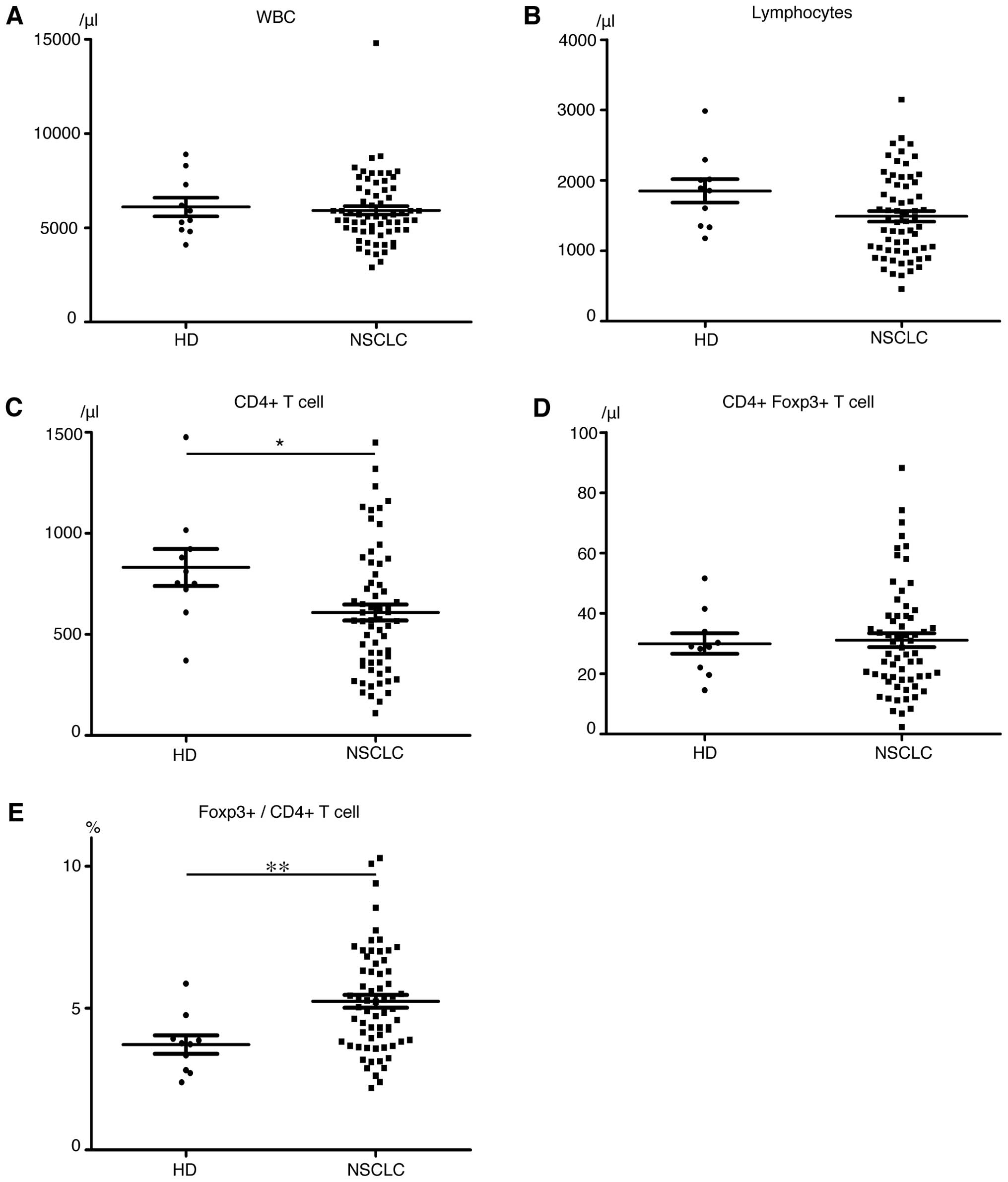
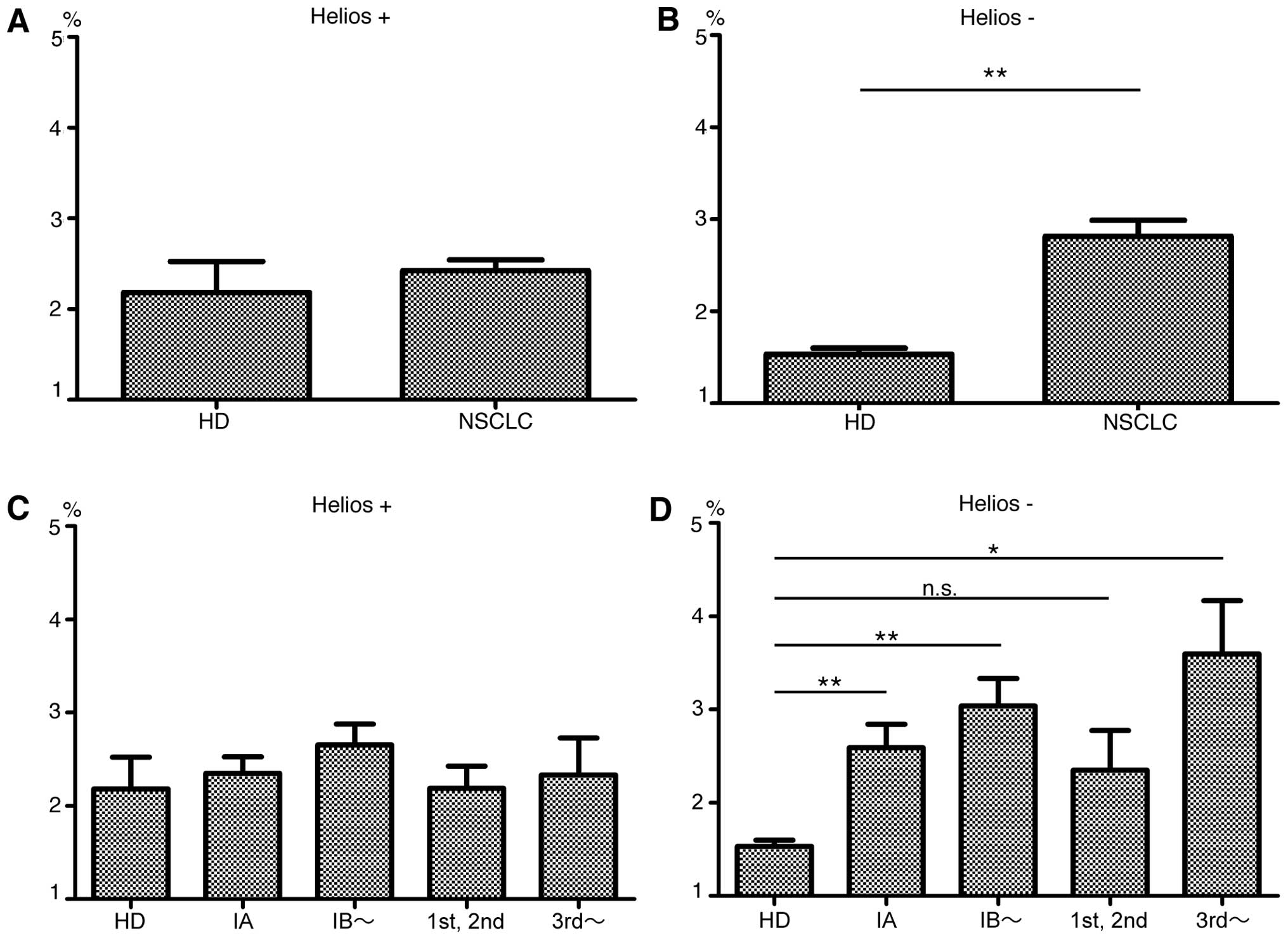
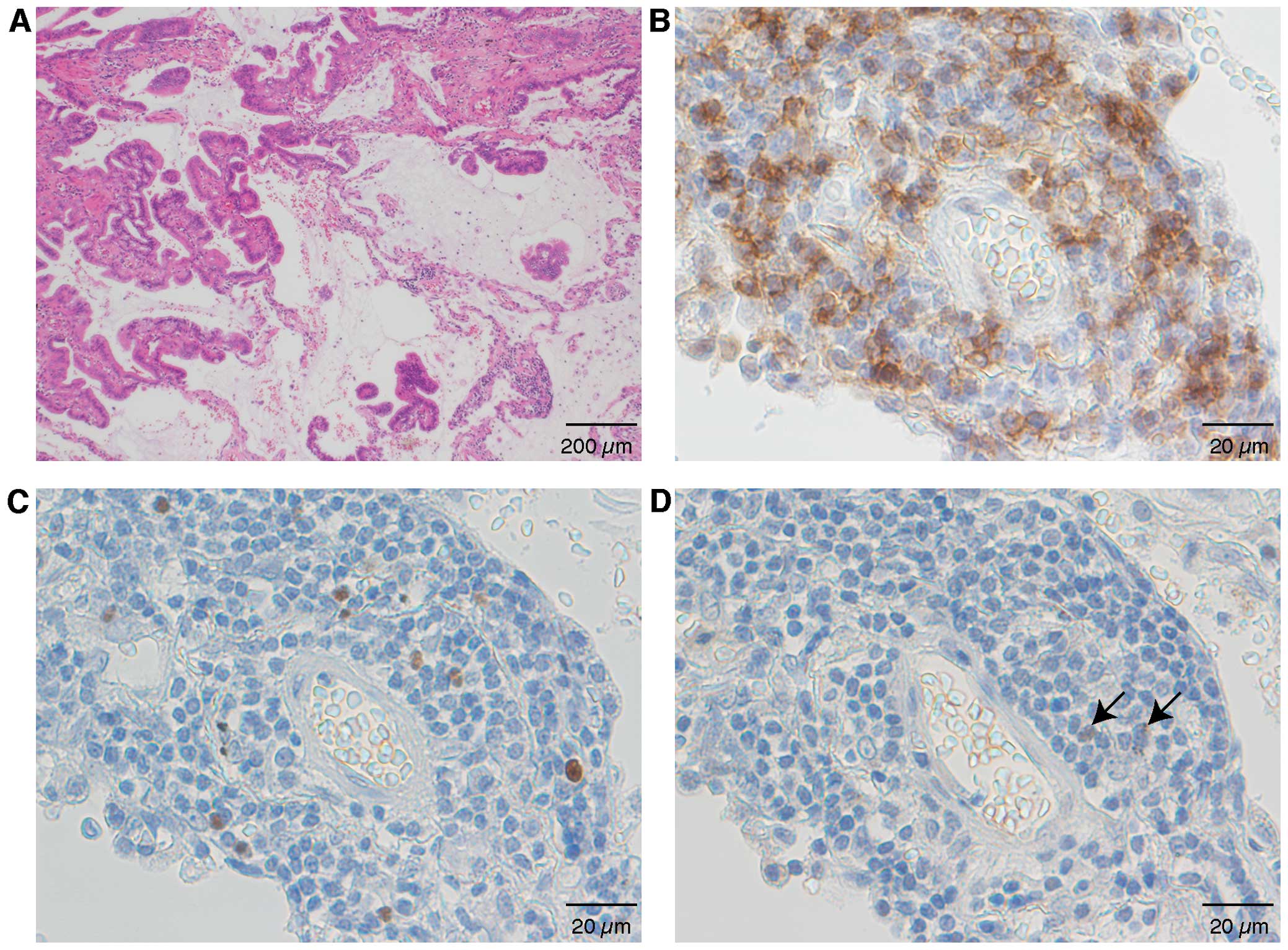
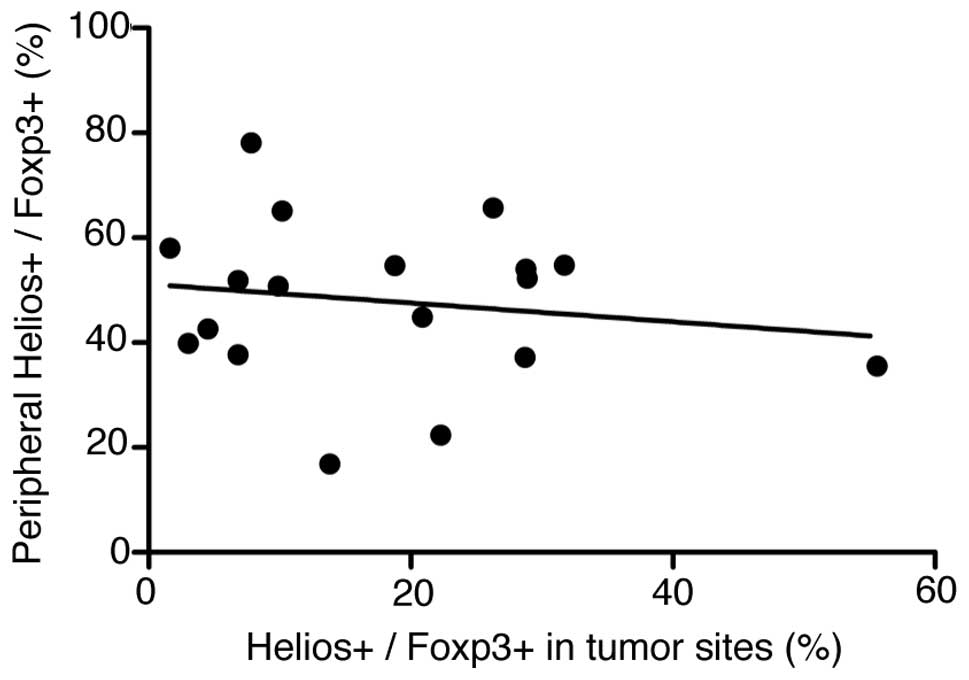
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mitsudomi T, Morita S, Yatabe Y, Negoro S,
Okamoto I, Tsurutani J, Seto T, Satouchi M, Tada H, Hirashima T, et
al; West Japan Oncology Group. Gefitinib versus cisplatin plus
