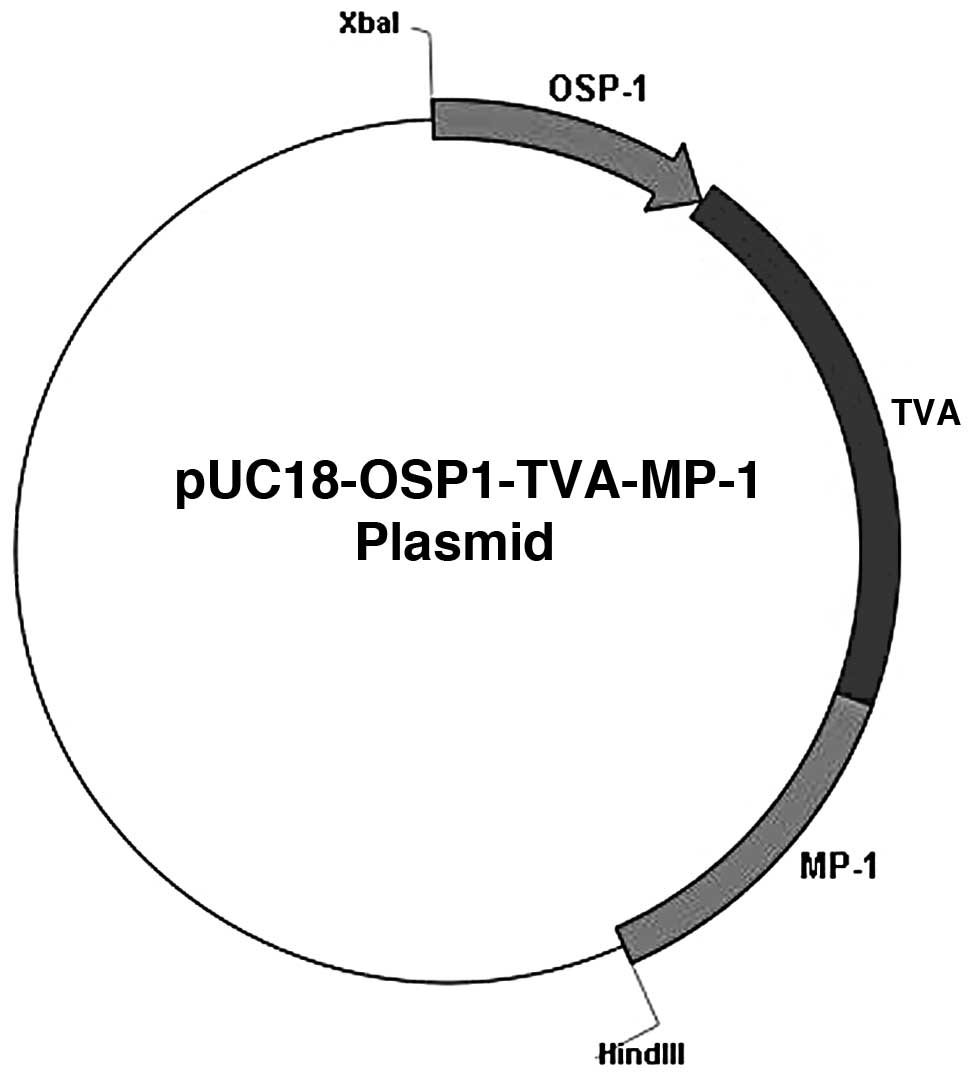
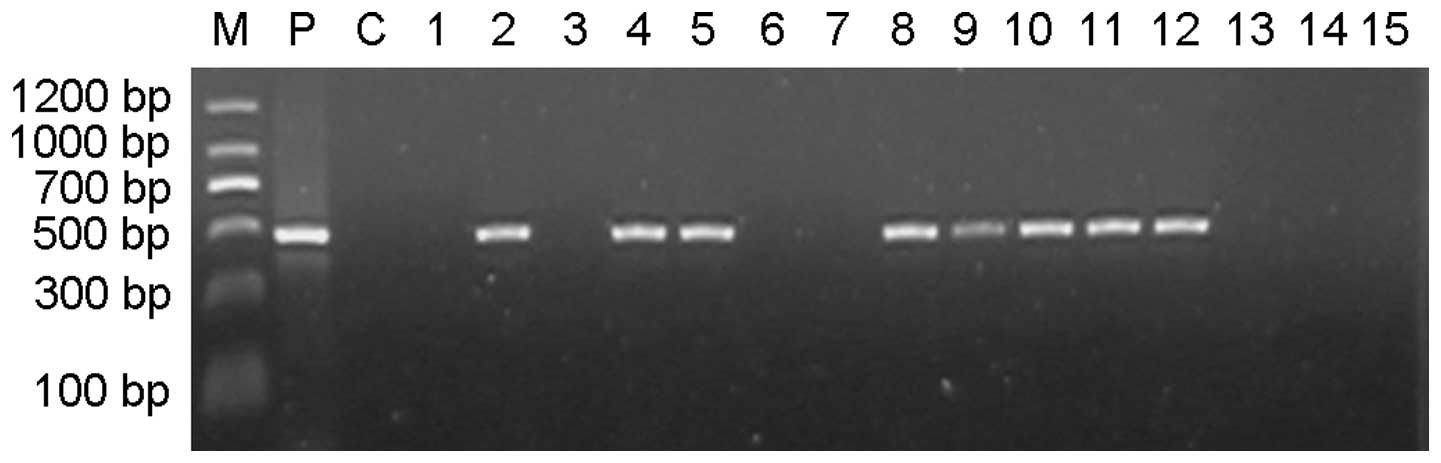
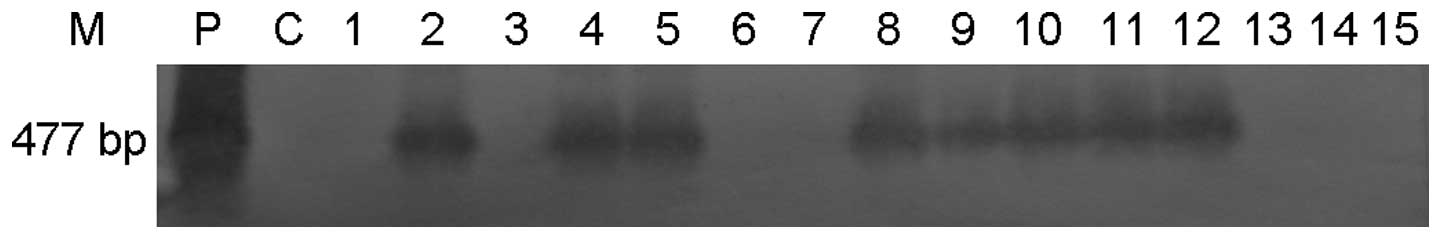
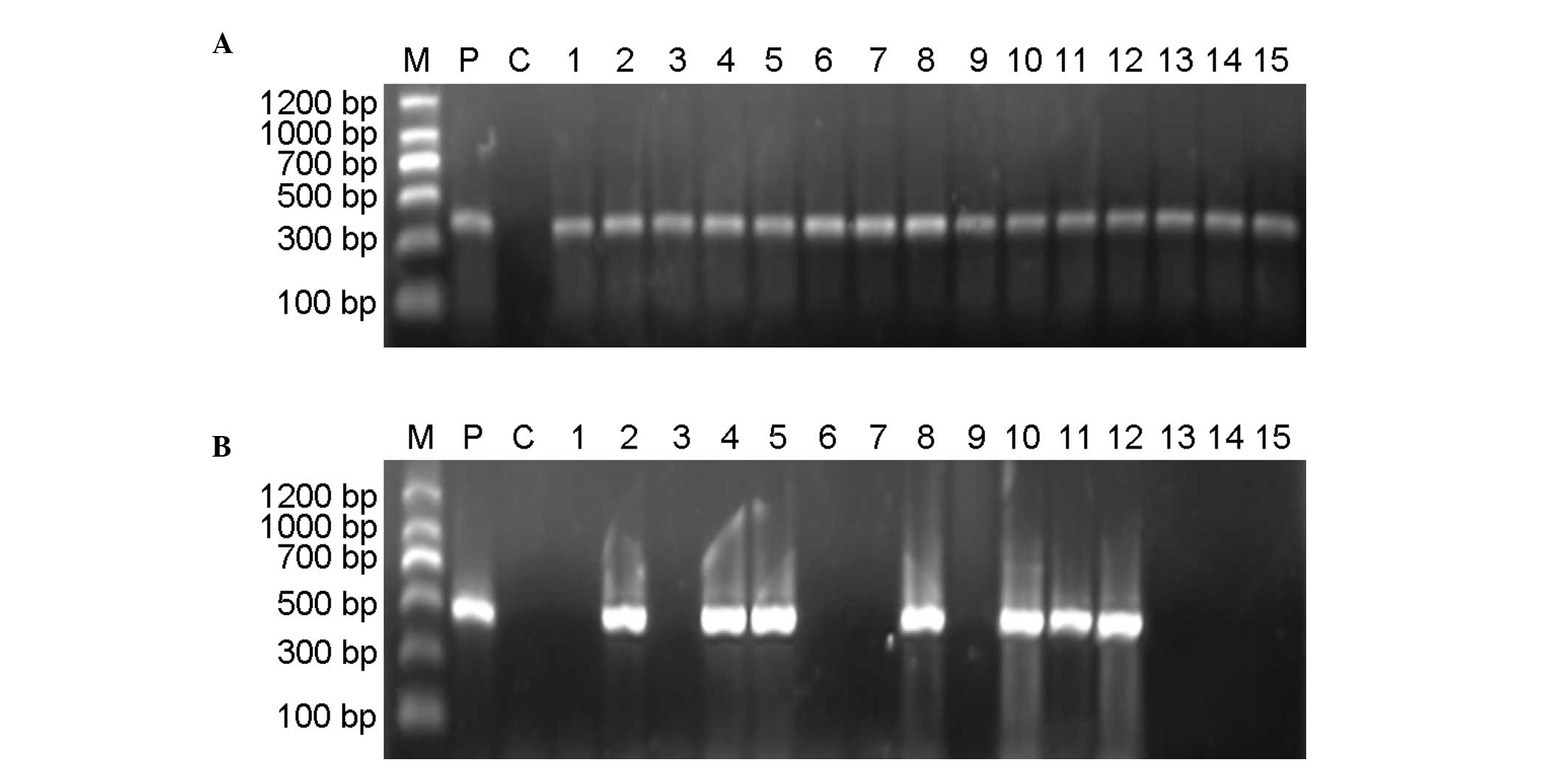
|
1
|
Gordon JW, Scangos GA, Plotkin DJ, Barbosa
JA and Ruddle FH: Genetic transformation of mouse embryos by
microinjection of purified DNA. Proc Natl Acad Sci USA.
77:7380–7384. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chan AW: Transgenic animals: current and
alternative strategies. Cloning. 1:25–46. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mullins LJ and Mullins JJ: Transgenesis in
the rat and larger mammals. J Clin Invest. 97:1557–1560. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wall RJ: New gene transfer methods.
Theriogenology. 57:189–201. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Niu Y and Liang S: Progress in gene
transfer by germ cells in mammals. J Genet Genomics. 35:701–714.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Moreira PN, Pozueta J, Pérez-Crespo M,
Valdivieso F, Gutiérrez-Adán A and Montoliu L: Improving the
generation of genomic-type transgenic mice by ICSI. Transgenic Res.
