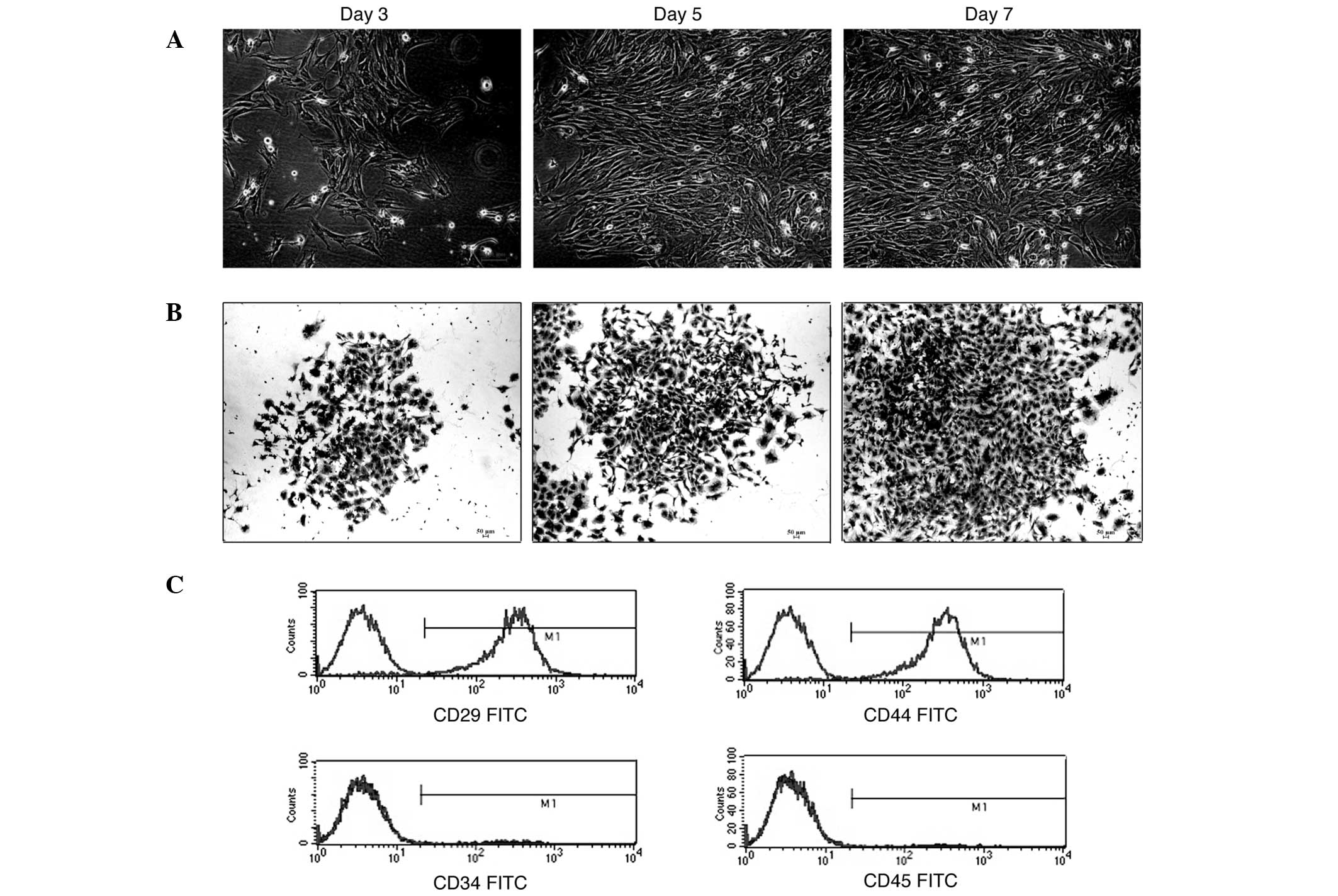
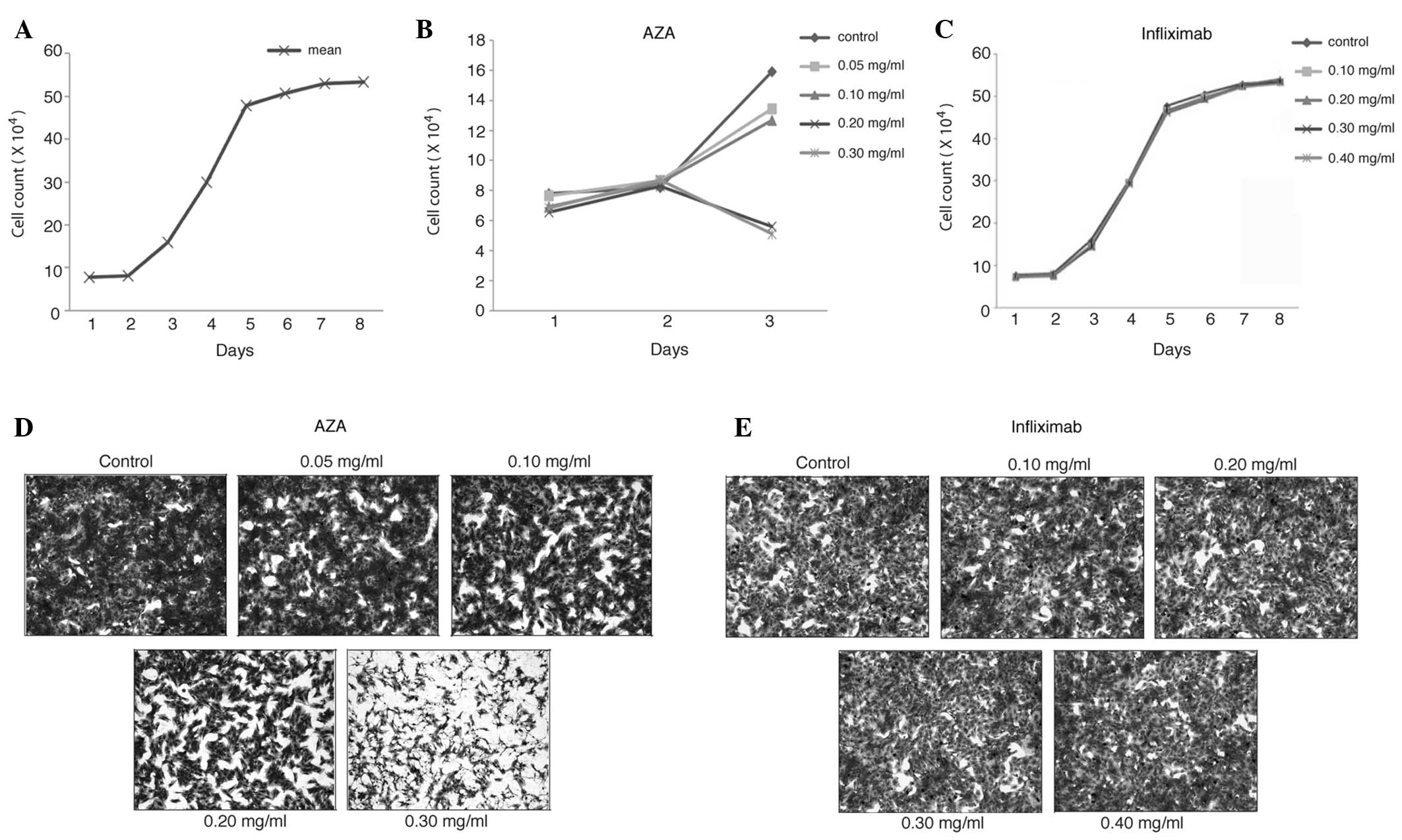
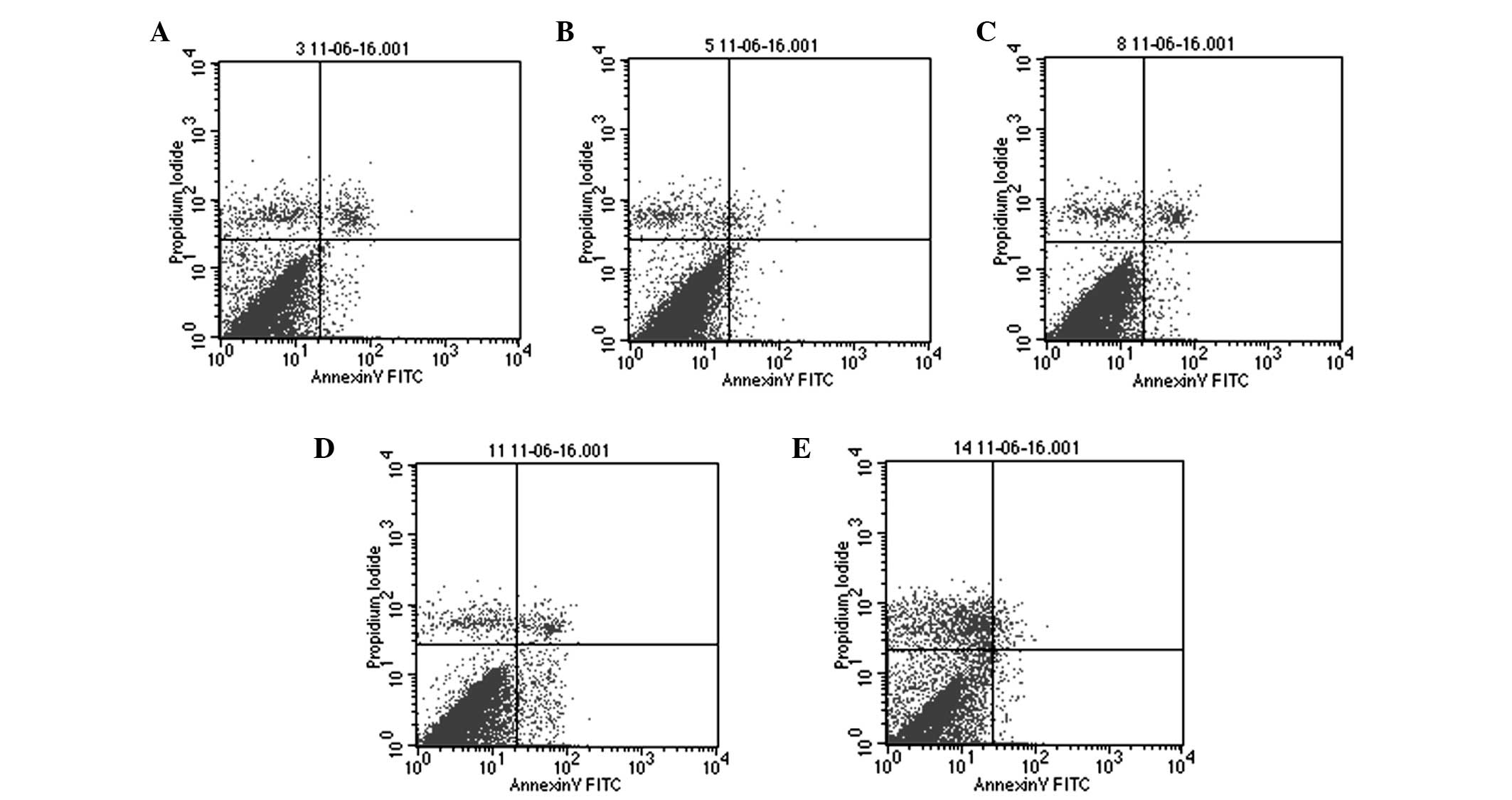
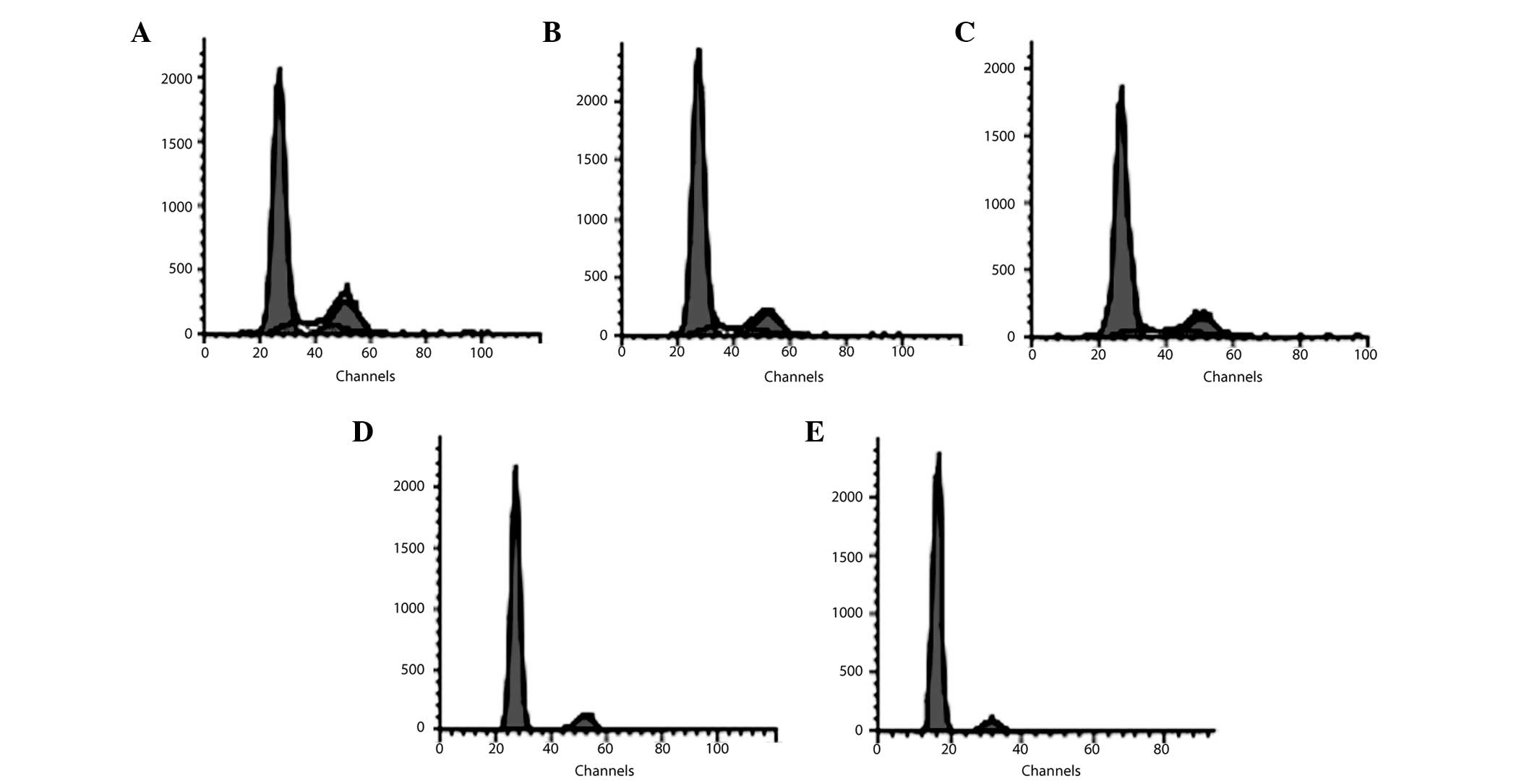
|
1
|
Beyer Nardi N and da Silva Meirelles L:
Mesenchymal stem cells: isolation, in vitro expansion and
characterization. Handb Exp Pharmacol. 174:249–282. 2006.
|
|
2
|
Li YP, Paczesny S, Lauret E, et al: Human
mesenchymal stem cells license adult CD34+ hemopoietic
progenitor cells to differentiate into regulatory dendritic cells