docetaxel in patients with non-small-cell lung cancer harbouring
mutations of the epidermal growth factor receptor (WJTOG3405): An
open label, randomised phase 3 trial. Lancet Oncol. 11:121–128.
2010. View Article : Google Scholar
|
|
3
|
Maemondo M, Inoue A, Kobayashi K, Sugawara
S, Oizumi S, Isobe H, Gemma A, Harada M, Yoshizawa H, Kinoshita I,
et al; North-East Japan Study Group. Gefitinib or chemotherapy for
non-small-cell lung cancer with mutated EGFR. N Engl J Med.
362:2380–2388. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhou C, Wu YL, Chen G, Feng J, Liu XQ,
Wang C, Zhang S, Wang J, Zhou S, Ren S, et al: Erlotinib versus
chemotherapy as first-line treatment for patients with advanced
EGFR mutation-positive non-small-cell lung cancer (OPTIMAL,
CTONG-0802): A multicentre, open-label, randomised, phase 3 study.
Lancet Oncol. 12:735–742. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Solomon BJ, Mok T, Kim DW, Wu YL, Nakagawa
K, Mekhail T, Felip E, Cappuzzo F, Paolini J, Usari T, et al;
PROFILE 1014 Investigators. First-line crizotinib versus
chemotherapy in ALK-positive lung cancer. N Engl J Med.
