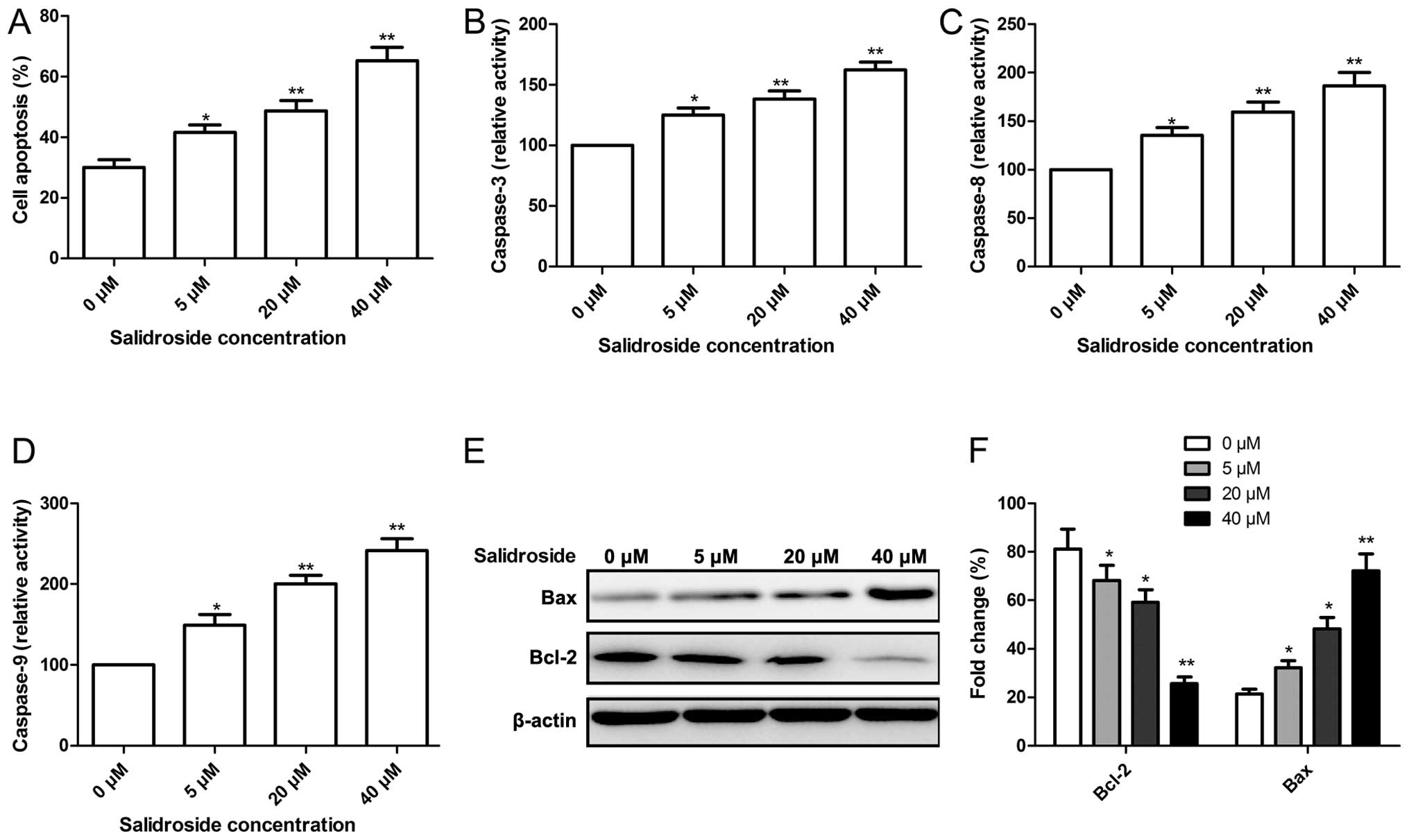
To determine whether the growth inhibitory effect of
salidroside is associated with the induction of apoptotic cell
death, flow cytometry was performed. MCF-7 cells were exposed to
various concentrations (0, 5, 20 and 40 μM) of salidroside
for 48 h and analyzed by flow cytometry. As shown in Fig. 2A, we observed a dose-dependent
increase in apoptotic cells in the presence of salidroside in MCF-7
cells.
To examine the contribution of caspases in the
salidroside-induced apoptosis, the role of caspase-3, -8 and -9 was
investigated. The results demonstrated that treatment of MCF-7
cells with salidroside resulted in a significant increase in the
activities of caspase-3, -8 and -9 in a dose-dependent manner
(Fig. 2B–D).
To determine the potential mechanism involved in the
effect on cell apoptosis by salidroside, expression levels of
apoptosis-related proteins, Bax and Bcl-2, were examined by western
blot analysis. Western blot analysis displayed a significant
upregulation of Bax expression, and downregulation of Bcl-2
expression in the MCF-7 cells following treatment with salidroside
in a dose-dependent manner (P<0.05, Fig. 2E and F).
