Introduction
Breast cancer is one of the most prevalent cancers
worldwide and is the main cause of cancer-related mortality among
females (1). Conventional treatment
strategies mainly include surgery, systemic agents (hormone
therapy, chemotherapy and targeted therapy) and radiotherapy.
Radiation is a critical component of breast cancer treatment
modalities and is highly effective in improving locoregional
control and allowing for breast conservation without any
detrimental effects on survival (2). However, its curative effect is
sometimes limited by radioresistance of the cancer cells, in
addition to radiation toxicity to normal tissue (2,3). As is
well established, the four factors that influence radioresistance
are i) repair of DNA, ii) reoxygenation, iii) redistribution of the
cell cycle and iv) repopulation (the '4 R′s'). Thus, any
pharmacologic agents aimed at sensitizing cells to radiotherapy
would need to target one or more of these factors. While many
agents have been explored based on this principle, the side-effects
of the agent itself often prohibits its successful use as a
radio-sensitizer (4–6). Thus, finding novel radiosensitizing
agents with minimal side-effects has become an area of interest for
radiation oncology investigators. More recently, the interest in
plant-derived natural products for breast cancer treatment and
symptom management has increased (7–10).
Given the multiple antitumor effects of many traditional Chinese
medicines (TCMs), exploring these agents as radiosensitization
agents is of interest.
Trametes robiniophila Murr (Huaier) is
classified as an officinal fungus which has been used in China as a
traditional medicine for many years (11). Previous investigations suggest that
Huaier displays multiple antitumor effects against hepatocarcinoma
(12,13), gastrointestinal (14), breast (15,16)
and lung cancer (17). Several
mechanisms for the antitumor activities of Huaier have been
suggested including the induction of apoptosis, anti-angiogenesis,
reversal of drug-resistance, anti-metastasis and activation of the
immune system (15,18–22).
In addition, in recent years, as a complementary drug, Huaier has
been routinely used in liver and breast cancer.
Previously, our research group investigated the
mechanisms of action of Huaier and demonstrated the multi-targeted
antitumor effects of Huaier (23).
We found that a microarray assay of MCF-7 cells treated with Huaier
demonstrated that the effects were related to the cell cycle, cell
division and DNA repair. Consequently, Huaier may have a
radiosensitization effect on breast cancer and the present study
was carried out to assess the combined effects of Huaier and
radiotherapy on breast cancer.
Materials and methods
Cell lines and reagents
The human breast cancer cell lines, MCF-7 and
MDA-MB-468, were obtained from the American Type Culture Collection
(ATCC; Rockefeller, MD, USA), and were routinely cultured in
Dulbecco's modified Eagle's medium (DMEM; Gibco, Rockville, MD,
USA) with 10% fetal bovine serum (FBS; Clark Bioscience, Seabrook,
MD, USA), 100 U/ml penicillin and 100 µg/ml streptomycin in
5% CO2 at 37°C. The antibodies utilized included
anti-Ku70, -Ku86 and -RAD51 (Santa Cruz Biotechnology Inc., Santa
Cruz, CA, USA), mouse anti-γ-H2AX antibody (Millipore, Billerica,
MA, USA), Rhodamine-labeled secondary antibody (Jackson
ImmunoResearch, West Grove, PA, USA) and HRP-labeled secondary
antibodies (KPL, Gaithersburg, MD, USA). Huaier aqueous extract, a
kind gift from Gaitianli Medicine Co. Ltd. (Jiangsu, China), was
dissolved in DMEM in a storage concentration of 100 mg/ml. All
other chemicals were obtained from Sigma-Aldrich (St. Louis, MO,
USA) unless specifically described.
Cell treatment and human transcriptome
array 2.0 (HTA 2.0) assay
MCF-7 cells were treated with Huaier aqueous extract
at a concentration of 8 mg/ml for 72 h or treated with DMEM as a
control, and total RNA was isolated according to the manufacturer's
protocol using TRIzol (Invitrogen, Carlsbad, CA, USA). Equal
amounts of 3 samples of RNAs were pooled together to obtain an
average. Gene alterations were detected using Affymetrix Human
Transcriptome Array 2.0 (HTA 2.0) assay and fold-change was used to
identify differentially expressed genes (DEGs). Next, DEGs were
analyzed by Gene Ontology (GO) analysis, which organizes genes into
hierarchical categories based on biological processes and molecular
function (24,25).
