Introduction
Gastric cancer is one of the most serious diseases
threatening human health. It is the fourth most common cancer and
the second leading cause of cancer-related death in the world
(1,2). According to the Chinese Ministry of
Health in 2006, over 400,000 new cases of gastric cancer are
diagnosed and approximately 300,000 deaths in China are reported
each year.
Interaction between the host immune response and
gastric cancer cells results in the occurrence and development of
gastric cancer. This involves multilateral factors, such as host
effector cells, stromal cells and tumor cells (3–5).
Shortage of co-stimulation molecules on the surface of tumor cells
is the main reason for immune evasion among all of the factors.
There are two types of co-stimulation molecules discovered at
present, which include positive and negative co-stimulation
molecules. The former influences the activation and proliferation
of T lymphocytes and secretion of cytokines by combining with
lymphocyte receptors, while the latter induces T cells anergy
through two ways as follows. One is to induce inhibitory factors to
express on the effector cells and the other is to elicit them to
express on tumor cells (6).
Immunoglobulin-like transcripts (ILTs), also called
leukocyte immunoglobulin-like receptors (LIRs/LILRs) or CD85
(7), are a group of membrane
receptors coded by more than 10 genes located on the 19q13.4
chromosome (8,9). Nakajima et al (10) identified a new class of
immunoglobulin superfamily, whose members were named ILT1 and ILT2,
and other ILT family members were later found and identified
(11–14). Characterized by different
transmembrane and cytoplasmic domains, the members of the ILT
family are divided into activating receptors, inhibitory receptors
and soluble receptors (15). ILT2
and ILT3 as the two primary types of inhibitory receptors, which
regulate the functions of cells involved in the immune system
(16). ILT2 is detected on the
surface of a proportion of NK cells (~75%) and T lymphocytes
(4–20%), on all B lymphocytes, monocytes, DCs and macrophages
(17). However, ILT3 is expressed
only on monocytes, DCs and endothelial cells, but not on T and B
lymphocytes (18,19). They play an extensive role by
transmitting inhibitory signals, for instance, they restrain the
killer activity of killer cells, and regulate signal transmission
between DCs and T lymphocytes (20–23).
ILT3 also induces the generation of CD8+ T suppressor
cells and renders antigen-presenting cells tolerogenic (22,24).
In addition, it has been reported that ILT2 and ILT3 integrate with
MHC-I molecules, especially HLA-G, and deliver negative signals by
immunoreceptor tyrosine-based inhibitory motifs (ITIM) to repress
the activation of CD4+ and CD8+ T lymphocytes
and down-regulate recognition of antigens by CD8+ T
lymphocytes, which leads to the immune escape of tumor cells
(25–27).
The mechanisms through which tumor cells escape from
host immunity are not yet clear. However, one important reasons is
that ILTs induce immunological tolerance. Lefebvre et al
(28) discovered that partial
tumor infiltrating lymphocytes in human breast cancer express ILT2.
Yet, the role of ILT receptors in tumors is not clear and many
issues require further study. Our research aimed to investigate the
expression and role of ILT2 and ILT3 in gastric cancer via in
vivo and in vitro experiments.
Materials and methods
Cell lines and culture
Six human gastric cancer cell lines were used, which
included the well-differentiated gastric cancer cell line MKNI,
moderately differentiated cell lines SGC7901 and AGS, the poorly
differentiated cell line MGC803 and undifferentiated cell lines
HGC27 and BCG823, respectively. Human NK92MI cells were used as
effector cells. All gastric cancer cell lines and human NK92MI were
from Shandong Province Key Laboratory for Tumor Target Molecules in
Jinan Central Hospital which is affiliated to Shandong University.
All cell lines were maintained in RPMI-1640 or F-12 medium (Gibco
Co., USA) supplemented with 10% heat-inactivated fetal calf serum
(FCS), 100 IU/ml penicillin and 100 IU/ml streptomycin at 37°C in a
humidified atmosphere with 5% CO2.
Patients and information
Tumor specimens were obtained from 50 primary
gastric cancer patients (37 men and 13 women; aged 38–76 years;
mean age, 53.3 years) without having received any preoperative
therapy at Qilu Hospital of Shandong University, China, between
February, 2008 and August, 2009. The main clinicopathological
variables of the patients are recorded in Tables I and II. According to pathological
Tumor-Node-Metastasis International System and Lauren
classification, 31 cases were characterized as gastric
adenocarcinomas, 10 were mucinous adenocarcinomas and 9 were signet
ring cell cancers. Considering pathological grading, 20 were staged
as well differentiated and 30 as poorly differentiated. Two-thirds
of the patients with lymph node metastasis were validated by
conventional postoperative pathological examination. There were 19
cases with tumor size >5 cm, and 31 cases with tumor size <5
cm. We obtained informed consent from each patient, and the study
protocol was approved by the Ethics Committee of Qilu Hospital of
Shandong University and was performed in accordance with the
ethical standards of the 2000 Declaration of Helsinki as well as
the Declaration of Istanbul 2008.
 | Table IRelationship between ILT2 expression
in the tumor cells and clinicopathological variables. |
Table I
Relationship between ILT2 expression
in the tumor cells and clinicopathological variables.
