Introduction
Dendritic cells (DCs) are antigen-presenting cells
that regulate the immune response via interactions between major
histocompatibility complex (MHC) molecules on DCs and T-cell
receptors on T cells alongside costimulatory molecules (1). Cancer vaccines using DCs loaded with
anti-genic peptides, tumor lysates, mRNA, DNA or whole tumor cells
are an approved approach for enhancing a patient's own immune
system to eradicate tumor cells (2). In clinical trials, MHC class
I-restricted peptide-loaded DCs have been most commonly used to
induce antigen-specific CD8+ cytotoxic T lymphocyte
(CTL) responses and, in some cases, have been associated with
clinical benefits; however, clinical efficacy has been limited
(3,4). It has been reported that activation
of CD4+ T helper type 1 (Th1) cell responses via MHC
class II molecules on mature DCs (mDCs) is essential for supporting
CD8+ CTL priming, memory formation, recruitment to tumor
tissue and tumor cell recognition (5-7).
Therefore, several long peptides targeting tumor-associated antigen
(TAA)-specific CD4+ cells have been developed for the
treatment of patients with cancer (5-10).
Our previous study reported the clinical benefits of using MHC
class I and II peptide-pulsed mDC vaccines to simultaneously induce
TAA-specific CD4+ Th1 cells and CD8+ CTLs
(4).
At present, numerous TAAs, such as Wilms' tumor 1
(WT1), HER-2/neu, gp100 and tyrosinase, have been isolated and
incorporated into cancer vaccine preparations in the form of
peptides (3-10). A promising target for cancer
vaccines, WT1 was ranked as the top antigen among 75 TAAs due to
its high immunogenicity (11). WT1
is highly expressed in various types of tumor and serves oncogenic
roles in their formation (12-14).
Additionally, WT1 has been successfully applied to induce both
CD4+ Th1 cells and CD8+ CTLs (15,16).
WT1-specific CD4+ Th1 cells contribute to the induction
and maintenance of WT1-specific CD8+ CTLs by
establishing the required cytokine milieu via cytokine secretion,
including interleukin (IL)-2 and interferon-γ (IFN-γ) (15). In our previous study, mDCs were
pulsed with three types of MHC class I- or class II-restricted WT1
peptide: i) 126-134 (amino acid sequence, RMFPNAPYL) for human
leukocyte antigen (HLA)-A*02:01/02:06; ii) 235-243 (amino acid
sequence, CYTWNQMNL) for HLA-A*24:02; and iii) 332-347 (amino acid
sequence, KRYFKLSHLQMHSRKH) for MHC class II (4). Patients with advanced pancreatic
ductal adenocarcinoma (PDA) were then treated with pulsed mDCs in
combination with standard chemotherapy. In the clinical trial, 4 of
7 patients with PDA exhibited strongly positive WT1-specific
immunity, resulting in prolongation of overall survival. However,
WT1-specific immunity was not induced in the remaining 3 patients.
Therefore, a novel WT1 helper peptide with increased immunogenicity
may be essential for the induction and maintenance of antitumor
immunity in clinical trials to generate more successful
results.
Recently, a novel WT1 helper peptide (WT1
HP34-51; amino acid sequence, WAPVLDFAPPGASAYGSL) was
identified (17). Of note, WT1
HP34-51 contains a killer WT1 peptide (WT1
KP37-45; amino acid sequence, VLDFAPPGA) that has
affinity for HLA-A*02:01 (18).
Accordingly, the novel peptide WT1 HP34-51 may induce
not only WT1-specific CD4+ Th1 cells but also
CD8+ CTLs simultaneously, at least partially, in an
HLA-A*02:01-restricted manner. Thus far, the function of WT1
HP34-51 remains unclear. In the present study, the
impact of mDCs pulsed with the novel peptide WT1 HP34-51
(mDC/WT1 HP34-51) on the induction of WT1-reactive
antitumor immunity was assessed.
Materials and methods
Prediction of peptide-MHC class I binding
or peptide-MHC class II molecules
WT1 peptide-MHC class I binding affinity was
predicted by the NetMHC 4.0 server-prediction program (http://www.cbs.dtu.dk/services/NetMHC-4.0). Moreover,
WT1 peptide-MHC class II binding affinity was predicted by the
NetMHCII-2.3 server-prediction program (https://services.healthtech.dtu.dk/service.php?NetMHCII-2.3).
Patient characteristics
The present study was jointly reviewed and approved
by the Ethics Committee of the Jikei Institutional Review Board,
Jikei University School of Medicine and the Clinical Study
Committee of Jikei University Kashiwa Hospital [approval no. 29-063
(8679)]. All patients provided written informed consent for the use
of their samples in scientific research before samples were
collected. To assess the stimulatory ability of mDC/WT1
HP34-51 for the induction of CD8+ CTLs
restricted by HLA-A2, 14 patients (7 male, 7 female; aged 37-88
years) with various types of cancer (cancer of the oropharynx,
lungs, esophagus, stomach, breast, pancreas, ovary, gallbladder,
biliary duct, prostate or uterus) expressing an HLA-A2 allele
[02:01 (n=5), 02:06 (n=7) or 02:07 (n=2)] were enrolled in the
Tokyo Midtown Center for Advanced Medical Science and Technology
between August 2017 and April 2019 (Table I).
 | Table IPatient characteristics. |
Table I
Patient characteristics.
| Patient | Sex | Age, years | Cancer | HLA-A |
|---|
| 1 | Male | 69 | Lung and
esophagus | 02:01/26:01 |
| 2 | Female | 49 | Breast | 02:01/11:01 |
| 3 | Female | 82 | Stomach | 02:01/24:02 |
| 4 | Male | 45 | Pancreas | 02:01/24:02 |
| 5 | Male | 59 | Gallbladder and
bile duct | 02:01/24:02 |
| 6 | Male | 52 | Oropharynx | 02:06/24:02 |
| 7 | Male | 88 | Stomach | 02:06/26:03 |
| 8 | Female | 47 | Ovary | 02:06/24:02 |
| 9 | Female | 47 | Ovary | 02:06/11:01 |
| 10 | Female | 70 | Pancreas | 02:06/24:02 |
| 11 | Male | 65 | Prostate | 02:06 |
| 12 | Female | 60 | Ovary | 02:06/24:02 |
| 13 | Female | 37 | Uterus | 02:07/24:02 |
| 14 | Male | 46 | Pancreas | 02:07/24:02 |
Preparation of WT1-pulsed DCs
Peripheral blood mono-nuclear cells (PBMCs) were
collected from the 14 patients, none of whom had received cancer
vaccines, via leukapheresis and cryopreserved at the Tokyo Midtown
Center for Advanced Medical Science and Technology. PBMCs were
isolated using a Ficoll-Plaque Premium (Cytiva) density gradient
solution, cryopreserved and stored for future experiments. After
cryopreserved PBMCs were thawed and washed, their viability was
assessed using trypan blue solution (Sigma-Aldrich; Merck KGaA).
Trypan blue staining was performed according to the manufacturer's
protocols. Cell viability was assessed under a CK40-F100 light
microscope (magnification, ×100; Olympus Corporation).
Subsequently, the cells were incubated in serum-free AIM-V medium
(Gibco; Thermo Fisher Scientific, Inc.) in a 10-cm Primaria cell
culture dish with surface-modified polystyrene for enhanced cell
culture (Corning Inc.) under 5% CO2 at 37°C in a
humidified incubator. After >30 min, plastic-adherent monocytes
and nonadherent (NAD) cells were isolated by washing with gentle
pipetting. NAD cells were cryopreserved in Bambanker solution
(Nippon Genetics Co., Ltd.) and used for induction of WT1-reactive
T cells. Adherent monocytes were cultured in serum-free AIM-V
medium containing granulocyte macrophage colony-stimulating factor
(GM-CSF; 50 ng/ml; Primmune Inc.) and IL-4 (50 ng/ml; R&D
Systems, Inc.) under 5% CO2 at 37°C in a humidified
incubator for 5 days to generate immature DCs (imDCs). Autologous
imDCs were cryopreserved until they were pulsed with WT1 peptides
or cocultured with NAD cells.
The viability and function of mDCs were not altered
by whether fresh or cryopreserved PBMCs or imDCs were used for mDC
production (19). Therefore,
cryopreserved imDCs were used to prepare WT1-pulsed DCs in the
present study. To generate WT1 peptide-pulsed autologous mDCs,
cryopreserved imDCs (1×105) were thawed and incubated
under 5% CO2 at 37°C in a humidified incubator for 24 h
with 100 µg WT1 HP34-51 or WT1 KP37-45
(all peptides obtained from Greiner Bio-One GmbH) in the presence
of GM-CSF (5 ng/ml), prostaglandin E2 (50 ng/ml; Daiichi Fine
Chemical Co., Ltd.) and lyophilized preparations of a
penicillin-killed, low-virulence strain of Streptococcus
pyogenes (OK-432; 10 µg/ml; Chugai Pharmaceutical Co.,
Ltd.). Autologous imDCs (1×104) were also cultured with
25 µg WT1 HP34-51 or WT1 KP37-45 under
5% CO2 at 37°C in a humidified incubator for 24 h to
generate WT1 peptide-pulsed imDCs, which were used as a second
stimulator.