Salidroside induces G0/G1 phase cell
cycle arrest in MCF-7 cell
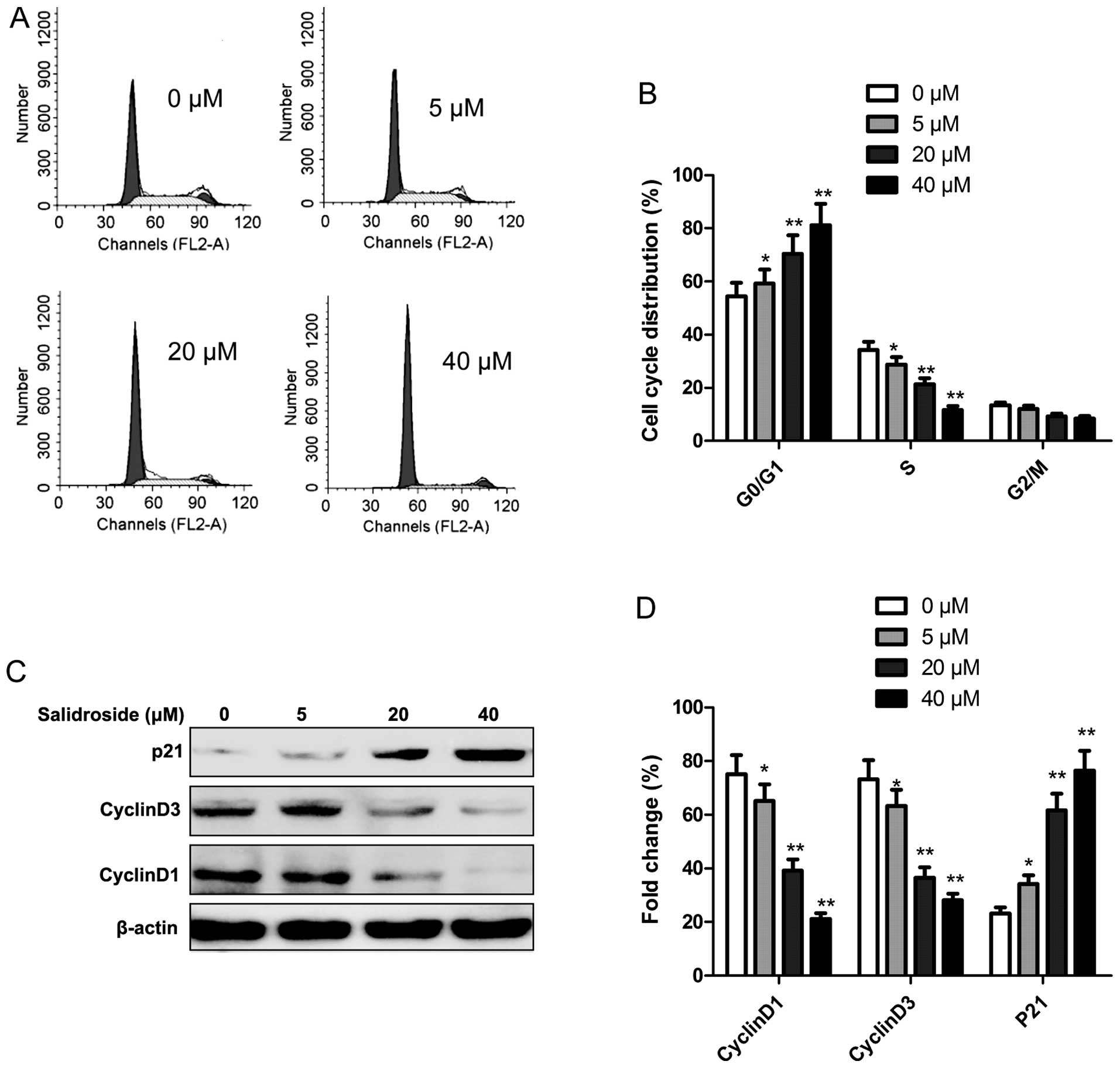
To determine whether the growth inhibitory effect of
salidroside is associated with cell cycle arrest, flow cytometry
was performed to determine the cell cycle distribution. The MCF7
cells were treated with various concentrations of salidroside (0,
5, 20 and 40 μM) for 48 h. It was found that salidroside
significantly increased the percentage of cells in the G0/G1 phase
in a dose-dependent manner (P<0.05), while the percentage of
cells in the S phase significantly decreased following salidroside
treatment (P<0.05, Fig. 3A and
B).
