|
1
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clin. 64:9–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Liu S, Goldstein RH, Scepansky EM and
Rosenblatt M: Inhibition of rho-associated kinase signaling
prevents breast cancer metastasis to human bone. Cancer Res.
69:8742–8751. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Thiery JP, Acloque H, Huang RY and Nieto
MA: Epithelial-mesenchymal transitions in development and disease.
Cell. 139:871–890. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yilmaz M and Christofori G: EMT, the
cytoskeleton, and cancer cell invasion. Cancer Metastasis Rev.
28:15–33. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Son H and Moon A: Epithelial-mesenchymal
transition and cell invasion. Toxicol Res. 26:245–252. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gonzalez DM and Medici D: Signaling
mechanisms of the epithelial-mesenchymal transition. Sci Signal.
7:re82014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chen Q, He H, Li P, Zhu J and Xiong M:
Identification and quantification of atractylenolide I and
atractylenolide III in Rhizoma Atractylodes macrocephala by liquid
chromatographyion trap mass spectrometry. Biomed Chromatogr.
27:699–707. 2013. View
Article : Google Scholar
|
|
8
|
Kang TH, Bang JY, Kim MH, Kang IC, Kim HM
and Jeong HJ: Atractylenolide III, a sesquiterpenoid, induces
apoptosis in human lung carcinoma A549 cells via
mitochondria-mediated death pathway. Food Chem Toxicol. 49:514–519.
2011. View Article : Google Scholar
|
|
9
|
Zhang NN, Park DK and Park HJ: The