| | ILT2
expression | | |
|---|
| |
| | |
|---|
| Clinicopathological
variables | No. of
patients | Negative | Positive | χ2 | P-value |
|---|
| Gender | | | | 0.002 | 0.968 |
| Male | 37 | 14 | 23 | | |
| Female | 13 | 5 | 8 | | |
| Age (years) | | | | 0.085 | 0.771 |
| >60 | 25 | 9 | 16 | | |
| ≤60 | 25 | 10 | 15 | | |
| Tumor location | | | | 0.656 | 0.720 |
| Ridge 1/3 | 30 | 11 | 19 | | |
| Medi 1/3 | 13 | 6 | 7 | | |
| Bottom 1/3 | 7 | 2 | 5 | | |
|
Differentiation | | | | 4.089 | 0.043a |
|
Well/moderately | 20 | 11 | 9 | | |
| Poorly | 30 | 8 | 22 | | |
| Muscular
infiltration | | | | 0.195 | 0.659 |
| No | 14 | 6 | 8 | | |
| Yes | 36 | 13 | 23 | | |
| Metastasis | | | | 0.110 | 0.740 |
| Negative | 17 | 7 | 10 | | |
| Positive | 33 | 12 | 21 | | |
| Tumor size | | | | 6.417 | 0.011a |
| ≤5 cm | 31 | 16 | 15 | | |
| >5 cm | 19 | 3 | 16 | | |
 | Table IIRelationship between ILT3 expression
on tumor cells and clinicopathological variables. |
Table II
Relationship between ILT3 expression
on tumor cells and clinicopathological variables.
| | ILT3
expression | | |
|---|
| |
| | |
|---|
| Clinicopathological
variables | No. of
patients | Negative | Positive | χ2 | P-value |
|---|
| Gender | | | | 0.081 | 0.755 |
| Male | 37 | 30 | 7 | | |
| Female | 13 | 11 | 2 | | |
| Age (years) | | | | 0.136 | 0.712 |
| >60 | 25 | 20 | 5 | | |
| ≤60 | 25 | 21 | 4 | | |
| Tumor location | | | | 2.414 | 0.087 |
| Ridge 1/3 | 30 | 24 | 6 | | |
| Medi 1/3 | 13 | 11 | 2 | | |
| Bottom 1/3 | 7 | 6 | 1 | | |
|
Differentiation | | | | 0.203 | 0.652 |
|
Well/moderately | 20 | 17 | 3 | | |
| Poorly | 30 | 24 | 6 | | |
| Infiltration
muscular | | | | 0.182 | 0.670 |
| No | 14 | 12 | 2 | | |
| Yes | 36 | 29 | 7 | | |
| Metastasis | | | | 0.002 | 0.963 |
| Negative | 17 | 14 | 3 | | |
| Positive | 33 | 27 | 6 | | |
| Tumor size | | | | 0.101 | 0.750 |
| ≤5 cm | 31 | 25 | 6 | | |
| >5 cm | 19 | 16 | 3 | | |
Total-RNA isolation and RT-PCR
The gastric cancer cells (3–5×105) with
various degrees of differentiation were collected respectively, and
the total-RNA was extracted using the TRIzol RNA isolation kit
(Invitrogen, USA). Total-RNA (1 μg) was reverse transcribed using
High-Capacity cDNA reverse transcription kits (Applied Biosystems,
USA) according to the manufacturer’s instructions and then was
detected by SYBR-Green I (Invitrogen) real-time PCR. The ILT
primers were synthesized by Sangon Biological Engineering
Technology and Services Co., Ltd. (Shanghai, China). The primers
for ILT2, ILT3 and ACTB (internal control) were designed as
follows: ILT2 primer, forward, 5′-GGGGTTGTGATCGGCATCTT and reverse
sequence, 5′-CTGGCCTGGACTCGATGTC; ILT3 primer, forward,
5′-CATCCATGACAGAGGACTATGC and reverse sequence,
5′-GGGCTGAAAGGGTGGGTTTA; ACTB forward, 5′-TTGCCGACAGGATGCAGAA and
reverse sequence, 5′-GCCGATCCACACGGAGTACT. The lengths of the
amplified fragments were 189, 311 and 101 bp, respectively. The
FQ-PCR system contained SYBR-Green I 10 μl, PCR forward and reverse
primers (5 μM) 1 μl, respectively, cDNA 2 μl, ddH2O 6
μl. There were 2 μl DEPC in the negative control instead of cDNA.
The reaction was incubated for 40 cycles at 95°C for 10 sec, 60°C
for 10 sec and 72°C for 10 sec.
Flow cytometry
We collected and counted the gastric cancer cells in
the media which were in the logarithmic phase, and adjusted the
consistency of the cells to 1–2.5×105 cells/ml. The
cells were washed twice with phosphate-buffered saline (PBS, pH
7.4), and incubated with mouse anti-human ILT2 (R&D, USA; clone
292305) and ILT3 antibody (R&D; clone 293623) for 30 min. After
washing thoroughly with PBS, the cells were stained with PE-labeled
goat anti-mouse antibodies (R&D) for 30 min at 4°C. Cells were
washed twice with PBS and analyzed by FACSCalibur flow cytometry
with CellQuest software and associated software (BD Company, USA).
Meanwhile, non-specific staining was determined using control goat
immunoglobin.