Next, we analyzed the effects of salidroside on the
expression of cell cycle-associated proteins, such as cyclin D1,
cyclin D3 and p21 by western blotting. As shown in Fig. 3C and D, salidroside treatment
markedly increased p21 expression, while cyclin D1 and cyclin D3
expression was significantly decreased in the MCF-7 cells in
dose-dependent manner (P<0.05).
Salidroside inhibits cell migration and
invasion in MCF-7 cells
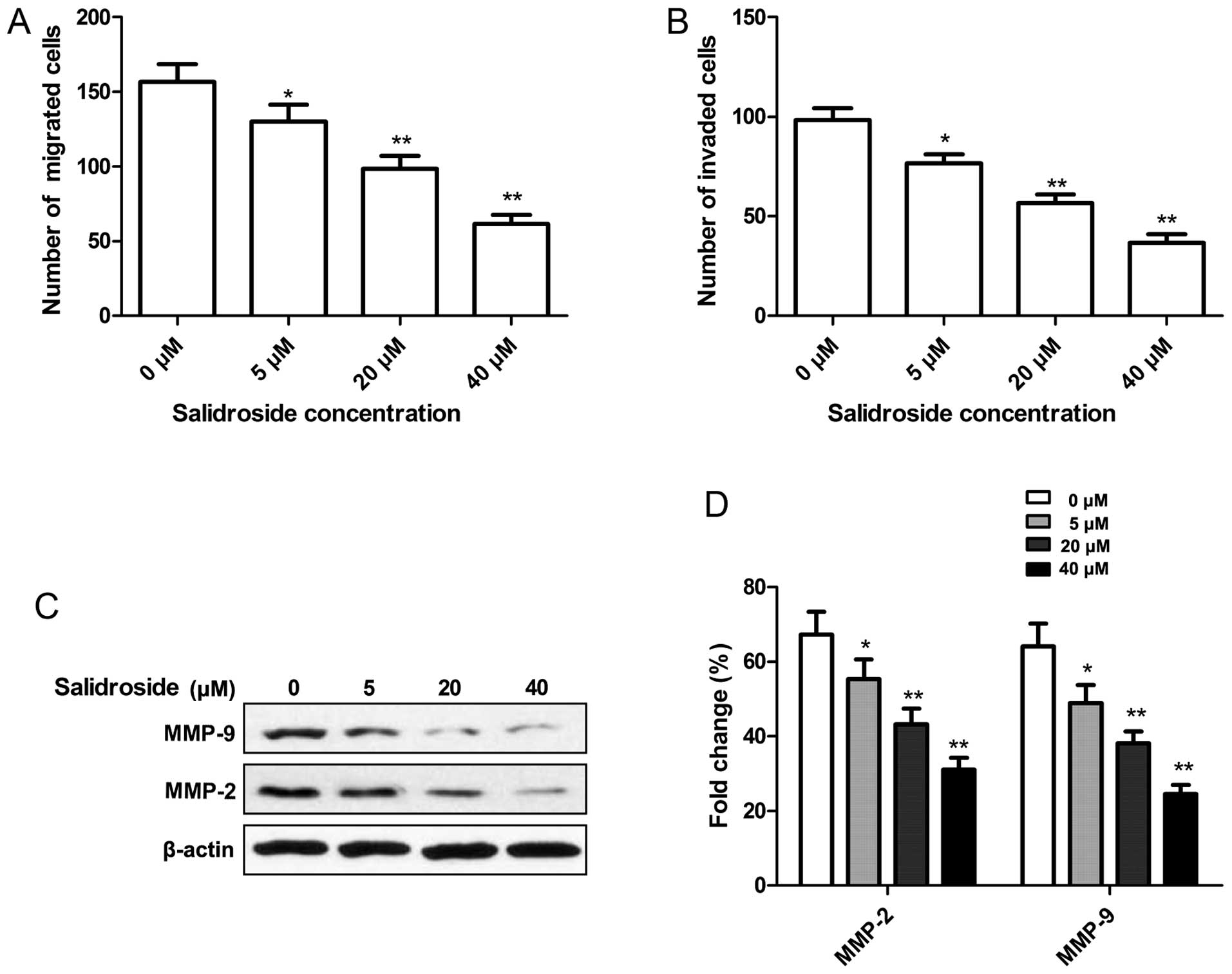
We were particularly interested in whether
salidroside affects cell vitality, demonstrated by migration or
invasion activity. Thus, cell migration and invasion assays were
performed by Transwell assay. It was found that salidroside
significantly decreased the migration of MCF-7 cells in a
dose-dependent manner (P<0.05, Fig.
4A). The ability of salidroside to reduce the invasiveness of
MCF-7 cells was further investigated. Transwell matrix penetration
(coated with Matrigel) assay showed that salidroside markedly
reduced the invasiveness of MCF-7 cells in dose-dependent manner
(P<0.05, Fig. 4B).