16:163–168. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ivics Z, Izsvák Z, Medrano G, Chapman KM
and Hamra FK: Sleeping Beauty transposon mutagenesis in rat
spermatogonial stem cells. Nat Protoc. 6:1521–1535. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Dhup S and Majumdar SS: Transgenesis via
permanent integration of genes in repopulating spermatogonial cells
in vivo. Nat Methods. 5:601–603. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lois C, Hong EJ, Pease S, Brown EJ and
Baltimore D: Germline transmission and tissue-specific expression
of transgenes delivered by lentiviral vectors. Science.
295:868–872. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yang SY, Wang JG, Cui HX, et al: Efficient
generation of transgenic mice by direct intraovarian injection of
plasmid DNA. Biochem Biophys Res Commun. 358:266–271. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bu W, Chen J, Morrison GD, et al: Keratin
6a marks mammary bipotential progenitor cells that can give rise to
a unique tumor model resembling human normal-like breast cancer.
Oncogene. 30:4399–4409. 2011. View Article : Google Scholar
|
|
12
|
Du Z and Li Y: RCAS-TVA in the mammary
gland: an in vivo oncogene screen and a high fidelity model for
breast transformation? Cell Cycle. 6:823–826. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Du Z, Podsypanina K, Huang S, et al:
Introduction of oncogenes into mammary glands in vivo with an avian
retroviral vector initiates and promotes carcinogenesis in mouse
models. Proc Natl Acad Sci USA. 103:17396–17401. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hambardzumyan D, Amankulor NM, Helmy KY,
Becher OJ and Holland EC: Modeling adult gliomas using RCAS/t-va
technology. Transl Oncol. 2:89–95. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lewis BC, Klimstra DS and Varmus HE: The
c-myc and PyMT oncogenes induce different tumor types in a somatic
mouse model for pancreatic cancer. Genes Dev. 17:3127–3138. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Loftus SK, Larson DM, Watkins-Chow D,
Church DM and Pavan WJ: Generation of RCAS vectors useful for
functional genomic analyses. DNA Res. 8:221–226. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Niu Y and Liang SL: Mammalian models based
on RCAS-TVA technique. Zoological Res. 3:335–345. 2008.