Ionizing radiation treatment
Cells were irradiated using a radiotherapy linear
accelerator PRIMUS HI (Siemens, Germany) at room temperature. The
X-rays were filtered through a 12-mm thick special organic glass
for dose built-up. The irradiation dose rate was 200 cGy/min.
Colony formation assay
The radiosensitization effects of Huaier on breast
cancer cells were assessed by colony formation assay. Briefly, the
cells were treated with 4 mg/ml Huaier or complete DMEM as a
control for 24 h, and then irradiated with incremental doses of
radiation (0–4 Gy) at room temperature. The cells were trypsinized,
suspended in complete medium, counted and plated in 6-well plates
and incubated for 14–21 days to collect cell colonies. Cell
colonies were fixed with methanol and stained with crystal violet.
Only colonies containing >50 cells were counted. The survival
fraction of each given dose (SFD) was calculated using the
linear-quadratic formula: S(D) = S(0)e(−αD − βD2) and
the α and β values were obtained to describe survival curve
characteristics relative to radiation dose (26).
Cell cycle analysis by flow
cytometry
The cell cycle distribution was analyzed using a
flow cytometer. Briefly, 5×105 cells were seeded in a 25
cm2 flask, and then treated with 4 mg/ml Huaier or DMEM
as control for 24 h before irradiation. The cells were then
trypsinized, washed with phosphate-buffered saline (PBS), incubated
with 1 ml propidium iodide (PI) (Liankebio, Zhejiang, China) in the
dark for 30 min at room temperature, and then analyzed using a flow
cytometer (Becton-Dickinson, Franklin Lakes, NJ, USA). The data
were analyzed using ModFit LT V2.0 software (Becton-Dickinson).
Immunofluorescence staining
Conditions of γ-H2AX foci were detected using
immunofluorescence staining. The cells were grown on coverslips in
24-well plates and pretreated with 4 mg/ml Huaier or DMEM (control)
for 24 h before irradiation. The cells were washed in PBS, and then
fixed with 4% paraformaldehyde for 15 min followed by treatment
with 0.2% Triton X-100 for 8 min. Then, the cells were washed in
PBS, blocked with 10% normal goat serum in PBS for 1 h, and
incubated with mouse anti-γ-H2AX antibody overnight at 4°C. After
washing in PBS, the cells were incubated with Rhodamine-conjugated
anti-mouse secondary antibody in the dark for 1 h. The cells were
then washed in PBS, and stained with 4′,6-diamidino-2-phenylindole
(DAPI). Then after washing in PBS for 3 times, the coverslips were
mounted on glass slides with anti-fading medium (Beyotime Institute
of Biotechnology, Jiangsu, China). The fluorescence signal was
acquired using a fluorescence microscope (Olympus, Tokyo,
Japan).
Western blot analysis
Cells were lysed with radioimmunoprecipitation assay
(RIPA) and phenylmethanesulfonyl fluoride (PMSF) (Biocolors,
Shanghai, China), and quantified by the BCA method (Merck,
Darmstadt, Germany). Equal amounts of protein were loaded on
SDS-PAGE gel, and then transferred to PVDF membranes (Millipore,
Bedford, MA, USA). After blocking in 5% defatted milk for 1 h at
room temperature, the membranes were incubated with the
corresponding primary antibody overnight at 4°C. The next day, the
membranes were washed in TBST three times prior to incubation with
the corresponding secondary antibody for 2 h. Signals were detected
using Luminescent Image Analyzer (GE Healthcare Life Sciences,
Sweden). β-actin was used as the loading control.
Statistical analysis
The data are expressed as the mean ± standard error
of the mean (SEM), and each experiment was conducted for a minimum
of 3 times. The SPSS 18.0 soft-ware (SPSS, Inc., Chicago, IL, USA)
was used for statistical analysis. Student's t-test and linear
regression analysis were used to analyze the statistical
significance. A p-value <0.05 was utilized as the threshold for
statistical significance.
