Introduction
Breast cancer is the most commonly diagnosed cancer
worldwide in women in 2020, with an estimated 2.3 million new cases
(11.7% of cancer cases in female patients), followed by lung cancer
(11.4%) (1). Acinic cell carcinoma
(ACC) is characterized by the presence of cancer cells with diffuse
serous differentiation (2,3). ACC can occur in the salivary glands,
pancreas, lung, stomach and breast (2–8).
Breast ACC is a rare subtype of breast cancer that has a benign
course in the majority of cases (8), although occasionally it can progress
into high-grade triple-negative breast cancer (TNBC) (9,10).
Although typically characterized by the presence of cells with
diffuse serous differentiation, breast ACCs can differ both
histologically and molecularly (8,9).
Breast ACCs are frequently found to comprise cytologically bland
cells with abundant eosinophilic cytoplasm, containing coarse or
fine zymogen-like cytoplasmic granules (8,9). These
granules tend to arrange themselves into a mixture of infiltrative
microglandular and nested growth patterns (10).
A previous genomic analysis study reported that
breast ACCs are not related to their salivary gland counterparts,
instead being closer to microglandular adenosis in addition to
sharing a similar molecular profile to TNBC (11). Tumor protein p53 (TP53)
mutations appear to be the most consistent molecular event in ACC
(11). PI3Kα mutations,
BRCA1 DNA repair-associated gene alterations, such as mutations and
gene deletions, and MutL homolog 1 germline mutations have also
been described in a subset of ACC cases (11,12).
Compared with open biopsy, image-guided core needle
biopsy (CNB) is highly sensitive and specific for the diagnosis of
breast cancer, leading to its widespread application (13). However, due to the low incidence
rate of ACC and lack of clinicopathological information present in
the biopsy tissues, breast ACC can be difficult to differentiate
from invasive breast carcinoma of no special type (NST) and other
types of TNBC pre-operatively (14). ACCs frequently exhibit a TNBC
phenotype but less aggressive clinical behavior compared with the
NST subtype (15). Therefore, the
present study aimed to analyze the morphological and
immunohistochemical features of breast ACC tumor samples obtained
by CNB and surgical excision. The association between the genomic
landscape and the morphological features of breast ACCs was also
investigated using next-generation sequencing (NGS).
Patients and methods
Patients
The clinical data from the medical records of 2
patients (case 1 and case 2) diagnosed with breast ACC were
retrospectively reviewed. Image-guided CNB was performed before
surgical treatment between January 1, 2015 and December 31, 2022.
The cases originated from the Department of Pathology of the
People's Liberation Army 989 Hospital (Pingdingshan, China) or the
Fenlan Laboratory (Hangzhou, China). The cases were reviewed by
five of the authors and samples were anonymized prior to analysis.
The present study was approved by 989 Hospital Medical Ethics
Committee (approval no. WZLL-2023-016) and Fenlan Lab Medical
Ethics Committee (2023–06). Patient consent was obtained if
required by the protocols. All tissues were fixed in 10% formalin
at room temperature for 24 h. For each tissue specimen, 4-µm
sections were cut. Hematoxylin and eosin-stained sections at room
temperature for 15 min of each case were reviewed by three
pathologists (LCY, LFY and LGX) under the BX53 light microscope
(Olympus Corporation). The inclusion criterion was a diagnosis in
accordance with breast ACC.
Immunohistochemistry (IHC)
Biopsy and surgical tissue sections (4 µm) from
paraffin blocks were stained immunohistochemically using a BOND-MAX
Automated IHC/ISH Stainer (Leica Microsystems GmbH). All tissue
blocks were fixed in 10% formalin at room temperature and embedded
in paraffin for 24 h. Sections were mounted onto slides, air dried
for 20 min and heated at 60°C for 20 min. Sections were dewaxed
twice with dewaxed solution (Celnovte Biotechnology Co., Ltd.) at
72°C for 2 min, hydrated three times with anhydrous ethanol at room
temperature for 2 min, and washed four times with washing solution
(Celnovte Biotechnology Co., Ltd.) at room temperature for 6 min,
The heat (98°C)-induced antigen retrieval method was performed
using Tris-EDTA buffer (1X; cat. no. K0071; Shanghai Jiehao
Biotechnology Co., Ltd.) and endogenous peroxidase activity was
quenched with 3% peroxidase-blocking reagent (Celnovte
Biotechnology Co., Ltd.) at 37°C for 5 min. The slides were
incubated with the following commercially available antibodies at
37°C for 30 min: Estrogen receptor (ER) (cat. no. CEM-0081; 1:500),
progesterone receptor (PR) (cat. no. CPM-0365; 1:500), HER2 (cat.
no. CCM-0844; 1:100); cytokeratin (CK)5/6 (cat. no. CCR-0982;
1:500), S100 (cat. no. CSM-0101; 1:500), GATA binding protein 3
(GATA3; cat. no. CGM-0130; 1:500), CK7 (cat. no. CCM-0992; 1:500),
p63 (cat. no. CPM-0160; 1:100) and Ki-67 (cat. no. CKM-0032; 1:500;
Table I). All antibodies were
purchased from Celnovte Biotechnology Co., Ltd. Tissue sections
were then washed twice with washing solution (Celnovte
Biotechnology Co., Ltd.) for 6 min and incubated with Microstacker™
+ Linker (cat. no. SD5300; 1:20; Celnovte Biotechnology Co., Ltd)
at room temperature for 15 min for signal amplification. After
washing twice with TBS for 6 min, samples were incubated with
Microstacker™ Flex HRP-polymer detection reagent (cat. no. SD5100;
1:20; Celnovte Biotechnology Co., Ltd) at 37°C for 30 min. After
incubation with the polymer reagent, tissue sections were
thoroughly washed three times with TBS buffer for 6 min and
incubated with Microstacker™ DAB + Chromogen (cat. no. SD5300;
1:20; Celnovte Biotechnology Co., Ltd) at 37°C for 6 min. Slides
were washed twice with TBS buffer for 6 min, then counterstained
with hematoxylin at 37°C for 2 min and washed with TBS and
dH2O for 6 min, respectively. Dehydration using graded
ethanol solutions and 90% xylene was performed, then sections were
mounted in synthetic resin and observed under a BX53 light
microscope (Olympus Corporation).
 | Table I.Primary antibodies and dilutions used
for the immunohistochemical analyses and observed results. |
Table I.