|
|
18
|
Reddy JP and Li Y: The RCAS-TVA system for
introduction of oncogenes into selected somatic mammary epithelial
cells in vivo. J Mammary Gland Biol Neoplasia. 14:405–409. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sausville J, Molinolo AA, Cheng X, et al:
RCAS/SCL-TVA animal model allows targeted delivery of polyoma
middle T oncogene to vascular endothelial progenitors in vivo and
results in hemangioma development. Clin Cancer Res. 14:3948–3955.
2008. View Article : Google Scholar
|
|
20
|
Orsulic S: An RCAS-TVA-based approach to
designer mouse models. Mamm Genome. 13:543–547. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Xing D and Orsulic S: A genetically
defined mouse ovarian carcinoma model for the molecular
characterization of pathway-targeted therapy and tumor resistance.
Proc Natl Acad Sci USA. 102:6936–6941. 2005. View Article : Google Scholar
|
|
22
|
Bao R, Selvakumaran M and Hamilton TC:
Targeted gene therapy of ovarian cancer using an ovarian-specific
promoter. Gynecol Oncol. 84:228–234. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Selvakumaran M, Bao R, Crijns AP, Connolly
DC, Weinstein JK and Hamilton TC: Ovarian epithelial cell
lineage-specific gene expression using the promoter of a
retrovirus-like element. Cancer Res. 61:1291–1295. 2001.PubMed/NCBI
|
|
24
|
He X, Qi B, Liu G, Yu W and Chen Q: A
novel method to transfer gene in vivo system. Prog Biochem Biophys.
33:685–690. 2006.
|
|
25
|
Chang KT, Ikeda A, Hayashi K, et al:
Production of transgenic rats and mice by the testis-mediated gene
transfer. J Reprod Dev. 45:29–36. 1999. View Article : Google Scholar
|
|
26
|
Shen W, Li L, Pan Q, Min L, Dong H and
Deng J: Efficient and simple production of transgenic mice and
rabbits using the new DMSO-sperm mediated exogenous DNA transfer
method. Mol Reprod Dev. 73:589–594. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Sato M, Ishikawa A and Kimura M: Direct
injection of foreign DNA into mouse testis as a possible in vivo
gene transfer system via epididymal spermatozoa. Mol Reprod Dev.
61:49–56. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
28
|
Valenzuela M, Relloso M and Esponda P: In
vivo transfection of the mouse vas deferens. J Exp Zool.
293:532–540. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Celebi C, Auvray P, Benvegnu T,
Plusquellec D, Jégou B and Guillaudeux T: Transient transmission of
a transgene in mouse offspring following in vivo transfection of
male germ cells. Mol Reprod Dev. 62:477–482. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yonezawa T, Furuhata Y, Hirabayashi K,
Suzuki M, Takahashi M and Nishihara M: Detection of transgene in
progeny at different developmental stages following testis-mediated
gene transfer. Mol Reprod Dev. 60:196–201. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Coward K, Kubota H, Hibbitt O, McIlhinney
J, Kohri K and Parrington J: Expression of a fluorescent
recombinant form of sperm protein phospholipase C zeta in mouse
epididymal sperm by in vivo gene transfer into the testis. Fertil
Steril. 85(Suppl 1): 1281–1289. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hibbitt O, Coward K, Kubota H, et al: In
vivo gene transfer by electroporation allows expression of a
fluorescent transgene in hamster testis and epididymal sperm and
has no adverse effects upon testicular integrity or sperm quality.
Biol Reprod. 74:95–101. 2006. View Article : Google Scholar
|
|
33
|
Kubota H, Hayashi Y, Kubota Y, Coward K
and Parrington J: Comparison of two methods of in vivo gene
transfer by electroporation. Fertil Steril. 83(Suppl 1): 1310–1318.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Gilbert SF: Developmental Biology. 8th
edition. Sinauer Associates; Sunderland, MA: pp. 613–646. 2006
|
|
35
|
Schlatt S: Spermatogonial stem cell
preservation and transplantation. Mol Cell Endocrinol. 187:107–111.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ju H, Bai L, Ren H, Mu Y, Yang S and Li K:
Production of transgenic mice by type-A spermatogonia-mediated gene
transfer. Agri Sci Chin. 10:431–437. 2011. View Article : Google Scholar
|