Cytotoxicity assay
Two types of target cells (1×105/ml) in a
logarithmic growth phase, ILT2lowILT3low
human well differentiated gastric cancer MKNI cells and
ILT2highILT3high undifferentiated gastric
cancer HGC27 cells, were seeded in 96 well culture plates at 37°C,
in a 5% CO2 incubator for 4 h, respectively. Each well
contained 100 μl RPMI-1640 medium supplemented with 10% fetal
bovine serum (FBS). Whereafter, NK92MI cells were added in a volume
of 100 μl at effector and target cell ratios of 10:1, 5:1, 2.5:1
and 1.25:1. Blank controls, target cell controls (T) and effector
cell controls (E) were set synchronously. The cell mixtures were
then incubated at 37°C, in 5% CO2 for 24 h, after which
20 μl MTT (5 mg/ml) was added and cultured for a further 4 h. The
absorbance was determined at 570 nm by an ELISA reader. The
percentage of kill (% kill) was calculated using the following
equation: % kill = [1 - (ODE+T -
ODE)/ODT] × 100%.
Immunohistochemistry
Immunohistochemical staining was performed using the
biotin-streptavidin-peroxidase method with a Vectastain ABC kit
(Invitrogen). Resected tissue specimens were collected and fixed in
formalin, embedded in paraffin, and cut into 4-μm serial sections.
Sections were incubated with mouse anti-ILT2 and ILT3 antibodies
(diluted 1:300) in a humidty chamber at 4°C overnight. After
washing with PBS, the sections were incubated with biotinylated
goat anti-mouse antibodies for 30 min and then washed three times
with PBS, and then incubated with streptavidin-conjugated
peroxidase for 30 min. The sections were then visualized by
incubation with 3,3′-diaminobenzidine solution (0.3% hydrogen
peroxide and 0.05% 3,3′-diaminobenzidine) and stained with
hematoxylin. Negative controls were carried out by substituting a
normal mouse IgG for the primary antibody. Histological analysis
was performed by two investigators simultaneously by microscopy
without knowledge of the patient clinical records, and the number
of ILT2- and ILT3-stained tumor cells among 1,000 tumor cells in
each section was determined. Cell counts were performed at a
magnification ×400 in at least five fields in randomly selected
tumor areas.
Statistical analysis
Statistical analysis was performed using SPSS 10.0.
Comparison of the means between the two groups was different
analyzed using the t-test, and comparison of the means among the
groups was assayed by variance analysis and SNK test. The result of
the immunohistochemistry was analyzed using the χ2 test,
and the relationship between the expression of ILT2 and ILT3 and
clinical parameters was assessed by the Fisher’s test. Data were
represented as the means ± standard deviation. Values of P<0.05
were considered to denote statistical signification.
Results
Expression of ILT2 and ILT3 mRNA in
gastric cancer cells
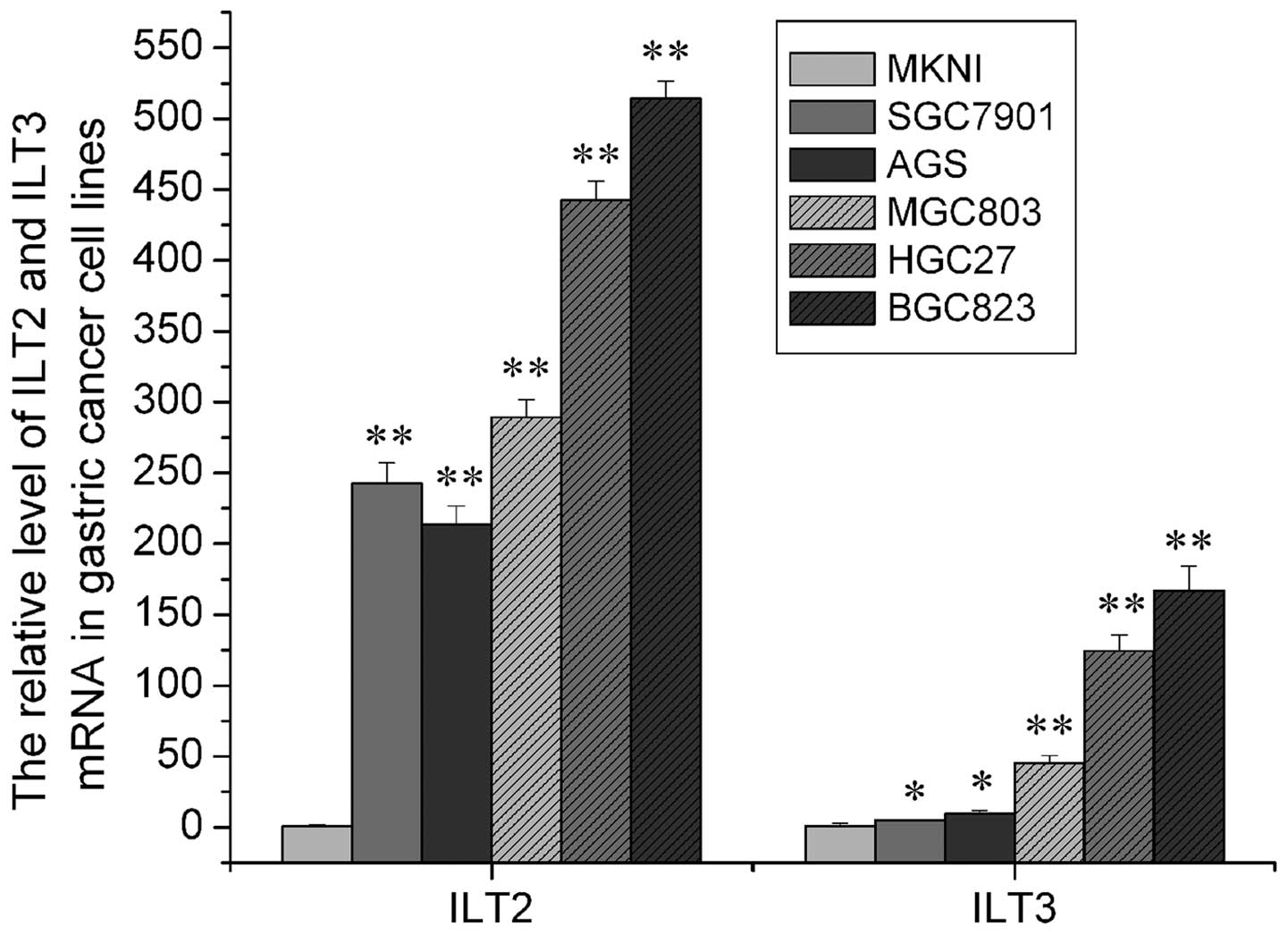
We detected ILT2 and ILT3 mRNA expression in the 6
gastric cancer cell lines using FQ-PCR SYBR-Green I. The relative
expression of ILT2 and ILT3 mRNA is shown in Fig. 1. Due to the absence of a negative
standard control, we used the expression of ILT2 and ILT3 mRNA in
the MKNI cell line as cardinal number. Relative expression of ILT2
and ILT3 mRNA in the other groups was derived from values compared
with the cardinal number. As shown in Fig. 1, it was demonstrated that ILT2 and
ILT3 mRNA expression increased along with increased
differentiation, and the difference among them was significant
(P<0.05).