Primary antibodies and dilutions used
for the immunohistochemical analyses and observed results.
|
|
|
|
| Results |
|---|
|
|
|
|
|
|
|---|
| Antibody | Clone | Working
dilution | Pretreatment | Case 1 | Case 2 |
|---|
| ER | C6H7 | RTU | EDTA (pH=9.0) | Negative | Negative |
| PR | C4D10 | RTU | EDTA (pH=9.0) | Negative | Negative |
| HER2 | C1F7 | RTU | EDTA (pH=9.0) | 2+ | 2+ |
| CK5/6 | C6H1/C1C8 | RTU | EDTA (pH=9.0) | Positive | Positive |
| S100 | 4C4.9 | RTU | EDTA (pH=9.0) | Positive | Positive |
| GATA3 | L50-823 | RTU | EDTA (pH=9.0) | Positive | Negative |
| P63 | C2C10 | RTU | EDTA (pH=9.0) | Negative | Negative |
| Ki-67 | C3G4 | RTU | EDTA (pH=9.0) | 30%+ | 55%+ |
IHC results were scored by two independent observers
according to the percentage of positively stained cells: i) 0+,
1–25% staining; ii) 1+, 26–50% staining; iii) 2+, 51–75% staining;
and iv) 3+, 76–100% staining.
Fluorescence in situ hybridization
(FISH)
HER2 FISH was performed using the Kanglu HER2 DNA
Probe Kit (Wuhan HealthCare Biotechnology Co., Ltd.) according to
the manufacturer's protocols. Tissue blocks were fixed in 10%
formalin at room temperature and embedded in paraffin for 24 h. For
each tissue specimen, 4-µm sections were cut, deparaffinized,
rehydrated in ethanol gradient and heated in a pretreatment
solution of 2-(N-morpholino) ethanesulphonic acid at 95°C for 10
min before being soaked twice in Tris/HCl buffer at room
temperature for 3 min. The specimens were then digested in 200 µl
pepsin (4 mg/ml; Guangzhou Anbiping Medical Technology Co., Ltd.)
working solution at 37°C for 45 min and agitated twice in Tris/HCl
buffer at room temperature for 3 min. Slides were then dehydrated
in an ascending ethanol series at room temperature and air-dried.
Probe (10 µl; 500 ng/µl) was applied to each slide at 85°C for 5
min and the hybridization area was covered with a coverslip.
Co-denaturation of the probe and specimen was then performed at
85°C for 5 min, before the slides were incubated at 37°C for 10 h.
The slides were washed in 2X saline-sodium citrate/0.1% NP-40 at
37°C for 10 min and 10 µl DAPI counterstain was applied at 85°C for
5 min before sealing. Slides were imaged using a Zeiss Imager Z2
fluorescence microscope using FISH imaging systems (V 1.3;
GuangZhou MM Photoelectric Techology Co., Ltd.) software
(Fluorescence microscope MF43-M (medical device model) + micro
camera MC50-S/MS23+ fluorescent light source MG-100/MG-120/MG-200).
The fluorescent signal in 20 cells from each slide was analyzed by
two independent observers. FISH results were recorded as either
negative or positive according to the American Society of Clinical
Oncology/College of American Pathologists guideline by the two
experienced pathologists independently in 20 tumor cells in a
blinded manner (16).
For ETS variant transcription factor 6 (ETV6)
FISH, 4-µm 10% formalin-fixed at room temperature for 24 h,
paraffin-embedded tumor sections were heated at 60°C for 1 h,
rinsed in 100% ethanol and pretreated with 0.2 N HCl for 20 min at
room temperature. Slides pretreatment, hybridization, washing and
counterdyeing steps followed: Place slides in a 65±5°C incubator
for 24 h, room temperature in xylene twice, 10 min each time → 100%
ethanol at room temperature for 10 min → 90% ethanol at room
temperature for 3 min → 70% ethanol at room temperature for 3 min →
purified water at room temperature for 3 min → purified water at
100°C for 25 min → pepsin (4 mg/ml; Guangzhou Anbiping Medical
Technology Co., Ltd.) working solution at 37°C for 15 min → 2×SSC
at room temperature for 3 min → 70% ethanol at room temperature for
2 min → 90% ethanol at room temperature for 2 min → 100% ethanol at
room temperature for 2 min → add probe (500 ng/µl; Guangzhou
Anbiping Medical Technology Co., Ltd.) to the slides→ denature at
85°C for 5 min → hybridization at 37°C for 18 h → 0.1% NP-40/2X SSC
at 37°C for 5 min → 70% ethanol for at room temperature 3 min →
hybrid blue staining solution at room temperature. Slides were
imaged using a Zeiss fluorescence microscope (Carl Zeiss AG).
IMSTAR software (version 2.0; ProgRes CT3, Jena, Germany) was used
to analysis the results.