To determine the potential mechanism involved in the
effect on cell migration and invasion by salidroside, expression of
MMP-2 and MMP-9 protein was determined by western blotting. Western
blot analysis showed that salidroside significantly reduced MMP-2
and MMP-9 expression in the MCF-7 cells in a dose-dependent manner
(P<0.05, Fig. 4C and D).
Effect of salidroside on the
intracellular ROS formation and MAPK signaling pathway in MCF-7
cells
ROS are produced particularly when cells undergo
chemical or environmental stress and could be one of the factors
leading to cell apoptosis. Therefore, the ROS level after a 48-h
treatment with salidroside was examined in the breast cancer cells
by the fluorescent probe DCFH-DA. It was found that salidroside
treatment significantly reduced the intracellular ROS level in the
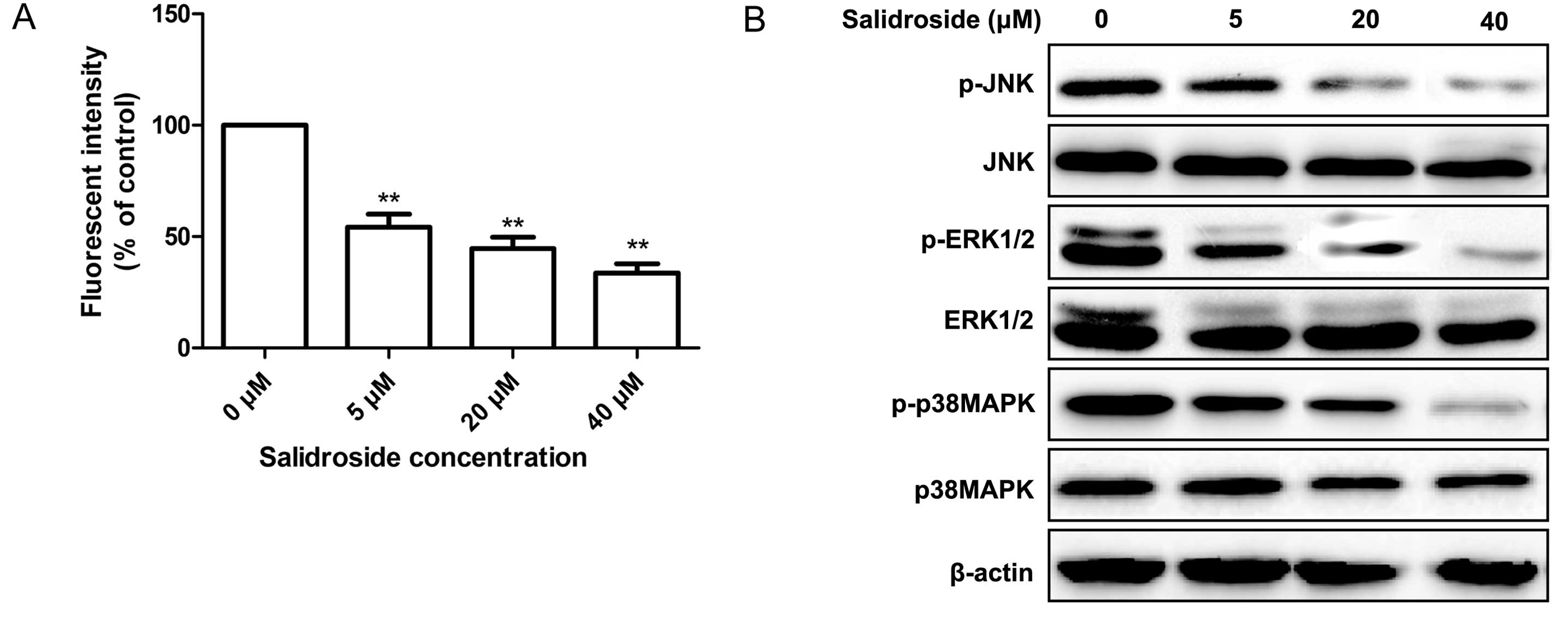
MCF-7 cells in a dose-dependent manner (Fig. 5A).
It is well known that the MAPK signaling pathway
plays a crucial role in cell proliferation and survival in various
cancers. In addition, ROS production has been shown to be coupled
with the sustained activation of the MAPK signaling pathway for a
variety of cellular effects (26).
Therefore, in the present study, we next evaluated the effect of
salidroside on several key downstream molecules involved in the
MAPK signaling pathway. Measurements of the
phosphorylation/activation pattern of p38MAPK, ERK1/2 and JNK were
performed by western blotting 12 h after treatment with
salidroside. It was found that salidroside treatment resulted in a
marked reduction in phosphorylated p38MAPK, ERK1/2 and JNK in the
MCF-7 cells in a dose-dependent manner, without altering the total
protein levels of p38MAPK, ERK1/2 and JNK at different
concentrations of salidroside (Fig.