Expression of ILT2 and ILT3 protein in
gastric cancer cells (Fig. 2)
The positive rate of ILT2 and ILT3 protein
expression in the wel- differentiated gastric cancer cell line MKN1
was low, which was significantly different from the expression
levels in the moderately or poorly differentiated gastric cancer
cell lines and the undifferentiated tumor cell lines as well
(P<0.05). Among the 6 gastric cancer cell lines, BCG823 hardly
expressed the ILT3 protein while it expressed a high level of the
ILT2 protein. The positive rates of ILT3 protein expression were
lower than those of the ILT2 protein in the five cell lines (MKN1,
SGC7901, AGC, MGC803 and HGC27), and expression of the ILT3 protein
did not vary obviously between the moderately or the poorly
differentiated and undifferentiated cell line (P>0.05). However,
the positive rate of ILT2 protein expression in poorly
differentiated and undifferentiated gastric cancer cells was much
higher than that in the moderately and well-differentiated cancer
cells (P<0.05).
Differential expression of ILT2 and ILT3
in gastric cancer cells affected NK92MI cytotoxicity
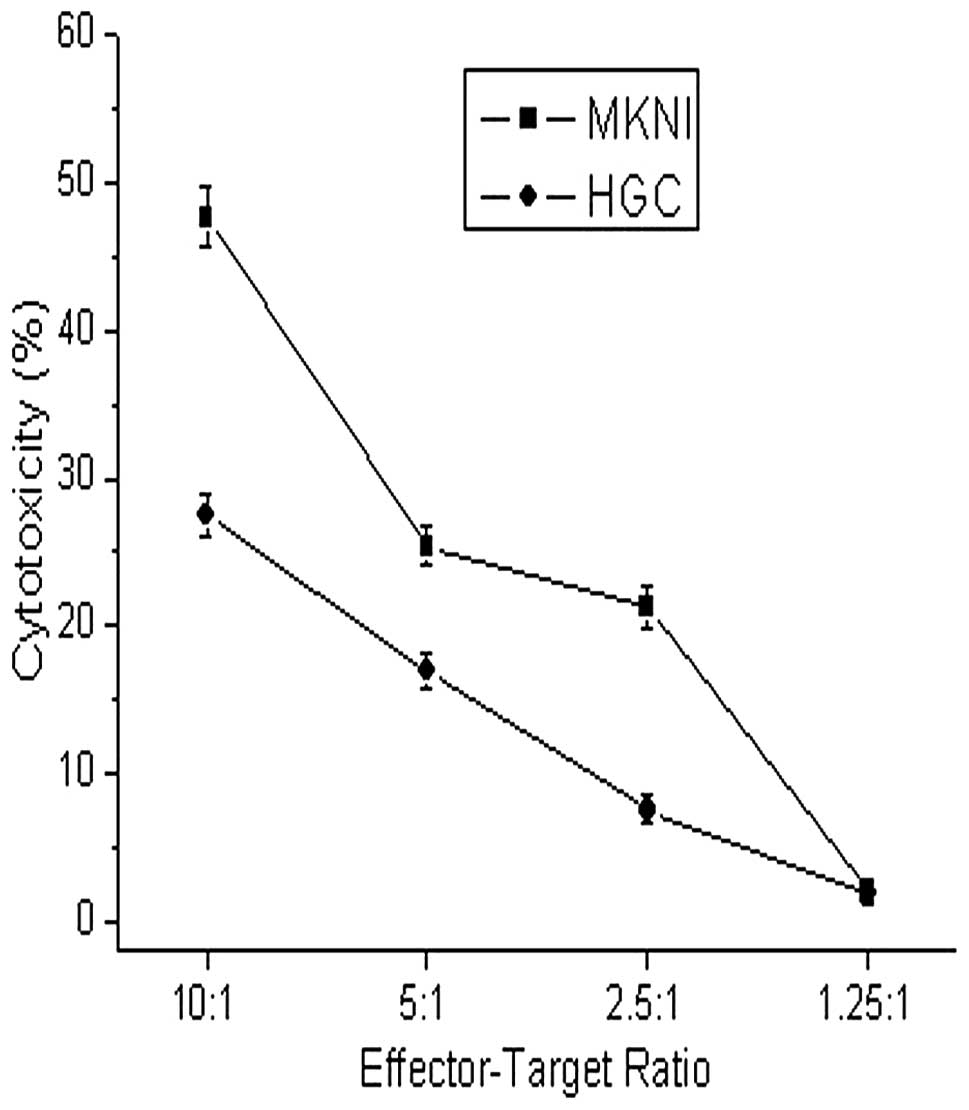
Here, we chose the
ILT2lowILT3low well-differentiated gastric
cancer cell line MKNI and the
ILT2highILT3high undifferentiated gastric
cancer cell line HGC27 as target cells, and the NK92MI as effector
cells. The cytotoxicity of NK92MI to MKNI and HGC27 was detected,
respectively, using different effector-target ratios. The
cytotoxicity of NK92MI to difference MKNI at effector-target ratios
from 10:1 to 2.5:1 was stronger than that to HGC27 after 24 h of
co-culture, and a significant difference was noted (P<0.05)
(Fig. 3). We conclude that the
different expression of ILT2 and ILT3 in gastric cancer cells
influenced the killer activity of NK92MI cells.
