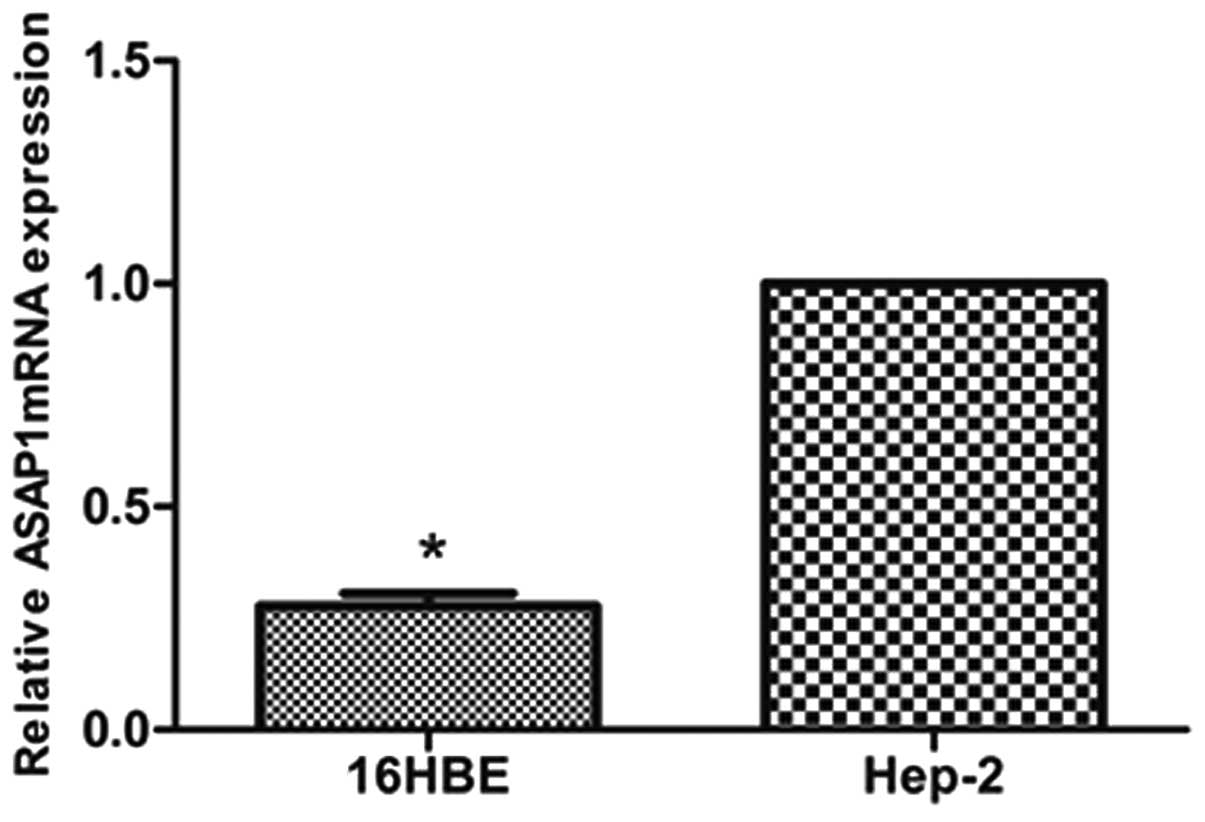
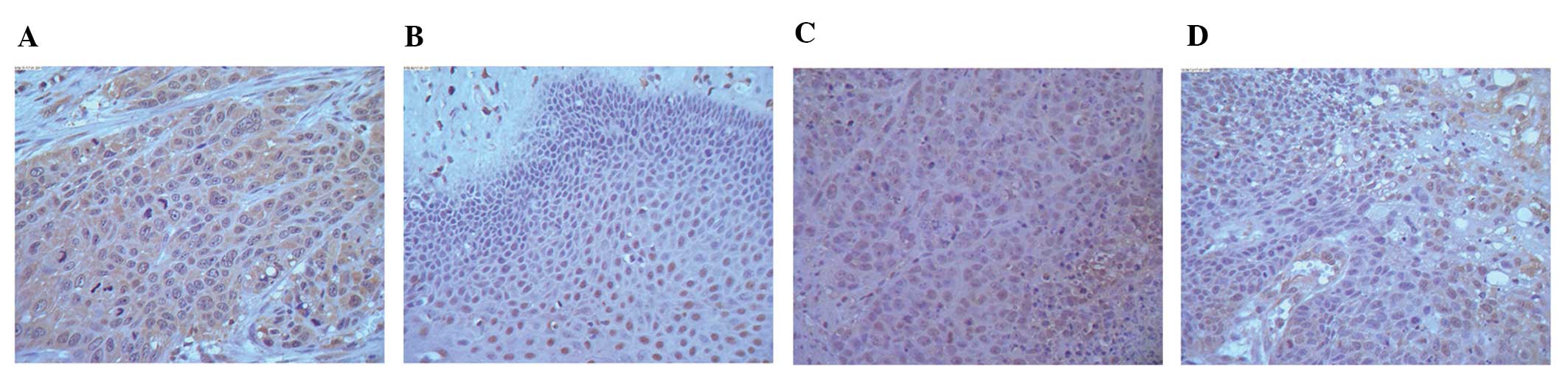
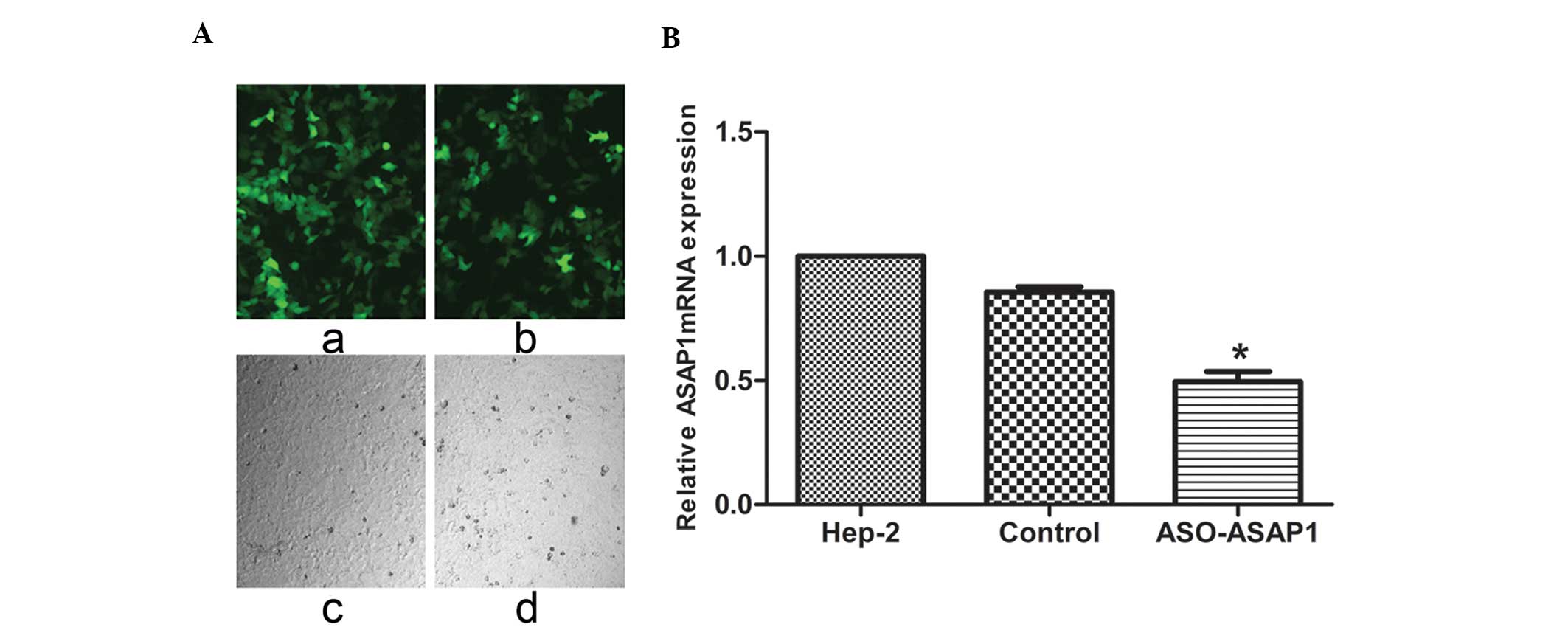
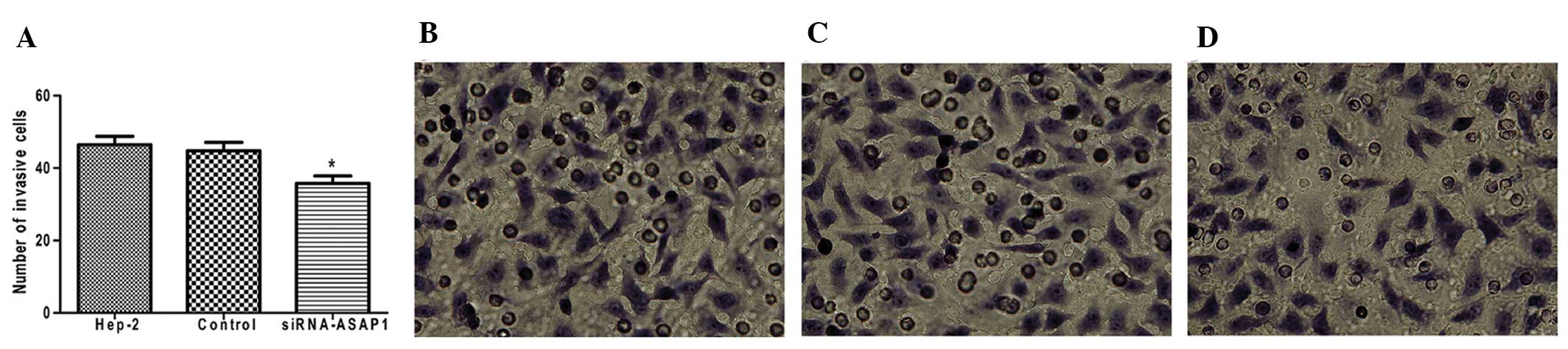
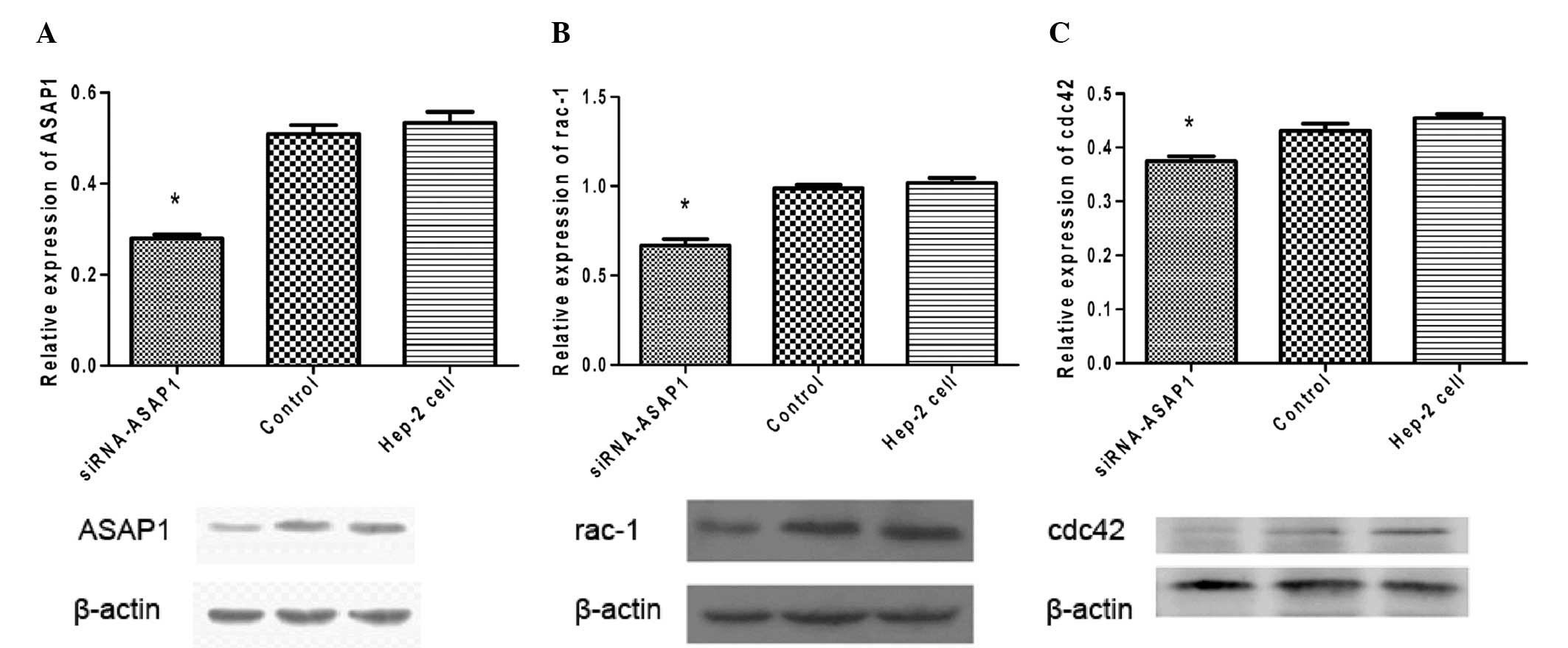