NGS
The two patients underwent molecular analysis using
the Illumina NGS platform (Illumina, Inc.), which can detect
mutations, copy number variations and fusions in 685 genes that are
known to be oncogenic drivers (17). NGS was performed for the two
patients using DNA from both tumor samples. NGS was performed
according to Illumina's standard protocol. DNA was extracted using
the MeiJi Tissue & Blood DNA Kit (cat. no. IVD3101-200;
Guangzhou Magen Biotechnology Co., Ltd.) according to the
manufacturer's protocol. A paired-end DNA library was prepared
according to the manufacturer's protocol (Agilent Technologies,
Inc.). Samples with >1 µg DNA were used for library preparation.
Qubit quantification was used to verify the quality and integrity
of samples and that the extraction amount was ≥50 ng. The loading
concentration of the final library was 0.7 nM. The concentration of
Cng/ul was measured by Qubit, and the average size of the library
was double base pair) analyzed using the Agilent 4200 bioanalyzer
(Agilent Technologies, Inc.). The molar concentration was
calculated according to the formula: M=(Cng/µl
×106)/(660 g/mol × D) nmol/l. Genomic DNA was fragmented
using a sonicator (Covaris, LLC) to a length of 200–300 bp. The
ends of the DNA fragments were then repaired, before the Illumina
Adaptor was added (Fast Library Prep Kit; iGeneTech Bioscience Co.,
Ltd.) Using the 2003200-P2 Unique Dual Index Adapters (2 nmol) and
KAPA Library Construction kit kk8514 (F. Hoffmann-La Roche Ltd;
cat. no. 07962428001) KAPA Hyperplus Kit. The DNA fragments were
end-polished, A-tailed and ligated with the full-length adapter.
After the sequencing library was constructed, whole exomes were
captured with the NadPrep Hybrid Capture Reagents (Nanodigmbio
Biotechnology Co., Ltd.) and double-ended double-label sequenced
using a Nextseq 6000 sequencer (Illumina, Inc.) with the NovaSeq
6000 S1 Reagent Kit v1.5 (300 cycles; cat. no. 20028317; Illumina,
Inc.). The maximum sequencing read length was 2×150 bp (Paired-End
150 bp) and double-ended double-label sequencing was performed.
Fastp software (version 0.11.9) (18) was used for quality control and
preprocessing and the Burrows-Wheeler Aligner software (version
0.7.16) (19) was used to align
clean reads with the hg19 reference genome. Mutect software
(version 1.1.6) (20) was used to
detect somatic variants in the tumor samples. Single nucleotide
variants were annotated and filtered using snpEff software (version
5.1) (21).
Results
Clinical findings
The clinical data of the 2 patients were recorded
(Table II). Both of the patients
were female, and the patients were aged 42 and 69 years old at the
time of diagnosis. Both patients reported the presence of a mass
with no other complications. The preoperative duration was 3 days