5B).
Salidroside suppresses tumor growth in a
nude mouse model
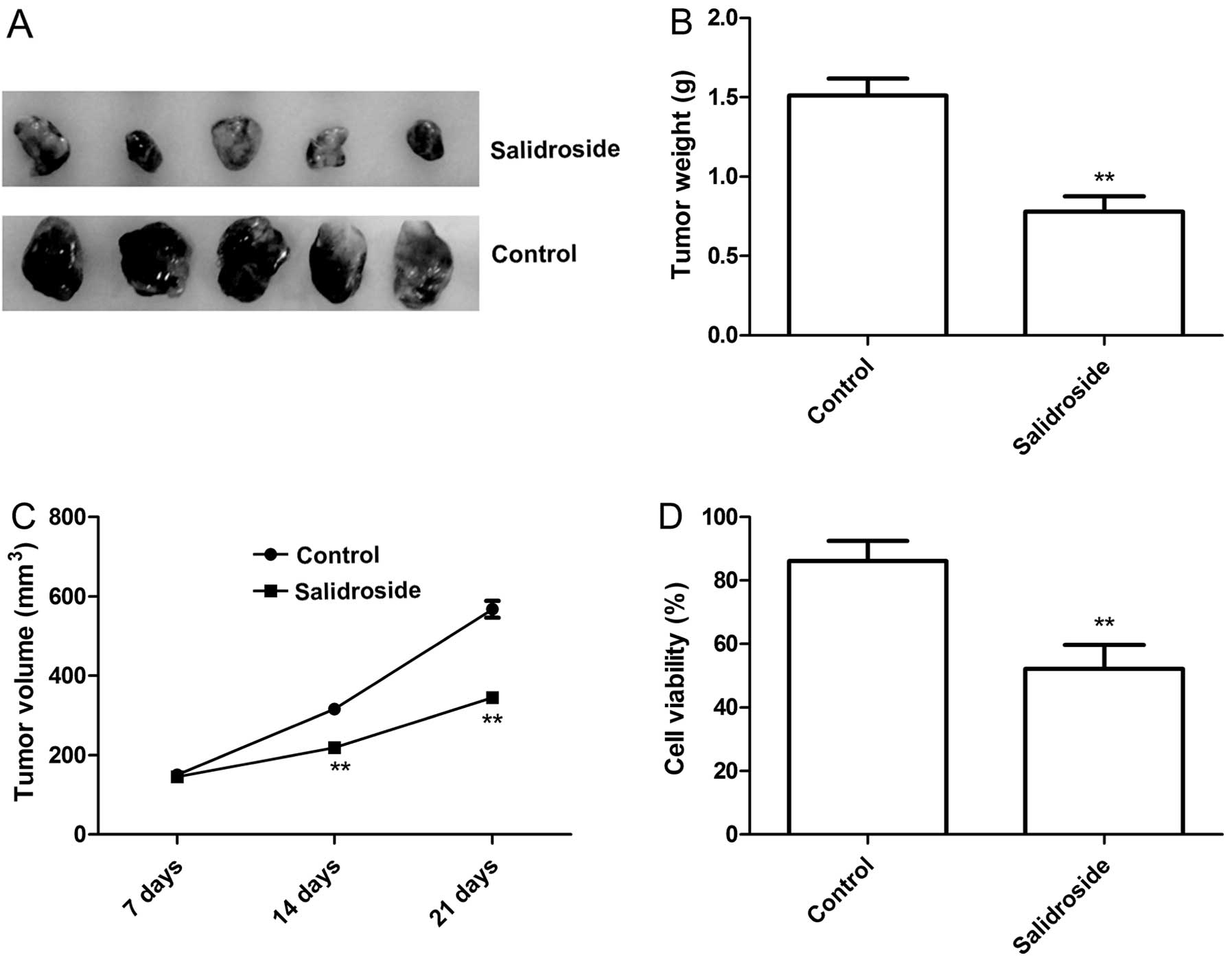
We aimed to ascertain whether salidroside treatment
inhibits tumor growth in a xenograft tumor model. Tumor growth was
monitored for 3 weeks. On day 21, mice were sacrificed, and the
tumor weight was measured. It was found that tumor weight were
significantly lower in the salidroside treatment group relative to
the control group (untreated group) (P<0.05, Fig. 6A and B). In addition, tumor volume
was also determined at different times. The tumor volume in the
salidroside treatment group was significantly diminished when
compared with that in the control group (Fig. 6A and C). In addition, we employed
MTT assays in modulating splenocyte proliferation to demonstrate
the antitumor activities of salidroside in vivo. It was
found that splenocyte cell proliferation in the salidroside
treatment group significantly decreased relative to the control
group (P<0.05, Fig. 6D). These
data suggest that salidroside treatment suppresses the tumor growth
of breast cancer in vivo.