Phenotypes of DCs and NAD cells
To assess DC phenotypes, imDCs or mDCs
(1×105/100 µl) were washed, incubated with Clear
Back human Fc receptor blocking reagent (dilution 1:20; cat. no.
MTG-001; MBL Life Science) for 5 min at 4°C and then stained with
the following monoclonal antibodies (mAbs) at 4°C for 30 min:
Fluorescein isothiocyanate (FITC)-conjugated anti-human HLA-ABC
(dilution 1:20; cat. no. 311404; clone W6/32), anti-CD80 (dilution
1:20; cat. no. 305206; clone 2D10), anti-CD40 (dilution 1:20; cat.
no. 334306; clone 5C3; all from BioLegend, Inc.); FITC-conjugated
anti-CD14 (dilution 1:20; cat. no. 11-0149-42; clone 61D3;
eBioscience; Thermo Fisher Scientific, Inc.); and phycoerythrin
(PE)-conjugated anti-human HLA-DR (dilution 1:20; cat. no. 307606;
clone L243), anti-CD11c (dilution 1:20; cat. no. 301606; clone
3.9), anti-CD83 (dilution 1:20; cat. no. 305308; clone HB15e) and
anti-CD86 (dilution 1:20; cat. no. 305406; clone IT2.2; all from
BioLegend, Inc.). In addition, to assess NAD pheno-types, NAD cells
(1×105/100 µl) were washed, incubated with Clear
Back human Fc receptor blocking reagent for 5 min at 4°C and then
stained with the following mAbs at 4°C for 30 min: FITC-conjugated
anti-human CD14 (dilution 1:20; cat. no. 11-0149-42; clone 61D3;
eBioscience; Thermo Fisher Scientific, Inc.); FITC-conjugated
anti-CD8 (dilution 1:60; cat. no. 301006; clone RPA-T8),
PE-conjugated anti-human CD19 (dilution 1:40; cat. no. 302208;
clone HIB19), allophycocyanin (APC)-conjugated anti-human CD56
(dilution 1:20; cat. no. 318310; clone HCD56) and APC/cyanine 7
(Cy7)-conjugated anti-human CD4 (dilution 1:60; cat. no. 317418;
clone OKT4; all from BioLegend, Inc.). Phenotypes were analyzed
using an Attune NxT flow cytometer (Thermo Fisher Scientific, Inc.)
and FlowJo analysis software (version 7.6.5; Tree Star, Inc.).
Prior to using the analyzer, 4 µg/ml propidium iodide
(Sigma-Aldrich; Merck KGaA) was added to the samples to exclude
dead cells at 4°C for ~5 min.
Induction of WT1-reactive CD4+
and CD8+ T cells
Cryopreserved NAD cells were thawed, washed and
incubated under 5% CO2 at 37°C in a humidified incubator
for 24 h in 24-well plates with RPMI-1640 medium (Sigma-Aldrich;
Merck KGaA) supplemented with 1% minimum essential medium
nonessential amino acids, 1 mM sodium pyruvate, 50 µM
2-mercaptoethanol (all from Gibco; Thermo Fisher Scientific, Inc.)
and 10% heat-deactivated fetal calf serum (Cytiva) in the presence
of IL-2 (20 U/ml; Shionogi & Co., Ltd.) and IL-7 (20 ng/ml;
PeproTech, Inc.). The next day (day 1), WT1
HP34-51-pulsed mDCs (mDC/WT1 HP34-51;
1×105) or WT1 KP37-45-pulsed mDCs (mDC/WT1
KP37-45; 1×105) were cocultured with NAD
cells (1×106) in RPMI-1640 medium containing 1% minimum
essential medium nonessential amino acids, 1 mM sodium pyruvate, 50
µM 2-mercaptoethanol and 10% heat-deactivated fetal calf
serum in the presence of low doses of IL-2 (10 U/ml) and IL-7 (10
ng/ml) under 5% CO2 at 37°C in a humidified incubator
for 7 days. At day 8, the stimulated cells (1×106) were
washed and cocultured again with freshly prepared mDC/WT1
HP34-51 (1×105) or mDC/WT1 KP37-45
(1×105) in the fresh culture medium containing IL-2 (10
U/ml) and IL-7 (10 ng/ml) for 7 days. To generate WT1-reactive T
cells effectively, the stimulation was repeated in the same manner
for a total of 4 times (day 1, 8, 15 and 22). In addition, as a
control (no stimulation group), NAD cells (1×106) were
maintained with IL-2 (10 U/ml) and IL-7 (10 ng/ml) in 7-day
intervals for a total of 4 times under 5% CO2 at 37°C in
a humidified incubator.
Microscopic analysis of cell
clusters
On day 4 of the final 7-day simulation interval (day
26), cell cluster formation was examined under a EVOS™ XL core
imaging system (magnification, ×4; cat. no. AMEX1000; Thermo Fisher
Scientific, Inc.).
Detection of CD4+ or
CD8+ T cells producing IFN-γ in response to
WT1-KP37-45
To assess the impact of mDC/WT1 HP34-51
on the activation of WT1-specific T cells, WT1-reactive T cells
(1×105) induced by 4 total stimulations with mDC/WT1
HP34-51 or mDC/WT1 KP37-45 were restimulated
once with WT1 HP34-51-pulsed imDCs (imDC/WT1
HP34-51), WT1 KP37-45-pulsed imDCs (imDC/WT1
KP37-45) or imDCs (1×104) alone in 96-well
U-bottomed plates (Corning Inc.) under 5% CO2 at 37°C in
a humidified incubator for 6 h using BD GolgiStop (BD Biosciences).
After stimulation, the cells were washed, incubated with Clear Back
human Fc receptor blocking reagent for 5 min at 4°C and then
stained with a PE/Cy5-conjugated anti-human CD8 mAb (dilution 1:20;
cat. no. 15-0088-71; clone RPA-T8; eBioscience; Thermo Fisher
Scientific, Inc.) and an APC/Cy7-conjugated anti-human CD4 mAb
(dilution 1:60; cat. no. 317418; clone OKT4; BioLegend, Inc.).
Thereafter, they were washed, fixed at 4°C for 20 min and then
permeabilized using a BD Cytofix/Cytoperm Plus
Fixation/Permeabilization kit with BD GolgiStop (BD Biosciences).
The cells were then incubated with an APC-conjugated anti-human
IFN-γ mAb (dilution 1:25; cat. no. 506510; clone B27; BioLegend,
Inc.). Lymphocytes were gated on an forward scatter-side scatter
(FSC-SSC) dot plot, and CD4+ or CD8+ T cells
were regated on a CD4/IFN-γ or CD8/IFN-γ dot plot, respectively.
The percentages of CD4+IFN-γ+ cells and
CD8+IFN-γ+ cells among the CD4+ or
CD8+ T cell population, respectively, were analyzed
using the same equipment and software as were used to analyze
DC/NAD phenotypes.
WT1-specific memory cells in
CD4+ or CD8+ T cell populations induced by
mDC/WT1 HP34-51
To assess the memory cell subsets of activated
WT1-reactive CD4+ or CD8+ T cells, NAD cells
were first stimulated with mDC/WT1 HP34-51 or mDC/WT1
KP37-45 for 4 times, followed by restimulation with
imDC/WT1 KP37-45 at a ratio of 10:1 under 5%
CO2 at 37°C in a humidified incubator. NAD cells without
any stimulation were maintained and used as a control. After
stimulation, the cells were washed, incubated with Clear Back human
Fc receptor blocking reagent as previously described and then
stained with an FITC-conjugated anti-human exon 4 splice variant of
the tyrosine phosphatase (CD45RA) mAb (dilution 1:40; cat. no.
304106; clone HI100; BioLegend, Inc.), a PE-conjugated anti-human
chemokine receptor type 7 (CCR7) mAb (dilution 1:10; cat. no.
FAB-197P; clone 150503; R&D Systems, Inc.), or a
PE/Cy5-conjugated anti-human CD8 mAb and an APC/Cy7-conjugated
anti-human CD4 mAb at 4°C for 30 min. Lymphocytes were gated on an
FSC-SSC dot plot, CD4+ or CD8+ T cells were
regated, and CD45RA and CCR7 were then analyzed using the same
equipment and software as were used to analyze DC/NAD phenotypes.