through activation of the Notch pathway. J Immunol. 180:1598–1608.
2008.PubMed/NCBI
|
|
3
|
Powell DW, Mifflin RC, Valentich JD, Crowe
SE, Saada JI and West AB: Myofibroblasts. II Intestinal
subepithelial myofibroblasts. Am J Physiol. 277:C183–C201.
1999.PubMed/NCBI
|
|
4
|
Lazebnik LB, Konopliannikov AG, Kniazew
OV, et al: Use of allogeneic mesenchymal stem cells in the
treatment of intestinal inflammatory disease. Ter Arkh. 82:38–43.
2010.(In Russian).
|
|
5
|
Gonzalez-Rey E, Anderson P, Gonzalez MA,
Rico L, Büscher D and Delgado M: Human adult stem cells derived
from adipose tissue protect against experimental colitis and
sepsis. Gut. 58:929–939. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Suarez-Almazor ME, Spooner C and Belseck
E: Azathioprine for treating rheumatoid arthritis. Cochrane
Database Syst Rev. 4:CD0014612000.PubMed/NCBI
|
|
7
|
Sandborn WJ: Azathioprine: state of the
art in inflammatory bowel disease. Scand J Gastroenterol. (Suppl
225): 92–99. 1998. View Article : Google Scholar
|
|
8
|
Patel AA, Swerlick RA and McCall CO:
Azathioprine in dermatology: the past, the present, and the future.
J Am Acad Dermatol. 55:369–389. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Rumbo C, Emerick KM, Emre S and Shneider
BL: Azathioprine metabolite measurements in the treatment of
autoimmune hepatitis in pediatric patients: a preliminary report. J
Pediatr Gastroenterol Nutr. 35:391–398. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Abu-Shakra M and Shoenfeld Y: Azathioprine
therapy for patients with systemic lupus erythematosus. Lupus.