Discussion
Although recent advances in diagnosis and treatment,
breast cancer mortality rates remain high. Conventional anticancer
chemotherapy are associated with occurrence of side effects induced
by the non-specific targeting of both normal and cancer cells
(27). Therefore, development of
novel agents for the prevention and treatment of human breast
cancer is urgently needed. As a valuable source for novel
chemotherapeutic agents, natural plant compounds exhibit effective
anticancer activities with few side effect (6,28).
Salidroside is a phenylpropanoid glycoside isolated from a popular
traditional Chinese medicinal plant, Rhodiola rosea L, and
has been proven to induce cell apoptosis and cell cycle arrest in
breast cancer cells (23), while
the effect of salidroside on cancer cell migration and invasion has
not yet been reported. The present study demonstrated for the first
time that salidroside treatment effectively inhibited the migration
and invasion of MCF-7 cells, and suppressed the tumor growth of
breast cancer in vivo.
Growing evidence has shown that inhibition of
anti-apoptotic members or activation of pro-apoptotic members of
the Bcl-2 family usually leads to an altered mitochondrial membrane
permeability, which allows the release of cytochrome c into
the cytosol and the subsequent activation of caspase-3 and -9,
leading to apoptotic cell death (29). Caspases (a family of proteases) are
one of the essential executioners of apoptosis, and their cleavage
and subsequent activation are considered as the primary hallmarks
of apoptosis (30). To understand
the potential antitumor mechanisms, the relative levels of
expression of Bcl-2 and Bax following salidroside treatment were
detected by western blot analysis, and the caspase-3, -8 and -9
activities were measured by a caspase kit. The present study showed
that salidroside downregulated the expression of Bcl-2, and
upregulated the expression of Bax, as well as increased caspase-3,
-8 and -9 activity in the MCF-7 cells in a concentration-dependent
manner (P<0.05). These results imply that salidroside induced
cell apoptosis by inhibiting Bcl-2 expression, leading to
activation of caspase-3, -8 and -9.
MMPs are the main family of proteolytic enzymes that
facilitate tumor cell migration by degrading the basement membrane,
and other components of the extracellular matrix (ECM) play a key
role in tumor cell invasion, migration and tumor angiogenesis
(31,32). MMP-2 and MMP-9 are important members
of the MMP family, and downregulation of the expression of MMP-2
and MMP-9 contributes to inhibit cancer cell invasion and
metastasis (33,34). Recently a report showed that
salidroside significantly suppressed MMP-2 and MMP-9 activity, and
increased tissue inhibitor of metalloproteinase-2 (TIMP-2)
expression in a dose-dependent manner in HT1080 cells (35). Consistent with this result, our
present study demonstrated that salidroside decreased MMP-2 and
MMP-9 activity in MCF-7 cells. Thus, salidroside may suppress MMP-2
and MMP-9 activity to reduce the metastatic capabilities of MCF-7
cells.
The MAPK signaling cascade, including ERK1/2, JNK,
and p38, has been reported to play a crucial role in signal
transduction and mediates cellular proliferation, differentiation,
inflammation and apoptosis (36).
It was known that a high level of ROS destroys the integrity of the
plasma membrane, affects the dynamics of the actin cytoskeleton,
and causes DNA damage, cumulatively known as oxidative stress,
leading to disruptions in normal mechanisms of cellular signaling
(37,38). ROS production has been shown to be
coupled with the sustained activation of the ERK signaling pathway
for a variety of cellular effects, such as apoptosis and
phagocytosis (26). Recently, Wang
et al reported that salidroside inhibited the intracellular
ROS formation and phosphorylation of p38 in A549 cells in a
dose-dependent manner (19).
Panossian et al found that salidroside reduced the
phosphorylation of JNK to fight against fatigue and stress in
rabbits (39). Sun et al
showed that salidroside treatment significantly decreased the
intracellular ROS level and inhibited phosphorylation of ERK1/2
expression in HT1080 cells (35).
Consistent with these results, we showed that salidroside treatment
inhibited the intracellular ROS formation, and suppressed
phosphorylation of p38, JNK and ERK1/2 expression in MCF-7 cells in
a dose-dependent manner. These findings suggest that salidroside
treatment inhibited human breast cancer growth, which may be due to
its downregulation of intracellular ROS formation and attenuation
of the activation of the MAPK family signaling pathway.
In conclusion, the findings of the present study
provide evidence that salidroside significantly inhibits breast
cell proliferation, migration and invasion, and induces cancer cell
apoptosis in vitro, as well as suppresses tumor growth in
vivo. In addition, salidroside treatment significantly
inhibited the intracellular ROS formation and MAPK pathway
activation, which may contribute to the inhibition of tumor growth
and decreased oxidative stress. Thus, salidroside may be a
promising natural compound for human breast cancer chemoprevention
or chemotherapy.