Activated CD4+ or CD8+ T cells can be
classified into 4 groups based on CD45RA and CCR7:
CD45RA+CCR7+ naïve,
CD45RA−CCR7+ central memory (CM),
CD45RA−CCR7− effector memory (EM) and
CD45RA+CCR7− terminally differentiated
effector memory (EMRA) cells.
Statistical analysis
Data are presented as the mean ± SD. Comparisons of
the difference in the percentage of cell surface markers between
imDCs and mDCs were performed with a paired t-test or a Wilcoxon
signed-rank test. P<0.05 was considered to indicate a
statistically significant difference. Statistical analyses were
performed using Microsoft Office Excel 2011 (Microsoft Corporation)
with the add-in software Statcel 3 (OMS Publishing Inc.).
Results
Binding of WT1 peptides to MHC class I or
MHC class II molecules
According to the NetMHC 4.0 server-prediction
program, WT1 KP37-45 within WT1 HP34-51
exhibited high affinity for HLA-A*02:01 and A*02:07. Moreover,
according to the NetMHCII-2.3 server-prediction program, WT1
HP34-51 and WT1 KP37-45 exhibited high
affinity for MHC class II (data not shown).
Characterization of imDCs, mDCs or NAD
cells
Frozen and stored PBMCs were used, and the yield of
mDCs at the end of the production process was 2.47±1.86% (n=3). As
not enough cells were available for patients 4 and 9, the
immunophenotype results of the imDCs and mDCs from the 12 cases are
presented in Tables SI and SII.
Both imDCs and mDCs generated from 12 of the analyzed cancer
patients displayed a characteristic phenotype comprising MHC class
I (HLA-ABC), MHC class II (HLA-DR), CD14, CD11c, CD80, CD86, CD40
and CD83 expression (Fig. S1, Tables
SI and SII). In addition, the percentages of mDCs expressing
CD80, CD86, CD40 and CD83 were significantly increased compared
with imDCs (P<0.001, P=0.002, P=0.021 and P<0.001,
respectively), indicating that mDCs were activated by exposure of
imDCs to OK-432 (Tables SI and
SII). In contrast, the NAD cell composition (n=5) included
lymphocytes (CD8+ cells, 27.14±8.44%; CD4+
cells, 54.49±10.38%; CD19+ cells, 0.78±0.42%;
CD56+ cells, 9.80±3.86%) and monocytes (CD14+
cells, 2.24±1.20%; data not shown).
WT1-reactive T cells generated by mDC/WT1
HP34-51 stimulation
To assess the stimulatory capacity of mDC/WT1
HP34-51 for induction of HLA-A*02:01-restricted
CD8+ T cells, NAD cells from patient 4
(HLA-A*02:01/24:02; Table I) were
first stimulated 4 times in 7-day intervals with mDC/WT1
HP34-51. As a control, NAD cells were also first
cultured with mDC/WT1 KP37-45, or not stimulated but
maintained with low doses of IL-2 and IL-7 4 times in 7-day
intervals. The cells were then restimulated with imDC/WT1
HP34-51, imDC/WT1 KP37-45 or imDCs alone
(Fig. 1A). At 4 days after the
final stimulation with mDC/WT1 HP34-51 or mDC/WT1
KP37-45 (Fig. 1A), cell
clusters had formed (Fig. 1B). In
contrast, there was no cluster formation by cells in the no
stimulation group (Fig. 1B). These
results indicated that both mDC/WT1 HP34-51 and mDC/WT1
KP37-45 exhibited the ability to stimulate these
cells.
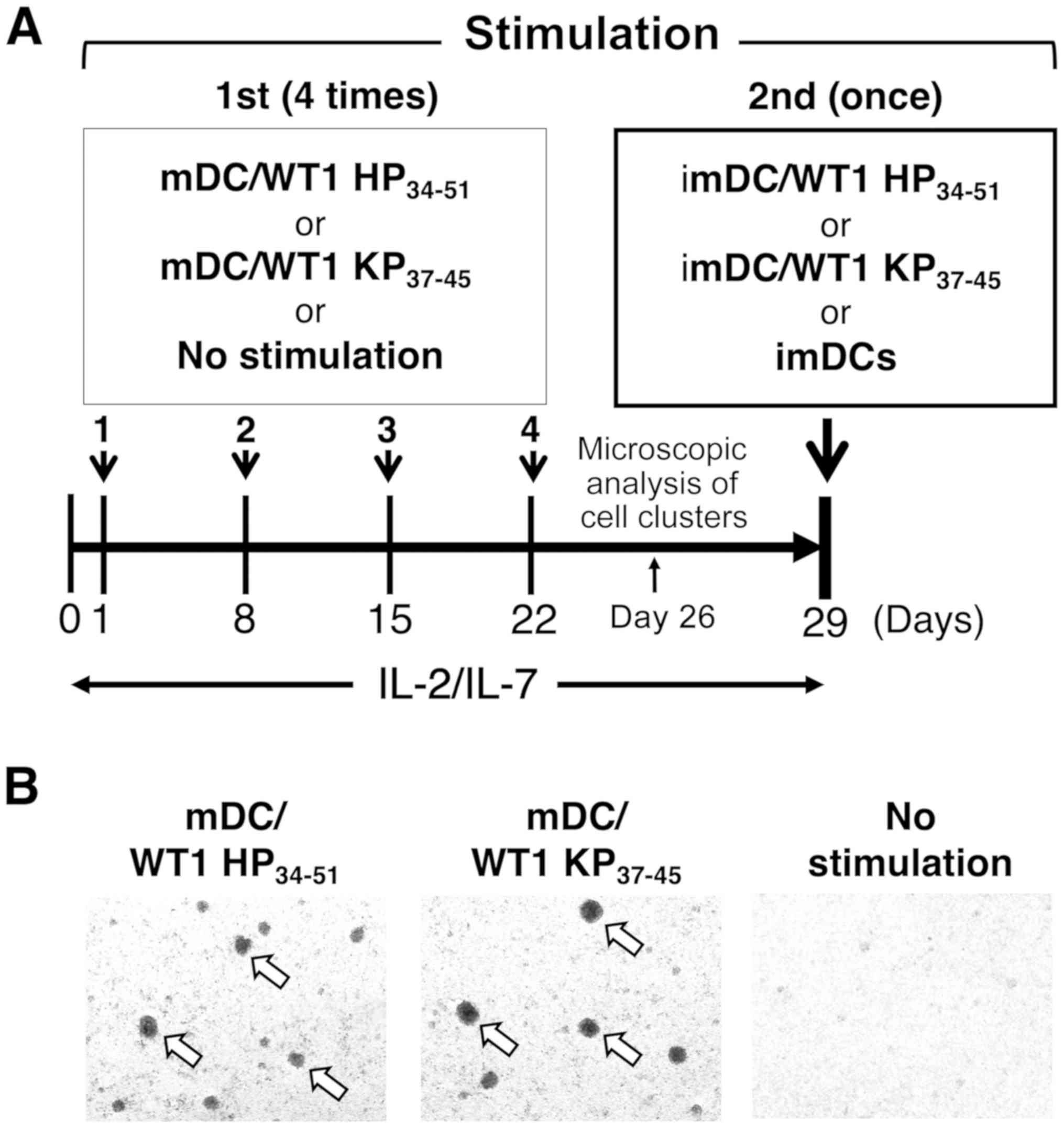 | Figure 1Stimulation of T cells with mDC/WT1
HP34-51 or mDC/WT1 KP37-45. (A) Schematic
representation of methods to induce WT1-reactive CD4+
and CD8+ T cells. (B) At 4 days after 4 times
stimulation with mDC/WT1 HP34-51 (left) or mDC/WT1
KP37-45 (middle) in 7-day intervals (day 26), but not
incubation without stimulation (right), the formation of cell
clusters (white arrows) was observed (magnification, ×4).