and 1 month for case 1 and case 2, respectively. Follow-up
information was available for case 1 and case 2 for 16 and 14
months, respectively. After surgery, case 1 was administered with
adjuvant chemotherapy of the TC taxel + Cyclophosphamide) scheme
(120 mg/m2 docetaxel and 800 mg/m2
cyclophosphamide) intravenously every 3 weeks for 4 cycles.
Subsequently, 1 year after surgery, the patient (case 1) developed
a liver metastasis (Fig. S1),
accompanied by lumbar vertebra and sacral vertebra metastasis. In
addition, 15 months after surgery, the patient presented with
ascites. Case 2 declined any further therapy and showed no
recurrence or metastasis after surgery. Patients denied the use of
novel antibody-drug conjugates (ADCs).
 | Table II.Clinical and histopathologic
characteristics of patients with breast ACC. |
Table II.
Clinical and histopathologic
characteristics of patients with breast ACC.
| Clinical
characteristic | Case 1 | Case 2 |
|---|
| Patient sex | Female | Female |
| Age, years | 42 | 69 |
| Imaging size |
|
|
| Lymph node
status | 0/9 | 0/23 |
| Follow-up duration,
months | 16 | 14 |
| Surgical
treatment | Mastectomy and
ALND | Mastectomy and
ALND |
| Preoperative
duration | 3 days | 1 month |
| Chemotherapy |
|
|
|
Neoadjuvant | No | No |
|
Adjuvant | Yes | No |
| Adjuvant
radiation | No | No |
| Growth pattern |
Solid-trabecular |
Solid-trabecular |
| DCIS | No | No |
| Recurrence | No | No |
| Site of distant
metastasis | Liver, lumbar
vertebra and sacral vertebra | Negative |
| Patient
outcome | Alive | Alive |
| Tumor size, mm | 50 | 25 |
| Diagnosis | Pure ACC | Pure ACC |
Imaging findings
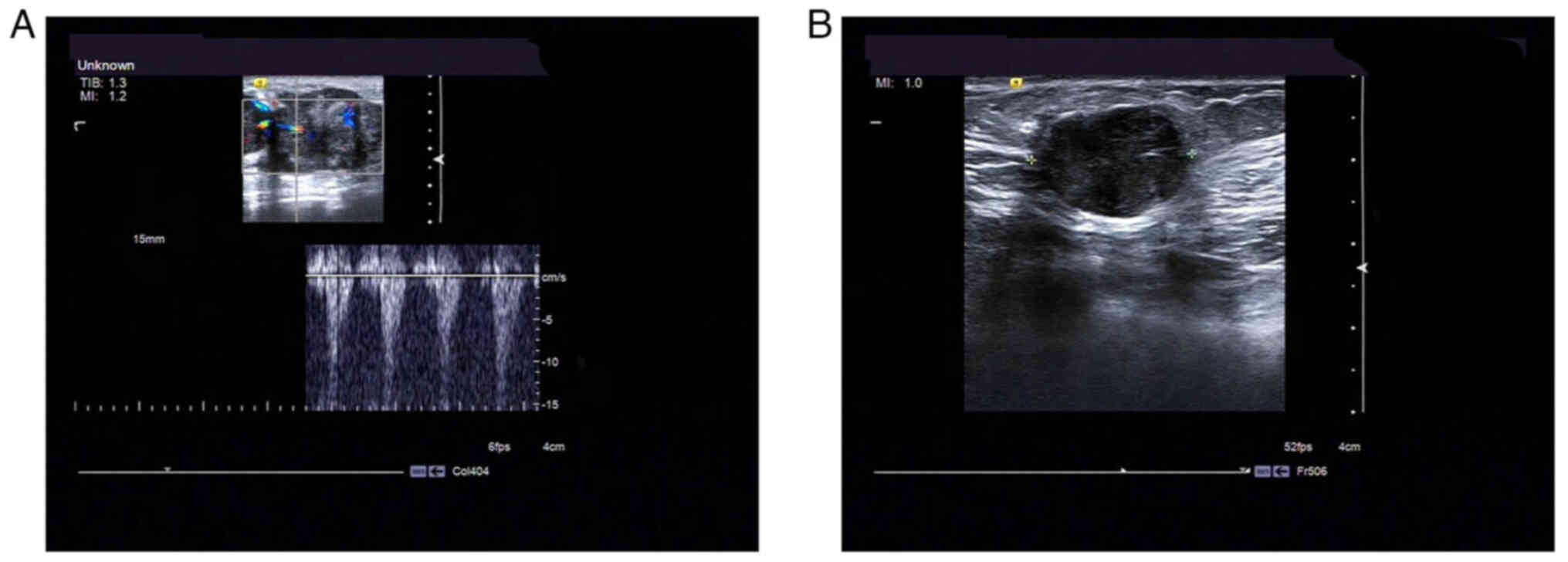
In case 1, a hypoechoic nodule of irregular shape
with dimensions of ~4.6×2.3×2.7 cm was found in the left breast (‘6
o'clock’) beneath the nipple with unclear boundaries (Fig. 1A). The lesion was characterized as
Breast Imaging Reporting and Data System (BI-RADS) category 4
(22).
In case 2, a hypoechoic mass measuring ~1.9×1.4×1.9
cm could be observed in the right breast (‘11 o'clock’), 2 cm away
from the nipple. The tumor boundary was visible, and the shape was
regular and was accompanied by a lateral sound shadow with a strong
echo. Blood flow signals could be detected in the mass. The lesion
was characterized as BI-RADS category 4 (Fig. 1B).
Pathological features
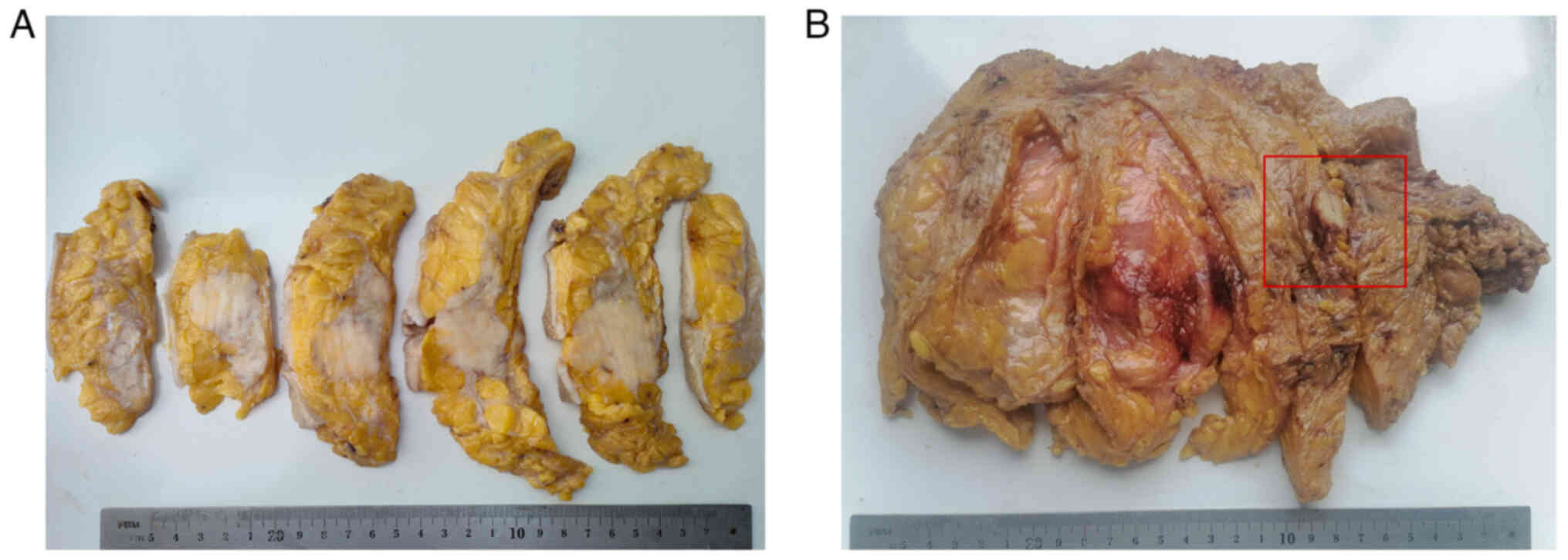
Macroscopic features
On gross examination, the tumor in case 1 was
55×40×30 mm in size, with a gray-white and rubbery consistency, had
an ill-defined border and was moderately firm-hard to the touch
(Fig. 2A). In case 2, there was a
30×25×20-mm tumor with a well-defined border that was moderately
firm-hard and white-grey on the cut surface (Fig. 2B). There were no skin ulcerations,
nipple changes or separate skin nodules in cases 1 and 2.