Results
Huaier alters biological activities
related to cell cycle, cell division and DNA repair
Microarray profiling was obtained and GO analysis
was used to detect the altered biological function of MCF-7 cells
after treatment of Huaier at 8 mg/ml for 72 h. As shown in Fig. 1A, the top altered categories were
genes mainly related to cell cycle, cell division, cell cycle
phases and DNA repair, with its -Lg (p-value) listed on the left
and DEG count to the right of the figure, respectively. Table I shows the top 10 GO categories
affected by Huaier treatment in comparison to the control
cells.
 | Table ITop 10 GO categories altered in the
Huaier-treated MCF-7 cells compared to the control cells. |
Table I
Top 10 GO categories altered in the
Huaier-treated MCF-7 cells compared to the control cells.
| GO name | P-value | FDR |
|---|
| Cell division | 9.28252E-51 | 6.45599E-48 |
| DNA replication | 1.92587E-50 | 8.92961E-48 |
| G1/S transition of
mitotic cell cycle | 2.89469E-39 | 1.00663E-36 |
| Mitotic
prometaphase | 7.24882E-38 | 2.01662E-35 |
| M phase of mitotic
cell cycle | 4.05311E-36 | 9.39646E-34 |
| S phase of mitotic
cell cycle | 1.86134E-34 | 3.69876E-32 |
| DNA repair | 7.23594E-32 | 1.25815E-29 |
| DNA strand elongation
involved in DNA replication | 1.06698E-30 | 1.64908E-28 |
| Mitosis | 1.17413E-27 | 1.63322E-25 |
| Mitotic anaphase | 2.69998E-26 | 3.41424E-24 |
Huaier sensitizes breast cancer cells to
irradiation
The cell cycle and DNA repair are closely related to
irradiation, thus in order to ascertain whether Huaier has a
radiosensitization effect on breast cancer cells, a colony
formation assay was carried out. Fig.
1B shows the effect of Huaier on MCF-7 and MDA-MB-468 breast
cancer cells relative to radiation. The survival fraction at 2 Gy
(SF2) was used to evaluate the radiosensitivity. In the MCF-7
cells, SF2 was 28.55±0.03% for cells pretreated with Huaier and
54.54±0.12% for the control, with a significant reduction of
~47.65% (p=0.018). Next, the linear quadratic formula was used to
analyze the α and β components. The α and β values were
0.529±0.095/Gy and 0.033±0.037/Gy2 for cells pretreated
with Huaier, −0.094±0.349/Gy and 0.034±0.054/Gy2 for the
control cells, suggesting two significantly different curves using
linear regression analysis (p<0.01). For the MDA-MB-468 cells,
SF2 was 30.19±0.08% for cells pretreated with Huaier, and