Representative images of two independent experiments are presented.
mDC, mature dendritic cell; imDC, immature dendritic cell; WT1,
Wilms' tumor 1; HP34-51, helper peptide;
KP37-45, killer peptide; mDC/WT1 HP34-51,
mDCs pulsed with WT1 HP34-51; mDC/WT1
KP37-45, mDCs pulsed with WT1 KP37-45; IL,
interleukin. |
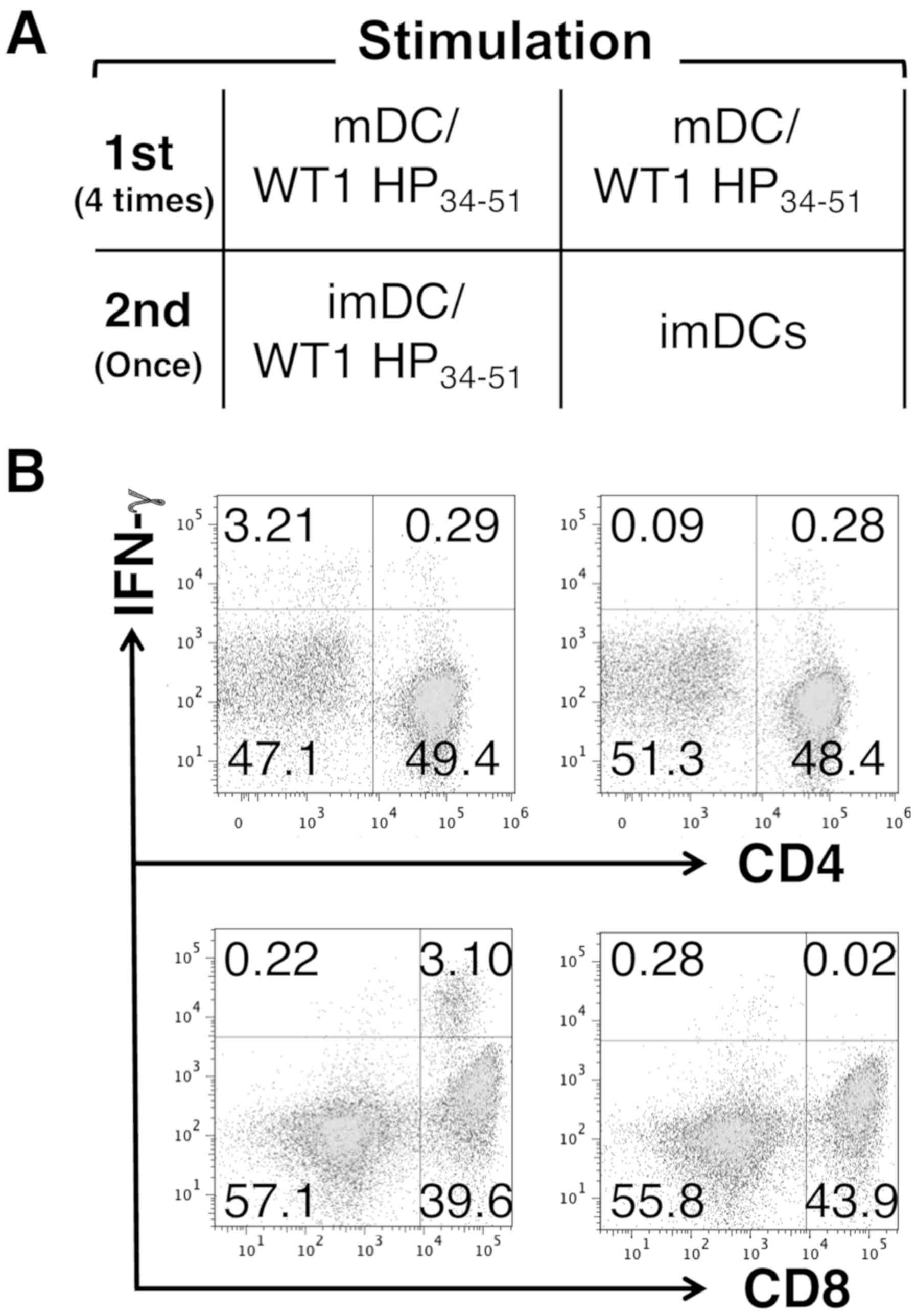
Then, it was evaluated as to whether mDC/WT1
HP34-51 induced WT1-reactive T cells with the functional
capacity to produce IFN-γ by monitoring the activity of
CD4+ and CD8+ T cells in response to imDC/WT1
HP34-51 (Fig. 2A). The
percentages of CD4+IFN-γ+ T cells among total
CD4+ T cells were 0.58% [0.29/(0.29+49.4)] and 0.58%
[0.28/(0.28+48.4)] after restimulation with imDC/WT1
HP34-51 or imDCs alone, respectively (Fig. 2B). In contrast to CD4+ T
cells, the percentages of CD8+IFN-γ+ T cells
among total CD8+ T cells after restimulation with
imDC/WT1 HP34-51 or imDCs alone were 7.26%
[3.10/(3.10+39.6)] and 0.05% [0.02/(0.02+43.9)], respectively
(Fig. 2B). These results suggested
that mDC/WT1 HP34-51 possessed the ability to stimulate
CD8+ T cells that can produce IFN-γ.
Whether mDC/WT1 HP34-51 had the capacity
to induce IFN-γ-producing CD8+ T cells similar to that
of mDC/WT1 KP37-45 was also evaluated (Fig. 3A). The percentages of
CD4+IFN-γ+ T cells among total
CD4+ T cells stimulated with mDC/WT1 HP34-51
or mDC/WT1 KP37-45 or left unstimulated, all of which
were subsequently restimulated once with imDC/WT1
KP37-45, were 0.68% [0.34/(0.34+49.3)], 1.00%
[0.51/(0.51+50.3)], and 0.19% [0.08/(0.08+41.9)], respectively
(Fig. 3B). Moreover, imDCs were
used as a control for the second stimulator, and the percentages of
CD4+IFN-γ+ T cells among total
CD4+ T cells stimulated with mDC/WT1 HP34-51
or mDC/WT1 KP37-45 following restimulation with imDCs
alone were 0.58% [0.28/(0.28+48.4); Fig. 2B] and 0.68% [0.34/(0.34+49.6);
Fig. S2], respectively. These
results suggested that mDC/WT1 HP34-51 and mDC/WT1
KP37-45 exhibited a capacity to induce IFN-γ-producing
CD4+ T cells after restimulation with imDC/WT1
KP37-45 when compared with no stimulation. In contrast
to CD4+ T cells, the percentages of
CD8+IFN-γ+ T cells among total
CD8+ T cells stimulated 4 times with mDC/WT1
HP34-51 or mDC/WT1 KP37-45 and restimulated
once with imDC/WT1 KP37-45 were 7.96% [3.39/(3.39+39.2)]
and 5.13% [2.64/(2.64+48.8)], respectively (Fig. 3B). Furthermore, the percentages of
CD8+IFN-γ+ T cells among total
CD8+ T cells stimulated with mDC/WT1 HP34-51
or mDC/WT1 KP37-45 following restimulation with imDCs
alone were 0.05% [0.02/(0.02+43.9); Fig. 2B] and 0.10% [0.05/(0.05+52.2);
Fig. S2], respectively. In
addition, 0.60% [0.30/(0.30+50.0)] of total CD8+ T cells
were CD8+IFN-γ+ following no stimulation
during 29 days of culture and then one stimulation with imDC/WT1
KP37-45 (Fig. 3B). Of
note, CD8+ T cells stimulated 4 times with mDC/WT1
HP34-51 produced IFN-γ at higher levels than those
stimulated 4 times with mDC/WT1 KP37-45 after one
restimulation with imDC/WT1 KP37-45 (Fig. 3B). These results suggested that the
combination treatment of mDC/WT1 HP34-51 and imDC/WT1
KP37-45 exhibited the capacity to induce IFN-γ-producing
CD8+ T cells more frequently than that of mDC/WT1
KP37-45 and imDC/WT1 KP37-45.
 | Figure 3Comparison of the efficiencies of
mDC/WT1 HP34-51 and mDC/WT1 KP + + 37-45 in
activating WT1-specific CD4 or CD8 T cells. (A) Nonadherent cells
were first stimulated with mDC/WT1 HP34-51, mDC/WT1
KP37-45 or IL-2/IL-7 alone. The activated T cells were
restimulated with imDC/WT1 KP37-45. (B) Dot plots of
IFN-γ-producing CD4+ (top) and CD8+ T cells
(bottom) derived from a patient with pancreatic ductal
adenocarcinoma after stimulation with mDC/WT1 HP34-51
(left), mDC/WT1 KP37-45 (middle) or no stimulation
(right) and restimulation with imDC/WT1 KP37-45.