References
|
1
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2013. CA Cancer J Clin. 63:11–30. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Clark O, Botrel TE, Paladini L and
Ferreira MB: Targeted therapy in triple-negative metastatic breast
cancer: a systematic review and meta-analysis. Core Evid. 9:1–11.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
DeSantis C, Ma J, Bryan L and Jemal A:
Breast cancer statistics, 2013. CA Cancer J Clin. 64:52–62. 2014.
View Article : Google Scholar
|
|
4
|
Murphy IG, Dillon MF, Doherty AO,
McDermott EW, Kelly G, O’Higgins N and Hill AD: Analysis of
patients with false negative mammography and symptomatic breast
carcinoma. J Surg Oncol. 96:457–463. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Nicolin V, Fancellu G and Valentini R:
Effect of tanshinone II on cell growth of breast cancer cell line
type MCF-7 and MD-MB-231. Ital J Anat Embryol. 119:38–43.
2014.PubMed/NCBI
|
|
6
|
Butler MS: The role of natural product
chemistry in drug discovery. J Nat Prod. 67:2141–2153. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Paterson I and Anderson EA: Chemistry. The
renaissance of natural products as drug candidates. Science.
310:451–453. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Maison W: Natural products research:
renaissance with strengthened integration of biology and chemistry.
Angew Chem Int Ed Engl. 45:3000–3002. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Xu YC, Wang HX, Tang L, Ma Y and Zhang FC:
A systematic review of vinorelbine for the treatment of breast
cancer. Breast J. 19:180–188. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Loo WT, Jin LJ, Chow LW, Cheung MN and
Wang M: Rhodiola algida improves chemotherapy-induced oral
mucositis in breast cancer patients. Expert Opin Investig Drugs.
19(Suppl 1): S91–S100. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Xu MC, Shi HM, Wang H and Gao XF:
Salidroside protects against hydrogen peroxide-induced injury in
HUVECs via the regulation of REDD1 and mTOR activation. Mol Med
Rep. 8:147–153. 2013.PubMed/NCBI
|
|
12
|
Zhu Y, Shi YP, Wu D, Ji YJ, Wang X, Chen
HL, Wu SS, Huang DJ and Jiang W: Salidroside protects against
hydrogen peroxide-induced injury in cardiac H9c2 cells via PI3K-Akt
dependent pathway. DNA Cell Biol. 30:809–819. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tan CB, Gao M, Xu WR, Yang XY, Zhu XM and
Du GH: Protective effects of salidroside on endothelial cell
apoptosis induced by cobalt chloride. Biol Pharm Bull.
32:1359–1363. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yuan Y, Wu SJ, Liu X and Zhang LL:
Antioxidant effect of salidroside and its protective effect against
furan-induced hepatocyte damage in mice. Food Funct. 4:763–769.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chen X, Zhang Q, Cheng Q and Ding F:
Protective effect of salidroside against
H2O2-induced cell apoptosis in primary
culture of rat hippocampal neurons. Mol Cell Biochem. 332:85–93.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang H, Ding Y, Zhou J, Sun X and Wang S:
The in vitro and in vivo antiviral effects of salidroside from
Rhodiola rosea L. against coxsackievirus B3. Phytomedicine.
16:146–155. 2009. View Article : Google Scholar
|
|
17
|
Chen X, Liu J, Gu X and Ding F:
Salidroside attenuates glutamate-induced apoptotic cell death in
primary cultured hippocampal neurons of rats. Brain Res.
1238:189–198. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wu T, Zhou H, Jin Z, Bi S, Yang X, Yi D
and Liu W: Cardio-protection of salidroside from
ischemia/reperfusion injury by increasing N-acetylglucosamine
linkage to cellular proteins. Eur J Pharmacol. 613:93–99. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wang J, Li JZ, Lu AX, Zhang KF and Li BJ:
Anticancer effect of salidroside on A549 lung cancer cells through
inhibition of oxidative stress and phospho-p38 expression. Oncol
Lett. 7:1159–1164. 2014.PubMed/NCBI
|
|
20
|
Liu Z, Li X, Simoneau AR, Jafari M and Zi
X: Rhodiola rosea extracts and salidroside decrease the growth of
bladder cancer cell lines via inhibition of the mTOR pathway and
induction of autophagy. Mol Carcinog. 51:257–267. 2012. View Article : Google Scholar
|
|
21
|
Zhang L, Yu H, Sun Y, Lin X, Chen B, Tan
C, Cao G and Wang Z: Protective effects of salidroside on hydrogen
peroxide-induced apoptosis in SH-SY5Y human neuroblastoma cells.