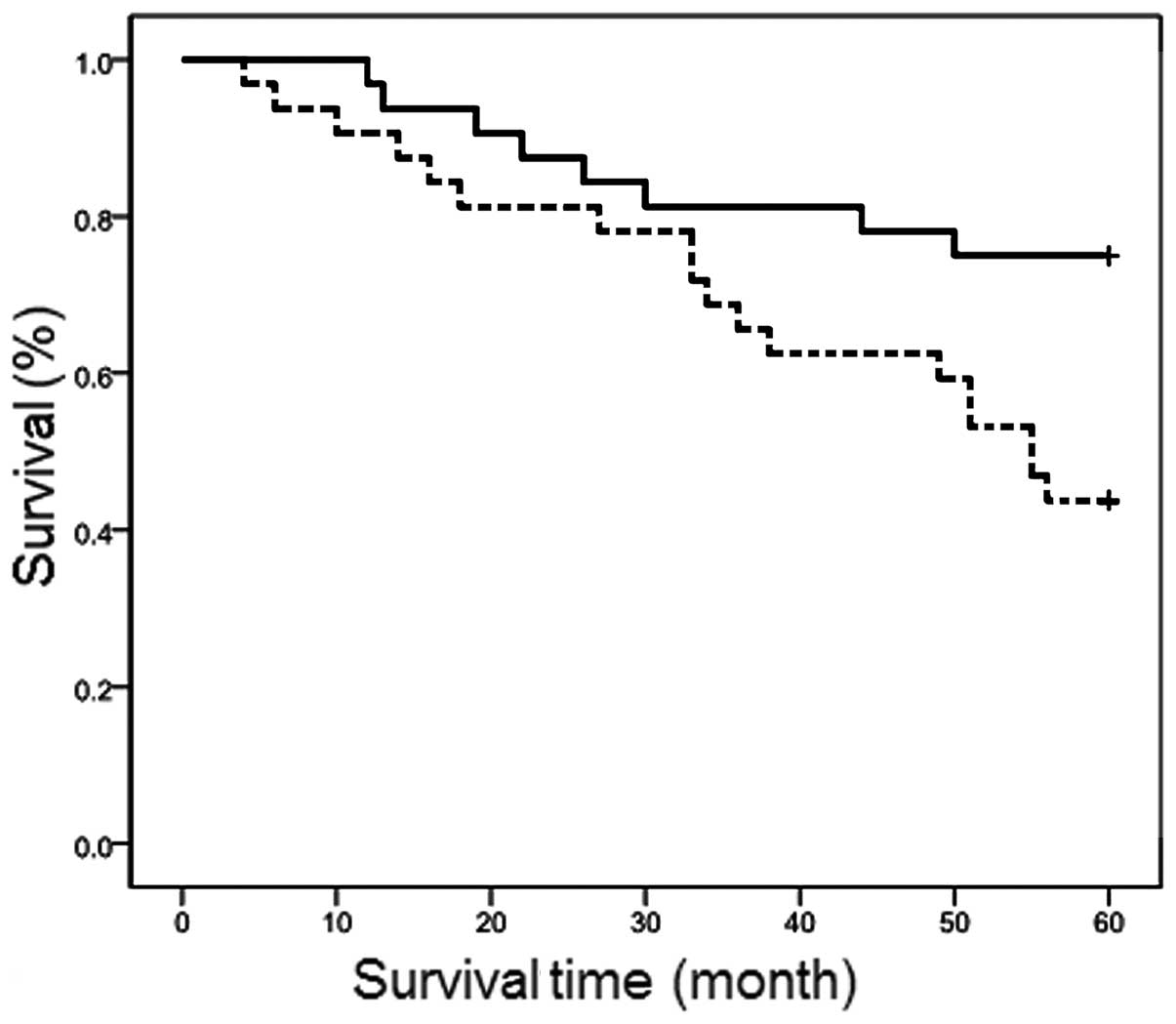
|
1
|
Inoue H and Randazzo PA: Arf GAPs and
their interacting proteins. Traffic. 8:1465–1475. 2007. View Article : Google Scholar
|
|
2
|
Buffart TE, Coffa J, Hermsen MA, et al:
DNA copy number changes at 8q11–24 in metastasized colorectal
cancer. Cell Oncol. 27:57–65. 2005.
|
|
3
|
Martin RK and Jackson TR: Centaurin β4 in
cancer. Biochem Soc Trans. 33:1282–1284. 2005.
|
|
4
|
Inoue H, Ha VL, Prekeris R and Randazzo
PA: Arf GTPase-activating protein ASAP1 interacts with Rab11
effector FIP3 and regulates pericentrosomal localization of
transferrin receptor-positive recycling endosome. Mol Biol Cell.