Representative plots of two independent experiments are presented.
mDC, mature dendritic cell; imDC, immature dendritic cell; WT1,
Wilms' tumor 1; HP34-51, helper peptide;
KP37-45, killer peptide; mDC/WT1 HP34-51,
mDCs pulsed with WT1 HP34-51; mDC/WT1
KP37-45, mDCs pulsed with WT1 KP37-45;
imDC/WT1 KP37-45, imDCs pulsed with WT1
KP37-45; IL, interleukin; IFN-γ, interferon-γ. |
WT1-specific memory cells in activated
CD4+ or CD8+ T cells stimulated with mDC/WT1
HP34-51
As mDC/WT1 HP34-51 and mDC/WT1
KP37-45 stimulated WT1 KP37-45-reactive
CD4+ T cells, patient 4 was selected for an analysis of
the memory cell subsets of CD4+ T cells (Fig. 4A). The percentages of EM cells
among total CD4+ T cells stimulated with mDC/WT1
HP34-51, mDC/WT1 KP37-45 or left unstimulated
were 78.3, 73.7 and 30.8%, respectively (Fig. 4B and C). The percentages of EMRA
cells among total CD4+ T cells stimulated with mDC/WT1
HP34-51 or mDC/WT1 KP37-45 were 8.29 and
6.67%, respectively (Fig. 4B and
C). These percentages were higher than those in the no
stimulation group (3.54%). The results suggested that mDC/WT1
HP34-51 and mDC/WT1 KP37-45 induced EM and
EMRA CD4+ T cells. Additionally, the percentage of naïve T cells
among total CD4+ T cells was decreased after stimulation
with mDC/WT1 HP34-51 or mDC/WT1 KP37-45
(Fig. 4B and C).
 | Figure 4Memory cell subsets of
CD4+ T cells stimulated with mDC/WT1 HP34-51
or mDC/WT1 KP37-45. (A) Nonadherent cells were first
stimulated 4 times with mDC/WT1 HP34-51, mDC/WT1
KP37-45 or IL-2/IL-7 alone. The activated T cells were
restimulated with imDC/WT1 KP37-45. (B) Dot plots of the
memory cell subsets in CD4+ T cells derived from a
patient with pancreatic ductal adenocarcinoma activated by mDC/WT1
HP34-51 (left), mDC/WT1 KP37-45 (middle) or
no stimulation (right), followed by restimulation with imDC/WT1
KP37-45. (C) Percentage of four subgroups (N, CM, EM or
EMRA) of memory CD4+ T cells activated by mDC/WT1
HP34-51 (left), mDC/WT1 KP37-45 (middle) or
no stimulation (right), and restimulation with imDC/WT1
KP37-45. mDC, mature dendritic cell; imDC, immature
dendritic cell; WT1, Wilms' tumor 1; HP34-51, helper
peptide; KP37-45, killer peptide; mDC/WT1
HP34-51, mDCs pulsed with WT1 HP34-51;
mDC/WT1 KP37-45, mDCs pulsed with WT1
KP37-45; imDC/WT1 KP37-45, imDCs pulsed with
WT1 KP37-45; IL, interleukin; N, naïve; CM, central
memory; EM, effector memory; EMRA, terminally differentiated
effector memory; CCR7, chemo-kine receptor 7; CD45RA, human exon 4
splice variant of the tyrosine phosphatase. |
As mDC/WT1 HP34-51 and mDC/WT1
KP37-45 also stimulated WT1 KP37-45-reactive
CD8+ T cells, patient 4 was also selected for an
analysis of the memory cell subsets of CD8+ T cells
(Fig. 5A). In contrast to
CD4+ T cells, the percentages of EM cells among total
CD8+ T cells stimulated with mDC/WT1 HP34-51,
mDC/WT1 KP37-45 or left unstimulated after one
restimulation with imDC/WT1 KP37-45 were 51.3, 53.2 and
56.4%, respectively (Fig. 5B and
C). The percentages of EMRA cells among total CD8+ T
cells stimulated with mDC/WT1 HP34-51, mDC/WT1
KP37-45 or left unstimulated following one restimulation
with imDC/WT1 KP37-45 were 45.0, 42.4 and 37.6%,
respectively (Fig. 5B and C).
These results suggested that the frequencies of CD8+
memory T cell subsets from a patient with PDA showed no notable
differences between the mDC/WT1 HP34-51, mDC/WT1
KP37-45 and no stimulation groups.
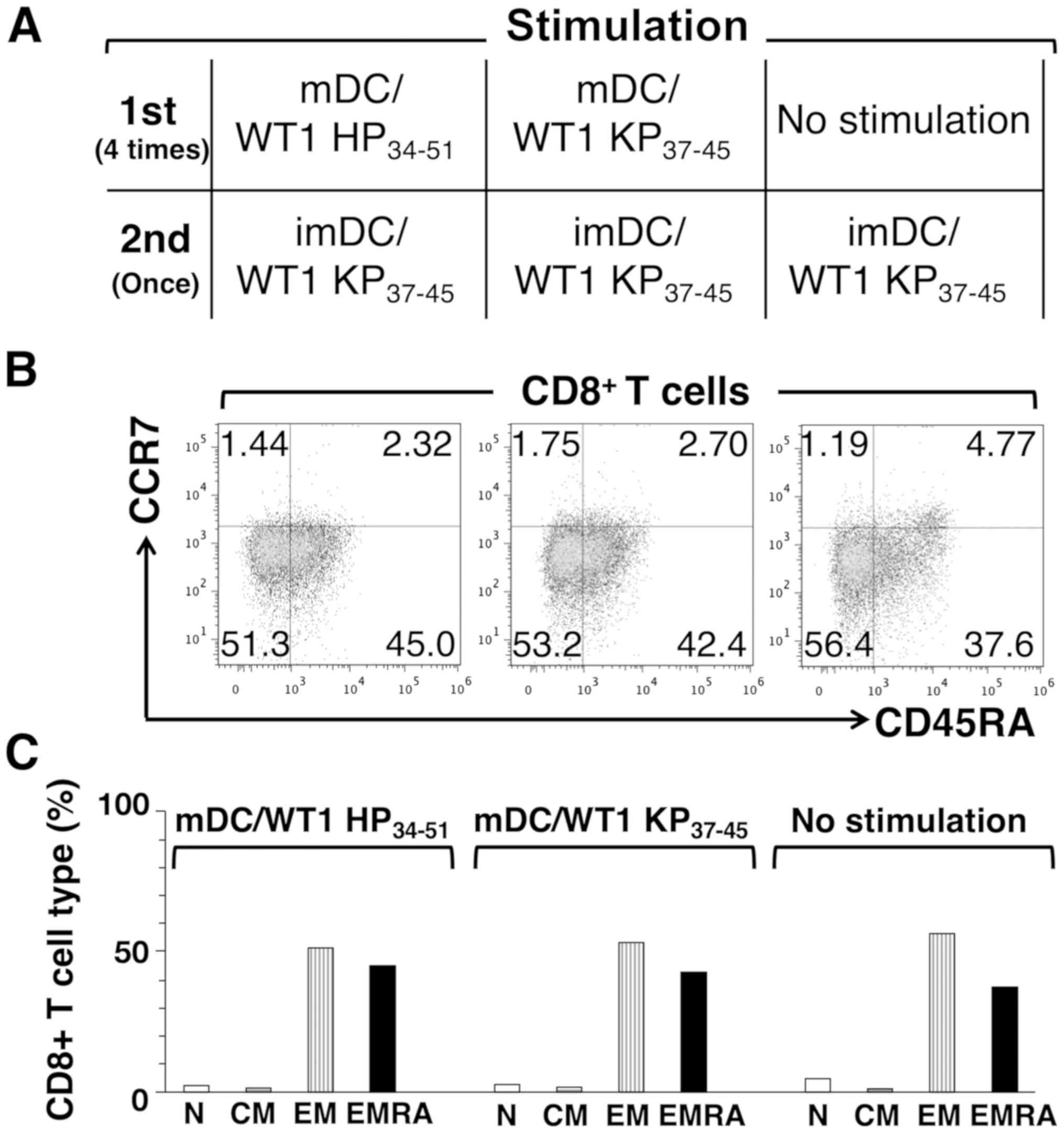 | Figure 5Memory cell subsets of
CD8+ T cells stimulated with mDC/WT1 HP34-51
or mDC/WT1 KP37-45. (A) Nonadherent cells were first
stimulated 4 times with mDC/WT1 HP34-51, mDC/WT1
KP37-45 or IL-2/IL-7 alone. The activated T cells were
restimulated with imDC/WT1 KP37-45. (B) Dot plots of the
memory cell subsets in CD8+ T cells derived from a
patient with pancreatic ductal adenocarcinoma activated by mDC/WT1
HP34-51 (left), mDC/WT1 KP37-45 (middle) or
no stimulation (right), followed by restimulation with imDC/WT1
KP37-45. (C) Percentage of four subgroups (N, CM, EM or
EMRA) of memory CD8+ T cells activated by mDC/WT1
HP34-51 (left), mDC/WT1 KP37-45 (middle) or
no stimulation (right), and restimulation with imDC/WT1
KP37-45. mDC, mature dendritic cell; imDC, immature
dendritic cell; WT1, Wilms' tumor 1; HP34-51, helper
peptide; KP37-45, killer peptide; mDC/WT1
HP34-51, mDCs pulsed with WT1 HP34-51;
mDC/WT1 KP37-45, mDCs pulsed with WT1
KP37-45; imDC/WT1 KP37-45, imDCs pulsed with
WT1 KP37-45; IL, interleukin; N, naïve; CM, central
memory; EM, effector memory; EMRA, terminally differentiated
effector memory; CCR7, chemokine receptor 7; CD45RA, human exon 4
splice variant of the tyrosine phosphatase. |
Induction of WT1-reactive CD4+
or CD8+ T cells derived from patients with different
types of cancer by mDC/WT1 HP34-51
The ability of mDC/WT1 HP34-51 to induce
WT1-reactive CD4+ or CD8+ T cells obtained
from patients with different types of cancer and HLA-A2 alleles
[02:01 (n=5), 02:06 (n=7), 02:07 (n=2)] was then subsequently
assessed. For 10 (patients 1, 3, 5, 6, 7, 10, 11, 12, 13 and 14) of
the 14 patients with cancer (71.4%), >1% of the total
CD4+ T cell population expressed IFN-γ after four rounds
of stimulation with mDC/WT1 HP34-51 followed by one
restimulation with imDCs (Table
II). Furthermore, for 10 (patients 1, 2, 4, 6, 7, 8, 9, 10, 13
and 14) of the 14 patients examined (71.4%), >10% of the total
CD4+ T cell population expressed IFN-γ after four rounds
of stimulation with mDC/WT1 HP34-51 and restimulation
once with imDC/WT1 KP37-45, which was higher compared