Eur J Pharmacol. 564:18–25. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhang Y, Yao Y, Wang H, Guo Y, Zhang H and
Chen L: Effects of salidroside on glioma formation and growth
inhibition together with improvement of tumor microenvironment.
Chin J Cancer Res. 25:520–526. 2013.PubMed/NCBI
|
|
23
|
Hu X, Zhang X, Qiu S, Yu D and Lin S:
Salidroside induces cell-cycle arrest and apoptosis in human breast
cancer cells. Biochem Biophys Res Commun. 398:62–67. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hamzeloo-Moghadam M, Aghaei M, Fallahian
F, et al: Britannin, a sesquiterpene lactone, inhibits
proliferation and induces apoptosis through the mitochondrial
signaling pathway in human breast cancer cells. Tumour Biol. Epub
ahead of print Oct 24, 2014.
|
|
25
|
Abe S, Nishimoto Y, Isu K, Ishii T and
Goto T: Japanese Musculoskeletal Oncology Group: Preoperative
cisplatin for initial treatment of limb osteosarcoma: its local
effect and impact on prognosis. Cancer Chemother Pharmacol.
50:320–324. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lee HB, Yu MR, Song JS and Ha H: Reactive
oxygen species amplify protein kinase C signaling in high
glucose-induced fibronectin expression by human peritoneal
mesothelial cells. Kidney Int. 65:1170–1179. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Head J and Johnston SR: New targets for
therapy in breast cancer: farnesyltransferase inhibitors. Breast
Cancer Res. 6:262–268. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
28
|
Newman DJ and Cragg GM: Natural products
as sources of new drugs over the 30 years from 1981 to 2010. J Nat
Prod. 75:311–335. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Green DR and Kroemer G: The
pathophysiology of mitochondrial cell death. Science. 305:626–629.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Jänicke RU, Sprengart ML, Wati MR and
Porter AG: Caspase-3 is required for DNA fragmentation and
morphological changes associated with apoptosis. J Biol Chem.
273:9357–9360. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Rajoria S, Suriano R, George A, Shanmugam
A, Schantz SP, Geliebter J and Tiwari RK: Estrogen induced
metastatic modulators MMP-2 and MMP-9 are targets of
3,3′-diindolylmethane in thyroid cancer. PLoS One. 6:e158792011.
View Article : Google Scholar
|
|
32
|
Yan L, Lin B, Gao L, Gao S, Liu C, Wang C,
Wang Y, Zhang S and Iwamori M: Lewis (y) antigen overexpression
increases the expression of MMP-2 and MMP-9 and invasion of human
ovarian cancer cells. Int J Mol Sci. 11:4441–4452. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Braicu EI, Gasimli K, Richter R, et al:
Tumor Bank Ovarian Cancer (TOC); German North Eastern Society for
Gynecological Oncology (NOGGO): Role of serum VEGFA, TIMP2, MMP2
and MMP9 in monitoring response to adjuvant radiochemotherapy in
patients with primary cervical cancer - results of a companion
protocol of the randomized NOGGO-AGO phase III clinical trial.
Anticancer Res. 34:385–391. 2014.PubMed/NCBI
|
|
34
|
Lai WW, Hsu SC, Chueh FS, Chen YY, Yang
JS, Lin JP, Lien JC, Tsai CH and Chung JG: Quercetin inhibits
migration and invasion of SAS human oral cancer cells through
inhibition of NF-κB and matrix metalloproteinase-2/-9 signaling
pathways. Anticancer Res. 33:1941–1950. 2013.PubMed/NCBI
|
|
35
|
Sun C, Wang Z, Zheng Q and Zhang H:
Salidroside inhibits migration and invasion of human fibrosarcoma
HT1080 cells. Phytomedicine. 19:355–363. 2012. View Article : Google Scholar
|
|
36
|
Dhillon AS, Hagan S, Rath O and Kolch W:
MAP kinase signalling pathways in cancer. Oncogene. 26:3279–3290.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Fiers W, Beyaert R, Declercq W and
Vandenabeele P: More than one way to die: apoptosis, necrosis and
reactive oxygen damage. Oncogene. 18:7719–7730. 1999. View Article : Google Scholar
|
|
38
|
Simon HU, Haj-Yehia A and Levi-Schaffer F:
Role of reactive oxygen species (ROS) in apoptosis induction.
Apoptosis. 5:415–418. 2000. View Article : Google Scholar
|
|
39
|
Panossian A, Hambardzumyan M, Hovhanissyan
A and Wikman G: The adaptogens rhodiola and schisandra modify the
response to immobilization stress in rabbits by suppressing the
increase of phosphorylated stress-activated protein kinase, nitric
oxide and cortisol. Drug Target Insights. 2:39–54. 2007.
|