Relationship between the expression of
ILT2 and ILT3 in patients with gastric cancer and clinicopathologic
variables
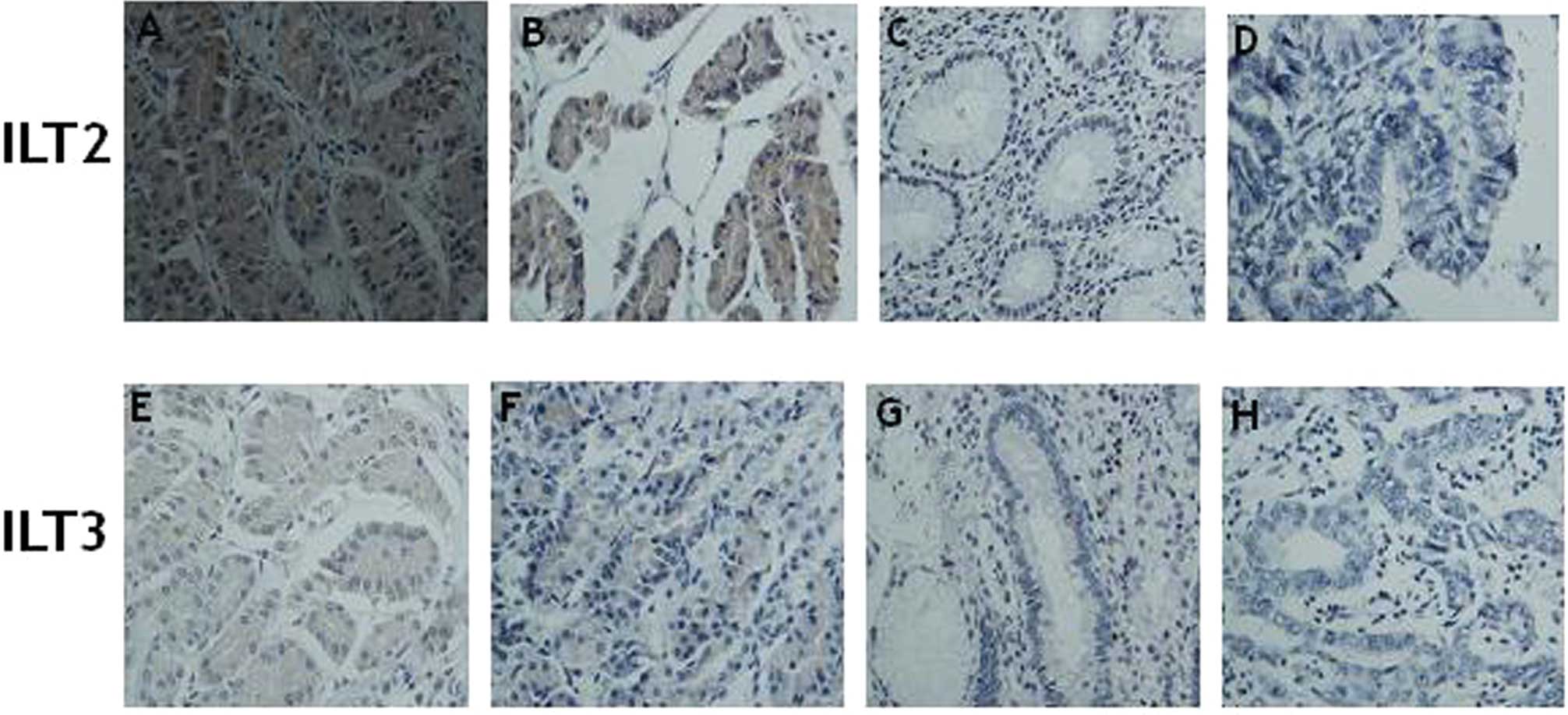
Expression of ILT2 and ILT3 in gastric cancer cells
from 50 patient pathological tissues with diverse degrees of
differentiation and pathological types were found to be dissimilar
by immunohistochemistry, and the positive rate of ILT2 and ILT3
expression was 62.0% (31/50) and 18.0% (9/50), respectively, which
indicates that there was a significant difference between ILT2 and
ILT3 in the gastric cancer cells (P<0.01). ILT2 and ILT3 protein
were expressed either in the cytoplasm or the cell membrane of the
gastric cancer cells, but no ILT2 and ILT3 expression was noted in
the normal stomach tissue (Fig.