with the percentage after restimulation once with imDCs alone
(Table II). In contrast to the
results for WT1-reactive CD4+ T cells, >1% of the
total CD8+ T cell population from 10 (patients 1, 3, 5,
6, 7, 8, 10, 11, 12 and 13) of the 14 patients (71.4%) examined
produced IFN-γ after four rounds of stimulation with mDC/WT1
HP34-51 followed by one stimulation with imDCs (Table III). For 6 (patients 2, 3, 4, 7,
9 and 14) of the 14 patients with cancer (42.9%), a >10%
increase in the IFN-γ-producing CD8+ T cell population
among total CD8+ T cells was detected after
restimulation once with imDC/WT1 KP37-45 compared with
restimulation once with imDCs alone (Table III). WT1-reactive CD8+
T cells from patient 14 with the HLA-A*02:07 allele exhibited
greater IFN-γ production than the cells from the other patient with
the HLA-A*02:07 allele. In summary, for 5 of the 12 patients with
cancer (41.7%) with the HLA-A*02:01 or HLA-A*02:06 allele,
WT1-reactive CD8+ T cells stimulated with mDC/WT1
HP34-51 exhibited enhanced levels of WT1
KP37-45-induced IFN-γ production, with a >10%
increase following one restimulation with imDC/WT1
KP37-45 in this experimental setting. These results
suggested that mDC/WT1 HP34-51 generated from patients
with different types of cancer may induce WT1-specific
CD8+ T cells at least in part in an HLA-A*02:01-,
HLA-A*02:06- or HLA-A*02:07-restricted manner.
 | Table IIPercentage of IFN-γ-producing
CD4+ T cells among total CD4+ T cells by
stimulation with mDC/WT1 HP34-51. |
Table II
Percentage of IFN-γ-producing
CD4+ T cells among total CD4+ T cells by
stimulation with mDC/WT1 HP34-51.
| Case | HLA-A | Restimulation
|
|---|
| imDC/WT1
KP37-45 | imDCs |
|---|
| 1 | 02:01 | 4.97a | 3.78 |
| 2 | 02:01 | 1.01a | 0.64 |
| 3 | 02:01 | 6.93 | 6.99 |
| 4 | 02:01 | 0.68a | 0.58 |
| 5 | 02:01 | 1.03 | 1.11 |
| 6 | 02:06 | 3.69a | 2.34 |
| 7 | 02:06 | 10.26a | 7.96 |
| 8 | 02:06 | 0.69a | 0.58 |
| 9 | 02:06 | 0.85a | 0.23 |
| 10 | 02:06 | 3.31a | 2.81 |
| 11 | 02:06 | 2.5 | 2.43 |
| 12 | 02:06 | 3.17 | 3.72 |
| 13 | 02:07 | 1.79a | 1.46 |
| 14 | 02:07 | 3.23a | 1.48 |
 | Table IIIPercentage of IFN-γ-producing
CD8+ T cells among total CD8+ T cells by
stimulation with mDC/WT1 HP34-51. |
Table III
Percentage of IFN-γ-producing
CD8+ T cells among total CD8+ T cells by
stimulation with mDC/WT1 HP34-51.
| Case | HLA-A | Restimulation
|
|---|
| imDC/WT1
KP37-45 | imDCs |
|---|
| 1 | 02:01 | 3.11 | 3.87 |
| 2 | 02:01 | 1.84a | 0.7 |
| 3 | 02:01 | 32.96a | 11.07 |
| 4 | 02:01 | 7.96a | 0.05 |
| 5 | 02:01 | 0.92 | 1.11 |
| 6 | 02:06 | 10.18 | 12.39 |
| 7 | 02:06 | 18.16a | 12.61 |
| 8 | 02:06 | 0.72 | 1.22 |
| 9 | 02:06 | 0.62a | 0.46 |
| 10 | 02:06 | 4.39 | 4.15 |
| 11 | 02:06 | 2.31 | 2.78 |
| 12 | 02:06 | 5.86 | 7.8 |
| 13 | 02:07 | 3.53 | 3.57 |
| 14 | 02:07 | 1.0a | 0.8 |
Discussion
The present study indicated that mDC/WT1
HP34-51 exhibited the ability to induce not only
WT1-reactive EM CD4+ Th1 cells but also CD8+
CTLs producing IFN-γ in an HLA-A*02:01- or HLA-A*02:06-restricted
manner in an in vitro human model. The combination of
mDC/WT1 HP34-51 and imDC/WT1 KP37-45 induced
higher levels of IFN-γ-producing CD8+ T cells than
stimulation with mDC/WT1 KP37-45 and imDC/WT1
KP37-45.
The impact of mDC/WT1 HP34-51 on the
activation of WT1-specific T cells was assessed by monitoring IFN-γ
production following restimulation with imDC/WT1 HP34-51
or imDC/WT1 KP37-45. As restimulation with mDCs alone
induced a degree of nonspecific IFN-γ production in the present
study, WT1-loaded or unloaded imDCs were used as restimulators.
CD4+ and CD8+ T cells were stimulated with
mDC/WT1 HP34-51 together at the same time, resulting in
IFN-γ production after restimulation with imDC/WT1
HP34-51 or imDC/WT1 KP37-45, but not imDCs
alone. Our previous phase I study with mDCs pulsed with a mixture
of three WT1 peptides, including WT1 HP332-347 and WT1
KP, observed that simultaneous induction of WT1-specific
CD4+ and CD8+ T cells was associated with
improved long-term survival (4).
The novel peptide WT1 HP34-51 was subsequently developed
to enhance WT1 peptide immunogenicity, and early clinical trials of
a cocktail WT1 peptide vaccine containing the novel peptide WT1
HP34-51 and WT1 KP have been conducted in patients with
pediatric brain cancer or myelodysplastic syndromes (20,21).
The results demonstrated WT1-specific CTL induction in clinical
responders, suggesting that WT1 HP34-51 has effective
activity. Moreover, vaccination of a mouse model with both WT1
HP34-51 and WT1 KP also induced strong infiltration of
WT1-specific CD4+ Th1 cells and CD8+ CTLs
into tumor microenvironments (17).
In general, simultaneous activation of WT1-specific
CD4+ Th1 cells and CD8+ CTLs may be essential
when treating patients with advanced-stage cancer. Notably, the
novel WT1 HP34-51 amino acid sequence includes
WT137-45, which has binding affinity for HLA-A*02:01
molecules (18). Therefore, the
novel peptide WT1 HP34-51 may stimulate not only
WT1-specific CD4+ T cells but also CD8+ T
cells in a manner, at least in part, restricted by HLA-A*02:01
molecules. To evaluate this hypothesis, a patient with PDA
possessing HLA-A*02:01/24:02 alleles was initially selected for
further analysis. Notably, mDC/WT1 HP34-51 activated not
only CD4+ T cells but also CD8+ T cells to
produce IFN-γ after restimulation with imDC/WT1 KP37-45,
which according to the NetMHC 4.0 server-prediction program has a
high affinity for HLA-A*02:01 but not for HLA-A*24:02. Thus,
WT1-specific CD8+ T cells from patient 4 induced by
mDC/WT1 HP34-51 may be HLA-A*02:01-restricted.