50.96±0.06% for the control cells respectively, with a reduction of
40.76% (p=0.026). The α and β values were calculated at
0.703±0.170/Gy and −0.019±0.052/Gy2 for cells pretreated
with Huaier, and 0.297±0.034/Gy and 0.007±0.010/Gy2 for
the control cells, again, demonstrating significant differences
with and without Huaier (p<0.01), suggesting the
radiosensitizing potential of Huaier.
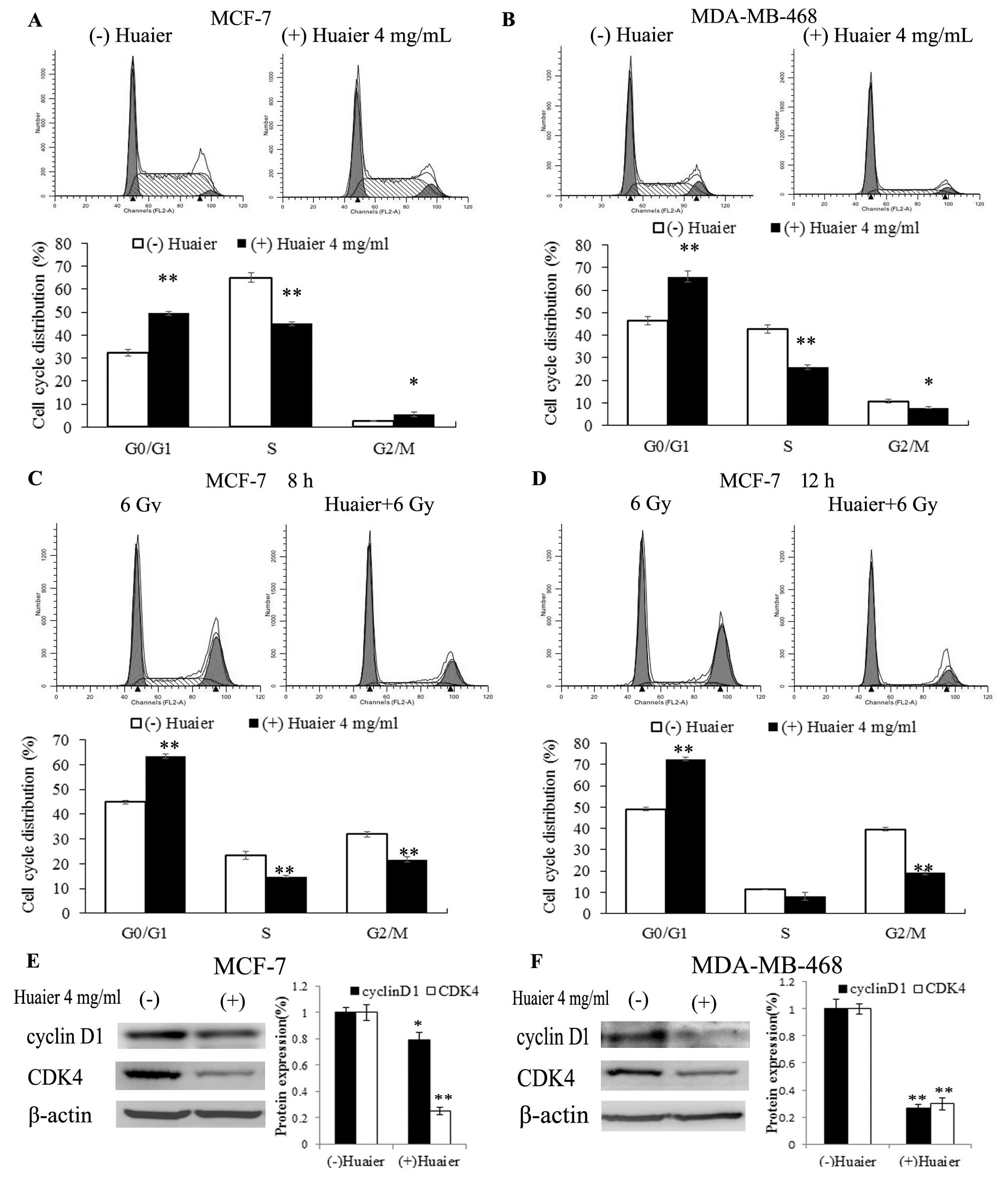
Huaier treatment causes G0/G1 arrest
Flow cytometry was used to analyze cell cycle
progression. As shown in Fig. 2A and
B, when cells were treated with Huaier at 4 mg/ml for 24 h, a
significant increase in the percentage of cells in the G0/G1 phase
was observed (from 32.33±1.64 to 49.49±0.86% in MCF-7 cells,
p<0.01; from 46.44±1.89 to 66.04±2.35% in MDA-MB-468 cells,
p<0.01), accompanied by an obvious decrease in the percentage of
cells in the S phase (from 65.07±1.89 to 45.04±0.85% in MCF-7 cells
with p<0.01, and from 42.79±2.01 to 26.02±1.05% in MDA-MB-468
cells with p<0.01). Within 24 h after irradiation, the MCF-7
radiated cells (6 Gy) were found to have a block at the G2/M phase,
whereas in cells pre-treated with Huaier this radiation-induced
G2/M phase block was not obvious (Fig.
2C and D). To determine the mechanism of G0/G1 arrest, we
detected the protein levels of cell cycle-regulating proteins by
western blotting (as shown) and were able to demonstrate that two
important cell cycle-regulating proteins, cyclin D1 and CDK4, were
obviously decreased in both cell lines following treatment with
Huaier, which may explain the G0/G1 cell cycle arrest for both cell
lines (Fig. 2E and F).
Huaier pre-treatment prolongs the
persistence of DNA double-strand breaks (DSBs)
γ-H2AX foci were detected using immunofluorescence
staining to evaluate conditions of DNA double-strand breaks (DSBs)
(27) at 0, 0.5, 4, 8 and 12 h
after 1 Gy irradiation in both the MCF-7 and MDA-MB-468 cells. Both
cell lines displayed similar phenomenon as shown in Fig. 3. In cells without Huaier treatment,
γ-H2AX foci appeared most prominently at 0. 5 h, decreased at 4 h,
and almost cleared at 8 h, while in cells pretreated with Huaier,
γ-H2AX foci were not completely cleared even at 12 h. Thus,
pre-treatment with Huaier prior to radiation appears to be
associated with a longer interval of DSBs, which may contribute to
its radiosensitization effect.