4).
Further information concerning the pathological
characteristics and the clinical data of these patients were
analyzed (Tables I and II). It was demonstrated that there was
no significant different between ILT2 and ILT3 expression and
factors such as cancer origin, invasive depth and lymphatic
metastasis (P>0.05). Yet, a distinct relationship was noted
between ILT2 and the differentiation and size of the tumor. The
positive rate of ILT2 expression was 73.3% (22/30) and 45% (9/20),
respectively, in the poorly differentiated cancers including
adenocarcinoma, signet ring cell cancer and well differentiated
cancer including mastoid adenocarcinoma, well- and moderately
differentiated tubular adenocarcinoma (χ2=4.089,
P<0.05). The positive rate of ILT2 expression was 84.2% when the
tumor size was >5 cm, which was higher than that of 48.4% when
the tumor size was <5 cm (χ2=6.417, P=0.011).
Nevertheless, no significantly statistical relation was noted
between ILT3 expression and the differentiation degree and the size
of the tumors.
Discussion
ILT2 and ILT3 belong to a family of inhibitory
receptors, which are characterized by long cytoplasm tails that
contain three immunoreceptor tyrosine-based inhibitory motifs
(ITIMs) and either two or four homologous extracellular C-2 type
Ig-like domains (8,29,30).
It has been suggested that the interaction of ILT2 or ILT3 with
HLA-G inhibits the activation of T cells, natural killer cells and
myelomonocytic cells (9,31,32).
They also regulate CD8+ T cell activation by blocking
CD8 binding as well as by recruiting inhibitory molecules through
their ITIM motifs (33). Recent
studies have demonstrated that monocytes and dendritic cells (DCs)
exposed to T suppressor or regulatory cells acquire a tolerogenic
phenotype characterized by upregulation of ILT2 and ILT3 expression
(34–36). Antigen-presenting cells (APCs),
which express high levels of these receptors, can become
tolerogenic and induce anergy in CD4+ T helper cells
(32,37). Then the antigen-presenting function
of monocytes and DCs is modulated by the level of expression of
ILT2 and ILT3. Studies have also shown that ILT3 on the surface of
B lymphocytes is related to tumor-specific T lymphocyte tolerance
in B-cell chronic lymphatic leukemia, and regardless of whether
ILT3 was membrane (mILT3) or soluble (sILT3), they all had strong
immunosuppressive activity (38).
In addition, high expression of ILT2 and ILT3 was found to highly
induce Th cells and CTLs into Treg and Ts cells, respectively,
in vitro while ILT was expressed in DCs.
CD8+CD28− Ts cells upregulated the expression
of HLA-G in tumor cells, which result in immune escape (25,39).
Yet, no report exists to date concerning ILT2 and ILT3 expression
in human tumor tissues and to our knowledge this is the first
description of the expression of these molecules in primary gastric
cancer.
Our studies demonstrate that ILT2 and ILT3 mRNA and
protein were expressed to a different extent in the gastric cancer
cells with different degrees of differentiation. Moreover, there
was a significant relationship between the expression of ILT2 and
ILT3 and the pathological grade of the gastric cancer. We also
found that the cytotoxicity of NK cells against gastric cancer
cells with different expression of ILT2 and ILT3 was distinct in
vitro. It was previously reported that inhibitory receptors
influence the function and state of T lymphocytes, NK cells and
DCs, and regulate immunological tolerance (40,41).
Thus, we speculated that tumor cells express inhibitory receptors
via certain pathways and affect the killer activity of
immunocytes.
We found that ILT2 and ILT3 were expressed to some
extent in the gastric cancer pathological tissues, and their
expression in cancer tissues was characterized by a scattered
pattern and was located in both the plasma membrane and the
cytoplasm of the cancer cells. The intensity and positive rates of
ILT2 were higher than that of ILT3, and the expression of ILT2 was
obviously related to the differentiation and size of the tumors,
which demonstrated that expression of this inhibitory receptor by
the tumor cells restrained the immune response. Sun et al
(42) discovered that ILT4 was
expressed in non-small cell lung cancer cells in vitro as
well. By assessing clinical pathological tissues, they also found
that there was a significant relation between the expression of
ILT4 and surrounding tumor infiltrating lymphocytes (TILs), which
suggested that the tumor cells induced infiltrating lymphocyte
tolerance by contacting them indirectly after upregulating
inhibitory ILT4. In conclusion, our research indicates that ILT2
and ILT3 expression in gastric cancer cells and tissues may play an
important role in tumor development, and ILT2 and ILT3 may be
applied as targets for the diagnosis and therapy of gastric cancer.
However, further research is needed to understand the specific
mechanism.
Acknowledgements
This study was supported in part by grants from the
Shandong Province Science Foundation for Key Programs
(2008GG30002017 and 2010GSF10274) and the University Innovation
Program from Jinan, Shandong Province (201004050).