inhibitory activity of atractylenolide III, a sesquiterpenoid, on
IgE-mediated mast cell activation and passive cutaneous anaphylaxis
(PCA). J Ethnopharmacol. 145:278–285. 2013. View Article : Google Scholar
|
|
10
|
Li CQ, He LC and Jin JQ: Atractylenolide I
and atractylenolide III inhibit lipopolysaccharide-induced
TNF-alpha and NO production in macrophages. Phytother Res.
21:347–353. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu C, Zhao H, Ji ZH and Yu XY:
Neuroprotection of atractylenolide III from Atractylodis
macrocephalae against glutamate-induced neuronal apoptosis via
inhibiting caspase signaling pathway. Neurochem Res. 39:1753–1758.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang KT, Chen LG, Wu CH, Chang CC and Wang
CC: Gastroprotective activity of atractylenolide III from
Atractylodes ovata on ethanol-induced gastric ulcer in vitro and in
vivo. J Pharm Pharmacol. 62:381–388. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kang TH, Han NR, Kim HM and Jeong HJ:
Blockade of IL-6 secretion pathway by the sesquiterpenoid
atractylenolide III. J Nat Prod. 74:223–227. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
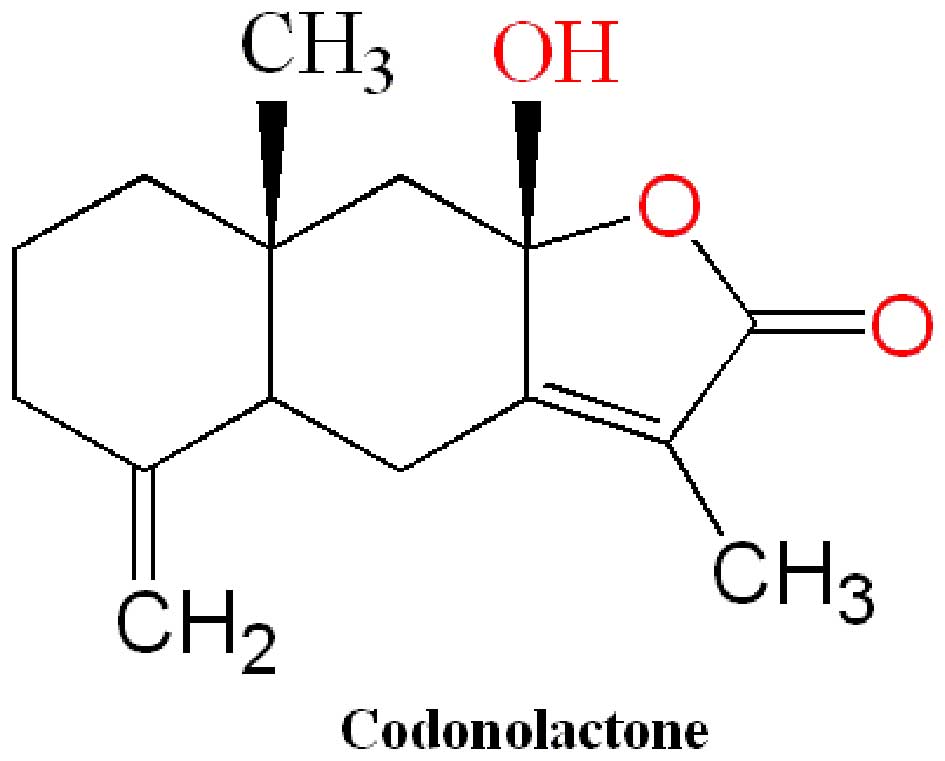
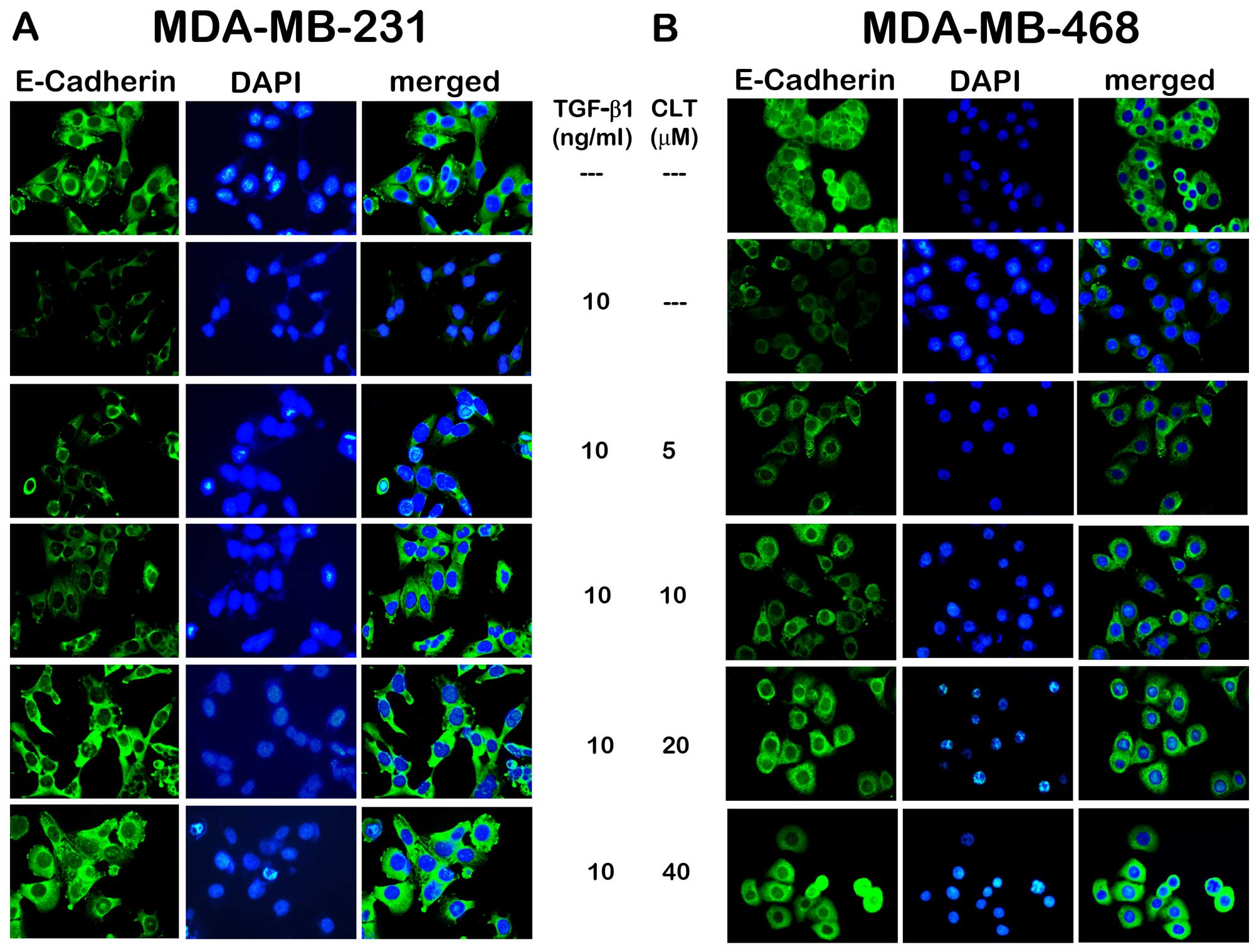
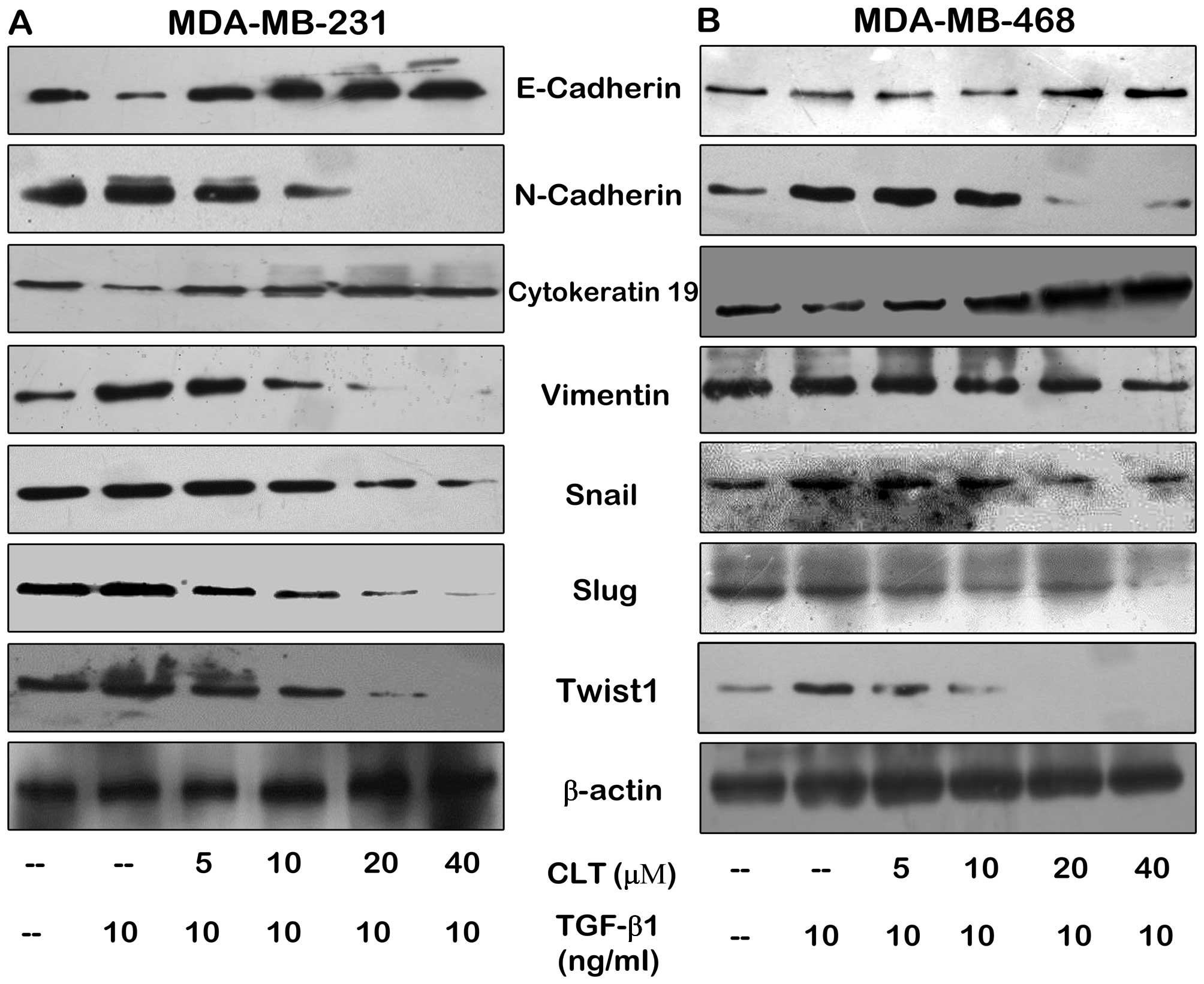
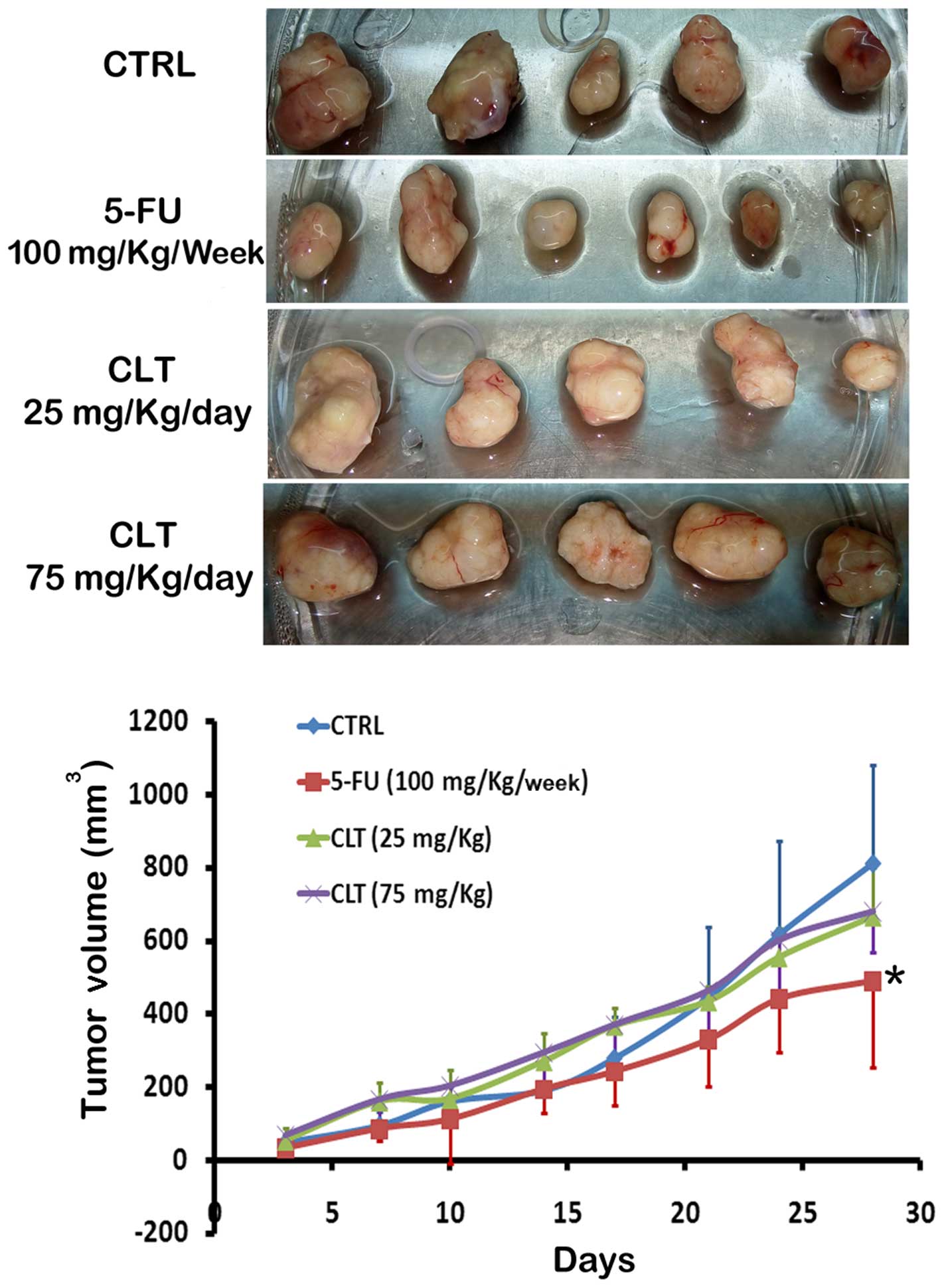
Wang W, Chen B, Zou R, Tu X, Tan S, Lu H,
Liu Z and Fu J: Codonolactone, a sesquiterpene lactone isolated
from Chloranthus henryi Hemsl, inhibits breast cancer cell
invasion, migration and metastasis by downregulating the
transcriptional activity of Runx2. Int J Oncol. 45:1891–1900.