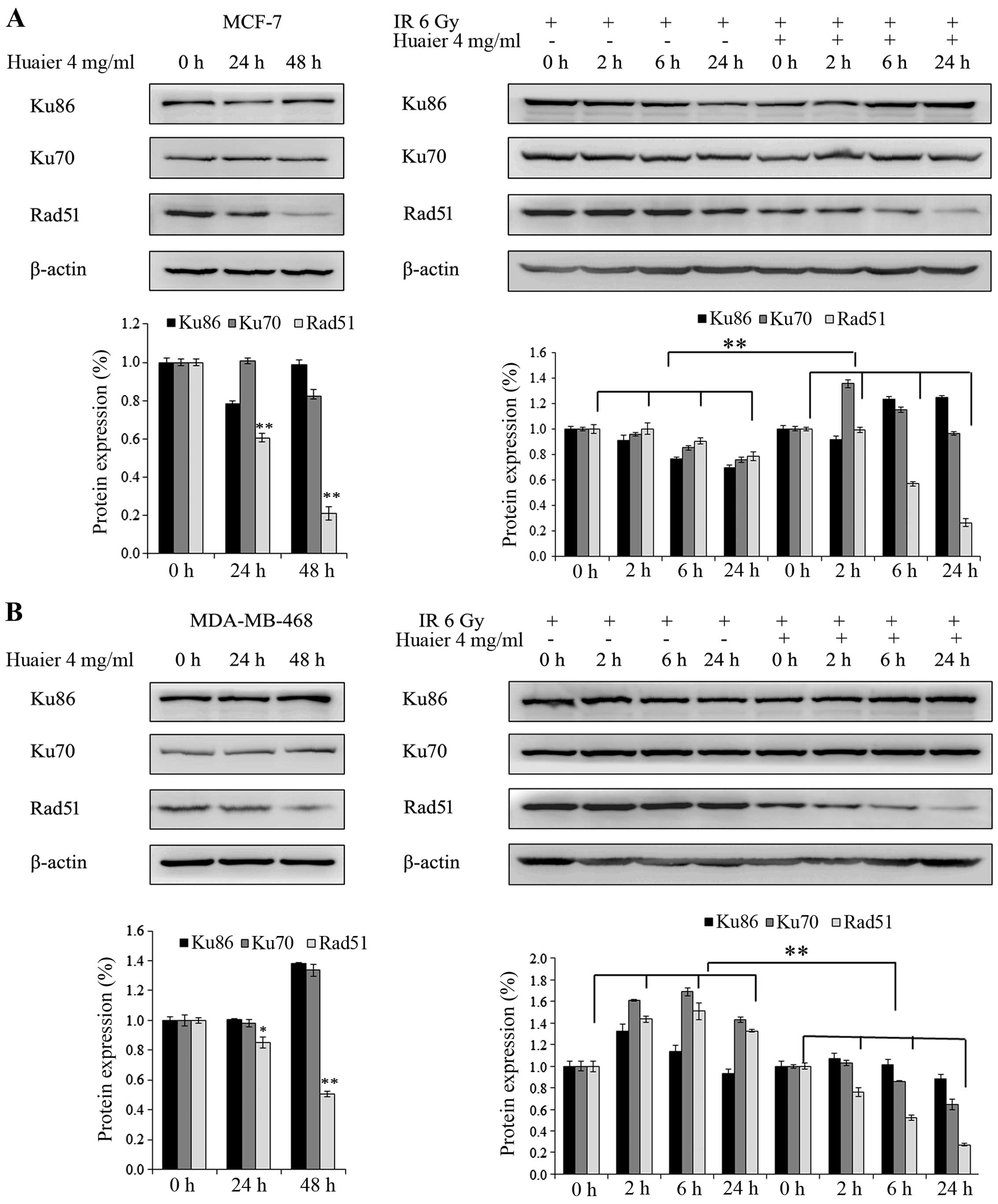
Huaier interferes with DNA repair through
the homologous recombination (HR) pathway
Ku70 and Ku86 are markers of non-homologous end
joining (NHEJ), while RAD51 is related to homologous recombination
(HR). Fig. 4 shows the protein
levels of Ku70, Ku86 and RAD51. In the MCF-7 cells, pre-treatment
of 4 mg/ml Huaier led to a decrease in RAD51 but not Ku70 and Ku86.
In cells pre-treated with Huaier compared to the control cells,
treatment with 6 Gy irradiation resulted in decreased RAD51 levels
in a time-dependent fashion (0, 2, 6 and 24 h). However, protein
levels of Ku70 and Ku86 were not changed in either group (Fig. 4A). These results were similarly
found in the MDA-MB-468 cells (Fig.