References
|
1
|
Y YonemuraY EndouT SasakiM HiranoA
MizumotoT MatsudaN TakaoM IchinoseM MiuraY LiSurgical treatment for
peritoneal carcinomatosis from gastric cancerEur J Surg
Oncol3611311138201010.1016/j.ejso.2010.09.00620933363
|
|
2
|
A JemalR SiegelE WardY HaoJ XuMJ
ThunCancer statistics, 2009CA Cancer J
Clin59225249200910.3322/caac.20006
|
|
3
|
H KawaidaK KonoA TakahashiH SugaiK MimuraN
MiyagawaH OmataA OoiH FujiiDistribution of
CD4+CD25(high) regulatory T-cells in tumor-draining
lymph nodes in patients with gastric cancerJ Surg
Res1241511572005
|
|
4
|
S GrossP WaldenImmunosuppressive
mechanisms in human tumors: Why we still cannot cure cancerImmunol
Lett116714200810.1016/j.imlet.2007.11.01218164076
|
|
5
|
K HirokawaM UtsuyamaT IshikawaY KikuchiM
KitagawaY FujiiH NariuchiH UetakeK SugiharaDecline of T
cell-related immune functions in cancer patients and an attempt to
restore them through infusion of activated autologous T cellsMech
Ageing Dev1308691200910.1016/j.mad.2008.05.00118555517
|
|
6
|
JK PettyK HeCL CorlessJT VettoAD
WeinbergSurvival in human colorectal cancer correlates with
expression of the T-cell costimulatory molecule OX-40 (CD134)Am J
Surg183512518200210.1016/S0002-9610(02)00831-012034383
|
|
7
|
N TedlaCW LeeL BorgesCL GeczyJP
ArmDifferential expression of leukocyte immunoglobulin-like
receptors on cord-blood-derived human mast cell progenitors and
mature mast cellsJ Leukoc
Biol83334343200810.1189/jlb.050731417998301
|
|
8
|
CA GleissnerTJ DenglerInduction of ILT
expression on nonprofessional antigen presenting cells: clinical
applicationsHum
Immunol70357359200910.1016/j.humimm.2009.01.02519405175
|
|
9
|
A Monsivais-UrendaP Nino-MorenoC
Abud-MendozaL BarandaE Layseca-EspinosaM Lopez-BotetR
Gonzalez-AmaroAnalysis of expression and function of the inhibitory
receptor ILT2 (CD85j/LILRB1/LIR-1) in peripheral blood mononuclear
cells from patients with systemic lupus erythematosus (SLE)J
Autoimmun2997105200710.1016/j.jaut.2007.05.00317601702
|
|
10
|
H NakajimaJ SamaridisL AngmanM
ColonnaCutting edge: human myeloid cells express an activating ILT
receptor (ILT1) that associates with Fc receptor gamma-chainJ
Immunol1625819999886363
|
|
11
|
M CellaC DohringJ SamaridisM DessingM
BrockhausA LanzavecchiaM ColonnaA novel inhibitory receptor (ILT3)
expressed on monocytes, macrophages, and dendritic cells involved
in antigen processingJ Exp
Med18517431751199710.1084/jem.185.10.1743
|
|
12
|
X XuP ZouL ChenG JinH ZhouIL-10 enhances
promoter activity of ILT4 gene and up-regulates its expression in
THP-1 cellsJ Huazhong Univ Sci Technolog Med
Sci30594598201010.1007/s11596-010-0548-821063840
|
|
13
|
H WendeA VolzA ZieglerExtensive gene
duplications and a large inversion characterize the human leukocyte
receptor
clusterImmunogenetics51703713200010.1007/s00251000018710941842
|
|
14
|
M TorkarZ NorgateM ColonnaJ TrowsdaleMJ
WilsonIsotypic variation of novel immunoglobulin-like
transcript/killer cell inhibitory receptor loci in the leukocyte
receptor complexEur J
Immunol2839593967199810.1002/(SICI)1521-4141(199812)28:12%3C3959::AID-IMMU3959%3E3.0.CO;2-29862332
|
|
15
|
Y ChenF ChuF GaoB ZhouGF GaoStability
engineering, biophysical, and biological characterization of the
myeloid activating receptor immunoglobulin-like transcript 1
(ILT1/LIR-7/LILRA2)Protein Expr
Purif56253260200710.1016/j.pep.2007.08.010
|
|
16
|
AH BanhamM ColonnaM CellaKJ MicklemK
PulfordAC WillisDY MasonIdentification of the CD85 antigen as ILT2,
an inhibitory MHC class I receptor of the immunoglobulin
superfamilyJ Leukoc Biol65841845199910380908
|
|
17
|
E MorelT BellonAmoxicillin conjugates to
HLA class I molecules and interferes with signalling through the
ILT2/LIR-1/CD85j inhibitory
receptorAllergy62190196200710.1111/j.1398-9995.2006.01285.x
|
|
18
|
AI ColovaiL TsaoS WangH LinC WangT SekiJG
FisherM MenesG BhagatB AlobeidN Suciu-FocaExpression of inhibitory
receptor ILT3 on neoplastic B cells is associated with lymphoid
tissue involvement in chronic lymphocytic leukemiaCytometry B Clin
Cytom72354362200710.1002/cyto.b.2016417266150
|
|
19
|
S Kim-SchulzeT SekiG VladL ScottoJ FanRC
ColomboJ LiuR CortesiniN Suciu-FocaRegulation of ILT3 gene
expression by processing of precursor transcripts in human
endothelial cellsAm J
Transplant67682200610.1111/j.1600-6143.2005.01162.x16433759
|
|
20
|
JV RavetchLL LanierImmune inhibitory
receptorsScience2908489200010.1126/science.290.5489.84
|
|
21
|
CS ChuiD LiRole of immuno-lglobulin-like
transcript family receptors and their ligands in suppressor