Stimulation of T cells by 4 incubations with
mDC/WT1 HP34-51, followed by one restimulation with
imDC/WT1 HP34-51 or imDC/WT1 KP37-45 resulted
in IFN-γ production by CD8+ T cells. When imDCs are
pulsed with WT1 HP34-51, WT1 HP34-51 may be
taken up by imDCs, which can be activated by OK-432. Both imDCs and
mDCs may be able to process WT1 HP34-51 and load WT1
KP37-45 onto HLA-A*02:01 molecules in the endoplasmic
reticulum, resulting in their surface expression of WT1
KP37-45/HLA-A*02:01 complexes for presentation to
CD8+ T cells. Endogenously processed WT1 in DCs has
relatively good access to MHC class I and II molecules, resulting
in the efficient induction of antitumor immunity (22). Thus, mDC/WT1 HP34-51,
imDC/WT1 HP34-51 or imDC/WT1 KP37-45 may also
stimulate CD8+ T cells through WT1
KP37-45/HLA-A*02:01 complexes, resulting in the
production of IFN-γ.
Indeed, a >10% increase in IFN-γ-producing cells
among total CD8+ T cells was detected in 3 of 5 patients
with the HLA-A*02:01 allele and 2 of 7 patients with the
HLA-A*02:06 allele when their NAD cells were stimulated 4 times
with mDC/WT1 HP34-51 followed by restimulation with
imDC/WT1 KP37-45 compared with restimulation with imDCs
alone. Therefore, WT1 HP34-51 may have the ability to
induce WT1-specific CD8+ CTLs in patients with cancer
carrying the HLA-A*02:01 or HLA-A*02:06 allele. Nonetheless, 1 of 2
cancer patients with the HLA-A*02:07 allele exhibited IFN-γ
production by CD8+ T cells. According to the NetMHC 4.0
server-prediction program, WT1 KP37-45 within WT1
HP34-51 has a relatively high affinity for HLA-A*02:07.
However, as HLA-A*02:07+ patients are not a major
population according to the national marrow donor program (23), only two patients with HLA-A*02:07
were enrolled in the present study. Accordingly, further studies
are required to assess whether mDC/WT1 HP34-51
consistently induces HLA-A*02:07-restricted WT1-specific
CD8+ T cells in patients with cancer.
Among the total CD8+ T cell populations
from patients with cancer, WT1-specific IFN-γ-producing
CD8+ T cells were more frequently induced by four rounds
of stimulation with mDC/WT1 HP34-51 followed by one
stimulation with imDC/WT1 KP37-45 compared with four
rounds of stimulation with mDC/WT1 KP37-45 followed by
one stimulation with imDC/WT1 KP37-45. Previous reports
indicate that CD4+ and CD8+ T cell
stimulation occurs when peptide vaccination with combined WT1 HP
and WT1 KP is performed; however, vaccination with WT1 HP alone
does not strongly activate T cells (4,17).
It has also been reported that activation of CD4+ T
cells is essential for CD8+ CTL induction, potentiating
CD8+ CTL proliferation and maintaining effector
functions in the tumor microenvironment (5,24).
Therefore, effective cancer vaccines must include a mechanism to
activate CD4+ T cells. Simultaneous activation of
CD4+ and CD8+ T cells by DCs pulsed with both
WT1 HP34-51 and WT1 KP37-45 may improve the
efficient induction of WT1-specific CD8+ T cells that
produce IFN-γ. In fact, for 10 of the 14 patients in the present
study, a >10% increase in the IFN-γ-producing CD4+ T
cell population among total CD4+ T cells was induced by
four rounds of stimulation with mDC/WT1 HP34-51 followed
by one stimulation with imDC/WT1 KP37-45. According to
the NetMHCII-2.3 server-prediction program, WT1 KP37-45,
which is a part of WT1 HP34-51, has a relatively high
affinity for MHC class II. Thus, WT1 KP37-45 may also be
present in MHC class II with imDC/WT1 KP37-45, resulting
in stimulation of CD4+ T cells producing IFN-γ. As one
of the major subtypes of HLA-A molecule is HLA-A*24:02 (23), mDCs may be pulsed with a cocktail
of WT1 HP34-51 and WT1 KP235-243 for
HLA-A*24:02 (4) to maximize the
induction and maintenance of CD4+ Th1 cells and
CD8+ CTLs, at least in part, in an HLA-A*02:01-,
HLA-A*02:06- or HLA-A*24:02-restricted manner for application in a
wider cohort of patients with cancer.
WT1-reactive CD4+ T cells from patient 4
induced by mDC/WT1 HP34-51 or mDC/WT1 KP37-45
stimulation were almost all EM T cells, whereas those maintained
with low doses of IL-2 and IL-7 were not. In contrast, cancer
patient CD4+ T cells in the no stimulation group were
almost all naïve. Naïve T cells differentiated into EM T cells via
the effector T cell route (25) by
undergoing several rounds of stimulation with mDC/WT1
HP34-51 or mDC/WT1 KP37-45. It has been
reported that peptide-specific memory T cell frequencies are
increased in vaccine responders with a positive delayed-type
hypersensitivity (DTH) test (4,26-28).
Our previous study also demonstrated that in super-responders with
WT1-specific DTH induced by vaccination with mDCs pulsed with WT1
HP and WT1 KP, continuous elicitation of WT1-specific long-lived EM
and CM T cells occurred and could be boosted; these long-lived
WT1-specific EM T cells patrol and recognize tumor cells and are
therefore associated with a clinical benefit (4). Although EM CD4+ T cells
were induced by mDC/WT1 HP34-51 stimulation, compared
with EM CD4+ T cells, the EM CD8+ T cell frequency was
not strongly increased. As some WT1 overexpression occurs in
various types of solid tumor, WT1-specific CD8+ T cells
are spontaneously induced (11-13),
suggesting that some memory CD8+ T cells may be detected
without any stimulation in vitro. As memory CD4+
T cells particularly support the priming and maintaining of
antigen-specific CD8+ T cells (29,30),
the increase in memory CD4+ T cell frequency induced by
mDC/WT1 HP34-51 stimulation may support an increase in
WT1-reactive CD8+ T cell frequency. In summary, T cells
stimulated with mDC/WT1 HP34-51 may have the capacity to
elicit WT1-specific EM CD4+ T cells and CD8+
CTLs, which was at least in part restricted by HLA-A*02:01 or
HLA-A*02:06, both of which produce IFN-γ. However, the memory cell
subsets were only analyzed in a single case, a patient with PDA, as
a sufficient number of cells was not available. The induction and
maintenance of the durable memory type of CD4+ Th1 cells
by mDC/WT1 HP34-51 may produce promising clinical
activity to prolong overall survival in advanced-stage cancer
patients.
As a sufficient number of PBMCs and autologous
tumor cells were not available, the killing activity of WT1-ractive
T cells induced by mDC/WT1 HP34-51 could not be assessed
in this preclinical study. A phase I clinical trial involving
patients with advanced pancreatic cancer who express HLA-A*02:01,
HLA-A*02:06 or HLA-A*24:02 is being performed using mDCs pulsed
with a novel peptide cock-tail containing WT1 HP34-51
and HLA-A*24:02-restricted WT1 KP235-243 combined with
standard chemotherapy; the results of this clinical trial will be
available soon. Additionally, future studies should optimize cancer
vaccines incorporating mDCs loaded with an immunogenic novel WT1
peptide cocktail in combination with an immune checkpoint-blocking
antibody.
Supplementary Data
Acknowledgments
Not applicable.
Funding
This work was supported by Grants-in-Aid for
Scientific Research from the Ministry of Education, Culture,
Sports, Science and Technology of Japan (grant no. 19K08383) and
the BioLegend Research Grant (2018; no grant no.).
Availability of data and materials
All data generated or analyzed during this study
are included in this published article.
Authors' contributions
SKa and TB performed the experiments. SKa, TB and
SKo analyzed the data. MS and JT collected patient samples, and
analyzed and interpreted data. SKa, SS, TO, HS and SKo conceived
and designed the experimental study. SKa and SKo wrote the
manuscript. All authors discussed, read and approved the final
manuscript.
Ethics approval and consent to
participate
The present study was reviewed and approved by the
Ethics Committee of the Jikei Institutional Review Board, Jikei
University School of Medicine and the Clinical Study Committee of
Jikei University Kashiwa Hospital [approval no. 29-093 (8679)].
Patient consent for publication
Not applicable.
Competing interests
HS is an inventor on the patents for the WT1
peptides used in the present study (PCT/JP02/02794 and
PCT/2010/057149), which are held by the International Institute of
Cancer Immunotherapy.