Microscopic features and IHC
findings
Both of the tumors exhibited solid or nested growth
patterns. The solid or nested growth patterns interconnected to
form the burrowing labyrinthine network, ‘hand-holding-hand’
pattern or crawling phenomenon, which is similar to the carcinoma
cuniculatum in the mandible (22)
or the crawling carcinoma in the stomach (23) (Figs.
3A-E and 4A-E). The neoplastic
cells exhibited abundant basophilic cytoplasm with
centrally-located nuclei and prominent nucleoli, but also
occasionally exhibited a clear cytoplasm (Figs. 3F and 4F). Atypia of tumor cells was prominent
with marked mitotic figures (Figs.
3F and 4F insert). The cancer
cells had high-grade nuclei with moderate-to-severe pleomorphism,
coarse chromatin and a prominent singular nucleolus (Figs. 3F and 4F). An in situ component was not
present either within or around the tumor. There were no invasive
lymphocytes in the tumor tissues (Figs.
3A and 4A). Tumors were
positive for S100 protein (Figs. 3G
and 4G) in both patients. The
neoplastic cells exhibited moderately positive (2+) HER2 expression
(Fig. 3H and 4H). Both of the cases were CK5/6-
(Fig. S2A and B) and CK7-positive
(Fig. S2C and D). The tumor was
positive for GATA3 in case 1 (Fig.
S2E), but negative in case 2 (Fig.
S2F). Staining for p63 (Fig. S2G
and H), ER (Fig. S3A and B)
and PR (Fig. S3C and D) was
negative in both tumors. The Ki-67 index was 30% (Fig. S3E) and 55% (Fig. S3F) in the tumors from case 1 and
case 2, respectively.
 | Figure 3.Staining results of the tumor tissue
from case 1. Imaging of the tumor tissue demonstrated (A) invasive
carcinoma (magnification, ×18), (B) the ‘hand-holding-hand’ pattern
of tumor cells (red box; magnification, ×66), (C) the fibrotic
focus (red box; magnification, ×10), (D) the ‘hand-holding-hand’
pattern (red box; magnification, ×50), (E) the ‘hand-holding-hand’
pattern (red box; magnification, ×50) in another area, (F) cell
characteristics of obvious atypia and mitotic figures (insert;
magnification, ×240), (G) positive S100 staining highlighting the
‘hand-holding-hand’ pattern (magnification, ×78) and (H) human
epidermal growth factor receptor-2 immunohistochemistry staining
scored as 2+ (magnification, ×150). |
 | Figure 4.Staining results of the tumor tissue
from case 2. Imaging of the tumor tissue demonstrated (A) invasive
carcinoma (magnification, ×20), (B) the ‘hand-holding-hand’ pattern
of tumor cells (red box; magnification, ×120), (C) the clear
boundary of the tumor (magnification, ×010), (D) the
‘hand-holding-hand’ pattern (red box; magnification, ×35), (E) the
‘hand-holding-hand’ pattern (red box; magnification, ×35) in
another area, (F) cell characteristics of obvious atypia and
mitotic figures (insert; magnification, ×260), (G) positive S100
staining and the ‘hand-holding-hand’ pattern (magnification, ×78)
and (H) human epidermal growth factor receptor-2
immunohistochemistry staining scored as 2+ (magnification,
×150). |
Molecular analysis demonstrated
mutations in mediator of RNA polymerase II transcription, subunit
12 homolog (MED12) exon 27
Targeted NGS results from case 1 demonstrated the
presence of mutations in the tumor protein p53 binding protein 1
(NM_001141980.1; c.1362_1367del; p.I455_P456del), partner and
localizer of breast cancer 2 (NM_024675. 3; c.2799del;
p.C933Wfs*2), fibroblast growth factor 3 (NM_005247.2; c.542G>A;
p.R181H), α thalassemia/mental retardation syndrome X-linked
(NM_000489.4; c.5690A>G; p.E1897G), mTOR (NM_004958.3;
c.6414G>T; p.L2138F), Kelch-like protein 6 (NM_130446.2;
c.1589C>G; p.A530G), cyclin E1 (NM_001238.3; c.479A>G;
p.K160R), MED12 (NM_005120.2; c.3817G>T; p.A1273S;
abundance, 2.17%) and E74-like ETS transcription factor 3
(NM_004433.4; c.485G>A; p.G162D) genes.
The gene mutations present in case 2 included
TP53 (NM_000546.5; c.323del; p.G108Vfs*15), nuclear
receptor-binding SET domain 1 (NM_022455.4; c.3157A>T;
p.K1053*), spen family transcription repressor (NM_015001.2;
c.7480C>T; p.P2494S), disruptor of telomeric silencing 1-like
(NM_032482.2; c.3580G>A; p.E1194K), rearranged in transfection
(NM_020975.4; c.538C>T; p.R180*), janus kinase 3 (NM_000215.3;
c.3118T>A; p.C1040S), mitogen-activated protein kinase kinase
kinase 14 (NM_003954.4; c.115G>A; p.V39I), MED12
(NM_005120.2; c.3817G>T; p.A1273S; abundance, 3.25%) and lysine
methyltransferase 2C (NM_170606.2; c.1814-1G>A; p.*). The
MED12 exon 27 mutation was found in both cases (Tables III and IV).
 | Table III.Gene mutations detected in case
1. |
Table III.