2014.PubMed/NCBI
|
|
15
|
Ferrari N, McDonald L, Morris JS, Cameron
ER and Blyth K: RUNX2 in mammary gland development and breast
cancer. J Cell Physiol. 228:1137–1142. 2013. View Article : Google Scholar
|
|
16
|
Lee KS, Kim HJ, Li QL, Chi XZ, Ueta C,
Komori T, Wozney JM, Kim EG, Choi JY, Ryoo HM, et al: Runx2 is a
common target of transforming growth factor beta1 and bone
morphogenetic protein 2, and cooperation between Runx2 and Smad5
induces osteoblast-specific gene expression in the pluripotent
mesenchymal precursor cell line C2C12. Mol Cell Biol. 20:8783–8792.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Phimphilai M, Zhao Z, Boules H, Roca H and
Franceschi RT: BMP signaling is required for RUNX2-dependent
induction of the osteoblast phenotype. J Bone Miner Res.
21:637–646. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bonnomet A, Syne L, Brysse A, Feyereisen
E, Thompson EW, Noël A, Foidart JM, Birembaut P, Polette M and
Gilles C: A dynamic in vivo model of epithelial-to-mesenchymal
transitions in circulating tumor cells and metastases of breast
cancer. Oncogene. 31:3741–3753. 2012. View Article : Google Scholar
|
|
19
|
Zhang Q, Yu N and Lee C: Vicious cycle of
TGF-β signaling in tumor progression and metastasis. Am J Clin Exp
Urol. 2:149–155. 2014.
|
|
20
|
Borthwick LA, Gardner A, De Soyza A, Mann
DA and Fisher AJ: Transforming growth factor-β1 (TGF-β1) driven
epithelial to mesenchymal transition (EMT) is accentuated by tumour
necrosis factor α (TNFα) via crosstalk between the SMAD and NF-κB
pathways. Cancer Microenviron. 5:45–57. 2012. View Article : Google Scholar
|
|
21
|
Savagner P: The epithelial-mesenchymal
transition (EMT) phenomenon. Ann Oncol. 21(Suppl 7): vii89–vii92.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lamouille S, Xu J and Derynck R: Molecular
mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell
Biol. 15:178–196. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zeisberg M and Neilson EG: Biomarkers for
epithelial-mesenchymal transitions. J Clin Invest. 119:1429–1437.
2009. View
Article : Google Scholar : PubMed/NCBI
|
|
24
|
Angst BD, Marcozzi C and Magee AI: The
cadherin superfamily: Diversity in form and function. J Cell Sci.
114:629–641. 2001.PubMed/NCBI
|
|
25
|
Gheldof A and Berx G: Cadherins and
epithelial-to-mesenchymal transition. Prog Mol Biol Transl Sci.
116:317–336. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Eriksson JE, Dechat T, Grin B, Helfand B,
Mendez M, Pallari HM and Goldman RD: Introducing intermediate
filaments: From discovery to disease. J Clin Invest. 119:1763–1771.