371:2167–2177. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Shaw AT, Yeap BY, Solomon BJ, Riely GJ,
Gainor J, Engelman JA, Shapiro GI, Costa DB, Ou SH, Butaney M, et
al: Effect of crizotinib on overall survival in patients with
advanced non-small-cell lung cancer harbouring ALK gene
rearrangement: A retrospective analysis. Lancet Oncol.
12:1004–1012. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Rosenberg SA, Yang JC and Restifo NP:
Cancer immunotherapy: Moving beyond current vaccines. Nat Med.
10:909–915. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Brahmer JR, Tykodi SS, Chow LQM, Hwu WJ,
Topalian SL, Hwu P, Drake CG, Camacho LH, Kauh J, Odunsi K, et al:
Safety and activity of anti-PD-L1 antibody in patients with
advanced cancer. N Engl J Med. 366:2455–2465. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Garon E, Balmanoukian A, Hamid O, Hui R,
Gandhi L and Leighi N: Preliminary clinical safety and activity of
MK-3475 monotherapy for the treatment of previously treated
patients with non-small cell lung cancer. In: IASLC 15th World
Conference on Lung Cancer; 27–31 October; Sydney, Australia.
2013
|
|
10
|
Herbst RS, Gordon MS and Fine GD: A study
of MPDL3280A, an engineered PD-L1 antibody in patients with locally
advanced or metastatic tumors. J Clin Oncol. 31:30002013.
|
|
11
|
Horn L, Herbst R and Spiegel D: An
analysis of the relationship of clinical activity to baseline EGFR
status PD-L1 expression and prior treatment history in patients
with non-small cell lung cancer (NSCLC) following PD-L1 blockade
with MPDL3280A (anti-PDL1). In: IASLC 14th World Conference on Lung
Cancer; July 3–7; Amsterdam, The Netherlands. 2012
|
|
12
|
Lynch TJ, Bondarenko I, Luft A,
Serwatowski P, Barlesi F, Chacko R, Sebastian M, Neal J, Lu H,
Cuillerot JM, et al: Ipilimumab in combination with paclitaxel and
carboplatin as first-line treatment in stage IIIB/IV non-small-cell
lung cancer: Results from a randomized, double-blind, multicenter
phase II study. J Clin Oncol. 30:2046–2054. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Creelan BC: Update on immune checkpoint
inhibitors in lung cancer. Cancer Control. 21:80–89. 2014.
|
|
14
|
deLeeuw RJ, Kost SE, Kakal JA and Nelson
BH: The prognostic value of FoxP3+ tumor-infiltrating
lymphocytes in cancer: A critical review of the literature. Clin
Cancer Res. 18:3022–3029. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hori S, Nomura T and Sakaguchi S: Control
of regulatory T cell development by the transcription factor Foxp3.
Science. 299:1057–1061. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fontenot JD, Gavin MA and Rudensky AY:
Foxp3 programs the development and function of
CD4+CD25+ regulatory T cells. Nat Immunol.
4:330–336. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Khattri R, Cox T, Yasayko SA and Ramsdell
F: An essential role for Scurfin in CD4+CD25+
T regulatory cells. Nat Immunol. 4:337–342. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hasegawa T, Suzuki H, Yamaura T, Muto S,
Okabe N, Osugi J, Hoshino M, Higuchi M, Ise K and Gotoh M:
Prognostic value of peripheral and local forkhead box
P3+ regulatory T cells in patients with non-small-cell
lung cancer. Mol Clin Oncol. 2:685–694. 2014.PubMed/NCBI
|
|
19
|
Curotto de Lafaille MA and Lafaille JJ:
Natural and adaptive foxp3+ regulatory T cells: More of
the same or a division of labor? Immunity. 30:626–635. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Apostolou I, Sarukhan A, Klein L and von
Boehmer H: Origin of regulatory T cells with known specificity for
antigen. Nat Immunol. 3:756–763. 2002.PubMed/NCBI
|
|
21
|
Bilate AM and Lafaille JJ: Induced CD4
Foxp3 regulatory T cells in immune tolerance. Annu Rev Immunol.