4B).
Discussion
Radiotherapy is an important and effective treatment
strategy for breast cancer although it is sometimes limited by
radio-resistance and radiotoxicity. Thus, the identification of a
radiosensitizer with a high benefit to risk ratio is needed.
Recently, a wide variety of TCMs have been gaining popularity, and
Huaier has been investigated by our group due to its seemingly low
toxicity and antitumor activity (23,28–30).
The biologic effects of Huaier are varied (23), and its use has widely increased as a
complementary treatment for cancers in recent years. In order to
understand its mechanism of action specifically in breast cancer, a
microarray assay of Huaier-treated MCF-7 breast cancer cells was
assessed, which indicated that among the categories of cell cycle
regulation, its effects are mainly related to cell cycle, cell
division and DNA repair, thus making it a reasonable agent to
investigate as a radiosensitizer. A colony formation assay was
utilized to validate the hypothesis using MCF-7 and MDA-MB-468
breast cancer cells.
Our findings demonstrated that the percentage of
cells in the S phase was significantly decreased in the
Huaier-treated cells compared with the control, which may, in part,
explain the radiosensitization phenomenon of Huaier. Moreover,
irradiated cells pretreated with Huaier exhibited no obvious G2/M
arrest, whereas irradiated cells had a significantly increased
signal of G2/M arrest, suggesting that Huaier combined with
radiation can alter the cell cycle distribution and significantly
alleviate G2/M arrest, increasing sensitization to radiotherapy.
These findings are consistent with other previously published
studies on G2/M arrest and irradiation sensitivity, which have
demonstrated the association between G2/M arrest and sensitization
to irradiation (31,32).
Cell cycle arrest is often related to cell
cycle-regulating proteins. Cyclin D1 is an important regulator in
cell cycle progression in G1 to S transition. By binding to CDK4,
it phosphorylates Rb to release E2F, which is required for
transition from the G1 to S phase (33). In the present study, we found that
the levels of cyclin D1 and CDK4 were significantly decreased in
both cell lines following treatment with Huaier, which may explain
the mechanism of G0/G1 arrest, particularly in the MDA-MB-468
cells. It is likely that Huaier regulates several cell
cycle-related proteins, ultimately resulting in induced G0/G1
arrest.
It is well-known that the main mechanism of action
of radiation for cell death is related to DNA damage (i.e.
double-strand DNA breaks), thus, cell mortality is proportional to
the unrepaired DNA damage (34).
Cells resistant to radiation develop various DNA repair pathways to
protect the damaged DNA. These protective mechanisms are strong,
and include DNA repair pathways such as nucleotide excision repair
(NER), mismatch repair (MMR), base excision repair (BER),
non-homologous end joining (NHEJ) and homologous recombination
(HR). In mammalian cells, HR and NHEJ are the main DNA repair
pathways. In the present study, the western blot assay demonstrated
that Huaier treatment decreased the protein level of RAD51 which is
an important player in HR, but not Ku70 and Ku86 which are members
of NHEJ. Yet, when irradiated with 6 Gy X-ray, the protein level of
RAD51 was decreased in a time-dependent manner. This phenomenon
indicates that Huaier treatment may inhibit the reparatory pathways
that are elicited with irradiation. Moreover, since γ-H2AX foci are
a biomarker of DNA double-strand breaks (35), as shown in the immunofluorescence
assays, the DNA damage in Huaier-treated cells persisted for a
longer period of time compared with that in the untreated cells.
Various recent studies support the contention that compared with
normal tissues, tumor cells have defects in DNA repair pathways in
familial breast cancer and other cancers (36,37).
Thus, the ability to diminish one (or more) of the pathways of DNA
repair may result in increased cell kill, and may be a potential
target for increasing radiosensitization.
In conclusion, our results showed that Huaier has
the ability to alter cell cycle progression by regulating protein
levels of cell cycle-regulating proteins, and disrupting DNA repair
through the HR pathway. These results suggest that Huaier should be
investigated further as a potential radiosensitizer for breast
cancer. While clinical correlation studies are needed to assess the
toxicity and efficacy of Huaier treatment, our current
investigation provides the laboratory basis to suggest that Huaier
is promising for further clinical investigation in breast cancer
patients.
Acknowledgments
The present study was supported by the National
Natural Science Foundation of China (nos. 81272903 and 81172529),
the Shandong Science and Technology Development Plan (no.