Gene mutations detected in case
1.
| Gene | Exon | Nucleic acid
variation | Amino acid
change | Abundance, % |
|---|
| TP53BP1 | 11 | NM_001141980.1;
c.1362_1367del | p.I455_P456del | 30.84 |
| PALB2 | 8 | NM_024675.3;
c.2799del | p.C933Wfs*2 | 29.65 |
| FGF3 | 3 | NM_005247. 2;
c.542G>A | p.R181H | 24.15 |
| ATRX | 23 | NM_000489.4;
c.5690A>G | p.E1897G | 22.16 |
| MTOR | 46 | NM_004958.3;
c.6414G>T | p.L2138F | 11.62 |
| KLHL6 | 7 | NM_130446.2;
c.1589C>G | p.A530G | 5.78 |
| CCNE1 | 7 | NM_001238.3;
c.479A>G | p.K160R | 3.09 |
| MED12 | 27 | NM_005120.2;
c.3817G>T | p.A1273S | 2.17 |
| ELF3 | 5 | NM_004433.4;
c.485G>A | p.G162D | 1.08 |
 | Table IV.Gene mutations detected in case
2. |
Table IV.
Gene mutations detected in case
2.
| Gene | Exon or intron | Nucleic acid
variation | Amino acid
change | Abundance, % |
|---|
| TP53 | Exon 4 | NM_000546.5;
c.323del | p.G108Vfs*15 | 78.19 |
| NSD1 | Exon 5 | NM_022455.4;
c.3157A>T | p.K1053* | 34.37 |
| SPEN | Exon 11 | NM_015001.2;
c.7480C>T | p.P2494S | 31.45 |
| DOT1L | Exon 25 | NM_032482.2;
c.3580G>A | p.E1194K | 30.84 |
| RET | Exon 3 | NM_020975.4;
c.538C>T | p.R180* | 19.38 |
| JAK3 | Exon 23 | NM_000215.3;
c.3118T>A | p.C1040S | 18.84 |
| MAP3K14 | Exon 2 | NM_003954.4;
c.115G>A | p.V39I | 9.40 |
| MED12 | Exon 27 | NM_005120.2;
c.3817G>T | p.A1273S | 3.25 |
| KMT2C | Intron 13 | NM_170606.2;
c.1814-1G>A | p.? | 3.17 |
FISH results
FISH showed the average copy number of the red
signal/average copy number of the green signal in case one and case
two was 1.0 and 1.11, respectively, and revealed that the tumor
tissues in the two case were negative for HER2 amplification
(Fig. S4A and B). The two cases
showed 2G(green signal)2R(red signal) signal mode and indicated no
ETV6 break-apart probe detection (Fig. S4C and D).
Discussion
Invasive breast carcinoma is an umbrella term used
for a large and heterogeneous group of malignant epithelial
neoplasms of the glandular elements in the breast (10). Breast ACC is a rare type of invasive
breast cancer that typically has a benign course (8,9).
However, occasionally breast ACC can progress into high-grade TNBC
(9,10). To the best of our knowledge, the
present study was the first to focus on the relationship between
the structure of breast ACC and gene alterations. In addition, the
microstructure of the breast ACC in CNB samples was probed,
supplementing the cellular features assessed, such that structures
in the CNB and surgically excised samples were compared. Beca et
al (12) previously reported
that breast ACCs were genetically heterogeneous and displayed
genomic features overlapping those of the common forms of TNBC
(12). Furthermore, breast ACCs
were not likely to be driven by a highly recurrent mutation or
oncogenic gene fusion events (12).
In the present study, the CNB samples and lumpectomy
specimens were compared. It was demonstrated that both cases showed
the same histological and IHC features. Specifically, both sets of
tissues exhibited a solid or nest interconnecting structures and
formed a burrowing labyrinthine network, similar to the carcinoma
cuniculatum of the oral cavity (23) or branching and anastomosing
structure, similar to ‘hand-shaking-hand’ pattern or crawling
phenomenon in well-differentiated gastric adenocarcinoma of
intestinal type (24). There was no
carcinoma in situ in the center or around the invasive
components, and no tubular or microglandular structures were found.
However, it was demonstrated that the cancer cells did have
high-grade nuclei of moderate-to-severe pleomorphism, coarse
chromatin and a prominent singular nucleolus. These tumor cells
also had uniform basophilic cytoplasm and the finely to coarsely
granular chromatin were not obvious. There were no or few
mononucleated lymphoid cells infiltrating into the tumor cells and
stroma, although there was marked hyalinization in the stromal
component. The tumors were negative for ER, PR and p63, and
positive for S100, CK5/6 and CK7. The tumor cells stained
moderately positive (2+) for HER2, but no HER2 amplification could
be detected by FISH. Therefore, burrowing labyrinthine networks,
hand-holding-hand pattern or crawling phenomenon as structures and
basophilic cytoplasm as a cytological feature are likely to be
unique to ACC CNB or surgically excised samples. Although ACC can
be misdiagnosed as a secretory carcinoma, it typically presents
with a bland nuclear morphology and harbors ETV6-neurotrophic
receptor tyrosine kinase 3 fusions, which were absent in the
present study.
According to the genomic analyses of the 2 patients
in the present study, the tumors demonstrated MED12 exon 27
mutations. MED12 is an X chromosome-linked gene that encodes
the protein mediator complex subunit 12 (25). Mediator complex subunit 12 is a
large multi-subunit complex that is critical for gene regulation
(24). MED12 mutations have
been identified in 80% of phyllodes tumors with little variance in
frequency among malignant, benign and borderline tumors (26). In addition, MED12 mutations
have been previously reported in breast cancer (27). Conlon et al (28) previously reported missense mutations
in the MED12 gene (D1204E) with lower allelic
frequencies in 1 case of breast ACC (27). However, both of the cases in the
present study displayed mutations in exon 27 (NM_005120.2;
c.3817G>T; p.A1273S). In addition, MED12 mutations
detected in these patients exhibited a similar abundance.