References
|
1
|
Steinman RM and Banchereau J: Taking
dendritic cells into medicine. Nature. 449:419–426. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Baldin AV, Savvateeva LV, Bazhin AV and
Zamyatnin AA Jr: Dendritic cells in anticancer vaccination:
Rationale for ex vivo loading or in vivo targeting. Cancers
(Basel). 12:5902020. View Article : Google Scholar
|
|
3
|
Rosenberg SA, Yang JC and Restifo NP:
Cancer immunotherapy: Moving beyond current vaccines. Nat Med.
10:909–915. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Koido S, Homma S, Okamoto M, Takakura K,
Mori M, Yoshizaki S, Tsukinaga S, Odahara S, Koyama S, Imazu H, et
al: Treatment with chemotherapy and dendritic cells pulsed with
multiple Wilms' tumor 1 (WT1)-specific MHC class I/II-restricted
epitopes for pancreatic cancer. Clin Cancer Res. 20:4228–4239.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Melseen M and Slingluff CL Jr: Vaccines
targeting helper T cells for cancer immunotherapy. Curr Opin
Immunol. 47:85–92. 2017. View Article : Google Scholar
|
|
6
|
Taniuchi I: CD4 helper and CD8 cytotoxic T
cell differentiation. Annu Rev Immunol. 36:579–601. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Fujiki F, Oka Y, Kawakatsu M, Tsuboi A,
Nakajima H, Elisseeva OA, Harada Y, Li Z, Tatsumi N, Kamino E, et
al: A WT1 protein-derived, naturally processed 16-mer peptide,
WT1(332), is a promiscuous helper peptide for induction of
WT1-specificTh1-type CD4(+) T cells. Microbiol Immunol. 52:591–600.
2008. View Article : Google Scholar
|
|
8
|
Koski GK, Koldovsky U, Xu S, Mick R,
Sharma A, Fitzpatrick E, Weinstein S, Nisenbaum H, Levine BL, Fox
K, et al: A novel dendritic cell-based immunization approach for
the induction of durable Th1-polarized anti-HER-2/neu responses in
women with early breast cancer. J Immunother. 35:54–65. 2012.
View Article : Google Scholar
|
|
9
|
Sharma A, Koldovsky U, Xu S, Mick R, Roses
R, Fitzpatrick E, Weinstein S, Nisenbaum H, Levine BL, Fox K, et
al: HER-2 pulsed dendritic cell vaccine can eliminate HER-2
expression and impact ductal carcinoma in situ. Cancer.
118:4354–4362. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Aarntzen EH, De Vries IJ, Lesterhuis WJ,
Schuurhuis D, Jacobs JF, Bol K, Schreibelt G, Mus R, De Wilt JH,
Haanen JB, et al: Targeting CD4(+) T-helper cells improves the
induction of antitumor responses in dendritic cell-based
vaccination. Cancer Res. 73:19–29. 2013. View Article : Google Scholar
|
|
11
|
Chever MA, Allison JP, Ferris AS, Finn OJ,
Hastings BM, Hecht TT, Mellman I, Prindiville SA, Viner JL, Weiner
LM, et al: The prioritization of cancer antigens: A national cancer
institute pilot project for the acceleration of translational
research. Clin Cancer Res. 15:5323–5337. 2009. View Article : Google Scholar
|
|
12
|
Huff V: Wilms' tumours: About tumour
suppressor genes, an oncogene and a chameleon gene. Nat Rev Cancer.
11:111–121. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Oka Y, Tsuboi A, Taguchi T, Osaki T, Kyo
T, Nakajima H, Elisseeva OA, Oji Y, Kawakami M, Ikegame K, et al:
Induction of WT1 (Wilms' tumor gene)-specific cytotoxic T
lymphocytes by WT1 peptide vaccinean the resultant cancer
regression. Proc Natl Acad Sci USA. 101:13885–13890. 2004.
View Article : Google Scholar
|
|
14
|
Takahara A, Koido S, Ito M, Nagasaki E,
Sagawa Y, Iwamoto T, Komita H, Ochi T, Fujiwara H, Yasukawa M, et
al: Gemcitabine enhances Wilms' tumor gene WT1 expression and
sensitizes human pancreatic cancer cells with WT1-specific
T-cell-mediated antitumor immune response. Cancer Immunol
Immunother. 60:1289–1297. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Krug LM, Dao T, Brown AB, Maslak P, Travis
W, Bekele S, Korontsvit T, Zakhaleva V, Wolchok J, Yuan J, et al:
WT1 peptide vaccinations induce CD4 and CD8 T cell immune responses
in patients with mesothelioma and non-small cell lung cancer.
Cancer Immunol Immunother. 59:1467–1479. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Keilholz U, Menssen HD, Gaiger A, Menke A,
Oji Y, Oka Y, Scheibenbogen C, Stauss H, Thiel E and Sugiyama H:
Wilms' tumour gene 1 (WT1) in human neoplasia. Leukemia.
19:1318–1323. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Nakata J, Nakajima H, Hayashibara H,
Imafuku K, Morimoto S, Fujiki F, Motooka D, Okuzaki D, Hasegawa K,
Hosen N, et al: Extremely strong infiltration of WT1-specific CTLs
into mouse tumor by the combination vaccine with WT1-specific CTL
and helper peptides. Oncotarget. 9:36029–36038. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Schmied S, Gostick E, Price DA, Abken H,
Assenmacher M and Richter A: Analysis of the functional
WT1-specific T cell repertoire in healthy donors reveals a
discrepancy between CD4(+) and CD8(+) memory formation. Immunology.
145:558–569. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hori S, Heike Y, Takei M, Maruyama M,
Inoue Y, Lee JJ, Kim HJ, Harada Y, Kawai H, Shimosaka A, et al:
Freeze-thawing procedures have no influence on the phenotypic and
functional development of dendritic cells generated from peripheral
blood CD14+ monocytes. J Immunother. 27:27–35. 2004. View Article : Google Scholar
|
|
20
|
Goto M, Nakamura M, Suginobe N, Takasu H,
Takanashi Y, Ban H and Li C: DSP-7888, a novel cocktail design of
WT1 peptide vaccine, and its combinational immunotherapy with
immune checkpoint-blocking antibody against PD-1. Blood. 128:4715.
2016. View Article : Google Scholar
|
|
21
|
Miyakoshi S, Usuki K, Matsumura I, Ueda Y,
Iwasaki H, Miyamoto T, Origuchi M, Tagashira S, Naoi I, Naoe T, et
al: Preliminary results from a phase 1/2 study of DSP-7888, a novel
WT1 peptide-based vaccine in patients with myelodysplastic syndrome
(MDS). Blood. 128:4335. 2016. View Article : Google Scholar
|
|
22
|
Rosalia RA, Quakkelaar ED, Redeker A, Khan
S, Camps M, Drijfhout JW, Silva AL, Jiskoot W, van Hall T, van
Veelen PA, et al: Dendritic cells process synthetic long peptides
better than whole protein, improving antigen presentation and
T-cell activation. Eur J Immunol. 43:2554–2565. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Maiers M, Graget L and Klitz W:
High-resolution HLA alleles and haplotypes in the United States
population. Hum Immunol. 68:779–788. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Mintern JD, Davey GM, Belz GT, Carbone FR
and Heath WR: Cutting edge: Precursor frequency affects the helper
dependence of cytotoxic T cells. J Immunol. 168:977–980. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lanzavecchia A and Sallusto F: Dynamics of
T lymphocyte responses: Intermediates, effectors, and memory cells.
Science. 290:92–97. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wedén S, Klemp M, Gladhaug IP, Møller M,
Eriksen JA, Gaudernack G and Buanes T: Long-term follow-up of
patients with resected pancreatic cancer following vaccination
against mutant K-ras. Int J Cancer. 128:1120–1128. 2011. View Article : Google Scholar
|
|
27
|
Nishida S, Koido S, Takeda Y, Homma S,
Komita H, Takahara A, Morita S, Ito T, Morimoto S, Hara K, et al:
Wilms tumor gene (WT1) peptide-based cancer vaccine combined with
gemcitabine for patients with advanced pancreatic cancer. J
Immunother. 37:105–114. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lesterhuis WJ, de Vries IJ, Schuurhuis DH,
Boullart AC, Jacobs JF, de Boer AJ, Scharenborg NM, Brouwer HM, van
de Rakt MW, Figdor CG, et al: Vaccination of colorectal cancer
patients with CEA-loaded dendritic cells: Antigen-specific T cell
responses in DTH skin tests. Ann Oncol. 17:974–980. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
MacLeod MK, Clambey ET, Kappler JW and
Marrack P: CD4 memory T cells: What are they and what can they do?
Semin Immunol. 21:53–61. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Krawczyk CM, Shen H and Pearce EJ: Memory
CD4 T cells enhance primary CD8 T-cell responses. Infect Immun.
75:3556–3560. 2007. View Article : Google Scholar : PubMed/NCBI
|