30:733–758. 2012. View Article : Google Scholar
|
|
22
|
Thornton AM, Korty PE, Tran DQ, Wohlfert
EA, Murray PE, Belkaid Y and Shevach EM: Expression of Helios, an
Ikaros transcription factor family member, differentiates
thymic-derived from peripherally induced Foxp3+ T
regulatory cells. J Immunol. 184:3433–3441. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Akimova T, Beier UH, Wang L, Levine MH and
Hancock WW: Helios expression is a marker of T cell activation and
proliferation. PLoS One. 6:e242262011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gottschalk RA, Corse E and Allison JP:
Expression of Helios in peripherally induced Foxp3+
regulatory T cells. J Immunol. 188:976–980. 2012. View Article : Google Scholar
|
|
25
|
Zabransky DJ, Nirschl CJ, Durham NM, Park
BV, Ceccato CM, Bruno TC, Tam AJ, Getnet D and Drake CG: Phenotypic
and functional properties of Helios+ regulatory T cells.
PLoS One. 7:e345472012. View Article : Google Scholar
|
|
26
|
Getnet D, Grosso JF, Goldberg MV, Harris
TJ, Yen HR, Bruno TC, Durham NM, Hipkiss EL, Pyle KJ and Wada S: A
role for the transcription factor Helios in human
CD4+CD25+ regulatory T cells. Mol Immunol.
47:1595–1600. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Raffin C, Pignon P, Celse C, Debien E,
Valmori D and Ayyoub M: Human memory Helios−
FOXP3+ regulatory T cells (Tregs) encompass induced
Tregs that express Aiolos and respond to IL-1β by downregulating
their suppressor functions. J Immunol. 191:4619–4627. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Elkord E, Sharma S, Burt DJ and Hawkins
RE: Expanded subpopulation of FoxP3+ T regulatory cells
in renal cell carcinoma co-express Helios, indicating they could be
derived from natural but not induced Tregs. Clin Immunol.
140:218–222. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Hatam LJ, Devoti JA, Rosenthal DW, Lam F,
Abramson AL, Steinberg BM and Bonagura VR: Immune suppression in
premalignant respiratory papillomas: Enriched functional
CD4+Foxp3+ regulatory T cells and
PD-1/PD-L1/L2 expression. Clin Cancer Res. 18:1925–1935. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Goldstraw P, Crowley J, Chansky K, Giroux
DJ, Groome PA, Rami-Porta R, Postmus PE, Rusch V and Sobin L;
International Association for the Study of Lung Cancer
International Staging Committee; Participating Institutions. The
IASLC Lung Cancer Staging Project: Proposals for the revision of
the TNM stage groupings in the forthcoming (seventh) edition of the
TNM Classification of malignant tumours. J Thorac Oncol. 2:706–714.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Roncador G, Brown PJ, Maestre L, Hue S,
Martínez-Torrecuadrada JL, Ling KL, Pratap S, Toms C, Fox BC,
Cerundolo V, et al: Analysis of FOXP3 protein expression in human
CD4+CD25+ regulatory T cells at the
single-cell level. Eur J Immunol. 35:1681–1691. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wainwright DA, Sengupta S, Han Y and
Lesniak MS: Thymus-derived rather than tumor-induced regulatory T
cells predominate in brain tumors. Neuro Oncol. 13:1308–1323. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Polansky JK, Schreiber L, Thelemann C,
Ludwig L, Krüger M, Baumgrass R, Cording S, Floess S, Hamann A and
Huehn J: Methylation matters: Binding of Ets-1 to the demethylated
Foxp3 gene contributes to the stabilization of Foxp3 expression in
regulatory T cells. J Mol Med Berl. 88:1029–1040. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kim YC, Bhairavabhotla R, Yoon J, Golding
A, Thornton AM, Tran DQ and Shevach EM: Oligodeoxynucleotides
stabilize Helios-expressing Foxp3+ human T regulatory
cells during in vitro expansion. Blood. 119:2810–2818. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Rossignol M, Pouysségur J and Klagsbrun M:
Characterization of the neuropilin-1 promoter; gene expression is
mediated by the transcription factor Sp1. J Cell Biochem.