T-cell-induced dendritic cell tolerizationHum
Immunol70686691200910.1016/j.humimm.2009.06.00319524004
|
|
22
|
DP BrownDC JonesKJ AndersonN LapaqueRA
BuerkiJ TrowsdaleRL AllenThe inhibitory receptor LILRB4 (ILT3)
modulates antigen presenting cell phenotype and, along with LILRB2
(ILT4), is upregulated in response to Salmonella infectionBMC
Immunol1056200910.1186/1471-2172-10-5619860908
|
|
23
|
LE PilsburyRL AllenM VordermeierModulation
of Toll-like receptor activity by leukocyte Ig-like receptors and
their effects during bacterial infectionMediators
Inflamm2010536478201010.1155/2010/53647820634939
|
|
24
|
G VladJ KingCC ChangZ LiuRA FriedmanAA
TorkamaniN Suciu-FocaGene profile analysis of CD8+
ILT3-Fc induced T suppressor cellsHum
Immunol72107114201110.1016/j.humimm.2010.10.01220974207
|
|
25
|
G VladR CortesiniN
Suciu-FocaCD8+ T suppressor cells and the ILT3 master
switchHum Immunol696816862008
|
|
26
|
JY ZhangAK SomaniKA SiminovitchRoles of
the SHP-1 tyrosine phosphatase in the negative regulation of cell
signallingSemin
Immunol12361378200010.1006/smim.2000.022310995583
|
|
27
|
M ColonnaH NakajimaM CellaA family of
inhibitory and activating Ig-like receptors that modulate function
of lymphoid and myeloid cellsSemin
Immunol12121127200010.1006/smim.2000.021410764620
|
|
28
|
S LefebvreM AntoineS UzanM McMasterJ
DaussetED CarosellaP PaulSpecific activation of the non-classical
class I histocompatibility HLA-G antigen and expression of the ILT2
inhibitory receptor in human breast cancerJ
Pathol196266274200210.1002/path.103911857488
|
|
29
|
B FedoricR KrishnanRapamycin downregulates
the inhibitory receptors ILT2, ILT3, ILT4 on human dendritic cells
and yet induces T cell hyporesponsiveness independent of FoxP3
inductionImmunol Lett1204956200810.1016/j.imlet.2008.06.009
|
|
30
|
B FavierJ LemaoultE LesportED
CarosellaILT2/HLA-G interaction impairs NK-cell functions through
the inhibition of the late but not the early events of the NK-cell
activating synapseFASEB
J24689699201010.1096/fj.09-13519419841038
|
|
31
|
V HofmeisterEH WeissHLA-G modulates immune
responses by diverse receptor interactionsSemin Cancer
Biol13317323200310.1016/S1044-579X(03)00022-114708711
|
|
32
|
XS JuC HackerB SchererV RedeckeT BergerG
SchulerH WagnerGB LipfordM ZenkeImmunoglobulin-like transcripts
ILT2, ILT3 and ILT7 are expressed by human dendritic cells and
down-regulated following
activationGene331159164200410.1016/j.gene.2004.02.01815094202
|
|
33
|
M ShiroishiK TsumotoK AmanoY ShirakiharaM
ColonnaVM BraudDS AllanA MakadzangeS Rowland-JonesB WillcoxEY
JonesPA van der MerweI KumagaiK MaenakaHuman inhibitory receptors
Ig-like transcript 2 (ILT2) and ILT4 compete with CD8 for MHC class
I binding and bind preferentially to HLA-GProc Natl Acad Sci
USA10088568861200310.1073/pnas.143105710012853576
|
|
34
|
JS ManavalanPC RossiG VladF PiazzaA
YarilinaR CortesiniD ManciniN Suciu-FocaHigh expression of ILT3 and
ILT4 is a general feature of tolerogenic dendritic cellsTranspl
Immunol11245258200310.1016/S0966-3274(03)00058-312967778
|
|
35
|
J WuA HoruzskoExpression and function of
immunoglobulin-like transcripts on tolerogenic dendritic cellsHum
Immunol70353356200910.1016/j.humimm.2009.01.02419405174
|
|
36
|
U SvajgerA VidmarM JerasNiflumic acid
renders dendritic cells tolerogenic and up-regulates inhibitory
molecules ILT3 and ILT4Int
Immunopharmacol89971005200810.1016/j.intimp.2008.03.00618486911
|
|
37
|
N Suciu-FocaR CortesiniCentral role of
ILT3 in the T suppressor cell cascadeCell
Immunol2485967200710.1016/j.cellimm.2007.01.01317923119
|
|
38
|
I TamirJM Dal PortoJC CambierCytoplasmic
protein tyrosine phosphatases SHP-1 and SHP-2: regulators of B cell
signal transductionCurr Opin
Immunol12307315200010.1016/S0952-7915(00)00092-310781410
|
|
39
|
LS TaamsJM van AmelsfortMM TiemessenKM
JacobsEC de JongAN AkbarJW BijlsmaFP LafeberModulation of
monocyte/macrophage function by human
CD4+CD25+ regulatory T cellsHum
Immunol66222230200510.1016/j.humimm.2004.12.00615784460
|
|
40
|
A MunitzInhibitory receptors on myeloid
cells: New targets for therapy?Pharmacol
Ther125128137201010.1016/j.pharmthera.2009.10.00719913051
|
|
41
|
J LeitnerK Grabmeier-PfistershammerP
SteinbergerReceptors and ligands implicated in human T cell
costimulatory processesImmunol
Lett1288997201010.1016/j.imlet.2009.11.00919941899
|
|
42
|
YP SunJ LiuP GaoYS WangCY LiuExpression of
Ig-like transcript 4 inhibitory receptor in human non-small cell
lung cancerChest134783788200810.1378/chest.07-110018625675
|