19:4224–4237. 2008. View Article : Google Scholar
|
|
5
|
Jian X, Brown P, Schuck P, et al:
Autoinhibition of Arf GTPase-activating protein activity by the BAR
domain in ASAP1. J Biol Chem. 284:1652–1663. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liu Y, Loijens JC, Martin KH, Karginov AV
and Parsons JT: The association of ASAP1, an ADP ribosylation
factor-GTPase activating protein, with focal adhesion kinase
contributes to the process of focal adhesion assembly. Mol Biol
Cell. 13:2147–2156. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Liu Y, Yerushalmi GM, Grigera PR and
Parsons JT: Mislocalization or reduced expression of Arf
GTPase-activating protein ASAP1 inhibits cell spreading and
migration by influencing Arf1 GTPase cycling. J Biol Chem.
280:8884–8892. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Brown MT, Andrade J, Radhakrishna H, et
al: ASAP1, a phospholipid-dependent arf GTPase-activating protein
that associates with and is phosphorylated by Src. Mol Cell Biol.
18:7038–7051. 1998.PubMed/NCBI
|
|
9
|
Bharti S, Inoue H, Bharti K, et al:
Src-dependent phosphorylation of ASAP1 regulates podosomes. Mol
Cell Biol. 27:8271–8283. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
King FJ, Hu E, Harris DF, Sarraf P,
Spiegelman BM and Roberts TM: DEF-1, a novel Src SH3 binding
protein that promotes adipogenesis in fibroblastic cell lines. Mol
Cell Biol. 19:2330–2337. 1999.PubMed/NCBI
|
|
11
|
Kam JL, Miura K, Jackson TR, et al:
Phosphoinositide-dependent activation of the ADP-ribosylation
factor GTase-activating protein ASAP1. Evidence for the pleckstrin
homology domain functioning as an allosteric site. J Biol Chem.
275:9653–9663. 2000. View Article : Google Scholar
|
|
12
|
Randazzo PA, Andrade J, Miura K, et al:
The Arf GTPase-activating protein ASAP1 regulates the actin
cytoskeleton. Proc Natl Acad Sci USA. 97:4011–4016. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Nie Z and Randazzo PA: Arf GAPs and
membrane traffic. J Cell Sci. 119:1203–1211. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Donaldson JG and Jackson CL: ARF family G
protein and their regulators: roles in membrane transport,
development and disease. Nat Rev Mol Cell Biol. 12:362–375. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Onodera Y, Hashimoto S, Hashimoto A, et
al: Expression of AMAP1, an ArfGAP, provides novel targets to
inhibit breast cancer invasive activities. EMBO J. 24:963–973.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Muller T, Stein U and Poletti A: ASAP1
promotes tumor cell motility and invasiveness, stimulates
metastasis formation in vivo, and correlates with poor survival in
colorectal cancer patients. Oncogene. 29:2393–2403. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ehlers JP, Worley L, Onken MD and Harbour
JW: DDEF1 is located in an amplified region of chromosome 8q
and is overexpressed in uveal melanoma. Clin Cancer Res.
11:3609–3613. 2005. View Article : Google Scholar
|
|
18
|
Lin D, Watahiki A, Bayani J, et al:
ASAP1, a gene at 8q24, is associated with prostate cancer
metastasis. Cancer Res. 68:4352–4359. 2008. View Article : Google Scholar
|
|
19
|
Randazzo PA, Inoue H and Bharti S: Arf
GAPs as regulators of the actin cytoskeleton. Biol Cell.
99:583–600. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Furman C, Short SM, Subramanian RR, Zetter
BR and Roberts TM: DEF-1/ASAP1 is a GTPase-activating protein (GAP)
for ARF1 that enhances cell motility through a GAP-dependent
mechanism. J Biol Chem. 277:7962–7969. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bar-Sagi D and Hall A: Ras and Rho
GTPases: a family reunion. Cell. 103:227–238. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wagner AC and Williams JA: Low molecular
weight GTP-binding proteins: molecular switches regulating diverse
cellular functions. Am J Physiol. 266:G1–G14. 1994.PubMed/NCBI
|
|
23
|
Waschke J, Burger S, Curry FR, Drenckhahn
D and Adamson RH: Activation of Rac-1 and Cdc42 stabilizes the
microvascular endothelial barrier. Histochem Cell Biol.