10:152–153. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tiede I, Fritz G, Strand S, et al:
CD28-dependent Rac1 activation is the molecular target of
azathioprine in primary human CD4+ T lymphocytes. J Clin
Invest. 111:1133–1145. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Waters OR and Lawrance IC: Understanding
the use of immunosuppressive agents in the clinical management of
IBD. Curr Drug Targets. 12:1364–1371. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chevaux JB, Peyrin-Biroulet L and Sparrow
MP: Optimizing thiopurine therapy in inflammatory bowel disease.
Inflamm Bowel Dis. 17:1428–1435. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Oh CJ, Das KM and Gottlieb AB: Treatment
with anti-tumor necrosis factor alpha (TNF-alpha) monoclonal
antibody dramatically decreases the clinical activity of psoriasis
lesions. J Am Acad Dermatol. 42:829–830. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Maini R, St Clair EW, Breedveld F, et al:
Infliximab (chimeric anti-tumour necrosis factor alpha monoclonal
antibody) versus placebo in rheumatoid arthritis patients receiving
concomitant methotrexate: a randomised phase III trial. ATTRACT
Study Group. Lancet. 354:1932–1939. 1999. View Article : Google Scholar
|
|
16
|
Braun J and Sieper J: Ankylosing
spondylitis. Lancet. 369:1379–1390. 2007. View Article : Google Scholar
|
|
17
|
Yamamoto-Furusho JK: Current and future
biological treatments in inflammatory bowel disease. Gene Therapy
Applications. Kang C: InTech; Rijeka: pp. 363–374. 2011
|
|
18
|
Van den Brande JM, Braat H, van den Brink
GR, et al: Infliximab but not etanercept induces apoptosis in
lamina propria T-lymphocytes from patients with Crohn’s disease.
Gastroenterology. 124:1774–1785. 2003.
|
|
19
|
Efron PA and Moldawer LL: Cytokines and
wound healing: the role of cytokine and anticytokine therapy in the
repair response. J Burn Care Rehabil. 25:149–160. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Böcker W, Docheva D, Prall WC, et al:
IKK-2 is required for TNF-alpha-induced invasion and proliferation
of human mesenchymal stem cells. J Mol Med (Berl). 86:1183–1192.
2008.PubMed/NCBI
|
|
21
|
Duijvestein M, Molendijk I, Roelofs H, et
al: Mesenchymal stromal cell function is not affected by drugs used
in the treatment of inflammatory bowel disease. Cytotherapy.
13:1066–1073. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zan H and Zhong YQ: The progress of
mesenchymal stem cells in the treatment of inflammatory bowel
disease. New Medicine. 4:211–215. 2011.(In Chinese).
|
|
23
|
Fischer UM, Harting MT, Jimenez F, et al:
Pulmonary passage is a major obstacle for intravenous stem cell
delivery: the pulmonary first-pass effect. Stem Cells Dev.
18:683–692. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Schrepfer S, Deuse T, Reichenspurner H,
Fischbein MP, Robbins RC and Pelletier MP: Stem cell
transplantation: the lung barrier. Transplant Proc. 39:573–576.
2007. View Article : Google Scholar
|
|
25
|
Lee RH, Pulin AA, Seo MJ, et al:
Intravenous hMSCs improve myocardial infarction in mice because
cells embolized in lung are activated to secrete the
anti-inflammatory protein TSG-6. Cell Stem Cell. 5:54–63. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lazebnik LB, Kniazev OV, Konopliannikov
AG, et al: Allogeneic mesenchymal stromal cells in patients with
ulcerative colitis: two years of observation. Eksp Klin
Gastroenterol. 11:3–15. 2010.(In Russian).
|
|
27
|
Baert F, Noman M, Vermeire S, Van Assche
G, D’Haens G, Carbonez A and Rutgeerts P: Influence of
immunogenicity on the long-term efficacy of infliximab in Crohn’s
disease. N Engl J Med. 348:601–608. 2003.
|
|
28
|
Sagynbaeva VÉ, Lazebnik LB, Kniazev OV and
Efremov LI: Antibodies to infliximab and antigens HLA I–II class as
the witnesses of immune response to the biological treatment of
inflammatory bowel disease. Eksp Klin Gastroenterol. 12:7–14.
2011.(In Russian).
|