Therefore, MED12 exon 27 mutations may be associated with
the burrowing labyrinthine network or ‘hand-holding-hand’ structure
of breast ACC.
According to IHC results, all invasive breast
cancers are grouped into the following biomarker-defined
subtypes/groups for treatment purposes on the basis of the HER2
status: i) ER-positive, HER2-negative; ii) ER-positive,
HER2-positive; iii) ER-negative, HER2-positive; or iv) ER-negative,
HER2-negative (10). HER2-negative
tumors are further categorized into HER2-0 (IHC staining score of
0) and HER2-low (IHC staining score of 1+ or 2+ combined with
FISH-negative results) (29). ADCs
have been used effectively in patients with low HER2 protein
expression (30). The patients in
the present study were grouped into the HER2-low breast cancer
category, although the tumors were HER2-negative according to the
FISH results. However, the patients refused the use of ADCs.
The prognosis of patients with breast ACC remains
uncertain. A previous case report suggested that breast ACC is
likely to have a good prognosis (15). However, it is clear that poorly
differentiated TNBC can become a component of breast ACC and
recurrence and mortality can occur (14). It could be suggested that prognosis
is predominantly driven by the presence of the poorly
differentiated component. The 2 cases in the present study
exhibited high-grade cell features. However, the patient from case
1 had a poor prognosis and the patient from case 2 appeared to have
a superior outcome. Although case 1 was administered adjuvant
chemotherapy of the TC scheme 10 days after surgery, and 1 year
after surgery, the patient developed a liver, lumbar vertebra and
sacral vertebra metastasis and 15 months later presented with
ascites after surgery. And Case two declined any further therapy
and showed no recurrence or metastasis after surgery. This diverse
prognostic observation may be related to the fibrotic focus
(31).
The present study had several limitations. The
present study was retrospectively designed, which could cause
certain selection bias. In addition, only 2 breast ACC cases were
described in the present study.
In summary, the present study demonstrated a rare
structure of breast ACC with a burrowing labyrinthine network or
‘hand-holding-hand’ feature. The key to accurate diagnosis is the
combination of knowledge of tumor type, recognition of the
characteristic morphology and IHC profile. The same gene mutation
with a similar abundance in MED12 exon 27 (NM_005120.2;
c.3817G>T; p.A1273S) was reported in both tumors. Therefore, the
MED12 exon 27 mutation may potentially be associated with
the formation of a burrowing labyrinthine network or the
‘hand-holding-hand’ feature observed in the present study. Future
studies with larger sample sizes are required to further
investigate this potential genotype-phenotype association in breast
ACC.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by a grant from the People's
Liberation Army 989 Hospital Foundation (grant no.
9892023YNKT-04A).
Availability of data and materials
The NGS datasets generated and/or analyzed during
the current study are available in the NCBI Sequence Read Archive
repository, https://www.ncbi.nlm.nih.gov/sra/PRJNA1025041. The
other datasets used and/or analyzed during the current study are
available from the corresponding author on reasonable request.
Authors' contributions
CL, GL, XX, ZL, FL and XY designed the study. ZL,
XX, MZ, YaL, XY and XG recruited the patients. CL, GL, FL, ZL, ZC,
CZ, XY, MZ, YaL, PW, YuL and XG analyzed the experimental data and
composed all figures and tables. CL, FL, ZL and HY wrote the
manuscript. ZC, XG, MZ and FL confirm the authenticity of all the
raw data. All authors read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
The present study was approved by the 989 Hospital
Medical Ethics Committee (Luoyang, China; ethics approval no.
WZLL-2023-016) and Fenlan Lab Medical Ethics Committee (Hangzhou,
China; ethics approval no. 2023-06). Written informed consent was
obtained from the patients at the time of the initial data
collection for participation in the study.
Patient consent for publication
The patients provided written informed consent for
publication of patient data and associated images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ordóñez NG: Pancreatic acinar cell
carcinoma. Adv Anat Pathol. 8:144–159. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Holen KD, Klimstra DS, Hummer A, Gonen M,
Conlon K, Brennan M and Saltz LB: Clinical characteristics and
outcomes from an institutional series of acinar cell carcinoma of
the pancreas and related tumors. J Clin Oncol. 5:4673–4678. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rodriguez J, Diment J, Lombardi L,
Dominoni F, Tench W and Rosai J: Combined typical carcinoid and
acinic cell tumor of the lung: A heretofore unreported occurrence.
Hum Pathol. 34:1061–1065. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hervieu V, Lombard-Bohas C, Dumortier J,
Boillot O and Scoazec JY: Primary acinar cell carcinoma of the
liver. Virchows Arch. 452:337–341. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ambrosini-Spaltro A, Potì O, De Palma M
and Filotico M: Pancreatic-type acinar cell carcinoma of the
stomach beneath a focus of pancreatic metaplasia of the gastric
mucosa. Hum Pathol. 40:746–749. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Pesci A, Castelli P, Facci E, Romano L and
Zamboni G: Primary retroperitoneal acinar cell cystadenoma. Hum
Pathol. 43:446–450. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Roncaroli F, Lamovec J, Zidar A and Eusebi
V: Acinic cell-like carcinoma of the breast. Virchows Arch.
429:69–74. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Guerini-Rocco E, Hodi Z, Piscuoglio S, Ng
CK, Rakha EA, Schultheis AM, Marchiò C, da Cruz Paula A, De Filippo
MR, Martelotto LG, et al: The repertoire of somatic genetic
alterations of acinic cell carcinomas of the breast: An
exploratory, hypothesis-generating study. J Pathol. 237:166–178.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
WHO Classification of Tumors Editorial
Board. Breast tumours. WHO Classification of Tumors. 5th edition.