2009. View
Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang Y, Shi J, Chai K, Ying X and Zhou BP:
The role of Snail in EMT and tumorigenesis. Curr Cancer Drug
Targets. 13:963–972. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Khan MA, Chen HC, Zhang D and Fu J: Twist:
A molecular target in cancer therapeutics. Tumour Biol.
34:2497–2506. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yang Z, Zhang X, Gang H, Li X, Li Z, Wang
T, Han J, Luo T, Wen F and Wu X: Up-regulation of gastric cancer
cell invasion by Twist is accompanied by N-cadherin and fibronectin
expression. Biochem Biophys Res Commun. 358:925–930. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Derynck R, Muthusamy BP and Saeteurn KY:
Signaling pathway cooperation in TGF-β-induced
epithelial-mesenchymal transition. Curr Opin Cell Biol. 31:56–66.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Thomson S, Petti F, Sujka-Kwok I, Mercado
P, Bean J, Monaghan M, Seymour SL, Argast GM, Epstein DM and Haley
JD: A systems view of epithelial-mesenchymal transition signaling
states. Clin Exp Metastasis. 28:137–155. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Xu J, Lamouille S and Derynck R:
TGF-beta-induced epithelial to mesenchymal transition. Cell Res.
19:156–172. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Peinado H, Quintanilla M and Cano A:
Transforming growth factor beta-1 induces snail transcription
factor in epithelial cell lines: Mechanisms for epithelial
mesenchymal transitions. J Biol Chem. 278:21113–21123. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Vincent T, Neve EP, Johnson JR, Kukalev A,
Rojo F, Albanell J, Pietras K, Virtanen I, Philipson L, Leopold PL,
et al: A SNAIL1-SMAD3/4 transcriptional repressor complex promotes
TGF-beta mediated epithelial-mesenchymal transition. Nat Cell Biol.
11:943–950. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Thuault S, Tan EJ, Peinado H, Cano A,
Heldin CH and Moustakas A: HMGA2 and Smads co-regulate SNAIL1
expression during induction of epithelial-to-mesenchymal
transition. J Biol Chem. 283:33437–33446. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yamashita M, Fatyol K, Jin C, Wang X, Liu
Z and Zhang YE: TRAF6 mediates Smad-independent activation of JNK
and p38 by TGF-beta. Mol Cell. 31:918–924. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Xie L, Law BK, Chytil AM, Brown KA, Aakre
ME and Moses HL: Activation of the Erk pathway is required for
TGF-beta1-induced EMT in vitro. Neoplasia. 6:603–610. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Stein GS, Lian JB, van Wijnen AJ, Stein
JL, Montecino M, Javed A, Zaidi SK, Young DW, Choi JY and Pockwinse
SM: Runx2 control of organization, assembly and activity of the
regulatory machinery for skeletal gene expression. Oncogene.
23:4315–4329. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Komori T: Runx2, a multifunctional
transcription factor in skeletal development. J Cell Biochem.
87:1–8. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Inman CK and Shore P: The osteoblast
transcription factor Runx2 is expressed in mammary epithelial cells
and mediates osteopontin expression. J Biol Chem. 278:48684–48689.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Gupta GP and Massagué J: Cancer
metastasis: Building a framework. Cell. 127:679–695. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Chimge NO, Baniwal SK, Little GH, Chen YB,
Kahn M, Tripathy D, Borok Z and Frenkel B: Regulation of breast
cancer metastasis by Runx2 and estrogen signaling: The role of
SNAI2. Breast Cancer Res. 13:R1272011. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Niu DF, Kondo T, Nakazawa T, Oishi N,
Kawasaki T, Mochizuki K, Yamane T and Katoh R: Transcription factor
Runx2 is a regulator of epithelial-mesenchymal transition and
invasion in thyroid carcinomas. Lab Invest. 92:1181–1190. 2012.
View Article : Google Scholar : PubMed/NCBI
|