125:397–406. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lamarche N, Tapon N, Stowers L, et al: Rac
and Cdc42 induce actin polymerization and G1 cell cycle progression
independently of p65PAK and the JNK/SAPK MAP kinase cascade. Cell.
87:519–529. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yang FC, Atkinson SJ, Gu Y, et al: Rac and
Cdc42 GTPases control hematopoietic stem cell shape, adhesion,
migration, and mobilization. Proc Natl Acad Sci USA. 98:5614–5618.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Huang M, Satchell L, Duhadaway JB,
Prendergast GC and Laury-Kleintop LD: RhoB links PDGF signaling to
cell migration by coordinating activation and localization of Cdc42
and Rac. J Cell Biochem. 112:1572–1584. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kaibuchi K, Kuroda S and Amano M:
Regulation of the cytoskeleton and cell adhesion by the Rho family
GTPases in mammalian cells. Annu Rev Biochem. 68:459–486. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Engers R, Springer E, Michiels F, Collard
JG and Gabbert HE: Rac affects invasion of human renal cell
carcinomas by up-regulating tissue inhibitor of metalloproteinases
(TIMP)-1 and TIMP-2 expression. J Biol Chem. 276:41889–41897. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Filipenko NR, Attwell S, Roskelley C and
Dedhar S: Integrin-linked kinase activity regulates Rac- and
Cdc42-mediated actin cytoskeleton reorganization via α-PIX.
Oncogene. 24:5837–5849. 2005.PubMed/NCBI
|
|
30
|
Fidler IJ: The pathogenesis of cancer
metastasis: the ‘seed and soil’ hypothesis revisited. Nat Rev
Cancer. 3:453–458. 2003.
|
|
31
|
Foley R, Hollywood D and Lawler M:
Molecular pathology of prostate cancer: the key to identifying new
biomarkers of disease. Endocr Relat Cancer. 11:477–488. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Miyata M, Raven JF, Baltzis D, Koromilas
AE and Sabe H: IRES-mediated translational control of AMAP1
expression during differentiation of monocyte U937 cells. Cell
Cycle. 7:3273–3281. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Jackson TR, Brown FD, Nie Z, et al: ACAPs
are arf6 GTPase-activating proteins that function in the cell
periphery. J Cell Biol. 151:627–638. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kondo A, Hashimoto S, Yano H, Nagayama K,
Mazaki Y and Sabe H: A new paxillin-binding protein,
PAG3/Papα/KIAA0400, bearing an ADP-ribosylation factor
GTPase-activating protein activity, is involved in paxillin
recruitment to focal adhesions and cell migration. Mol Biol Cell.
11:1315–1327. 2000.PubMed/NCBI
|
|
35
|
Sabe H, Hashimoto S, Morishige M, et al:
The EGFR-GEP100-Arf6-AMAP1 signaling pathway specific to breast
cancer invasion and metastasis. Traffic. 10:982–993. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Nam JM, Onodera Y, Mazaki Y, Miyoshi H,
Hashimoto S and Sabe H: CIN85, a Cbl-interacting protein, is a
component of AMAP1-mediated breast cancer invasion machinery. EMBO
J. 26:647–656. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Onodera Y, Nam JM, Hashimoto A, et al:
Rab5c promotes AMAP1-PRKD2 complex formation to enhance beta1
integrin recycling in EGF-induced cancer invasion. J Cell Biol.
197:983–996. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ridley AJ, Paterson HF, Johnston CL,
Diekmann D and Hall A: The small GTP-binding protein rac regulates
growth factor-induced membrane ruffling. Cell. 70:401–410. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Chianale F, Cutrupi S, Rainero E, et al:
Diacylglycerol kinase-alpha mediates hepatocyte growth
factor-induced epithelial cell scatter by regulating Rac activation
and membrane ruffling. Mol Biol Cell. 18:4859–4871. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Nobes CD and Hall A: Rho, rac, and cdc42
GTPases regulate the assembly of multimolecular focal complexes
associated with actin stress fibers, lamellipodia, and filopodia.
Cell. 81:53–62. 1995. View Article : Google Scholar
|