Lyon; IARC: 2019
|
|
11
|
Geyer FC, Berman SH, Marchio C, Burke KA,
Guerini-Rocco E, Piscuoglio S, Ng CK, Pareja F, Wen HY, Hodi Z, et
al: Genetic analysis of microglandular adenosis and acinic cell
carcinomas of the breast provides evidence for the existence of a
low-grade triple-negative breast neoplasia family. Mod Pathol.
30:69–84. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Beca F, Lee SSK, Pareja F, Da Cruz Paula
A, Selenica P, Ferrando L, Gularte-Mérida R, Wen HY, Zhang H,
Guerini-Rocco E, et al: Whole-exome sequencing and RNA sequencing
analyses of acinic cell carcinomas of the breast. Histopathology.
75:931–937. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Collins LC, Connolly JL, Page DL, Goulart
RA, Pisano ED, Fajardo LL, Berg WA, Caudry DJ, McNeil BJ and
Schnitt SJ: Diagnostic agreement in the evaluation of image-guided
breast core needle biopsies: Results from a randomized clinical
trial. Am J Surg Pathol. 28:126–31. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ajkunic A, Skenderi F, Shaker N, Akhtar S,
Lamovec J, Gatalica Z and Vranic S: Acinic cell carcinoma of the
breast: A comprehensive review. Breast. 66:208–216. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cima L, Kaya H, Marchiò C, Nishimura R,
Wen HY, Fabbri VP and Foschini MP: Triple-negative breast
carcinomas of low malignant potential: Review on diagnostic
criteria and differential diagnoses. Virchows Arch. 480:109–126.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wolff AC, Hammond MEH, Allison KH, Harvey
BE, Mangu PB, Bartlett JMS, Bilous M, Ellis IO, Fitzgibbons P,
Hanna W, et al: Human epidermal growth factor receptor 2 testing in
breast cancer: American Society of Clinical Oncology/College of
American pathologists clinical practice guideline focused update. J
Clin Oncol. 36:2105–2122. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Abdel-Salam EM, Faisal M, Alatar AA,
Qahtan AA and Alam P: Genome-wide transcriptome variation landscape
in Ruta chalepensis organs revealed potential genes responsible for
rutin biosynthesis. J Biotechnol. Jan. 325:43–56. 2021.PubMed/NCBI
|
|
18
|
Chen S, Zhou Y, Chen Y and Gu J: fastp: An
ultra-fast all-in-one FASTQ preprocessor. Bioinformatics.
34:i884–i890. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li H and Durbin R: Fast and accurate short
read alignment with Burrows-Wheeler transform. Bioinformatics.
25:1754–1760. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cibulskis K, Lawrence MS, Carter SL,
Sivachenko A, Jaffe D, Sougnez C, Gabriel S, Meyerson M, Lander ES
and Getz G: Sensitive detection of somatic point mutations in
impure and heterogeneous cancer samples. Nat Biotechnol.
31:213–219. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yen JL, Garcia S, Montana A, Harris J,
Chervitz S, Morra M, West J, Chen R and Church DM: A variant by any
name: Quantifying annotation discordance across tools and clinical
databases. Genome Med. 9:72017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Magny SJ, Shikhman R and Keppke AL: Breast
Imaging Reporting and Data System. 2022 Aug 29. StatPearls.
StatPearls Publishing; Treasure Island, FL: 2023
|
|
23
|
Thavaraj S, Cobb A, Kalavrezos N, Beale T,
Walker DM and Jay A: Carcinoma cuniculatum arising in the tongue.
Head Neck Pathol. 6:130–134. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ushiku T, Arnason T, Ban S, Hishima T,
Shimizu M, Fukayama M and Lauwers GY: Very well-differentiated
gastric carcinoma of intestinal type: Analysis of diagnostic
criteria. Mod Pathol. 26:1620–1631. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Gonzalez CG, Akula S and Burleson M: The
role of mediator subunit 12 in tumorigenesis and cancer
therapeutics. Oncol Lett. 23:742022. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yoshida M, Sekine S, Ogawa R, Yoshida H,
Maeshima A, Kanai Y, Kinoshita T and Ochiai A: Frequent MED12
mutations in phyllodes tumours of the breast. Br J Cancer.
112:1703–1708. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Banerji S, Cibulskis K, Rangel-Escareno C,
Brown KK, Carter SL, Frederick AM, Lawrence MS, Sivachenko AY,
Sougnez C, Zou L, et al: Sequence analysis of mutations and
translocations across breast cancer subtypes. Nature. 486:405–409.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Conlon N, Sadri N, Corben AD and Tan LK:
Acinic cell carcinoma of breast: morphologic and
immunohistochemical review of a rare breast cancer subtype. Hum
Pathol. 51:16–24. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tarantino P, Hamilton E, Tolaney SM,
Cortes J, Morganti S, Ferraro E, Marra A, Viale G, Trapani D,
Cardoso F, et al: HER2-Low breast cancer: Pathological and clinical
landscape. J Clin Oncol. 38:1951–1962. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Banerji U, van Herpen C, Saura C,
Thistlethwaite F, Lord S, Moreno V, Macpherson I, Boni V, Rolfo C,
de Vries E, et al: Trastuzumab duocarmazine in locally advanced and
metastatic solid tumours and HER2-expressing breast cancer: A phase
1 dose-escalation and dose-expansion study. Lancet Oncol.
20:1124–1135. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Jeong YJ, Park SH, Mun SH, Kwak SG, Lee SJ
and Oh HK: Association between lysyl oxidase and fibrotic focus in
relation with inflammation in breast cancer. Oncol Lett.
15:2431–2440. 2018.PubMed/NCBI
|