88:744–757. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yadav M, Louvet C, Davini D, Gardner JM,
Martinez-Llordella M, Bailey-Bucktrout S, Anthony BA, Sverdrup FM,
Head R, Kuster DJ, et al: Neuropilin-1 distinguishes natural and
inducible regulatory T cells among regulatory T cell subsets in
vivo. J Exp Med. 209:1713–1722. s1711–1719. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Weiss JM, Bilate AM, Gobert M, Ding Y,
Curotto de Lafaille MA, Parkhurst CN, Xiong H, Dolpady J, Frey AB,
Ruocco MG, et al: Neuropilin 1 is expressed on thymus-derived
natural regulatory T cells, but not mucosa-generated induced Foxp3
T reg cells. J Exp Med. 209:1723–1742. s17212012. View Article : Google Scholar :
|
|
38
|
Lin X, Chen M, Liu Y, Guo Z, He X, Brand D
and Zheng SG: Advances in distinguishing natural from induced
Foxp3+ regulatory T cells. Int J Clin Exp Pathol.
6:116–123. 2013.
|
|
39
|
Onizuka S, Tawara I, Shimizu J, Sakaguchi
S, Fujita T and Nakayama E: Tumor rejection by in vivo
administration of anti-CD25 (interleukin-2 receptor alpha)
monoclonal antibody. Cancer Res. 59:3128–3133. 1999.PubMed/NCBI
|
|
40
|
Gallimore A and Sakaguchi S: Regulation of
tumour immunity by CD25+ T cells. Immunology. 107:5–9.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Morse MA, Hobeika AC, Osada T, Serra D,
Niedzwiecki D, Lyerly HK and Clay TM: Depletion of human regulatory
T cells specifically enhances antigen-specific immune responses to
cancer vaccines. Blood. 112:610–618. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Sutmuller RP, van Duivenvoorde LM, van
Elsas A, Schumacher TN, Wildenberg ME, Allison JP, Toes RE,
Offringa R and Melief CJ: Synergism of cytotoxic T
lymphocyte-associated antigen 4 blockade and depletion of
CD25+ regulatory T cells in antitumor therapy reveals
alternative pathways for suppression of autoreactive cytotoxic T
lymphocyte responses. J Exp Med. 194:823–832. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Ko K, Yamazaki S, Nakamura K, Nishioka T,
Hirota K, Yamaguchi T, Shimizu J, Nomura T, Chiba T and Sakaguchi
S: Treatment of advanced tumors with agonistic anti-GITR mAb and
its effects on tumor-infiltrating
Foxp3+CD25+CD4+ regulatory T
cells. J Exp Med. 202:885–891. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
GlaxoSmithKline plc. Update on phase III
clinical trial of investigational MAGE-A3 antigen-specific cancer
immunotherapeutic in non-small cell lung cancer [Internet].
02–April. 2014, [cited 2015 March 24] Available from http://www.gsk.com/en-gb/media/press-releases/2014/update-on-phase-iii-clinical-trial-of-investigational-mage-a3-antigen-specific-cancer-immunother-apeutic-in-non-small-cell-lung-cancer/.
|
|
45
|
Suzuki H, Fukuhara M, Yamaura T, Mutoh S,
Okabe N, Yaginuma H, Hasegawa T, Yonechi A, Osugi J, Hoshino M, et
al: Multiple therapeutic peptide vaccines consisting of combined
novel cancer testis antigens and anti-angiogenic peptides for
patients with non-small cell lung cancer. J Transl Med. 11:972013.
View Article : Google Scholar : PubMed/NCBI
|