2013GRC31801), and the Special Support Plan for National High Level
Talents ('Ten Thousand Talents Program') to Q.Y.
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Brown LC, Mutter RW and Halyard MY:
Benefits, risks, and safety of external beam radiation therapy for
breast cancer. Int J Womens Health. 7:449–458. 2015.PubMed/NCBI
|
|
3
|
Gerweck LE, Vijayappa S, Kurimasa A, Ogawa
K and Chen DJ: Tumor cell radiosensitivity is a major determinant
of tumor response to radiation. Cancer Res. 66:8352–8355. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Uma Devi P: Normal tissue protection in
cancer therapy -progress and prospects. Acta Oncol. 37:247–252.
1998. View Article : Google Scholar
|
|
5
|
Thomson D, Yang H, Baines H, Miles E,
Bolton S, West C and Slevin N: NIMRAD - a phase III trial to
investigate the use of nimorazole hypoxia modification with
intensity-modulated radiotherapy in head and neck cancer. Clin
Oncol. 26:344–347. 2014. View Article : Google Scholar
|
|
6
|
Ang KK: More lessons learned from the
suffocation of hypoxia. J Clin Oncol. 28:2941–2943. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Cragg GM and Newman DJ: Plants as a source
of anti-cancer agents. J Ethnopharmacol. 100:72–79. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chen P, Li J, Jiang HG, Lan T and Chen YC:
Curcumin reverses cisplatin resistance in cisplatin-resistant lung
cancer cells by inhibiting FA/BRCA pathway. Tumour Biol.
36:3591–3599. 2015. View Article : Google Scholar
|
|
9
|
Shi XP, Miao S, Wu Y, Zhang W, Zhang XF,
Ma HZ, Xin HL, Feng J, Wen AD and Li Y: Resveratrol sensitizes
tamoxifen in antiestrogen-resistant breast cancer cells with
epithelial-mesenchymal transition features. Int J Mol Sci.
14:15655–15668. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Srivastava V, Negi AS, Kumar JK, Gupta MM
and Khanuja SP: Plant-based anticancer molecules: A chemical and
biological profile of some important leads. Bioorg Med Chem.
13:5892–5908. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li LYS, Wang Y and Tang Z: Progress on
experimental research and clinical application of Trametes
robiniophila. China Cancer. 16:110–113. 2006.
|
|
12
|
Zheng J, Li C, Wu X, Liu M, Sun X, Yang Y,
Hao M, Sheng S, Sun Y, Zhang H, et al: Huaier polysaccharides
suppress hepatocarcinoma MHCC97-H cell metastasis via inactivation
of EMT and AEG-1 pathway. Int J Biol Macromol. 64:106–110. 2014.
View Article : Google Scholar
|
|
13
|
Li C, Wu X, Zhang H, Yang G, Hao M, Sheng
S, Sun Y, Long J, Hu C, Sun X, et al: A Huaier polysaccharide
reduced metastasis of human hepatocellular carcinoma SMMC-7721
cells via modu-lating AUF-1 signaling pathway. Tumour Biol.
36:6285–6293. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang T, Wang K, Zhang J, Wang X, Chen Z,
Ni C, Qiu F and Huang J: Huaier aqueous extract inhibits colorectal
cancer stem cell growth partially via downregulation of the
Wnt/β-catenin pathway. Oncol Lett. 5:1171–1176. 2013.PubMed/NCBI
|
|
15
|
Wang X, Zhang N, Huo Q and Yang Q:
Anti-angiogenic and antitumor activities of Huaier aqueous extract.
Oncol Rep. 28:1167–1175. 2012.PubMed/NCBI
|
|
16
|
Zhang N, Kong X, Yan S, Yuan C and Yang Q:
Huaier aqueous extract inhibits proliferation of breast cancer
cells by inducing apoptosis. Cancer Sci. 101:2375–2383. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wu T, Chen W, Liu S, Lu H, Wang H, Kong D,
Huang X, Kong Q, Ning Y and Lu Z: Huaier suppresses proliferation
and induces apoptosis in human pulmonary cancer cells via
upregulation of miR-26b-5p. FEBS Lett. 588:2107–2114. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang F, Zhang Z and Liu Z: Effects of
Huaier aqueous extract on proliferation and apoptosis in the
melanoma cell line A875. Acta Histochem. 115:705–711. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Cui Y, Meng H, Liu W, Wang H and Liu Q:
Huaier aqueous extract induces apoptosis of human fibrosarcoma
HT1080 cells through the mitochondrial pathway. Oncol Lett.
9:1590–1596. 2015.PubMed/NCBI
|
|
20
|
Li C, Wu X, Zhang H, Yang G, Hao M, Sheng
S, Sun Y, Long J, Hu C, Sun X, et al: A Huaier polysaccharide
inhibits hepatocellular carcinoma growth and metastasis. Tumour
Biol. 36:1739–1745. 2015. View Article : Google Scholar
|
|
21
|
Sun Y, Sun T, Wang F, Zhang J, Li C, Chen
X, Li Q and Sun S: A polysaccharide from the fungi of Huaier
exhibits anti-tumor potential and immunomodulatory effects.
Carbohydr Polym. 92:577–582. 2013. View Article : Google Scholar
|
|
22
|
Yu Zhe WT and Yang Z: The reversal effects
of Trametes Robiniophila Murr. on multidrug resistance in resistant
human hepatocellular carcinoma cell line BEL-7402/5-Fu. J Clin
Oncol. 19:443–447. 2013.
|
|
23
|
Kong X, Ding X and Yang Q: Identification
of multi-target effects of Huaier aqueous extract via microarray
profiling in triple-negative breast cancer cells. Int J Oncol.
46:2047–2056. 2015.PubMed/NCBI
|
|
24
|
Gene Ontology Consortium: The Gene
Ontology (GO) project in 2006. Nucleic Acids Res. 34:D322–D326.
2006. View Article : Google Scholar :
|
|
25
|
Ashburner M, Ball CA, Blake JA, Botstein
D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT,
et al The Gene Ontology Consortium: Gene ontology: Tool for the
unification of biology. Nat Genet. 25:25–29. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Franken NA, Rodermond HM, Stap J, Haveman
J and van Bree C: Clonogenic assay of cells in vitro. Nat Protoc.
1:2315–2319. 2006. View Article : Google Scholar
|
|
27
|
Gerić M, Gajski G and Garaj-Vrhovac V:
γ-H2AX as a biomarker for DNA double-strand breaks in
ecotoxicology. Ecotoxicol Environ Saf. 105:13–21. 2014. View Article : Google Scholar
|
|
28
|
Cohen I, Tagliaferri M and Tripathy D:
Traditional Chinese medicine in the treatment of breast cancer.
Semin Oncol. 29:563–574. 2002. View Article : Google Scholar
|
|
29
|
Wong KY, Tan EY, Chen JJ, Teo C and Chan
PM: The use of traditional Chinese medicine among breast cancer
patients: Implications for the clinician. Ann Acad Med Singapore.
43:74–78. 2014.PubMed/NCBI
|
|
30
|
Wang X, Zhang N, Huo Q, Sun M, Lv S and
Yang Q: Huaier aqueous extract suppresses human breast cancer cell
proliferation through inhibition of estrogen receptor α signaling.
Int J Oncol. 43:321–328. 2013.PubMed/NCBI
|
|
31
|
Busse PM, Bose SK, Jones RW and Tolmach
LJ: The action of caffeine on X-irradiated HeLa cells. III.
Enhancement of X-ray-induced killing during G2 arrest. Radiat Res.
76:292–307. 1978. View
Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang J, Liu Q and Yang Q:
Radiosensitization effects of berberine on human breast cancer
cells. Int J Mol Med. 30:1166–1172. 2012.PubMed/NCBI
|
|
33
|
Giacinti C and Giordano A: RB and cell
cycle progression. Oncogene. 25:5220–5227. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Núñez MI, McMillan TJ, Valenzuela MT, Ruiz
de Almodóvar JM and Pedraza V: Relationship between DNA damage,
rejoining and cell killing by radiation in mammalian cells.
Radiother Oncol. 39:155–165. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kuo LJ and Yang LX: Gamma-H2AX - a novel
biomarker for DNA double-strand breaks. In Vivo. 22:305–309.
2008.PubMed/NCBI
|
|
36
|
Shibata D: When does MMR loss occur during
HNPCC progression? Cancer Biomark. 2:29–35. 2006.PubMed/NCBI
|
|
37
|
Powell SN and Kachnic LA: Therapeutic
exploitation of tumor cell defects in homologous recombination.
Anticancer Agents Med Chem. 8:448–460. 2008. View Article : Google Scholar : PubMed/NCBI
|